Back to Journals » OncoTargets and Therapy » Volume 13
GRK2 Suppresses Hepatocellular Carcinoma Metastasis and Invasion Through Down-Regulation of Prostaglandin E Receptor 2
Authors Li N, Wu JJ, Chen TT, Li XQ, Du JJ, Shan S, Wei W, Sun WY
Received 9 June 2020
Accepted for publication 20 August 2020
Published 28 September 2020 Volume 2020:13 Pages 9559—9571
DOI https://doi.org/10.2147/OTT.S266641
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr William C. Cho
Nan Li,* Jing-Jing Wu,* Ting-Ting Chen, Xiu-Qin Li, Jia-Jia Du, Shan Shan, Wei Wei, Wu-Yi Sun
Institute of Clinical Pharmacology, Anhui Medical University, Key Laboratory of Anti-Inflammatory and Immune Medicine, Ministry of Education, Anhui Collaborative Innovation Center of Anti-Inflammatory and Immune Medicine, Hefei 230032, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Wu-Yi Sun; Wei Wei
Institute of Clinical Pharmacology, Anhui Medical University, Key Laboratory of Anti-Inflammatory and Immune Medicine, Ministry of Education, Anhui Collaborative Innovation Center of Anti-Inflammatory and Immune Medicine, Meishan Road, Hefei 230032, Anhui Province, People’s Republic of China
Tel/Fax +86 551 65161206
Email [email protected]; [email protected]
Background: Hepatocellular carcinoma (HCC) is an aggressive form of human liver cancer and the fifth most common malignancy worldwide. Novel effective treatment strategies for HCC are urgently in clinical because of its poor response to conventional therapies. G protein-coupled receptor kinases (GRKs), including GRK2 and GRK3, are known that involves in various essential cellular processes and regulates numerous signaling pathways. However, the role of GRK2/3 in invasion and metastasis of HCC still remains unclear.
Materials and Methods: Immunohistochemistry, Western blot, laser confocal microscopy and qRT-PCR were used to detect the expression of GRK2/3 and EP2 in liver tissues of HCC patients and DEN-induced HCC mice. Wound healing and transwell assay were applied to measure the migration and invasion of HCC cells after transfected with GRK2 siRNA. The downstream pathway of Akt and ERK was verified by Western blot.
Results: The expression of GRK2 was significantly decreased, while GRK3 was not significantly changed in HCC tissues compared with noncancerous tissues of HCC patients. Moreover, GRK2 expression was reduced during liver tumorigenesis in diethylnitrosamine-induced liver tumor model. In addition, our in vitro study showed that GRK2 expression was gradually decreased with increasing HCC cell line metastatic potential, and GRK2 knockdown significantly promoted the migration and invasion of HCC cells. Furthermore, low GRK2 expression was associated with increased expression of EP2 receptor translocation to HCC cell membrane, and the activation of Akt pathway.
Conclusion: These data suggest that GRK2 inhibits HCC metastasis and invasion may be through regulating EP2 receptor translocation, and this effect appears to be mediated by Akt pathway.
Keywords: GRK2, hepatocellular carcinoma, invasion, migration
Introduction
Hepatocellular carcinoma (HCC) is the predominant histological subtype of primary liver cancer, accounts for the fifth most common malignant tumor and ranking third among various cancer death rates in the world.1,2 Although surgical liver cancer resection, radiofrequency ablation, and liver transplantation can improve survival in patients with early HCC, rapid invasion and metastasis of HCC have become the main causes of treatment failure.3 Therefore, exploring the mechanism of HCC invasion and metastasis is of profound significance for finding potential new targets for drug therapy.
G protein-coupled receptor kinases (GRKs) belong to serine/threonine kinases. Its classical role is to phosphorylate and rapidly desensitize G protein-coupled receptors (GPCRs).4,5 Among the seven subtypes of GRKs, of which only GRK2 and GRK3 are abundantly expressed in liver tissues. Accumulating studies have been demonstrated that GRKs are involved in the development of various diseases such as cardiovascular disease, arthritis and malignant tumors.6,7 Under neuroendocrine prostate cancer, GRK3 acts as a direct target of cyclic adenosine monophosphate response element binding protein regulation and promotes neuroendocrine differentiation.8 Our previous study found that GRK2 expression in liver fibrosis model rats is decreased.9 Combined with the above evidence, we speculate that GRK2 may play an important role in HCC invasion and metastasis, although how it participates mechanistically has not been thoroughly studied.
Prostaglandin E receptors (EP1-4) belong to GPCRs. It has been reported that among these 4 EP receptors, the expression of EP2 receptor was considerably upregulated in HCC tissues compared with the expression of EP1, EP3, and EP4 receptor, and predominately mediated the function of PGE2 in the progression of HCC.10 EP2 has proven to be beneficial to stimulate tumor vascular endothelial cell growth, promote tumor cell invasion and metastasis in the development of various tumors.11,12 In addition, our previous studies found that butaprost, a selective EP2 receptor agonist, can significantly stimulate the proliferation and invasion of HCC cells.13 However, whether the precise mechanism of GRK2 involving in HCC is related with EP2 receptor remain to be elucidated.
