Back to Journals » Therapeutics and Clinical Risk Management » Volume 15
Hepatorenal Syndrome Type 1: Current Challenges And Future Prospects
Authors Facciorusso A
Received 8 August 2019
Accepted for publication 5 November 2019
Published 27 November 2019 Volume 2019:15 Pages 1383—1391
DOI https://doi.org/10.2147/TCRM.S205328
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Garry Walsh
Antonio Facciorusso
Gastroenterology Unit, University of Foggia, Foggia, Italy
Correspondence: Antonio Facciorusso
Gastroenterology Unit, Department of Medical Sciences, University of Foggia, AOU Ospedali Riuniti, Viale Pinto, 1, Foggia 71100, Italy
Tel +39 0881732110
Fax +39 0881733545
Email [email protected]
Abstract: Renal dysfunction represents a dreadful complication of advanced liver cirrhosis. In addition to the traditional types of acute kidney injury (AKI) that can occur in the general population, cirrhotics might experience a different kind of renal dysfunction, called hepatorenal syndrome (HRS). The exact definition of HRS is a functional renal dysfunction caused by overactivity of the endogenous vasoactive systems (in particular intrarenal circulation) which lead to reduced renal perfusion. Type I HRS (HRS-1) is characterized by an abrupt deterioration in renal function (in less than 2 weeks), defined by a doubling of baseline sCr to >2.5 mg/dL or a 50% reduction in the initial 24 hrs creatinine clearance to
Keywords: kidney, liver cirrhosis, terlipressin, mortality
Introduction
Renal dysfunction represents a dreadful complication of advanced liver cirrhosis.1,2
The traditional definition of renal dysfunction in patients with liver disease is based on a serum creatinine (sCr) concentration of ≥1.5 mg/dl, and in this context, acute kidney injury (AKI) is diagnosed in the presence of the abrupt doubling of the baseline value of sCr beyond the threshold of 1.5 mg/dL.3,4
Classification Of Acute Kidney Injury In Cirrhotic Patients
In addition to the traditional types of AKI that can occur in the general population, namely, prerenal, intrarenal, and post-renal, cirrhotics might experience a different kind of renal dysfunction, called hepatorenal syndrome (HRS).5 Specifically, acute tubular necrosis occurs in 41.7% of cirrhotics, prerenal AKI in 38% of patients, while postrenal AKI is rare in patients with liver cirrhosis (0.3%).6 Therefore, while the recently published International Club of Ascites (ICA) guidelines have suggested that all cases of acute renal dysfunction in patients with cirrhosis should be classified under the broad heading of acute kidney injury (AKI), HRS is registered only in 20% of subjects with liver cirrhosis.7
Definition Of Hepatorenal Syndrome And Sub-Classification
The exact definition of HRS is a functional renal dysfunction caused by overactivity of the endogenous vasoactive systems (in particular intrarenal circulation) which leads to reduced renal perfusion.3,4
HRS has been classified traditionally into two different clinical types; type I HRS (HRS-1) is characterized by an abrupt deterioration in renal function (in less than 2 weeks), defined by a doubling of baseline sCr to >2.5 mg/dL or a 50% reduction in the initial 24 hrs creatinine clearance to <20 mL/min.3,4
Based on the aforementioned assumptions, in the presence of a cirrhotic patient with rapid renal function deterioration, several differential diagnoses should be ruled out before considering HRS, such as prerenal AKI due to hypovolemia (for example, after massive variceal bleeding), acute tubular necrosis due to sepsis, iatrogenic injury, diabetic nephropathy.8
Frequent precipitating events leading to HRS-1 are bacterial infections, gastrointestinal hemorrhage, or large-volume paracentesis without adequate albumin administration as well as massive diuretic use.7 On the other hand, type 2 HRS (HRS-2) is characterized by the slow occurrence of renal dysfunction and it is usually considered within the spectrum of refractory ascites.
Updated Definition And New Remarks
In 2015, the ICA revised the definitions and recommendations concerning HRS. In fact, creatinine levels are likely to remain low in cirrhotics even in the presence of advanced AKI mainly due to sarcopenia and this misleading diagnosis may negatively impact the earlier institution of therapy.5 Therefore, the use of a fixed threshold of sCr should be abandoned, while the dynamic changes of sCR proved to provide an accurate definition of AKI and HRS in these patients.
According to the new definition, AKI is defined by the increase in sCr ≥0.3 mg/dl within 48 hrs, or a percentage increase sCr ≥50% from baseline.5,9
In this context, the definition of HRS-1 is based on the following criteria: (a) diagnosis of cirrhosis and ascites, (b) diagnosis of AKI according to ICA-AKI criteria (see above), (c) no response after two consecutive days of diuretic withdrawal and plasma volume expansion with albumin 1 g per kg of body weight, (d) absence of shock and no current or recent use of nephrotoxic drugs, and (e) no macroscopic signs of structural kidney injury (defined as absence of proteinuria or microhaematuria, or normal findings on renal ultrasonography).5
The revised definition allows to adopt effective pharmacological therapy based on albumin and vasoconstrictors in an earlier stage thus not influenced anymore by the rigid sCr cut-off value of >2.5 mg/dl as in the previous definition of HRS-1. Although this aspect should lead theoretically to better therapeutic outcomes,10,11 however, trials testing vasoconstrictors in patients with HRS and lower values of sCr are lacking, thus calling for a particular note of caution in this setting.12
Pathophysiology
Activation of renin-angiotensin-aldosterone system (RAAS) plays a key role in the pathogenesis of hydroelectrolytic imbalances in cirrhotics. Activation of RAAS, as well as activation of sympathetic nervous system (SNS) and hyperincretion of anti-diuretic hormone (ADH), is triggered as a response to maintain arterial pressure within normal ranges in cirrhotic patients with ascites.13
Role Of Portal Hypertension
The pathophysiological mechanisms underlying the development of hepatorenal syndrome are described in Figure 1.
