Back to Journals » Therapeutics and Clinical Risk Management » Volume 15
Histological type of focal cortical dysplasia is associated with the risk of postsurgical seizure in children and adolescents
Authors Chen J, Huang Z, Li L, Ren L, Wang Y
Received 26 January 2019
Accepted for publication 14 June 2019
Published 11 July 2019 Volume 2019:15 Pages 877—884
DOI https://doi.org/10.2147/TCRM.S203039
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Deyun Wang
Jia Chen,1,2 Zhaoyang Huang,1,2 Liping Li,1,2 Liankun Ren,1,2 Yuping Wang1,2
1Department of Neurology, Xuanwu Hospital, Capital Medical University, Beijing 100053, People’s Republic of China; 2Beijing Key Laboratory of Neuromodulation, Beijing 100053, People’s Republic of China
Aim: Focal cortical dysplasia (FCD) is a common cause of refractory epilepsy in children and adolescents. Epilepsy surgery is a treatment option for FCD. This study aimed to investigate the relationship between postsurgical outcomes and FCD types according to the International League Against Epilepsy (ILAE) classification and assess prognostic factors in pediatric and adolescent epilepsy surgery.
Methods: We retrospectively analyzed 92 children and adolescents with a proven pathological diagnosis of FCD who underwent resective surgery at our epilepsy center between August 2012 and September 2015. The patients were followed up for at least 1 year to evaluate the surgical outcomes, and a multivariable regression analysis was performed to identify risk factors of seizure relapse.
Results: After surgery, 53.3% of the patients were completely seizure-free during the entire follow-up period. The FCD types, incomplete resection of the epileptic cortex, and use of intracranial electrode were independent risk factors of seizure recurrence. The patients with FCD type II had relatively favorable surgical outcomes compared to the patients with FCD type I. The difference in the postoperative outcome between patients with FCD types III and I was not significant.
Conclusion: There is a significant association between FCD types and surgical outcomes in children and adolescents with epilepsy. These findings provide guidance for the optimization of surgical strategies.
Keywords: epilepsy, focal cortical dysplasia, surgery, outcome, children, adolescent
Introduction
Focal cortical dysplasia (FCD) is a frequent cause of focal epilepsy in children and adolescents and is considered as a type of malformation in cortical development characterized by localized aberrant cortical architecture. Most FCD patients experience the onset of epilepsy by 16 years of age, and the seizures are often medically refractory.1
Over the past decades, various FCD classification systems have been proposed. The new three-tiered FCD classification proposed by the International League Against Epilepsy (ILAE) is based on Palmini’s FCD classification and distinguishes isolated forms from those associated with other epileptic lesions, such as hippocampal sclerosis and tumors, which are defined as FCD type III.2 Surgery is a therapeutic option for intractable epilepsy in patients with FCD. Several studies have focused on the prognosis of FCD surgery.3–7 The reported postoperative outcome of FCD patients is controversial, and the heterogeneity in the patient age among different studies could contribute to the inconsistent results.8 Therefore, an epilepsy surgery series focused on a specific age group is valuable for improving our understanding of the disease. Furthermore, most previous studies used Palmini’s FCD classification. The new ILAE classification proposed a new type III, ie, associated FCD, which is differentiated from isolated FCD. Outcome data regarding the new classification by the ILAE, particularly regarding FCD type III in children and adolescents are limited.8,9
Therefore, this study aimed to investigate the surgery outcomes in children and adolescent patients with FCD as the cause of epilepsy and prognostic factors of pediatric and adolescent epilepsy surgery. In addition, we investigated the relationship between postsurgical seizure outcomes and different FCD types using the new ILAE classification.
Methods
Patient selection
We retrospectively analyzed consecutive patients diagnosed with pharmacoresistant epilepsy who underwent resective epilepsy surgery at our center between August 2012 and September 2015. All patients had been treated with first-line antiepileptic drugs and fulfilled the criteria for drug resistance proposed by ILAE, which defines drug resistance as “failure of adequate trials of two tolerated and appropriately chosen anti-epileptic drugs (AEDs) schedules (whether as monotherapies or in combination) to achieve seizure freedom”.10 Patients who underwent previous resective epilepsy surgery were excluded. All patients underwent a thorough presurgical evaluation to assess the extent of the resection. This study was approved by Ethics Committee of Xuanwu Hospital, and was conducted in accordance with the Declaration of Helsinki. All patients or their parental or legal guardian provided written informed consent, including the use of their medical data for research.