In the current research, to assess the importance of GRKs in HCC, we explored GRK2 and GRK3 expression in tumor samples from HCC patients and diethylnitrosamine (DEN)-induced liver tumor models in vivo. We found that GRK2 expression was significantly decreased in HCC tissues compared with noncancerous tissues of HCC patients, while GRK3 expression was not significantly changed. Moreover, reduced GRK2 expression was associated with hepatocarcinogenesis in DEN-induced liver tumor mice. Furthermore, we employed HCC cell lines with stepwise metastatic potential demonstrated that GRK2 expression gradually decreased with increasing HCC cell line metastatic potential, and in vitro depletion experiments showed that decreased GRK2 significantly promoted the migration and invasion of HCC cells, and elevated EP2 expression in cell membrane, thus upregulating the downstream Akt pathway. Taken together, these findings suggest that GRK2 is a potential treatment target in invasion and metastasis of HCC.
Materials and Methods
Patients and Samples
Patients with primary HCC and intrahepatic biliary lithiasis were recruited from Anhui Provincial Hospital. This study protocol was approved by the research ethics committee of Anhui Medical University, and performed in accordance with the Declaration of Helsinki. All participants provided their written informed consent. Eighteen paired tumor tissues and adjacent noncancerous liver tissues used for qRT-PCR assay and Western blot analysis were acquired from HCC patients who underwent surgical resection. Fresh tissues were collected immediately upon tumor resection in the operating room, and stored at −80°C until required. The paraffin-embedded tissue specimens used for immunohistochemical staining were collected from 72 HCC patients and 17 patients with intrahepatic biliary lithiasis (control group) who were diagnosed and underwent surgery between January 2007 and December 2010.
Animals
Male C57BL/6J mice were purchased from the Animal Center of Anhui Medical University. All animals were housed under specific-pathogen-free (SPF) conditions. All mouse experiments were performed in accordance with the guidelines of the Animal Care and Use Committee of Anhui Medical University. And all experiments were subject to approval by the Ethics Review Committee for Animal Experimentation of the Institute of Clinical Pharmacology, Anhui Medical University.
The DEN (Sigma-Aldrich, St. Louis, MO)-induced liver tumor mice model was established as described previously.14,15 Mice at postnatal day 14 were administered single intraperitoneal injections of DEN, which was dissolved in saline at a dose of 20 mg/kg. Control mice were injected with equal volumes of saline. For tissue sampling, mice were fasted overnight and sacrificed at 8, 16, 24, 32 and 40 weeks after DEN injection. Immediately after euthanasia, the mice liver tissues were obtained and photographed. Part of the liver was rapidly fixed in buffered 10% formalin for 24 h for histological analysis.
Cell Lines
HCC cell lines (MHCC97L, MHCC97H and HCCLM3) with stepwise metastatic potential were purchased from Liver Cancer Institute, Zhongshan Hospital, Fudan University, Shanghai, China. The normal liver cell line L-02 and the HCC cell lines with relatively low metastatic potential HepG2 and SMMC-7721 were purchased from the Institute of Biochemistry and Cell Biology, Chinese Academy of Sciences, Shanghai, China. The present cell study has been approved by the Research Ethics Committee of Anhui Medical University. All the cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM, Gibco, USA) supplemented with 10% fetal bovine serum (FBS, Zhejiang Tianhang Biotechnology Co., Ltd., Hangzhou, China), and 1% penicillin-streptomycin solution (Beyotime Biotechnology, Shanghai, China). Butaprost (Peprotech, USA), a selective EP2 receptor agonist, was used to explore the potential role of GRK2 and EP2 concerned with tumor cell migration and invasion.
Quantitative Real-Time PCR
Real-time PCR was conducted to determine GRK2 and GRK3 gene expression in tumor and non-tumor liver tissue samples. Total RNA was extracted using Trizol reagent, and complementary DNA (cDNA) was synthesized using a RevertAid First Strand cDNA Synthesis Kit (Fermentas, Vilnius, Lithuania), according to the manufacturer’s recommendations. The gene expression analysis was carried out using a Real-time PCR Detection System (ABI 7500) with an SYBR GreenER qPCR SuperMix Universal Kit (Invitrogen, Carlsbad, CA). The 2−ΔΔCT method was used to quantify the relative expression of these genes. The sequences of primers were as follows: GRK2, 5ʹ-CTTCCAGCCATACATTGAGGAG-3ʹ (forward) and 5ʹ-TTCGATGCACACTGAAGTCATT-3ʹ (reverse); GRK3, 5ʹ-AGTGCCGACTGGTTCTCCCTG-3ʹ (forward) and 5ʹ-TCTCGCTGAAGCAAGCCCTC-3ʹ (reverse); GAPDH, 5ʹ-AGGTCGGAGTCAACGGATTTG-3ʹ (forward) and 5ʹ-CCTGGAAGATGGTGATGGGAT-3ʹ (reverse). The mRNA expression of GAPDH was used as an internal reference.