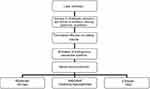 |
Figure 1 Pathogenesis of hepatorenal syndrome. Abbreviation: HRS, hepatorenal syndrome. |
Disruption of liver architecture occurring in cirrhotic patients increases the intrahepatic vascular resistance leading to worsening of portal pressure. This process releases several mediators including nitric oxide and endogenous cannabinoids causing the vasodilation of the splanchnic vascular bed.14 In advanced cirrhotic patients, the cardiac output can no longer compensate for the reduced systemic vascular resistance triggered by the splanchnic vasodilation, determining a decreased effective circulating volume.8,14,15 This, in turn, leads to activation of the SNS and RAAS as well as vasopressin to help maintain circulating volume, leading to renal vasoconstriction and hypoperfusion of these organs.16
As aforementioned, the consequence of solute-free water excretion impairment (due to ADH incretion and renal dysfunction) is the occurrence of hyponatremia, so-called dilutional hyponatremia as it develops due to the increased total body water and dilution of extracellular fluid volume.16,17
The development of renal vasoconstriction in cirrhotic patients with ascites is clinically relevant as it predisposes to the development of HRS.18
In fact, HRS occurrence is determined by a complex interplay due to the increased activity of vasoconstrictor factors (mainly plasma renin activity and norepinephrine) and reduced activity of renal vasodilator factors.19
Urinary And Blood Biomarkers
In recent years, several urinary AKI markers that played a role in trying to determine the etiology of AKI in patients with cirrhosis were reported. For example, urinary neutrophil gelatinase-associated lipocalin (NGAL) levels were found to be increased in patients with cirrhosis and acute tubular necrosis compared with patients who had prerenal AKI and HRS.20 Other urinary biomarkers tested are interleukin (IL)-18, kidney injury molecule-1, and liver-type fatty acid-binding protein.21
In addition to urinary biomarkers, also renal blood biomarkers have been evaluated to correctly estimate glomerular filtration rate (GFR), for example, cystatin C, b-trace protein, b-2 microglobulin, and dimethylarginines.22–26
However, in spite of the high number of studies testing new urinary and blood biomarkers, none of them are used routinely in the clinical practice due to the high costs and the scanty evidence supporting the superiority of a specific marker over the others in the early diagnosis and management of AKI and HRS.27
Management Of HRS
General Concepts
According to the aforementioned ICA consensus,5 a novel definition of response to treatment was provided. In particular, partial response is defined as regression of AKI stage with a reduction in sCr to ≥0.3 mg/dl above the baseline value, whereas complete response is defined as the return of sCr to a value within 0.3 mg/dl of the baseline value.
Patients with ascites and suspected HRS-1 should be immediately treated as follows: (a) reduction or withdrawal of diuretic therapy and withdrawal of all potentially nephrotoxic drugs, (b) effective plasma volume expansion in the case of hypovolaemia, and (c) prompt recognition and early treatment of bacterial infections when suspected.5,28
In the case of response (return of sCr to a value within 0.3 mg/dl of the baseline), patients should be followed closely for early identification of eventual recurrences.29 Alternatively, in the case of non-response, it should be considered the expansion of plasma volume with intravenous albumin at the dose of 1 g per kg body weight per day for two consecutive days, in order to treat pre-renal AKI and to allow differential diagnosis of AKI.5 Diagnosis of HRS should be formulated when all these therapeutic approaches result unsuccessful and after ruling out other causes of secondary nephropathy (Figure 2).
 |
Figure 2 Therapeutic approach to the renal disease in cirrhotic patients. Abbreviation: HRS, hepatorenal syndrome. |
Role Of Albumin And Non-Selective Beta-Blockers
Albumin, whose concentration is the higher among serum proteins, is of fundamental importance in both maintaining fluid distribution in the body and influencing immune response through modulation of pro-inflammatory molecules. In end-stage liver disease, both albumin concentration and functionality are significantly impaired.30 Therefore, patients with advanced cirrhosis and ascites might benefit from albumin infusions either in terms of the improved immune system with a consequent lower risk of experiencing severe complications such as spontaneous bacterial peritonitis (SBP) and renal perfusion.31,32
Non-selective beta-blockers (NSBBs) are the first-line pharmacological therapy for preventing variceal bleeding in both primary and secondary prophylaxis. Other proposed benefits of NSBB therapy which may lead to a decreased risk of infections might be the increased gut motility and reduced bacterial translocation.33,34 Furthermore, a subgroup analysis of the CANONIC study found that NSBBs therapy leads to decreased severity of systemic inflammation and prolonged survival in subjects with ACLF.35
Interestingly, the role of NSBBs in advanced cirrhotic patients has been recently questioned by some retrospective studies, in particular, in the presence of refractory ascites36 and SBP.37 However, these findings of reduced survival in cirrhotic patients treated with NSBBs were not confirmed by subsequent studies.38–41
Therefore, the “window hypothesis” was formulated where NSBBs are beneficial only within a narrow window in cirrhotic patients and might decrease survival in end-stage patients42 mainly due to reduced perfusion to vital organs (among them the kidney, hence the increased risk of HRS) caused by the excessive beta-adrenergic activity suppression. This interesting theory still needs to be supported by robust evidence and the exact time during the natural course of cirrhosis when that “window” should be considered closed is still unknown.43,44
In conclusion, due to the above-cited findings, NSBBs should not be withheld in cirrhotics even in an advanced stage as there is no definitive evidence on the increasing risk of developing HRS due to NSBB treatment.
