Back to Journals » Clinical Epidemiology » Volume 12
Herpes Zoster, Hepatitis C, and Tuberculosis Risk with Apremilast Compared to Biologics, DMARDs and Corticosteroids to Treat Psoriasis and Psoriatic Arthritis
Authors Hagberg KW , Persson R , Vasilakis-Scaramozza C , Niemcryk S, Peng M, Paris M, Lindholm A, Jick S
Received 22 November 2019
Accepted for publication 24 January 2020
Published 12 February 2020 Volume 2020:12 Pages 153—161
DOI https://doi.org/10.2147/CLEP.S239511
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Vera Ehrenstein
Katrina Wilcox Hagberg,1 Rebecca Persson,1 Catherine Vasilakis-Scaramozza,1 Steve Niemcryk,2 Michael Peng,2 Maria Paris,2 Anders Lindholm,2 Susan Jick1,3
1Boston Collaborative Drug Surveillance Program, Lexington, MA 02421, USA; 2Celgene Corporation, Summit, NJ 07901, USA; 3Boston University School of Public Health, Lexington, MA 02421, USA
Correspondence: Susan Jick
Boston Collaborative Drug Surveillance Program, 11 Muzzey Street, Lexington, MA 02421, USA
Tel +1-781-862-6660
Fax +1-781-862-1680
Email [email protected]
Purpose: Psoriasis and psoriatic arthritis (PsA) are associated with an increased infection risk. In this cohort study of patients with treated psoriasis or PsA, we used MarketScan (2014– 2018) to estimate rates of herpes zoster, hepatitis C (HepC) and tuberculosis (TB) with apremilast compared to other systemic treatments.
Materials and Methods: Patients were exposed from first apremilast [APR], DMARD, TNF-inhibitor [TNF], IL-inhibitor [IL], or corticosteroids [CS] prescription after March 21, 2014. Study exposures were APR, DMARDs only, TNF-only, IL-only, CS-only, DMARDs+CS, TNF+DMARDs and/or CS, IL+DMARDs and/or CS. Cases had treated herpes zoster, HepC, or TB event. We calculated incidence rates (IRs) [95% confidence intervals] per 1000 patient-years.
Results: The study population included 131,604 patients. For herpes zoster (N=2271), IRs were highest for users of DMARDs+CS (12.5 [9.8– 15.7]), CS-only (12.5 [10.4– 14.1]), and TNF+DMARDs and/or CS (11.9 [10.6– 13.4]), compared with DMARDs only (9.9 [8.7– 11.2]). IRs were lowest for users of IL-only (6.7 [5.8– 7.8]) and APR (7.0 [5.8– 8.4]). IRs of HepC (N=150) and TB (N=81) were low and between-treatment differences were not significant.
Conclusion: Rates of herpes zoster varied by treatment: highest among those who received polytherapy, lowest in users of apremilast only. IRs for HepC and TB were low for all exposures.
Keywords: apremilast, psoriasis, psoriatic arthritis, herpes zoster, hepatitis C, tuberculosis
Introduction
Apremilast is a drug marketed in the United States as of March 2014 for the treatment of psoriasis and psoriatic arthritis (PsA). It is an oral medication that inhibits phosphodiesterase-4, a protein found in immune cells associated with inflammation. In randomized clinical trials of apremilast for the treatment of psoriasis or PsA, serious infections were rare and comparable across the various exposure groups.1–6 No cases of active tuberculosis, herpes zoster, or reactivation of hepatitis C were reported in trials for apremilast, although active tuberculosis, history of tuberculosis, and/or positive hepatitis C antibodies at screening were the reason for exclusion from some of the studies.1–6 While the safety profile for apremilast in the clinical trials is reassuring, drugs used to treat autoimmune disorders have been shown to increase the risk of serious infection, though there is no consensus on the magnitude of the risk.7–26 Hepatitis C and tuberculosis are particularly rare and unlikely to occur in small selected clinical trial populations compared with larger epidemiological studies. Therefore, we conducted a post-marketing safety study to estimate rates of treated herpes zoster, hepatitis C, and tuberculosis in patients with psoriasis or PsA treated with apremilast compared with users of other systemic psoriasis and PsA treatments.