Histopathological Analyses
Mice liver tissues were obtained and fixed with 10% formalin for 24 h, and then embedded in paraffin and serially cut into 4 µm-thick sections. The sections were stained by hematoxylin and eosin (H&E) staining. In brief, the prepared liver slices were stained with hematoxylin for 5 min followed by eosin for 3 min. Pathological changes in liver tissues were observed in H&E-stained sectioned samples through a light BX53 microscopic (Olympus Corporation, Tokyo, Japan).
Immunohistochemical Staining
The paraffin section of the liver tissue was baked at 60°C for 2 h, then blocked with 5% BSA solution, as well as dewaxed with xylene and hydrated in a series of graded ethanol. Sections were heated in 10 mM sodium citrate buffer for antigen retrieval and incubated with endogenous peroxidase blocking solution (Zhongshan Goldenbridge Biotechnology Co., Ltd, Beijing, China). Slides were incubated with primary antibodies against GRK2, GRK3 and EP2 (Santa Cruz Biotechnology, CA, USA) overnight at 4°C. Sections were stained with DAB chromogenic kit (Zhongshan Goldenbridge Biotechnology Co., Ltd, Beijing, China). Then they were counterstained with hematoxylin, washing, dehydrated and sealed. The sections were viewed under an Olympus BX53 microscope. The positive result was based on the appearance of yellow particles, and semi quantitative analysis of image was conducted using Image-Pro Plus software. For each slide, five random fields were analysed.
Immunofluorescence Assay
Cells were seeded on coverslips and treated with butaprost for 10, 30, and 120 min, then were immersion-fixed in 4% paraformaldehyde for 40 min. All sections were immersed and incubated with primary antibody against GRK2 or EP2 at 4°C overnight in the dark. Negative control sections were incubated with phosphate-buffered saline (PBS) instead of primary antibody. To determine proteins expression and cellular distribution, sections probed with anti-mouse Alexa Fluor 488 or anti-rabbit Alexa Fluor 555 conjugated antibodies (Thermo Fisher, MA, USA). And 4′, 6-diamidino-2-phenylindole (DAPI, Beyotime Biotechnology, Shanghai, China) was used to stain the nuclei. Finally, immunofluorescence staining was assessed by Leica TCS SP5 confocal microscope (Leica Microsystems).
siRNA Transfection
For GRK2 knockdown, SMC7721 cells were seeded in six-well plates 24 h prior to transfection. The Lipofectamine 2000 (Invitrogen, Carlsbad, CA) transfection reagent was shaken well, equal volumes of GRK2 siRNA and transfection reagent were mixed and incubated for 20 min before application to cells. Cells were harvested for Western blot or migration, invasion assays 48 h after transfection. The sequences of the designed oligonucleotides against GRK2 were as follows: 5ʹ-GCAUCAUGCAUGGCUACAUTT-3ʹ (sense) and 5ʹ-AUGUAGCCAUGCAUGAUGCTT-3ʹ (antisense). A scrambled RNA duplex was used as a negative control. siRNA duplexes were purchased from GenePharma Company (Shanghai, China).
Wound-Healing Assay
After SMC7721 cells transfected with the indicated GRK2 siRNA for 48 h. Cell monolayer was wounded with a sterile 200 μL pipette tip and washed by PBS to remove cell debris. The linear wound at 0 h and 24 h was photographed using an Olympus IX-71 inverted microscope. Relative migration of cells under each condition was estimated through taking five representative images including the scratch areas.
Transwell Invasion Assay
Cells transfected with GRK2 siRNA were trypsinized, re-suspended, and then 5×104 cells were added to the upper chamber. However, the lower chamber was supplemented with medium containing 10% FBS as a chemo-attractant, then was incubated for 36 h. Migrated cells were fixed with paraformaldehyde for 5 min, stained with crystal violet in the lower chamber. Invasion cells were counted by a light microscope in five randomly selected regions.
Membrane Protein Isolation in Cells
Cells were isolated using lysis buffer (Beyotime Institute of Biotechnology, Shanghai, China) on the ice for 30 min. Cell lysates were centrifuged at 12,000×g for 20 min at 4°C, some of the supernatants were used to detect total protein expression. The other supernatants were centrifuged at 100,000×g for 1 h at 4°C.16,17 The supernatant was collected and added the protein loading buffer (5×) to measure the cytoplasm protein. The precipitated membrane proteins were re-suspended in cell lysis buffer, added the protein loading buffer (5×), and boiled for 8 min. Samples were stored at −20°C until immunblotting.