Spontaneous Bacterial Peritonitis
As SBP occurrence plays a fundamental role as a triggering event of HRS-1, primary and secondary prevention of SBP should be adopted when indicated. Traditional indications to antibiotic prevention are upper gastrointestinal hemorrhage, low protein concentration in the ascitic fluid (<1.5 g/dL) when combined with any of the following features: Child-Pugh score ≥B9, serum bilirubin level ≥3 mg/dL, impaired renal function or hyponatremia (≤130 mEq/L), and previous episodes of SBP.45
In the case of gastrointestinal bleeding, prophylaxis with intravenous ceftriaxone (1 g/day) should be started when bleeding occurs and continued for up to 7 days.45 Conclusion of a recent network meta-analysis supports with moderate-quality evidence the use of quinolones (both norfloxacin and ciprofloxacin) in patients with low protein content in the ascitic fluid and only with low-quality evidence the use of rifaximin over placebo.46
As a consequence of earlier studies,47 current guidelines recommend secondary lifelong prophylaxis with norfloxacin (400 mg/day) after the first occurrence of SBP.45
Of course, any eventual cause of the underlying liver disease should be treated. In particular, the great impact of novel antiviral therapies in viral cirrhosis48,49 is expected to play a fundamental role in the prevention of cirrhosis complications such as HRS.
Vasoconstrictor Agents
The rationale for the use of vasoconstrictor agents in HRS is related to the splanchnic vasodilation underlying the renal dysfunction.
Vasoconstrictors actually in use for the management of HRS are: terlipressin, noradrenaline and the combination of midodrine + octreotide. Terlipressin represents the most used and tested drug in the therapeutic algorithm of HRS. A number of studies showed the combination of terlipressin plus albumin to be more effective than albumin alone in these patients.50–53 The REVERSE trial compared 97 patients treated with terlipressin + albumin vs 99 patients treated with placebo (+ albumin) and did not find a significant benefit in terms of HRS reversal rate (19.6% with terlipressin and 13.1% with albumin alone, p=0.22), whereas the change in creatinine levels at end of treatment was significantly superior with terlipressin (−1.1 mg/dl vs −0.6 mg/dl, p<0.001).53 Of note, this landmark trial did not show any difference between the two arms in terms of transplant-free survival.53
Terlipressin can be administered both as intravenous boluses (from 0.5–1 mg every 4–6 hrs to 2 mg every 4 hrs) and as a continuous intravenous infusion (from 2 mg/day to 12 mg/day). The two administration regimens were found to be equally effective with the latter being associated with a significantly lower incidence of severe side effects such as diarrhea, ischemia, or circulatory overload in a recent Italian trial.54
In absence of response, terlipressin dose should be increased in a stepwise manner, while albumin should be administered at the dose of 20–40 g/day. Anyway, the treatment should be discontinued within 14 days. The recurrence rate after discontinuation is less than 20% and retreatment is frequently effective.5
In spite of the increasing body of evidence supporting its use, terlipressin has not been approved for use in the United States yet.
Midodrine (an α1-agonist drug) is usually administered in combination with octreotide (a somatostatin analogue) and albumin, and it represents the current standard of care in the United States. A single Italian randomized controlled trial showed that terlipressin plus albumin clearly outperformed midodrine plus octreotide and albumin in the management of HRS.55
The administration of norepinephrine (administered in continuous infusion at a dose of 0.5–3 mg/h) plus albumin has been investigated in the treatment of HRS-1, and it was showed to be as effective as terlipressin in the treatment of HRS in a number of trials (mainly small single-center series).56–59 Of note, noradrenaline has no effect on portal pressure (unlike terlipressin) and this aspect may be of interest in the management of HRS in acute-on-chronic liver failure (ACLF) patients.60
Currently, the use of norepinephrine in treating HRS is limited by the need for a central venous line and continuous monitoring, thus rendering the treatment unfeasible outside intensive care units.