Presurgical evaluation
For each patient, the clinical and investigatory data were considered in the presurgical evaluation, including clinical history, neurological examination, magnetic resonance imaging (MRI), and long-term scalp video-electroencephalography (EEG) monitoring. The MRI scans were obtained at 1.5 or 3.0 Tesla and included axial and coronal T1-weighted, axial T2-weighted, and axial fluid-attenuated inversion recovery (FLAIR) sequences. The MRI findings were divided according to the presence or absence of lesions (normal or nonspecific findings that could not explain the epilepsy were considered as the absence of lesions). The scalp EEG was acquired using a 10–20 International System. For statistical purposes, we also classified the epileptiform activity as regional, lateralized and generalized abnormalities. If the location of the epileptogenic zone was unclear, additional investigations, including single-photon-emission computed tomography (SPECT), positron emission tomography (PET), and magnetoencephalography (MEG), were performed. The presurgical evaluation findings were reviewed at a multidisciplinary seizure conference, and the decisions to proceed with surgery or perform an invasive evaluation were made. At our institution, we use a combination of subdural and deep electrodes for invasive intracranial monitoring.
Surgical procedure
Based on the results of the presurgical evaluation, decisions regarding the type and extent of surgery were made for each individual patient after a multidisciplinary case discussion. The resection areas included the lesions as indicated by MRI, EEG onset zone and adjacent epileptogenic areas. In certain cases, intraoperative electrocorticography (ECoG) was performed to avoid eloquent cortex resection. The surgeries were classified into the following 4 categories according to the resection extent: standard anterior temporal lobectomy, unilobar, multilobar, and hemispherical resection. All patients underwent a postoperative MRI to identify the completeness of excision. The complete excision of MRI lesions and seizure onset zones was considered a complete surgical resection. In cases of the area of the MRI lesion or seizure onset zone overlapped with an eloquent cortical region, the resection may be incomplete to avoid unacceptable neurological deficits. A histopathological analysis of each resected brain was performed, and the lesions were pathologically classified based on the histological abnormalities. The FCD abnormalities were classified as FCD types I, II or III according to the ILAE classification.2 After surgery patients received AEDs treatment and follow-up including EEG.
Outcome assessment
The surgical outcomes were annually assessed using the ILAE classification.11 The patients regularly visited the outpatient clinic or were contacted via telephone. Follow-up visits were scheduled at 3 months and 1 year after surgery, followed by annual visits for an outcome evaluation. In our study, only ILAE class 1 was considered seizure-free, and postoperative auras were considered as seizures. Seizures that occurred during the first 2 weeks after surgery were excluded. We assessed the outcomes until January 2017.
Statistical analysis
Statistical analysis was performed using medians and ranges for the continuous variables and frequencies for the categorical variables. For exploratory purposes, the variables were first screened by performing Wilcoxon rank-sum, chi-square, and Fisher’s exact tests to compare the seizure-free patients to those with seizure recurrence regardless of the follow-up time. The variables with a significance level of 10% based on the initial univariable analysis were then tested in a multivariable Cox regression analysis. Follow-up duration indicated the time from at 3 months after surgery till the latest yearly evaluation. First, a Kaplan-Meier survival analysis was performed to calculate the probability of seizure-free survival in the overall group, and then, the FCD types were considered. The differences between the FCD types were analyzed using a log-rank test. Significance was established at P<0.05, and the accepted confidence interval (CI) was 95%. All analyses were performed using SPSS version 18.0 (IBM Corporation, Armonk, NY, USA).
Results
Patient characteristics
Patients who met the following criteria were included in the analysis: age <18 years at epilepsy surgery, pathology-proven FCD without dual pathology, and complete postsurgical follow-up data for at least 12 months. In total, 92 patients (59 males and 33 females) were included in the study. Based on histological evaluation, 34 (37.0%) patients had FCD type I, 20 (21.7%) patients had FCD type II, and 38 (41.3%) patients had FCD type III. The seizure-onset zone was considered temporal in 36 (39.1%) cases and extratemporal in 56 (60.9%) cases. The other characteristics of our cohort are shown in Table 1.
 |
Table 1 Characteristics of patients |
Overall outcomes
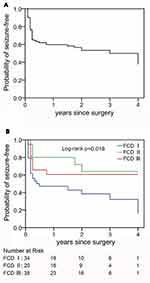
Of the 92 patients, 49 (53.3%) patients had not experienced seizure recurrence after surgery, and 43 (46.7%) patients had seizure relapse. The seizure outcomes of the 92 patients are presented in a Kaplan-Meier survival curve (Figure 1A). The probabilities of seizure-free survival at 1, 2, 3, and 4 years after surgery were 0.598 (95% CI, 0.498 to 0.697), 0.533 (95% CI, 0.425 to 0.641), 0.500 (95% CI, 0.380 to 0.620), and 0.375 (95% CI, 0.146 to 0.604), respectively.