Methods
Study Population
We conducted a cohort study within the IBM MarketScan Commercial Claims and Encounters Database (CCE) with Medicare supplement, a large United States (US)-based claims database containing data on over 50 million patients from over 150 large employers distributed throughout the US that covers employees and their dependent family members. It has been reported that the age, sex, and geographic distribution of patients who participated in an employer-sponsored private insurance survey are similar to the US population.27 The database contains basic demographics and information on pharmaceuticals (using National Drug Codes), diagnoses (using International Classification of Diseases (ICD)-9 and 10-CM), and procedures (using Current Procedural Terminology, Fourth Edition and the Healthcare Common Procedure Coding System).
The study population included all patients with a diagnosis of psoriasis and/or PsA who received ≥1 prescription for apremilast, a disease-modifying antirheumatic drug (DMARD), a tumor necrosis factor inhibitor (TNF-i) biologic, an interleukin-17 or −12/23 inhibitor (IL-i) biologic, or systemic corticosteroids between March 21, 2014 (when apremilast was marketed) and October 31, 2018 (end of study) (see Supplemental Table 1for a list of all study drugs). Because systemic corticosteroids are not indicated for treatment of psoriasis, patients with psoriasis only (ie, no PsA at any time) whose only study drug was a corticosteroid were not included in the study population. For patients with PsA, we required a PsA diagnosis on the same day as a corticosteroid injection or within 15 days before filling a prescription for oral corticosteroids because corticosteroids have multiple indications for use. Patients diagnosed with rheumatoid arthritis before psoriasis or PsA were excluded from the study population. Patients entered the study at the time of their first study drug prescription after March 21, 2014 (cohort entry date) and were followed through the censor date, defined as record end, date the patient became a case, or October 31, 2018 (end of the study period), whichever came first.
Exposure Definition
The study exposures of interest were apremilast, TNF-i biologics, IL-i biologics, conventional DMARDs, and systemic corticosteroids. Current use of each study drug was calculated using the length of each prescription claim plus 30 days. For those who received the drug by injection or infusion, current use was defined as 60 days (or 120 days for ustekinumab). If a new prescription for the same drug was received before the end of the previous prescription duration, the number of days was truncated and days were accumulated from the date of the new prescription. A patient was considered exposed to multiple study drugs if the duration of different study drug prescriptions overlapped, except for TNF-i and IL-i biologics which were not allowed to overlap. We assigned exposure for each day of follow-up to one of the following mutually exclusive categories: apremilast only, apremilast with other study drugs, TNF-i biologics only, IL-i biologics only, DMARDs only, corticosteroids only, DMARDs + corticosteroids, TNF-i biologics with DMARDs and/or corticosteroids, IL-i biologics with DMARDs and/or corticosteroids, or unexposed (defined as any days after “current use” and before a new study drug prescription).
Case Definition
Cases were treated herpes zoster, treated hepatitis C, or treated tuberculosis, and included both newly diagnosed and reactivated infections. A patient was required to have both a diagnosis and treatment for the study infection within 15 days of each other to be a case, and the infection diagnosis and treatment had to first occur at least 7 days after cohort entry. We further required at least 60 days of treatment for cases of hepatitis C and tuberculosis. See Supplemental Table 2 for treatments for herpes zoster, hepatitis C, and tuberculosis included in the study. Patients with active, treated infections at cohort entry (prevalent cases) were not considered as cases; however, they did remain in the study population and were followed forward from their initial diagnosis date to determine if they later qualified to be a reactivated case. To be considered a case of reactivated infection, a 6-month treatment-free period after the initial infection was required before the next episode of treated infection. Cases were censored at the first qualifying diagnosis code or treatment for that outcome, whichever came first. The electronic records of all hepatitis C cases, all tuberculosis cases, and a sample of herpes zoster cases were reviewed manually to confirm case status and index dates.