Table 1 reports the available trials testing pharmacological agents in cirrhotic patients with type-1 hepatorenal syndrome.
 |
Table 1 Characteristics And Outcomes Of Published Randomized Controlled Trials Comparing Different Pharmacological Interventions For Management Of Type 1 Hepatorenal Syndrome |
Comparative Efficacy Of Treatments For HRS-1
The efficacy of vasoconstrictor agents was confirmed in several meta-analyses.63–70 A meta-analysis by Gluud et al showed that vasoconstrictor drugs alone or with albumin reduce mortality compared with no intervention or albumin alone (risk ratio 0.82).63 In subgroup analyses, the effect on mortality was seen at 15 days but not at 30 days, 90 days, or 180 days.63
Conclusions of a recent network meta-analysis support with moderate-quality evidence a benefit of terlipressin in terms of improved survival in patients with HRS-1 and only with low-quality evidence the use of other pharmacological agents compared with placebo.66 The same review suggests the use of terlipressin over placebo (low-quality evidence) and over midodrine plus octreotide (moderate-quality evidence) in terms of HRS reversal whereas terlipressin and norepinephrine showed similar results, albeit the former was supported by the higher quality of evidence.66
Therefore, although current American guidelines still recommend the use of midodrine plus octreotide with albumin in these patients, only low-quality evidence was found to support this recommendation, without any significant benefit for short-term survival or in reversing hepatorenal syndrome.66 As clearly reported by Gines in the editorial to the above-cited network meta-analysis, it is time to consider terlipressin “ready for prime time” in the management of HRS-1 due to the great body of evidence and the unequivocal results supporting its use.71
Non-pharmacological treatments of HRS include renal replacement therapy, molecular adsorbent recirculating system (MARS), and transjugular intrahepatic portosystemic shunting (TIPS). In patients with irreversible HRS with no response to pharmacological agents, renal replacement therapy either in the form of hemodialysis or continuous veno-venous hemofiltration should be considered, particularly in presence of intractable fluid overload and acidosis, uremic symptoms, and electrolyte abnormalities.72–74
MARS resulted able to significantly decrease sCr level in patients with HRS on ACLF, although with no difference in 28-day mortality as compared to standard medical therapy.75
Another randomized controlled trial showed that MARS significantly reduces sCr in addition to standard medical treatment and hemodiafiltration.76
Although TIPS was traditionally contraindicated in patients with unresolved HRS-1, a recent meta-analysis including nine studies (128 patients) found pooled short-term and 1-year survival rates as high as 72% and 47% in HRS-1 with no lethal procedure-related complications observed.77 The pooled rate of renal function improvement after transjugular intrahepatic portosystemic shunt was 93% in type 1 hepatorenal syndrome and 83% in any type of hepatorenal syndrome.77
Liver Transplantation
Orthotopic liver transplantation (OLT) represents the best therapeutic option in these subjects regardless of their response to pharmacological therapy.78 However, there are still some unsolved issues concerning the use of OLT in HRS patients. First, mean sCr was found to be higher even after OLT in patients transplanted for HRS as compared to other cirrhotic patients.78,79 Therefore, simultaneous liver-kidney transplant should be considered in these subjects not only based on the concern of increased mortality post-transplant, but also due to the concern of lack of renal recovery after OLT.80,81
Identification and validation of predictors of renal recovery and the estimation of the extent of that recovery following liver transplantation represent an unsolved issue in the field.
Finally, concerning the priority in the waiting list, the paradoxical effect of pharmacological treatment in responders should be considered. In fact, effective treatment by lowering sCr can reduce the baseline MELD score, thus delaying the timing of OLT. This paradoxical effect of treatment in responders should be obviated either by considering only the baseline MELD, or by including eventual other treatments such as dialysis in the calculation of the MELD score according to the kind of response.82
Although renal recovery and patient survival after liver transplant were described to be significantly poorer for patients with AKI due to acute tubular necrosis than hepatorenal syndrome,83 correct prediction of reversal of kidney injury and the extent of that recovery following OLT represent still a challenge. In fact, a number of confounders should be considered, such as pre-existing comorbidities, undiagnosed primary renal disease, perioperative events, and post-transplant immunosuppression; therefore, it is difficult to delineate the contribution of each of these factors to the clinical course after liver transplantation.
Conclusion
Renal dysfunction is a common complication in patients with end-stage cirrhosis, with or without ACLF. Several issues remain to be addressed such as the impact of the management of AKI according to the new algorithm on the outcome of these patients and the role of the new biomarkers of renal tubular damage in predicting the progression and prognosis of HRS, and in the differential diagnosis of the different types of AKI.
In summary, the results of the latest consensus conference of the ICA introduce a new dynamic definition of AKI and HRS in patients with cirrhosis, representing a substantial change from the traditional criteria used so far in the definition of AKI and HRS-1.
Disclosure
The author reports no conflicts of interest in this work.