Risk factors of seizure relapse
First, we performed a univariable screening of potential risk factors. The histological type of FCD had a significant effect on the outcome, and the use of invasive EEG was significantly associated with surgical failure (Table 2). The other prognostic indicators significantly correlated with the postsurgical seizure recurrence included incomplete resection and absence of MRI lesions (Table 2). No significant difference was observed in the epilepsy duration, age at epilepsy onset, age at epilepsy surgery, and presurgical seizure frequency between the patients who were postoperatively seizure-free and those with seizure relapse.
 |
Table 2 Univariable analysis of risk factors for postoperative seizure |
To further verify all risk factors identified as potentially significant in the univariable analysis, we performed a multivariable analysis using a Cox proportional hazards regression. The variables that retained significance included incomplete resection and the use of invasive EEG (adjusted P-values of 0.007 and 0.029, respectively, Table 3). Patients with FCD type II were predicted to have a more favorable outcome than patients with FCD type I. The surgical outcome did not significantly differ between patients with FCD type I and III. The significant association between the absence of MRI lesions and seizure relapse was not confirmed by the multivariable analysis (adjusted P-value =0.503, Table 3).
 |
Table 3 Multivariable analysis of risk factors for postoperative seizure |
Outcome based on different FCD types
Kaplan-Meier survival curves of the seizure-free patients with different FCD types are shown in Figure 1B. The seizure-free rate among the patients with different FCD types rapidly declined within the first year. The probability of seizure-free survival at 1 year after surgery was 0.471 (95% CI, 0.302 to 0.640) in the patients with FCD type I, 0.605 (95% CI, 0.450 to 0.760) in the patients with FCD type II, and 0.640 (95% CI, 0.397 to 0.883) in the patients with FCD type III. After one year, the seizure-free rate in the patients with FCD type I continued to decline until the end of the follow-up year, whereas no seizure recurrence was observed in the patients with FCD type III after one year and in the patients with FCD type II after two years.
Of the patients with postoperative seizures within the first year (n=36), 29 patients had at least two years of follow-up. Of these patients, 3 patients had subsequent remission. Two patients with pathological findings of FCD types I and III were seizure-free during the second postoperative year. Another FCD type III patient became seizure-free during the third year.
Discussion
We report postoperative seizure outcomes in a large series of child and adolescent FCD patients. The overall seizure-free rate was 59.8% during the first year after surgery, and 53.3% of the patients were completely seizure-free throughout the entire follow-up duration. The risk factors of seizure recurrence included the histological types of FCD, incomplete resection and intracranial electrode implantation, whereas negative MRI findings were not predictive of poor postsurgical outcome.
Most previously published studies investigating seizure outcomes after epilepsy surgery have focused on patients with FCD types I and II according to Palmini’s classification, which correspond to isolated FCDs in the ILAE classification system. Engle’s class I outcomes were achieved in 40–75% of these FCD patients.12–14 Recently, several studies have analyzed the clinical characteristics and surgery outcomes of patients with the new FCD type III proposed by the ILAE.15–17 However, these studies compared specific FCD III subtypes with isolated epileptic lesions without FCD, and studies comparing the three FCD types are limited. In our cohort, FCD patients were classified into three pathological types based on the ILAE classification. FCD type I accounted for 37.0% of the patients, type II accounted for 21.7% of the patients, and type III accounted for 41.3% of the patients. The distribution of FCD types slightly differed from that reported in a recent study.9 In our study, the proportion of FCD type II was lower, likely due to the varied standards used in the patient selection and the different age groups.
According to our findings, the different histological types are associated with different postoperative seizure outcomes. FCD type II has a more favorable outcome than FCD type I, consistent with previous studies.4,6,18 FCD type I likely represents a more diffuse structural abnormality, and the extent of the epileptogenic zone is difficult to delineate, subsequently leading to incomplete resection. The localization of dysplastic lesions using MRI and EEG was also better in the patients with FCD type II, which may contribute to the better outcomes.