Covariates
We described the study population according to their exposure at cohort entry with respect to the presence of chronic infection risk factors (Table 1). We also described the presence of acute infection risk factors within the 60 days before the index date, including other immunosuppressant drugs (azathioprine, chloroquine, cyclophosphamide, hydroxychloroquine, leflunomide, minocycline, and sulfasalazine), fractures, major or minor surgical procedures, ventilator use, urinary catheter use, pressure ulcers, and diagnosis or treatment for malignant neoplasm. Duration of psoriasis or PsA was calculated using the date of the first diagnosis code in the patient record, categorized as <1 year, 1–2.9, 3–4.9, ≥5 years, and unknown (where the first psoriasis or PsA diagnosis was recorded less than 6 months after enrollment in MarketScan).
 |
Table 1 Characteristics of Study Population at Cohort Entry Date by Exposure |
Statistical Analyses
We calculated incidence rates (IRs) with 95% confidence intervals (CIs) of treated herpes zoster, treated hepatitis C, and treated tuberculosis separately for each exposure. For herpes zoster, we calculated incidence rate ratios (IRRs) with 95% CIs and adjusted IRRs controlling for age, sex, calendar year and presence of rheumatoid arthritis for each exposure category compared to DMARDs only using Poisson regression, as well as stratified by indication for use (ie, PsA or psoriasis). There were too few exposed cases of hepatitis C and tuberculosis to calculate crude or adjusted IRRs. Studies using MarketScan are exempt from IRB review because the data is compliant with the Health Insurance Portability and Accountability Act (HIPAA) to protect patient privacy.
Results
The study population encompassed 131,604 patients with psoriasis or PsA (Table 1). At cohort entry, 10,074 (7.7%) patients were exposed to apremilast (alone or in combination with other study drugs), while 47,361 (36.0%) were exposed to TNF-i biologics only, 24,678 (18.8%) were exposed to DMARDs only, 12,438 (9.5%) were exposed to IL-i biologics only, 20,692 (15.7%) were exposed to CS only, and 16,361 (12.4%) were exposed to multiple study drugs (DMARDs+CS, or TNF-i with DMARDs and/or CS, or IL-i with DMARDs and/or CS). Half of the population (53.3%) were between 40 and 59 years old at cohort entry, and half (50.5%) were female. At cohort entry, proportionally more apremilast users had an indication of psoriasis rather than PsA compared to users of the other study drugs.
We identified 2271 new or reactivated cases of treated herpes zoster (Table 2), yielding an IR in the entire study population and regardless of treatment of 8.9 (95% CI 8.5–9.3) per 1000 person-years (PY) (Table 3). IRs of treated herpes zoster were highest for current users of DMARDs+CS (12.5, 95% CI 9.8–15.7), CS only (12.5, 95% CI 10.4–14.1), TNF-i biologics with DMARDs and/or CS (11.9, 95% CI 10.6–13.4), and IL-i biologics with DMARDs and/or CS (10.3, 95% CI 7.6–13.7) (Table 3). IRs of treated herpes zoster were lowest for current users of IL-i biologics only (6.7, 95% CI 5.8–7.8) and apremilast alone or in combination (7.0, 95% CI 5.8–8.4). The crude and adjusted IRRs, with DMARDs only as the referent, were around 1.2 for current users of TNF-i biologics with DMARDs and/or CS, CS only, and DMARDs+CS, while the IRRs were close to or below 1.0 for all other exposure categories (Table 3). Compared to DMARDs only, the adjusted IRR for current use of apremilast only was 0.70 (95% CI 0.54–0.90) and 0.89 (95% CI 0.63–1.25) for current use of apremilast in combination with other study exposures. The results were similar when stratified by indication for use (ie, PsA or psoriasis) (Table 3).