References
1. Tujios SR, Hynan LS, Vazquez MA, et al. Acute Liver Failure Study Group. Risk factors and outcomes of acute kidney injury in patients with acute liver failure. Clin Gastroenterol Hepatol. 2015;13:352–359. doi:10.1016/j.cgh.2014.07.011
2. Moreau R, Jalan R, Gines P, et al. Acute-on-chronic liver failure is a distinct syndrome that develops in patients with acute decompensation of cirrhosis. Gastroenterology. 2013;144:1426–1437. doi:10.1053/j.gastro.2013.02.042
3. Arroyo V, Gines P, Gerbes AL, et al. Definition and diagnostic criteria of refractory ascites and hepatorenal syndrome in cirrhosis. Hepatology. 1996;23:164–176. doi:10.1002/hep.v23:1
4. Salerno F, Gerbes A, Gines P, et al. Diagnosis, prevention and treatment of the hepatorenal syndrome in cirrhosis a consensus workshop of the International Ascites Club. Gut. 2007;56:1310–1318. doi:10.1136/gut.2006.107789
5. Angeli P, Gines P, Wong F, et al. Diagnosis and management of acute kidney injury in patients with cirrhosis: revised consensus recommendations of the ICA. J Hepatol. 2015;62:968–974. doi:10.1016/j.jhep.2014.12.029
6. Moreau R, Lebrec D. Acute renal failure in patients with cirrhosis: perspectives in the age of MELD. Hepatology. 2003;37:233–243. doi:10.1053/jhep.2003.50084
7. Angeli P, Garcia-Tsao G, Nadim MK, Parikh CR. News in pathophysiology, definition and classification of hepatorenal syndrome: a step beyond the International Club of Ascites (ICA) consensus document. J Hepatol. 2019;
8. Facciorusso A, Nacchiero MC, Rosania R, et al. The use of human albumin for the treatment of ascites in patients with liver cirrhosis: item of safety, facts, controversies and perspectives. Curr Drug Saf. 2011;6(4):267–274. doi:10.2174/157488611798280906
9. Davenport A, Sheikh MF, Lamb E, Agarwal B, Jalan R. Acute kidney injury in acute-on-chronic liver failure: where does hepatorenal syndrome fit? Kidney Int. 2017;92(5):1058–1070. doi:10.1016/j.kint.2017.04.048
10. Boyer TD, Sanyal AJ, Garcia-Tsao G, et al. Predictors of response to terlipressin plus albumin in hepatorenal syndrome (HRS) type 1: relationship of serum creatinine to hemodynamics. J Hepatol. 2011;55:315–321. doi:10.1016/j.jhep.2010.11.020
11. Rodriguez E, Elia C, Solà E, et al. Terlipressin and albumin for type-1 hepatorenal syndrome associated with sepsis. J Hepatol. 2014;60:955–961. doi:10.1016/j.jhep.2013.12.032
12. Trawalé JM, Paradis V, Rautou PE, et al. The spectrum of renal lesions in patients with cirrhosis: a clinicopathological study. Liver Int. 2010;30:725–732. doi:10.1111/j.1478-3231.2009.02182.x
13. Iwakiri Y. Pathophysiology of portal hypertension. Clin Liver Dis. 2014;18(2):281–291. doi:10.1016/j.cld.2013.12.001
14. Cárdenas A, Ginès P. Portal hypertension. Curr Opin Gastroenterol. 2009;25(3):195–201. doi:10.1097/MOG.0b013e328329e154
15. Facciorusso A, Del Prete V, Crucinio N, et al. Angiotensin receptor blockers improve survival outcomes after radiofrequency ablation in hepatocarcinoma patients. J Gastroenterol Hepatol. 2015;30(11):1643–1650. doi:10.1111/jgh.2015.30.issue-11
16. Facciorusso A, Amoruso A, Neve V, Antonino M, Prete VD, Barone M. Role of vaptans in the management of hydroelectrolytic imbalance in liver cirrhosis. World J Hepatol. 2014;6(11):793–799. doi:10.4254/wjh.v6.i11.793
17. Ginès P, Berl T, Bernardi M, et al. Hyponatremia in cirrhosis: from pathogenesis to treatment. Hepatology. 1998;28:851–864. doi:10.1002/hep.510280337
18. Ginès A, Escorsell A, Ginès P, et al. Incidence, predictive factors, and prognosis of hepatorenal syndrome in cirrhosis. Gastroenterology. 1993;105:229–236. doi:10.1016/0016-5085(93)90031-7
19. Leithead JA, Hayes PC, Ferguson JW. Review article: advances in the management of patients with cirrhosis and portal hypertension-related renal dysfunction. Aliment Pharmacol Ther. 2014;39(7):699–711. doi:10.1111/apt.2014.39.issue-7
20. Fagundes C, Pepin MN, Guevara M, et al. Urinary neutrophil gelatinase-associated lipocalin as biomarker in the differential diagnosis of impairment of kidney function in cirrhosis. J Hepatol. 2012;57:267–273. doi:10.1016/j.jhep.2012.03.015
21. Belcher JM, Sanyal AJ, Peixoto AJ, et al. Kidney biomarkers and differential diagnosis of patients with cirrhosis and acute kidney injury. Hepatology. 2014;60:622–632. doi:10.1002/hep.v60.2
22. Mindikoglu AL, Opekun AR, Coarfa C, et al. Robust metabolomic signature is associated with altered renal hemodynamics in patients with cirrhosis. Gastroenterology. 2017;152(5):S1044. doi:10.1016/S0016-5085(17)33534-5