Western Blot Analysis
As usual, proteins from liver tissues or HCC cells were lysed for 30 min on ice, and centrifuged at 14,000×g for 20 min at 4°C. The supernatant was collected to detect the expression of proteins. The proteins were resolved by sodium dodecyl-sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and then transferred to polyvinylidene fluoride (PVDF) membranes (Millipore, Bedford, MA). Membranes were blocked with 5% dried skimmed milk in 0.05% Tween 20-PBS for 2 h and incubated with primary antibodies overnight at 4 °C. The primary antibodies used were as follows: GRK2, GRK3, EP2 (Santa Cruz Biotechnology, CA, USA), β-actin (Zhongshan Goldenbridge Biotechnology Co., Ltd, Beijing, China), Na+/K+ ATPase (Abcam, Cambridge, UK), phospho-Akt (p-Akt), Akt, phospho-ERK (p-ERK) and ERK (Cell Signaling, MA, USA). The membranes were treated with the secondary antibodies followed by further incubation for 2 h. Proteins were quantitated with Image Quant LAS 4000mini (GE Healthcare Bio-Sciences AB, Uppsala, Sweden) and analyzed by ImageJ software version 1.4.2b (NIH, Bethesda, MD, USA). β-actin and Na+/K+ ATPase expressions were used as a loading control for total and membrane fractions, respectively.
Statistical Analysis
A t-test and one-way analysis of variance (ANOVA) were applied for statistical analysis with SPSS software version 15.0 (SPSS, Chicago, IL). Values in the figures are given as means ± SD. P< 0.05 was considered to be significant.
Results
GRK2 Was Down-Regulated in HCC Patients
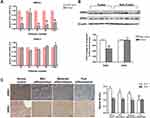
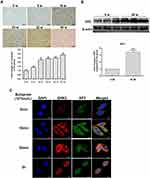
To verify the role of GRK2/3 in HCC, firstly we detected the expression of GRK2 and GRK3 in 18 pairs of tumor and adjacent noncancerous liver tissue samples from HCC patients. As shown in Figure 1A, GRK2 mRNA was down-regulated in tumor tissues compared with noncancerous liver tissues as determined by qRT-PCR. However, GRK3 mRNA had no significant change between tumor and non-tumor tissues. The results of Western blot further confirmed that GRK2 protein expression was lower in HCC tissues than in noncancerous liver tissues. Similar protein levels of GRK3 expression were detected in tumor and non-tumor tissues (Figure 1B). These results indicated that GRK2 was frequently down-regulated in HCC patients. We next examined the association between GRK2 dysregulation and HCC progression through immunohistochemical staining of paraffin-embedded HCC tumor samples with different histological grades (well differentiation, moderate differentiation, or poor differentiation) from 72 HCC patients and 17 histologically normal controls. Different HCC histological grades showed different GRK2 immunoreactivity (Figure 1C). The expression of GRK2 was significantly lower in HCC livers than in normal control livers. Furthermore, poorly differentiated HCC demonstrated lower GRK2 expression than the other HCC histological grades, as GRK2 expression increased from poorly differentiated to well-differentiated HCC. While there was no significant difference in GRK3 expression among different histological grade HCC tissues and normal liver tissues. These data collectively disclosed that GRK2 down-regulation may be associated with aggressive tumor behavior of HCC.
Decreased Expression of GRK2 During Hepatocarcinogenesis in DEN-Induced Liver Tumor Model
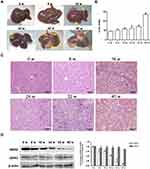
A DEN-induced liver tumor model was established to further explore the dynamic expression of GRK2 and GRK3 in vivo during liver tumorigenesis. As shown in Figure 2A, at 8 weeks after DEN injection, the liver size was obviously larger. At 16 weeks, mice liver became distinctly color changes, and there were displayed several tiny tumor nodules on the liver surface. From 24 to 40 weeks after DEN injection, a variable number of tumor nodules were present compared with 0 week, all of the tumor nodules were grayish white with an envelope, can be separated from surrounding tissues. With increasing severity of HCC, the size of the tumor nodules clearly increased. At 40 weeks after DEN injection, the tumor nodules occupied most of the liver surface. Meanwhile, the liver index reflected tumor burden in Figure 2B, the liver index significantly increased post DEN injection for 16 weeks. Histological examination showed the liver pathology of 0 week mice presented complete liver tissue structure, normal morphology and neat arrangement (Figure 2C). Similarly, the liver in 8 weeks had almost the same constructions excepted for some disorganized cell arrangement. However, significantly increased lobular inflammation and hepatocyte necrosis exhibited at 16 weeks post-DEN injection, and appeared fibrous tissue hyperplasia. At 32 weeks after DEN injection, carcinoma cells were microscopically nest-like distributed and accumulated irregularly without the normal hepatocytes constructions. The above results indicate that the mouse liver cancer model in this experiment was successfully established.
To investigate GRK2/3 expression in vivo, time-course expression of GRK2/3 was identified by Western blot. As shown in Figure 2D, compared with 0 week mice, the expression of GRK2 significantly decreased from 8 to 40 weeks after DEN injection. Nevertheless, the expression of GRK3 was observed no significant difference, as the severity of liver tumor increased. These findings revealed that decreased GRK2 expression is associated with hepatocarcinogenesis.