 |
Table 2 Characteristics of Cases at the Index Date |
 |
Table 3 Incidence Rates (IR) and Incidence Rate Ratios (IRR) for Treated Herpes Zoster by Exposure in Patients with Psoriasis or PsA |
We identified 150 new or reactivated cases of treated hepatitis C (Table 2), resulting in an IR for the entire study population, regardless of treatment, of 0.6 (95% CI 0.5–0.7) per 1000 PY (Table 4). IRs of treated hepatitis C were low for all exposure categories and there were only 12 cases with current exposure to apremilast. Finally, we identified 81 new or reactivated cases of treated tuberculosis (Table 2), yielding an IR in the entire study population, regardless of treatment, of 0.3 (95% CI 0.2–0.4) per 1000 person-years (PY) (Table 5). IRs of tuberculosis were low and there were only four cases were exposed to apremilast. The IRs were similar and not statistically different for all exposures. The crude IRRs were all below 1.0 (Table 5). There were too few exposed cases of hepatitis C and tuberculosis, especially in the reference group, to calculate stable IRRs and all estimates were based on small numbers.
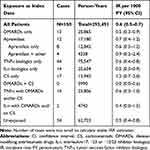 |
Table 4 Incidence Rates (IR) for Treated Hepatitis C by Exposure in Patients with Psoriasis or PsA |
 |
Table 5 Incidence Rates (IR) for Treated Tuberculosis by Exposure in Patients with Psoriasis or PsA |
Discussion
In this large study of patients with psoriasis and PsA, there was no suggestion that patients treated with apremilast were at higher risk of new or recurrent infections of treated herpes zoster, hepatitis C, or tuberculosis compared to patients with psoriasis or PsA treated with DMARDs only. The IRs and IRRs of herpes zoster among apremilast users were slightly lower compared with those of users of other treatments for psoriasis or PsA evaluated in this study, whereas the IRs of hepatitis C and tuberculosis were similar to those of patients treated with DMARDs and/or biologics. The results of this study provide reassurance that apremilast is not likely to be associated with a high risk of herpes zoster, hepatitis C, or tuberculosis in patients with psoriasis or PsA.
The incidence of herpes zoster for users of DMARDs and biologics in our study was similar to those reported in several other studies and systematic reviews.8–17 This provides confidence in the quality of the data and the ability to detect associations between these exposures and outcomes. This further provides reassurance that the incidence of herpes zoster among patients exposed to apremilast, which was slightly lower than users of DMARDS only, is not elevated compared with users of other psoriasis and PsA treatments.
The available literature describing hepatitis C among patients treated for rheumatoid arthritis, psoriasis or PsA is limited and consists primarily of case reports of hepatitis C reactivation or liver function changes among these patients.7,19–22,28 There was one retrospective cohort study that found that biologic treatment in patients with psoriasis increased the risk of reactivation of hepatitis C infection; however, there was no data on apremilast users in particular.23 Overall, we identified 150 cases of treated hepatitis C in our entire cohort, only 12 of which were exposed to apremilast (alone or in combination with other study drugs), indicating that hepatitis C is rare in patients with psoriasis or PsA and does not occur more often in apremilast users compared with users of other treatments.
Few studies have been conducted since the implementation of tuberculosis screening to assess the incidence of tuberculosis among patients with rheumatoid arthritis, psoriasis or PsA.24–26,29 We found a few cases of patients with treated tuberculosis in our cohort, four of whom were exposed to apremilast. The IRs for treated tuberculosis among users of DMARDs and biologics in our study fall within the range previously reported.23–26 Our results provide reassurance that apremilast is not likely to be associated with a high risk of reactivation of latent tuberculosis in patients with psoriasis or PsA.