23. Mindikoglu AL, Dowling TC, Magder LS, et al. Estimation of glomerular filtration rate in patients with cirrhosis by using new and conventional filtration markers and dimethylarginines. Clin Gastroenterol Hepatol. 2016;14:624–632 e2. doi:10.1016/j.cgh.2015.06.021
24. Stevens LA, Coresh J, Schmid CH, et al. Estimating GFR using serum cystatin C alone and in combination with serum creatinine: a pooled analysis of 3,418 individuals with CKD. Am J Kidney Dis. 2008;51:395–406. doi:10.1053/j.ajkd.2007.11.018
25. Cholongitas E, Ioannidou M, Goulis I, et al. Comparison of creatinine and cystatin formulae with 51 chromiumethylenediaminetetraacetic acid glomerular filtration rate in patients with decompensated cirrhosis. J Gastroenterol Hepatol. 2017;32:191–198. doi:10.1111/jgh.2017.32.issue-1
26. De Souza V, Hadj-Aissa A, Dolomanova O, et al. Creatinine versus cystatine C-based equations in assessing the renal function of candidates for liver transplantation with cirrhosis. Hepatology. 2014;59:1522–1531. doi:10.1002/hep.26886
27. Mindikoglu AL, Pappas SC. New developments in hepatorenal syndrome. Clin Gastroenterol Hepatol. 2018;16(2):162–177. doi:10.1016/j.cgh.2017.05.041
28. Crager S. Critically ill patients with end-stage liver disease. Emerg Med Clin North Am. 2019;37(3):511–527. doi:10.1016/j.emc.2019.03.008
29. Tsien CD, Rabie R, Wong F. Acute kidney injury in decompensated cirrhosis. Gut. 2013;62:131–137. doi:10.1136/gutjnl-2011-301255
30. Domenicali M, Baldassarre M, Giannone FA, et al. Posttranscriptional changes of serum albumin: clinical and prognostic significance in hospitalized patients with cirrhosis. Hepatology. 2014;60:1851–1860. doi:10.1016/j.cgh.2010.06.024
31. Caraceni P, Riggio O, Angeli P, et al. Long-term albumin administration in decompensated cirrhosis (ANSWER): an open-label randomised trial. Lancet. 2018;391(10138):2417–2429. doi:10.1016/S0140-6736(18)30840-7
32. Facciorusso A, Antonino M, Orsitto E, Sacco R. Primary and secondary prophylaxis of spontaneous bacterial peritonitis: current state of the art. Expert Rev Gastroenterol Hepatol. 2019;13(8):751–759. doi:10.1080/17474124.2019.1644167
33. Merli M, Lucidi C, Di Gregorio V, et al. The chronic use of beta-blockers and proton pump inhibitors may affect the rate of bacterial infections in cirrhosis. Liver Int. 2015;35(2):362–369. doi:10.1111/liv.2015.35.issue-2
34. Reiberger T, Ferlitsch A, Payer BA, et al. Non-selective betablocker therapy decreases intestinal permeability and serum levels of LBP and IL-6 in patients with cirrhosis. J Hepatol. 2013;58:911–921. doi:10.1016/j.jhep.2012.12.011
35. Mookerjee RP, Pavesi M, Thomsen KL, et al. Treatment with non-selective beta blockers is associated with reduced severity of systemic inflammation and improved survival of patients with acute-on-chronic liver failure. J Hepatol. 2016;64(3):574–582. doi:10.1016/j.jhep.2015.10.018
36. Sersté T, Francoz C, Durand F, et al. Beta-blockers cause paracentesis-induced circulatory dysfunction in patients with cirrhosis and refractory ascites: a cross-over study. J Hepatol. 2011;55:794–799. doi:10.1016/j.jhep.2011.01.034
37. Mandorfer M, Bota S, Schwabl P, et al. Nonselective b blockers increase risk for hepatorenal syndrome and death in patients with cirrhosis and spontaneous bacterial peritonitis. Gastroenterology. 2014;146:1680–1690.e1. doi:10.1053/j.gastro.2014.03.005
38. Bossen L, Krag A, Vilstrup H, et al. Non-selective β-blockers do not affect mortality in cirrhosis patients with ascites: post hoc analysis of three RCTs with 1198 patients. Hepatology. 2015. doi:10.1002/hep.28352
39. Bhutta AQ, Garcia-Tsao G, Reddy KR, et al. Beta-blockers in hospitalised patients with cirrhosis and ascites: mortality and factors determining discontinuation and reinitiation. Aliment Pharmacol Ther. 2018;47(1):78–85. doi:10.1111/apt.14366
40. Chirapongsathorn S, Valentin N, Alahdab F, et al. Nonselective β-blockers and survival in patients with cirrhosis and ascites: a systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2016;14(8):1096–1104. doi:10.1016/j.cgh.2016.01.012
41. Facciorusso A, Roy S, Livadas S, et al. Nonselective beta-blockers do not affect survival in cirrhotic patients with ascites. Dig Dis Sci. 2018;63(7):1737–1746. doi:10.1007/s10620-018-5092-6
42. Krag A, Wiest R, Albillos A, Gluud LL. The window hypothesis: haemodynamic and non-haemodynamic effects of betablockers improve survival of patients with cirrhosis during a window in the disease. Gut. 2012;61:967–969. doi:10.1136/gutjnl-2011-301348