GRK2 Down-Regulation Was Associated with HCC Metastasis
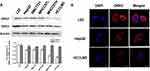
Based on the results of GRK2/3 expression in vivo, we examined the expression of GRK2/3 in human normal liver cell L-02 and five different HCC cell lines with stepwise metastatic potential (HepG2, SMC7721, MHCC97L, MHCC97H, HCCLM3) in vitro. According to the results of Western blot, compared with normal L-02 liver cells, the expression of GRK2 was gradually decreased in HCC cell lines with the increase of cell metastatic potential. Moreover, there was no evident change in GRK3 expression in each cell line (Figure 3A). Next, immunofluorescence staining was used to confirm GRK2 expression and subcellular localization. HCC cells with low metastatic potential (HepG2) and high metastatic potential (HCCLM3) were selected.18,19 As shown in Figure 3B, we found that the fluorescence intensity of GRK2 in normal hepatocytes L-02 was stronger than that in HCC cells, which were consistent with Western blot results. GRK2 was expressed predominantly in the cytoplasm and partially the cytoplasmic membrane. Therefore, combining the aforementioned data in vivo and vitro, we confirm that down-regulation of GRK2 rather than GRK3 is involved in HCC development.
Gene Silencing of GRK2 Enhanced Migration and Invasion of HCC Cells in vitro
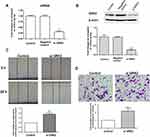
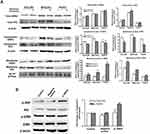
To investigate whether GRK2 is involved in the migration and invasion of HCC cells, siRNA targeting GRK2 mRNA was used to detect the effects of endogenous GRK2 expression on the migration and invasion of HCC cells. GRK2 siRNA was transfected into SMC7721 cells, which displayed relatively high endogenous expression of GRK2. The results of qRT-PCR and Western blot confirmed that GRK2 mRNA and protein expression was decreased in cells transfected with GRK2 siRNA compared with controls (Figure 4A and B). These data indicate that the transfection was succeeded. As shown in Figure 4C, wound-healing assay presented that the wound closure of SMC7721 cells was significantly increased after transfected with GRK2 siRNA compared with the control tumor cells. In addition, the results of transwell assay showed that when the GRK2 expression was reduced by GRK2 siRNA, the number of migrated cells was obviously increased (Figure 4D). The above results indicate that GRK2 silencing in HCC cells could promote the migration and invasion ability of cells. In brief, GRK2 was negatively correlated with the migration and invasion of HCC cells.
GRK2 Regulated HCC Cell Migration and Invasion via Modulation of EP2 Receptor and Downstream Akt Pathway
Considering that EP2, which is classified as GRCRs, plays an important role in cancer invasion.20 And our previous studies showed that butaprost, a selective EP2 receptor agonist, can significantly stimulate the proliferation and invasion of HCC cells.13 We next postulated that whether GRK2 regulated HCC cell migration and invasion through down-regulation of EP2. Firstly, we examined expression of EP2 in DEN-induced liver tumor model in vivo by immunohistochemical staining and Western blot. As shown in Figure 5A, EP2 staining intensity showed a clearly increasing trend in liver tissues with the progression of liver cancer after DEN injection. Moreover, the results of Western blot were consistent with immunohistochemical staining, which presented significantly increased EP2 protein expression in DEN-induced liver tumor mice (Figure 5B). These data indicated that EP2 expression was positively associated with the severity of disease.
Finally, we decided to study the putative role of the GRK2/EP2 in HCC cell migration and invasion. Butaprost, a selective EP2 agonist was used to stimulate HepG2 cells at 10−6 mol/L lasted for 2 h. The expression and subcellular localization of GRK2 and EP2 were confirmed by double-staining immunofluorescence. Immunofluorescent analysis showed that GRK2 protein was diffusely expressed predominantly in the cytoplasm of untreated HepG2 cells and co-expressed with EP2. However, expression of GRK2 was distributed in the cytoplasmic membrane of HepG2 cells after butaprost stimulation for up to 30 min (Figure 5C). These results suggest that co-expression of GRK2 and EP2 resulted in their co-localization in the cytoplasm of HCC cells. To determine whether GRK2 has a role in regulating EP2, we separated cytoplasmic and membrane proteins of HCC cells with different metastatic potential by ultracentrifugation.21 Western blot results showed that EP2 expression in cell membrane was significantly increased after butaprost stimulation in HCC cells (Figure 6A), which was in consistent with the results of immunofluorescent analysis. And upon butaprost stimulation, HCCLM3, the HCC cell line with the highest metastatic potential, exhibited significantly lower GRK2 expression and higher EP2 expression in cell membrane than HepG2 cells (the HCC cell line with relatively low metastatic potential). These findings indicate that low GRK2 expression may promote HCC cell invasion and metastasis by inhibiting EP2 desensitization. Next, we attempted to explore the signaling pathways involved in the GRK2 regulated migration and invasion of HCC cells. Since Akt and ERK signaling pathways have been demonstrated to participate in EP2-mediated signal cascades and tumor cell migration and invasion,22 we detected the expression of phosphorylated forms of ERK and Akt. As shown in Figure 6B, the Akt phosphorylation (p-Akt) was significantly increased in SMC7721 cells transfected with GRK2 siRNA compared with control cells. By contrast, the levels of ERK phosphorylation were not significantly changed in SMC7721 cells. These results suggest that interfering with expression of GRK2 may activate EP2-mediated Akt pathway, thus promote HCC cell migration and invasion.