Our study evaluated a large cohort of more than 10,000 apremilast exposed patients with psoriasis and PsA. We required all cases of herpes zoster, hepatitis C, and tuberculosis to have received treatment for the infection to minimize case misclassification. We were also able to classify all study exposures according to whether or not they were prescribed in combination with other treatments. There were small numbers of cases in some exposure categories, particularly for hepatitis C and tuberculosis, resulting in wide confidence intervals. While due to small numbers we were unable to include all infection risk factors in our adjusted models, there were enough cases of herpes zoster to evaluate the impact of important covariates (sex, age, calendar year, and rheumatoid arthritis) on the association of treatment and infection. Due to the nature of ICD coding in claims data, differentiating between screening, infection, and routine follow-up visits for patients with a history of hepatitis C or tuberculosis is challenging. To minimize case misclassification, we reviewed the records of every case and required each to have received at least 60 days of treatment; however, it remains possible that some of these infection cases may have received chemoprophylaxis and the event was not a true latent infection reactivation. Because the total number of hepatitis C (N=150) and tuberculosis (N=83) cases identified in this study were small, we only presented rates of these infections and did not calculate IRRs between exposure categories; thus, the presented results are not biased by differential case misclassification. Finally, at cohort entry proportionally more users of apremilast had an indication of psoriasis compared with the other study exposures, and due to small numbers, we were unable to adjust our estimates by indication for use. Thus, disease severity should be taken into consideration and the results of this study should be interpreted with caution.
Conclusion
These data suggest that rates of hepatitis C and tuberculosis were low among commercially insured US patients for all types of psoriasis and PsA treatment. Rates of herpes zoster varied by treatment and were lowest in users of apremilast only. The results of this study provide reassurance that apremilast is not likely to be associated with a high risk of reactivation of herpes zoster, hepatitis C, or latent tuberculosis in patients with psoriasis or PsA. However, the outcomes evaluated in this study are relatively rare events and some exposure groups had limited numbers of cases; thus, these results should be interpreted with caution.
Acknowledgments
This study was supported by Celgene Corporation (Summit, NJ 07901). The abstract entitled “Rates of Herpes Zoster, hepatitis C, and tuberculosis among patients with psoriasis treated with apremilast, biologics, DMARDs, and corticosteroids: a cohort study in the US MarketScan Database” was submitted for presentation at American Academy of Dermatology 2020 Annual Meeting. While related to the paper that we have submitted for publication, this abstract presents preliminary results on a subgroup of patients with psoriasis.
Disclosure
Ms Katrina Wilcox Hagberg reports grants from Celgene Corp, during the conduct of the study. Ms Rebecca Persson reports grants from Celgene Corporation, during the conduct of the study; grants from Celgene Corporation and AbbVie Inc., outside the submitted work. Dr Catherine Vasilakis-Scaramozza reports grants from Celgene Corporation, during the conduct of the study. Dr Steve Niemcryk is an employee of Celgene Corp, and owns stock in Celgene Corp. Dr Michael Peng, Dr Maria Paris, and Dr Anders Lindholm are employees of Celgene Corp. Dr Susan Jick reports grants from Celgene Corp, during the conduct of the study. The authors report no other conflicts of interest in this work.