43. Ge PS, Runyon BA. The changing role of beta-blocker therapy in patients with cirrhosis. J Hepatol. 2014;60(3):643–653. doi:10.1016/j.jhep.2013.09.016
44. Ge PS, Runyon BA. When should the β-blocker window in cirrhosis close? Gastroenterology. 2014;146(7):1597–1599. doi:10.1053/j.gastro.2014.04.028
45. European Association for the Study of the Liver. EASL clinical practice guidelines on the management of ascites, spontaneous bacterial peritonitis, and hepatorenal syndrome in cirrhosis. J Hepatol. 2010;53(3):397–417. doi:10.1016/j.jhep.2010.05.004
46. Facciorusso A, Papagiouvanni I, Cela M, Buccino VR, Sacco R. Comparative efficacy of long-term antibiotic treatments in the primary prophylaxis of spontaneous bacterial peritonitis. Liver Int. 2019;39(8):1448–1458. doi:10.1111/liv.14109
47. Ginés P, Rimola A, Planas R, et al. Norfloxacin prevents spontaneous bacterial peritonitis recurrence in cirrhosis: results of a double-blind, placebo-controlled trial. Hepatology. 1990;12(4 Pt 1):716–724. doi:10.1002/hep.1840120416
48. Facciorusso A, Garcia Perdomo HA, Muscatiello N, Buccino RV, Wong VW, Singh S. Systematic review with meta-analysis: change in liver stiffness during anti-viral therapy in patients with hepatitis B. Dig Liver Dis. 2018;50(8):787–794. doi:10.1016/j.dld.2018.05.005
49. Singh S, Facciorusso A, Loomba R, Falck-Ytter YT. Magnitude and kinetics of decrease in liver stiffness after antiviral therapy in patients with chronic hepatitis C: a systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2018;16(1):27–38. doi:10.1016/j.cgh.2017.04.038
50. Solanki P, Chawla A, Garg R, Gupta R, Jain M, Sarin SK. Beneficial effects of terlipressin in hepatorenal syndrome: a prospective, randomized placebo-controlled clinical trial. J Gastroenterol Hepatol. 2003;18:152–156. doi:10.1046/j.1440-1746.2003.02934.x
51. Neri S, Pulvirenti D, Malaguarnera M, et al. Terlipressin and albumin in patients with cirrhosis and type I hepatorenal syndrome. Dig Dis Sci. 2008;53:830–835. doi:10.1007/s10620-007-9919-9
52. Sanyal AJ, Boyer T, Garcia-Tsao G, et al. A randomized, prospective, double-blind, placebo-controlled trial of terlipressin for type 1 hepatorenal syndrome. Gastroenterology. 2008;134:1360–1368. doi:10.1053/j.gastro.2008.02.014
53. Boyer TD, Sanyal AJ, Wong F, et al. Terlipressin plus albumin is more effective than albumin alone in improving renal function in patients with cirrhosis and hepatorenal syndrome type 1. Gastroenterology. 2016;150:1579–1589. doi:10.1053/j.gastro.2016.02.026
54. Cavallin M, Piano S, Romano A, et al. Terlipressin given by continuous intravenous infusion versus intravenous boluses in the treatment of hepatorenal syndrome: a randomized controlled study. Hepatology. 2016;63:983–992. doi:10.1002/hep.28396
55. Cavallin M, Kamath PS, Merli M, et al. Terlipressin plus albumin versus midodrine and octreotide plus albumin in the treatment of hepatorenal syndrome: a randomized trial. Hepatology. 2015;62:567–574. doi:10.1002/hep.v62.2
56. Indrabi RA, Javid G, Zargar SA, et al. Noradrenaline is equally effective as terlipressin in reversal of type 1 hepatorenal syndrome: a randomized prospective study. J Clin Exp Hepatol. 2013;1:S97. doi:10.1016/j.jceh.2013.02.242
57. Sharma P, Kumar A, Shrama BC, Sarin SK. An open label, pilot, randomized controlled trial of noradrenaline versus terlipressin in the treatment of type 1 hepatorenal syndrome and predictors of response. Am J Gastroenterol. 2008;103:1689–1697. doi:10.1111/ajg.2008.103.issue-7
58. Singh V, Ghosh S, Singh B, et al. Noradrenaline vs. terlipressin in the treatment of hepatorenal syndrome: a randomized study. J Hepatol. 2012;56:1293–1298. doi:10.1016/j.jhep.2012.01.012
59. Alessandria C, Ottobrelli A, Debernardi-Venon W, et al. Noradrenalin vs terlipressin in patients with hepatorenal syndrome: a prospective, randomized, unblinded, pilot study. J Hepatol. 2007;47:499–505. doi:10.1016/j.jhep.2007.04.010
60. Arora V, Maiwall R, Rajan V, et al. Terlipressin is superior to noradrenaline in the management of acute kidney injury in acute on chronic liver failure. Hepatology. 2018. [Epub ahead of print]. doi:10.1002/hep.30208
61. Srivastava SShalimar Vishnubhatla S. Randomized controlled trial comparing the efficacy of terlipressin and albumin with a combination of concurrent dopamine, furosemide, and albumin in hepatorenal syndrome. J Clin Exp Hepatol. 2015;5(4):276–285.
62. Tavakkoli H, Yazdanpanah KMansourian M. Noradrenalin versus the combination of midodrine and octreotide in patients with hepatorenal syndrome: randomized clinical trial. Int J Prev Med. 2012;3(11):764–769.