Discussion
Worldwide, more than 90% of primary liver cancer cases are due to HCC.23 Its malignancy is high, easy to relapse and metastasis. The incidence and mortality of HCC in recent years are still rising, especially occurs in Eastern and South-Eastern Asia and Western Africa.24 However, HCC is highly resistant to traditional anticancer therapies such as radiotherapy and chemotherapy. Hence, it is still very urgent to explore the new pathological mechanism of HCC and drug target for HCC treatment.
There is mounting evidence suggests that GRKs are closely related to the occurrence and development of malignant tumors, including breast, prostate, and central nervous system cancer.25,26 GRK2 and GRK3 have recently been shown playing an important role in liver diseases. However, their clinical relevance in regard to HCC progression and metastasis has not been clarified. In combination with the current study, we found that GRK2 may play a more vital negative regulatory molecule in hepatocarcinogenesis, but not GRK3. In the present study, it is found that GRK2 was almost negative staining in patients with high malignancy, while in the well-differentiated tissues and normal control tissues, GRK2 showed obvious positive staining. The positive staining of GRK3 was not significantly changed in the tissues of HCC patients with different degrees of differentiation. Similar results were obtained from qRT-PCR and Western blot, GRK2 expression in liver tissues of HCC patients was lower than in non-tumor tissues. In addition, DEN-induced C57BL/6J mice liver cancer model was used to simulate the development of human HCC in vivo.27 We found that the expression of GRK2 gradually decreased when the degree of malignancy increased, while there was no significant change in GRK3 expression. In vitro, GRK2 expression was obviously decreased in HCCLM3 and MHCC97H cell lines with high metastatic potential than in SMC7721 and HepG2 cell lines with low metastatic potential. Taken together with clinical observations, these findings suggest that the expression of GRK2 in HCC is decreased, which may be related to the occurrence and development of liver cancer. In addition, it was confirmed that GRK2 knockdown can promote the migration and invasion of HCC cells by cell scratch assay and transwell assay.
EPs are usually coupled with G proteins to produce a second messenger, allowing extracellular signals to be introduced into cells, thereby causing a series of physiological or pathological effects.28 Low expression of EP2 is thought to significantly inhibit LN229 human glioma cell growth and migration.29 Activation of EP2 receptor can significantly encourage the invasion and migration of Huh7 cells.30 As expected, the results of our present study showed that the expression of EP2 receptor was obviously increased in DEN-induced HCC mice model, which suggested that EP2 expression was positively associated with the severity of disease. GRK2 with different subcellular localization acts different roles of intracellular signaling,31 and membranous fraction of EP2 leading to activation of its downstream signals and inducing laryngeal carcinomas progression.32 We detected the distribution changes of GRK2 and EP2 in HCC cells, and the membrane proteins were isolated from three HCC cell lines with stepwise metastatic potential through ultracentrifugation. The results of Western blot showed that membrane expression of EP2 was increased in HCC cells after stimulation with butaprost. Furthermore, HCC cell line with the highest metastatic potential exhibited significantly lower GRK2 expression and higher EP2 expression in cell membrane than HCC cell line with low metastatic potential. Since GRK2 has been discovered to be responsible for GPCRs desensitization and down-regulation of GPCRs signaling.33 Therefore, we speculate that low expression of GRK2 may promote HCC invasion and metastasis by inhibiting EP2 desensitization thus increasing its expression in the cell membranes.
Tumor metastasis constitutes one of the most significant biological characteristics of malignant tumors and the leading cause of death in clinical cancer patients.34 It has been reported that inhibition of the phosphoinositide 3-kinase (PI3K)-Akt signaling pathway may reduce the aggressiveness ability of gastric cancer cells.35 The PI3K/Akt signaling pathway also induces the expression of the epithelial-mesenchymal transition-related markers in breast cancer cells, promoting its metastasis.36 ERK signals can serve as a medium for tumor invasion and metastasis, and function by amplifying the signal.37–39 In PGE2-related malignancy, it has been reported that PGE2 stimulates SN12C renal cancer cells to promote EP2-mediated Akt signaling activation, thereby promoting the invasion of renal cancer cells.40 EP2 has been demonstrated to promote the activation of ERK signal and promote the formation of pancreatic cancer in mice.41 More interestingly, our results of Western blot showed that the Akt pathway was significantly activated, not ERK pathway after interfering with the expression of GRK2 in HCC cells by siRNA. The above results suggest that GRK2 may inhibit the invasion and metastasis of HCC by inhibiting the EP2 receptor-mediated Akt signaling pathway.
Conclusion
In summary, our data suggest that low GRK2 expression is indispensable for promoting HCC invasion and metastasis. We provide evidence that GRK2 may regulate Akt signaling mediated by EP2 receptor. Future research on the relationship between GRK2 dysregulation and liver cancer may provide a potential treatment target in invasion and metastasis of HCC.