The outcomes of the patients with FCD type III did not significantly differ from those of the patients with FCD type I. Our results are consistent with a previous study which showed no significant difference in the outcomes of epilepsy patients with FCD IIIa and isolated FCD I in the temporal lobe.16 In contrast, another study indicated that patients with refractory epilepsy secondary to a solitary tumor or with a tumor-associated FCD shared similar postoperative seizure outcomes but differed from patients with solitary FCD.15 These results are poorly comparable due to the varied subtypes of FCD III. Due to the limited number of patients with FCD type III in our series, comparisons among the different subtypes of FCD III could not be performed. Further studies focusing on the subtypes of FCD type III are needed to clarify the epileptogenic contributions of FCD I changes and other related lesions.
Early versus late surgery failure also showed variations in different FCD types. Seizure recurrence mostly occurred during the first 6–12 months in all FCD types. However, no additional failure occurred after one year among the patients with FCD type III or after two years among the patients with FCD type II, but the patients with FCD type I showed a gradual loss of their seizure freedom after one year until the final follow-up. Thus, late failure in the patients with FCD types II and III occurred less frequently than in patients with FCD type I, consistent with previous studies reporting that FCD type I is a risk factor of late failure in medically intractable epilepsy patients.18,19 Early postoperative seizure relapse has been attributed to inadequate resection of the epileptogenic zone, and late recurrence reflects the epileptogenesis of “pro-epileptic” lesions. Thus, our findings suggest that patients with FCD type I are more likely to develop neo-epileptogenesis after the complete resection of the initial epileptogenic FCD. However, why FCD type III including the pathological change of FCD I and additional related pathology has a lower rate of late failure than isolated FCD I is unclear. A possible explanation might be that the development of FCD I is attributed to other underlying pathological lesions, and the complete removal of this original lesion can prevent the re-emergence of FCD.20
The “running-down phenomenon” has been mostly described in temporal lobe epilepsies and is defined as a spontaneous vanishing of seizures in patients with initial postoperative seizure.21 This phenomenon was observed in three patients in our cohort as follows: two patients with FCD type III and one patient with FCD type I. Our results confirm the observations reported in earlier studies.3,8 In addition, we demonstrate that patients with early relapse also have an opportunity to become seizure-free without medical modification.
Recently, intraoperative MRI has been reported to be helpful in improving postoperative seizure freedom and reducing neurological deficits due to surgical damage.22 Intracranial electrode implantation was another significant predictive variable of unfavorable outcomes in our series. Invasive EEG usually reflects difficulties in precise epileptogenic zone localization merely based on scalp EEG and neuroimaging. The relationship between the use of an intracranial electrode and surgical failure implies that invasive EEG may not provide adequate information to fill the surgical outcome gap in these patients.
The present study has several limitations. First, this study was a retrospective study, and recall bias cannot be completely avoided. Second, because of the limited sample size, we could not perform subgroup analyses for each FCD subtype. Third, the duration of the follow-up was not sufficiently long to describe dynamic changes in the postoperative outcome. Fourth, we did not perform genetic analysis of the patients.
In conclusion, we report postsurgical seizure outcomes and risk factors of seizure relapse in child and adolescent patients with FCD undergoing comprehensive presurgical evaluation. Seizure freedom was obtained in more than half of the FCD patients. After dividing the histological findings into the different types according to the ILAE classification, the patients with FCD type II exhibited a better outcome than the patients with FCD type I, and no significant difference was observed between FCD type I and type III. The other determinants of a favorable postsurgical outcome in our study included complete resection and the non-use of intracranial electrode.