References
1. Kavanaugh A, Mease PJ, Gomez-Reino JJ, et al. Treatment of psoriatic arthritis in a Phase 3 randomised, placebo-controlled trial with apremilast, an oral phosphodiesterase 4 inhibitor. Ann Rheum Dis. 2014;73(6):1020–1026. doi:10.1136/annrheumdis-2013-205056
2. Papp K, Cather JC, Rosoph L, et al. Efficacy of apremilast in the treatment of moderate to severe psoriasis: a randomised controlled trial. Lancet. 2012;380(9843):738–746. doi:10.1016/S0140-6736(12)60642-4
3. Papp KA, Kaufmann R, Thaçi D, Hu C, Sutherland D, Rohane P. Efficacy and safety of apremilast in subjects with moderate to severe plaque psoriasis: results from a Phase II, multicenter, randomized, double-blind, placebo-controlled, parallel-group, dose-comparison study. J Eur Acad Dermatol Venereol. 2013;27(3):e376–e383. doi:10.1111/j.1468-3083.2012.04716.x
4. Paul C, Cather J, Gooderham M, et al. Efficacy and safety of apremilast, an oral phosphodiesterase 4 inhibitor, in patients with moderate-to-severe plaque psoriasis over 52 weeks: a Phase III, randomized controlled trial (ESTEEM 2). Br J Dermatol. 2015;173(6):1387–1399. doi:10.1111/bjd.14164
5. Schett G, Wollenhaupt J, Papp K, et al. Oral apremilast in the treatment of active psoriatic arthritis: results of a multicenter, randomized, double-blind, placebo-controlled study. Arthritis Rheum. 2012;64(10):3156–3167. doi:10.1002/art.34627
6. Kavanaugh A, Gladman DD, Edwards CJ, et al. Long-term experience with apremilast in patients with psoriatic arthritis: 5-year results from PALACE 1-3 polled analysis. Arthritis Res Ther. 2019;21:118. doi:10.1186/s13075-019-1901-3
7. De Keyser F. Choice of biologic therapy for patients with rheumatoid arthritis: the infection perspective. Curr Rheumatol Rev. 2011;7(1):77–87. doi:10.2174/157339711794474620
8. Dreiher J, Kresch FS, Comaneshter D, Cohen AD. Risk of Herpes zoster in patients with psoriasis treated with biologic drugs. J Eur Acad Dermatol Venereol. 2012;26(9):1127–1132. doi:10.1111/j.1468-3083.2011.04230.x
9. Zisman D, Bitterman H, Shalom G, et al. Psoriatic arthritis treatment and the risk of herpes zoster. Ann Rheum Dis. 2016;75(1):131–135. doi:10.1136/annrheumdis-2013-205148
10. Megna M, Napolitano M, Ayala F, Balato N. The risk of herpes zoster in patients with psoriasis: a retrospective records-based observational study. Indian J Dermatol Venereol Leprol. 2016;82(6):744. doi:10.4103/0378-6323.183630
11. Smitten AL, Choi HK, Hochberg MC, et al. The risk of herpes zoster in patients with rheumatoid arthritis in the United States and the United Kingdom. Arthritis Rheum. 2007;57(8):1431–1438. doi:10.1002/(ISSN)1529-0131
12. Strangfeld A, Listing J, Herzer P, et al. Risk of herpes zoster in patients with rheumatoid arthritis treated with anti-TNF-alpha agents. JAMA. 2009;301(7):737–744. doi:10.1001/jama.2009.146
13. Galloway JB, Mercer LK, Moseley A, et al. Risk of skin and soft tissue infections (including shingles) in patients exposed to anti-tumour necrosis factor therapy: results from the British Society for Rheumatology Biologics Register. Ann Rheum Dis. 2013;72(2):229–234. doi:10.1136/annrheumdis-2011-201108
14. Winthrop KL, Baddley JW, Chen L, et al. Association between the initiation of anti-tumor necrosis factor therapy and the risk of herpes zoster. JAMA. 2013;309(9):887–895. doi:10.1001/jama.2013.1099
15. Adelzadeh L, Jourabchi N, Wu JJ, Adelzadeh L, Jourabchi N, Wu JJ. The risk of herpes zoster during biological therapy for psoriasis and other inflammatory conditions. J Eur Acad Dermatol Venereol. 2014;28(7):846–852. doi:10.1111/jdv.12307