63. Gluud LL, Christensen K, Christensen E, Krag A. Systematic review of randomized trials on vasoconstrictor drugs for hepatorenal syndrome. Hepatology. 2010;51:576–584. doi:10.1002/hep.23286
64. Dobre M, Demirjian S, Sehgal AR, et al. Terlipressin in hepatorenal syndrome: a systematic review and meta-analysis. Int Urol Nephrol. 2011;43:175–184. doi:10.1007/s11255-010-9725-8
65. Fabrizi F, Dixit V, Messa P, et al. Terlipressin for hepatorenal syndrome: a meta-analysis of randomized trials. Int J Artif Organs. 2009;32:133–140. doi:10.1177/039139880903200303
66. Facciorusso A, Chandar AK, Murad MH, et al. Comparative efficacy of pharmacological strategies for management of type 1 hepatorenal syndrome: a systematic review and network meta-analysis. Lancet Gastroenterol Hepatol. 2017;2:94–102. doi:10.1016/S2468-1253(16)30157-1
67. Gifford FJ, Morling JR, Fallowfield JA. Systematic review with meta-analysis: vasoactive drugs for the treatment of hepatorenal syndrome type 1. Aliment Pharmacol Ther. 2017;45:593–603. doi:10.1111/apt.13912
68. Gluud LL, Christensen K, Christensen E, et al. Terlipressin for hepatorenal syndrome. Cochrane Database Syst Rev. 2012;9:CD005162.
69. Mattos AZ, Mattos AA, Ribeiro RA. Terlipressin versus noradrenaline in the treatment of hepatorenal syndrome: systematic review with meta-analysis and full economic evaluation. Eur J Gastroenterol Hepatol. 2016;28:345–351. doi:10.1097/MEG.0000000000000537
70. Sagi SV, Mittal S, Kasturi KS, et al. Terlipressin therapy for reversal of type 1 hepatorenal syndrome: a meta-analysis of randomized controlled trials. J Gastroenterol Hepatol. 2010;25:880–885. doi:10.1111/j.1440-1746.2009.06132.x
71. Gines P. Terlipressin for hepatorenal syndrome: ready for prime time. Lancet Gastroenterol Hepatol. 2017;2(2):75–76. doi:10.1016/S2468-1253(16)30211-4
72. Capling RK, Bastani B. The clinical course of patients with type 1 hepatorenal syndrome maintained on hemodialysis. Ren Fail. 2004;26:563–568. doi:10.1081/JDI-200035988
73. Keller F, Heinze H, Jochimsen F, et al. Risk factors and outcome of 107 patients with decompensated liver disease and acute renal failure (including 26 patients with hepatorenal syndrome): the role of hemodialysis. Ren Fail. 1995;17:135–146. doi:10.3109/08860229509026250
74. Witzke O, Baumann M, Patschan D, et al. Which patients benefit from hemodialysis therapy in hepatorenal syndrome? J Gastroenterol Hepatol. 2004;19:1369–1373. doi:10.1111/jgh.2004.19.issue-12
75. Banares R, Nevens F, Larsen FS, et al. Extracorporeal albumin dialysis with the molecular adsorbent recirculating system in acute-on-chronic liver failure: the RELIEF trial. Hepatology. 2013;57:1153–1162. doi:10.1002/hep.26185
76. Mitzner SR, Stange J, Klammt S, et al. Improvement of hepatorenal syndrome with extracorporeal albumin dialysis MARS: results of a prospective, randomized, controlled clinical trial. Liver Transpl. 2000;6:277–286. doi:10.1053/lv.2000.6355
77. Song T, Rössle M, He F, Liu F, Guo X, Qi X. Transjugular intrahepatic portosystemic shunt for hepatorenal syndrome: a systematic review and meta-analysis. Dig Liver Dis. 2018;50(4):323–330. doi:10.1016/j.dld.2018.01.123
78. Boyer TD, Sanyal AJ, Garcia-Tsao G, et al. Impact of liver transplantation on the survival of patients treated for hepatorenal syndrome type 1. Liver Transpl. 2011;17:1328–1332. doi:10.1002/lt.v17.11
79. Wong F, Leung W, Al Beshir M, Marquez M, Renner EL. Outcomes of patients with cirrhosis and hepatorenal syndrome type 1 treated with liver transplantation. Liver Transpl. 2015;21:300–307. doi:10.1002/lt.24049
80. Formica RN, Aeder M, Boyle G, et al. Simultaneous liver-kidney allocation policy: a proposal to optimize appropriate utilization of scarce resources. Am J Transplant. 2016;16:758–766. doi:10.1111/ajt.13631
81. Ruiz R, Barri YM, Jennings LW, et al. Hepatorenal syndrome: a proposal for kidney after liver transplantation (KALT). Liver Transpl. 2007;13:838–843. doi:10.1002/lt.21149
82. Martin-Llahi M, Pepin M-N, Guevara M, et al. Terlipressin and albumin vs albumin in patients with cirrhosis and hepatorenal syndrome: a randomized study. Gastroenterology. 2008;134(5):1352–1359.
83. Zafar S, Haque I, Tayyab GU, Khan G, Chaudry N. Role of terlipressin and albumin combination versus albumin alone in hepatorenal syndrome. American Journal of Gastroenterology. 2012;107:S175–S176.
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