Acknowledgments
This work was supported by grants from the National Natural Science Foundation of China (No. 81770605, 81300332), Program for Young Excellent Talents in Universities of Anhui Province (No. gxyqZD2018024), Anhui Provincial Natural Science Foundation (No. 2008085QH413). The authors acknowledge the help of the staff members of the Institute of Clinical Pharmacology, Anhui Medical University in conducting the study.
Disclosure
The authors declare that they have no conflicts of interest for this work.
References
1. Sun Y, Ma W, Yang Y, et al. Cancer nanotechnology: enhancing tumor cell response to chemotherapy for hepatocellular carcinoma therapy. Asian J Pharm Sci. 2019;14(6):581–594. doi:10.1016/j.ajps.2019.04.005
2. Bray F, Ferlay J, Soerjomataram I, et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424. doi:10.3322/caac.21492
3. Ikeda K. Recent advances in medical management of hepatocellular carcinoma. Hepatol Res. 2019;49(1):14–32. doi:10.1111/hepr.13259
4. Samji H, Yu A, Kuo M, et al. Late hepatitis B and C diagnosis in relation to disease decompensation and hepatocellular carcinoma development. J Hepatol. 2017;67(5):909–917. doi:10.1016/j.jhep.2017.06.025
5. Sun WY, Wu JJ, Peng WT, et al. The role of G protein-coupled receptor kinases in the pathology of malignant tumors. Acta Pharmacol Sin. 2018;39(11):1699–1705. doi:10.1038/s41401-018-0049-z
6. Peng WT, Sun WY, Li XR, et al. Emerging roles of G protein-coupled receptors in hepatocellular carcinoma. Int J Mol Sci. 2018;19(5):E1366. doi:10.3390/ijms19051366
7. Schumacher SM, Koch WJ. Noncanonical roles of G protein-coupled receptor kinases in cardiovascular signaling. J Cardiovasc Pharmacol. 2017;70(3):129–141. doi:10.1097/FJC.0000000000000483
8. Sang M, Hulsurkar M, Zhang X, et al. GRK3 is a direct target of CREB activation and regulates neuroendocrine differentiation of prostate cancer cells. Oncotarget. 2016;7(29):45171–45185. doi:10.18632/oncotarget.9359
9. Li R. Role of G Protein-Coupled Receptor Kinase in Signal Transduction of Hepatic Stellate Cells from Rats with Immunological Hepatic Fibrosis and the Effects of Paeoniflorin[D]. Anhui Medical University; 2010.
10. Zang S, Ma X, Wu Y, et al. PGE2 synthesis and signaling in malignant transformation and progression of human hepatocellular carcinoma. Hum Pathol. 2017;63:120–127. doi:10.1016/j.humpath.2017.02.018
11. Dietlmeier S, Ye Y, Kuhn C, et al. The prostaglandin receptor EP2 determines prognosis in EP3-negative and galectin-3-high cervical cancer cases. Sci Rep. 2020;10(1):1154. doi:10.1038/s41598-020-58095-3
12. Mizuno R, Kawada K, Sakai Y. Prostaglandin E2/ EP signaling in the tumor microenvironment of colorectal cancer. Int J Mol Sci. 2019;20(24):E6254. doi:10.3390/ijms20246254
13. Hu S, Sun W, Wei W, et al. Involvement of the prostaglandin E receptor EP2 in paeoniflorin-induced human hepatoma cell apoptosis. Anti-Cancer Drug. 2013;24(2):140–149. doi:10.1097/CAD.0b013e32835a4dac
14. Xu ZW, Yan SX, Wu HX, et al. Angiotensin II and tumor necrosis factor-α stimulate the growth, migration and invasion of BEL-7402 cells via down-regulation of GRK2 expression. Dig Liver Dis. 2019;51(2):263–274. doi:10.1016/j.dld.2018.06.007
15. Sun WY, Hu SS, Wu JJ, et al. Down-regulation of β-arrestin2 promotes tumour invasion and indicates poor prognosis of hepatocellular carcinoma. Sci Rep. 2016;6:35609. doi:10.1038/srep35609
16. Park J, Kawamoto J, Esaki N, et al. Identification of cold-inducible inner membrane proteins of the psychrotrophic bacterium, shewanella livingstonensis Ac10, by proteomic analysis. Extremophiles. 2012;16(2):227–236. doi:10.1007/s00792-011-0422-z
17. Yang X, Zhao Y, Jia X, et al. CP-25 combined with MTX/LEF ameliorates the progression of adjuvant-induced arthritis by the inhibition on GRK2 translocation. Biomed Pharmacother. 2019;110:834–843. doi:10.1016/j.biopha.2018.12.040
18. Li Y, Tang ZY, Ye SL, et al. Establishment of cell clones with different metastatic potential from the metastatic hepatocellular carcinoma cell line MHCC97. World J Gastroenterol. 2001;7(5):630–636. doi:10.3748/wjg.v7.i5.630