16. Marra F, Lo E, Kalashnikov V, Richardson K. Risk of herpes zoster in individuals on biologics, disease-modifying antirheumatic drugs, and/or corticosteroids for autoimmune disease: a systematic review and meta-analysis. Open Forum Infect Dis. 2016;3(4):205. doi:10.1093/ofid/ofw205
17. Shalom G, Zisman D, Bitterman H, et al. Systemic therapy for psoriasis and the risk of herpes zoster: a 500,000 person-year study. JAMA Dermatol. 2015;151(5):533–538. doi:10.1001/jamadermatol.2014.4956
18. Brunasso AM, Puntoni M, Gulia A, Massone C. Safety of anti-tumour necrosis factor agents in patients with chronic hepatitis C infection: a systematic review. Rheumatology (Oxford). 2011;50(9):1700–1711. doi:10.1093/rheumatology/ker190
19. Frider B, Bruno A, Ponte M, Amante M. Drug-induced liver injury caused by adalimumab: a case report and review of the bibliography. Case Rep Hepatol. 2013;2013:406901. doi:10.1155/2013/406901
20. Lin KM, Lin JC, Tseng WY, Cheng TT. Rituximab-induced hepatitis C virus reactivation in rheumatoid arthritis. J Microbiol Immunol Infect. 2013;46(1):65–67. doi:10.1016/j.jmii.2011.12.020
21. Pompili M, Biolato M, Miele L, Grieco A. Tumor necrosis factor-α inhibitors and chronic hepatitis C: a comprehensive literature review. World J Gastroenterol. 2013;19(44):7867–7873. doi:10.3748/wjg.v19.i44.7867
22. Salvi M, Macaluso L, Luci C, et al. Safety and efficacy of anti-tumor necrosis factors α in patients with psoriasis and chronic hepatitis C. World J Clin Cases. 2016;4(2):49–55. doi:10.12998/wjcc.v4.i2.49
23. Snast I, Atzmony L, Braun M, Hodak E, Pavlovsky L. Risk for hepatitis B and C virus reactivation in patients with psoriasis on biologic therapies: a retrospective cohort study and systematic review of the literature. J Am Acad Dermatol. 2017;77(1):88–97.e5. doi:10.1016/j.jaad.2017.01.037
24. Brassard P, Lowe AM, Bernatsky S, Kezouh A, Suissa S. Rheumatoid arthritis, its treatments, and the risk of tuberculosis in Quebec, Canada. Arthritis Rheum. 2009;61(3):300–304. doi:10.1002/art.24476
25. Gómez-Reino JJ, Carmona L, Angel DM, Biobadaser Group. Risk of tuberculosis in patients treated with tumor necrosis factor antagonists due to incomplete prevention of reactivation of latent infection. Arthritis Rheum. 2007;57(5):756–761. doi:10.1002/art.22768
26. Tubach F, Salmon D, Ravaud P, et al. Research axed on tolerance of biotherapies group. Risk of tuberculosis is higher with anti-tumor necrosis factor monoclonal antibody therapy than with soluble tumor necrosis factor receptor therapy: the three-year prospective French Research Axed on Tolerance of Biotherapies registry. Arthritis Rheum. 2009;60(7):1884–1894. doi:10.1002/art.24632
27. Pickens G, Moldwin E, Marder W. Healthcare spending index for employer-sponsored insurance: methodology and baseline results. Available from: http://truvenhealth.com/Portals/0/Assets/HealthInsights/TRU_15667_0415_HSI_ESI_WP.pdf.
28. Iannone F, La Montagna G, Bagnato G, Gremese E, Giardina A, Lapadula G. Safety of etanercept and methotrexate in patients with rheumatoid arthritis and hepatitis C virus infection: a multicenter randomized clinical trial. J Rheumatol. 2014;41:2. doi:10.3899/jrheum.130658
29. Stoll ML, Grubbs JA, Beukelman T, et al. Risk of tuberculosis among Alabama children and adolescents treated with tumor necrosis factor inhibitors: a retrospective study. Pediatr Rheumatol. 2017;15(1):1–6.
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