Acknowledgments
This work was supported by the National Key Research and Development Program of China (No. 2017YFC0907702).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Fauser S, Huppertz H-J, Bast T, et al. Clinical characteristics in focal cortical dysplasia: a retrospective evaluation in a series of 120 patients. Brain. 2006;129:1907–1916. doi:10.1093/brain/awl133
2. Blumcke I, Thom M, Aronica E, et al. The clinicopathologic spectrum of focal cortical dysplasias: a consensus classification proposed by an ad hoc task force of the ILAE diagnostic methods commission. Epilepsia. 2011;52:158–174. doi:10.1111/j.1528-1167.2010.02777.x
3. Phi JH, Cho BK, Wang KC, et al. Longitudinal analyses of the surgical outcomes of pediatric epilepsy patients with focal cortical dysplasia. J Neurosurg Pediatr. 2010;6:49–56. doi:10.3171/2010.3.PEDS09497
4. Kim YH, Kang HC, Kim DS, et al. Neuroimaging in identifying focal cortical dysplasia and prognostic factors in pediatric and adolescent epilepsy surgery. Epilepsia. 2011;52:722–727. doi:10.1111/j.1528-1167.2010.02950.x
5. Chen HH, Chen C, Hung SC, et al. Cognitive and epilepsy outcomes after epilepsy surgery caused by focal cortical dysplasia in children: early intervention maybe better. Childs Nerv Syst. 2014;30:1885–1895. doi:10.1007/s00381-014-2463-y
6. Choi SA, Kim SY, Kim H, et al. Surgical outcome and predictive factors of epilepsy surgery in pediatric isolated focal cortical dysplasia. Epilepsy Res. 2017;139:54–59. doi:10.1016/j.eplepsyres.2017.11.012
7. Rowland NC, Englot DJ, Cage TA, Sughrue ME, Barbaro NM, Chang EF. A meta-analysis of predictors of seizure freedom in the surgical management of focal cortical dysplasia. J Neurosurg. 2012;116:1035–1041. doi:10.3171/2012.1.JNS111105
8. Fauser S, Essang C, Altenmuller DM, et al. Long-term seizure outcome in 211 patients with focal cortical dysplasia. Epilepsia. 2015;56:66–76. doi:10.1111/epi.12876
9. Jin B, Wang J, Zhou J, Wang S, Guan Y, Chen S. A longitudinal study of surgical outcome of pharmacoresistant epilepsy caused by focal cortical dysplasia. J Neurol. 2016;263:2403–2410. doi:10.1007/s00415-016-8274-1
10. Kwan P, Arzimanoglou A, Berg AT, et al. Definition of drug resistant epilepsy: consensus proposal by the ad hoc task force of the ILAE commission on therapeutic strategies. Epilepsia. 2010;51:1069–1077. doi:10.1111/j.1528-1167.2009.02397.x
11. Wieser HG, Blume WT, Fish D, et al. ILAE Commission Report. Proposal for a new classification of outcome with respect to epileptic seizures following epilepsy surgery. Epilepsia. 2001;42:282–286.
12. Gaitanis JN, Donahue J. Focal cortical dysplasia. Pediatr Neurol. 2013;49:79–87. doi:10.1016/j.pediatrneurol.2012.12.024
13. Tripathi M, Singh MS, Padma MV, et al. Surgical outcome of cortical dysplasias presenting with chronic intractable epilepsy: a 10-year experience. Neurol India. 2008;56:138–143.
14. Widdess-Walsh P, Kellinghaus C, Jeha L, et al. Electro-clinical and imaging characteristics of focal cortical dysplasia: correlation with pathological subtypes. Epilepsy Res. 2005;67:25–33. doi:10.1016/j.eplepsyres.2005.07.013
15. Cossu M, Fuschillo D, Bramerio M, et al. Epilepsy surgery of focal cortical dysplasia-associated tumors. Epilepsia. 2013;54 Suppl 9:115–122. doi:10.1111/epi.12455
16. Fauser S, Essang C, Altenmuller DM, et al. Is there evidence for clinical differences related to the new classification of temporal lobe cortical dysplasia? Epilepsia. 2013;54:909–917. doi:10.1111/epi.12147
17. Gales JM, Jehi L, Nowacki A, Prayson RA. The role of histopathologic subtype in the setting of hippocampal sclerosis-associated mesial temporal lobe epilepsy. Hum Pathol. 2017;63:79–88. doi:10.1016/j.humpath.2017.02.013
18. Krsek P, Pieper T, Karlmeier A, et al. Different presurgical characteristics and seizure outcomes in children with focal cortical dysplasia type I or II. Epilepsia. 2009;50:125–137. doi:10.1111/j.1528-1167.2008.01682.x
19. McIntosh AM, Averill CA, Kalnins RM, et al. Long-term seizure outcome and risk factors for recurrence after extratemporal epilepsy surgery. Epilepsia. 2012;53:970–978. doi:10.1111/j.1528-1167.2012.03430.x
20. Messripour M, Mesripour A. Alteration of ADP-ribosylation in aging rat brain astrocytes. Biocell. 2019;43:37–40. doi:10.32604/biocell.2019.05865
21. Salanova V, Rasmussen T, Andermann F. The running down phenomenon in temporal lobe epilepsy. Adv Neurol. 1999;81:165–169.
22. Sacino MF, Ho CY, Murnick J, Keating RF, Gaillard WD, Oluigbo CO. The role of intraoperative MRI in resective epilepsy surgery for peri-eloquent cortex cortical dysplasias and heterotopias in pediatric patients. Neurosurg Focus. 2016;40:E16. doi:10.3171/2016.1.FOCUS15538
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.