19. Zhang L, Jia G, Shi B, et al. PRSS8 is downregulated and suppresses tumour growth and metastases in hepatocellular carcinoma. Cell Physiol Biochem. 2016;40(3–4):757–769. doi:10.1159/000453136
20. Lian S, Xia Y, Ung TT, et al. Prostaglandin E2 stimulates urokinase-type plasminogen activator receptor via EP2 receptor-dependent signaling pathways in human AGS gastric cancer cells. Mol Carcinog. 2017;56(2):664–680. doi:10.1002/mc.22524
21. Han CC, Liu Q, Zhang Y, et al. CP-25 inhibits PGE2-induced angiogenesis by down-regulating EP4/AC/cAMP/PKA-mediated GRK2 translocation. Clin Sci (Lond). 2020;134(3):331–347. doi:10.1042/CS20191032
22. Xing W, Xiao Y, Lu X, et al. GFI1 downregulation promotes inflammation-linked metastasis of colorectal cancer. Cell Death Differ. 2017;24(5):929–943. doi:10.1038/cdd.2017.50
23. Sayan M, Yegya-Raman N, Greco SH, et al. Rethinking the role of radiation therapy in the treatment of unresectable hepatocellular carcinoma: a data driven treatment algorithm for optimizing outcomes. Front Oncol. 2019;9:345. doi:10.3389/fonc.2019.00345
24. Heimbach JK, Kulik LM, Finn RS, et al. AASLD guidelines for the treatment of hepatocellular carcinoma. Hepatology. 2018;67(1):358–380. doi:10.1002/hep.29086
25. Ma Y, Han CC, Huang Q, et al. GRK2 overexpression inhibits IGF1-induced proliferation and migration of human hepatocellular carcinoma cells by downregulating EGR1. Oncol Rep. 2016;35(5):3068–3074. doi:10.3892/or.2016.4641
26. Nogues L, Palacios-Garcia J, Reglero C, et al. G protein-coupled receptor kinases (GRKs) in tumorigenesis and cancer progression: GPCR regulators and signaling hubs. Semin Cancer Biol. 2018;48:78–90. doi:10.1016/j.semcancer.2017.04.013
27. Romualdo GR, Prata GB, Da ST, et al. Fibrosis-associated hepatocarcinogenesis revisited: establishing standard medium-term chemically-induced male and female models. PLoS One. 2018;13(9):e203879. doi:10.1371/journal.pone.0203879
28. Kim JH, Kim YD, Lee M, et al. Modified PAGE-B score predicts the risk of hepatocellular carcinoma in Asians with chronic hepatitis B on antiviral therapy. J Hepatol. 2018;69(5):1066–1073. doi:10.1016/j.jhep.2018.07.018
29. Ba MC, Ba Z, Long H, et al. LncRNA AC093818.1 accelerates gastric cancer metastasis by epigenetically promoting PDK1 expression. Cell Death Dis. 2020;11(1):64. doi:10.1038/s41419-020-2245-2
30. Cheng SY, Zhang H, Zhang M, et al. Prostaglandin E2 receptor EP2 mediates snail expression in hepatocellular carcinoma cells. Oncol Rep. 2014;31(5):2099–2106. doi:10.3892/or.2014.3074
31. Yang X, Li S, Zhao Y, et al. GRK2 mediated abnormal transduction of PGE2-EP4-cAMP-CREB signaling induces the imbalance of macrophages polarization in collagen-induced arthritis mice. Cells. 2019;8(12):1596. doi:10.3390/cells8121596
32. Mochocki M, Morawski P, Kopta R, et al. Expression of prostaglandin E2 prostanoid receptor EP2 and interleukin-1β in laryngeal carcinoma – preliminary study. Contemp Oncol (Pozn). 2015;19(2):113–119. doi:10.5114/wo.2015.51417
33. Pfleger J, Gresham K, Koch WJ. G protein-coupled receptor kinases as therapeutic targets in the heart. Nat Rev Cardiol. 2019;16(10):612–622. doi:10.1038/s41569-019-0220-3
34. Millanta F, Asproni P, Canale A, et al. COX-2, mPGES-1 and EP2 receptor immunohistochemical expression in canine and feline malignant mammary tumours. Vet Comp Oncol. 2016;14(3):270–280. doi:10.1111/vco.12096
35. Phosri S, Arieyawong A, Bunrukchai K, et al. Stimulation of adenosine A2B receptor inhibits endothelin-1-induced cardiac fibroblast proliferation and alpha-smooth muscle actin synthesis through the cAMP/Epac/PI3K/Akt-signaling pathway. Front Pharmacol. 2017;8:428. doi:10.3389/fphar.2017.00428
36. Wang Y, Liu C, Xie Z, et al. Knockdown of TRIM47 inhibits breast cancer tumorigenesis and progression through the inactivation of PI3K/Akt pathway. Chem Biol Interact. 2020;317:108960. doi:10.1016/j.cbi.2020.108960
37. Agudo-Ibanez L, Crespo P, Casar B. Analysis of Ras/ERK compartmentalization by subcellular fractionation. Methods Mol Biol. 2017;1487:151–162. doi:10.1007/978-1-4939-6424-6_11
38. Ishibashi M, Takahashi R, Tsubota A, et al. SLAMF3-mediated signaling via ERK pathway activation promotes aggressive phenotypic behaviors in multiple myeloma. Mol Cancer Res. 2020;1541–7786. doi:10.1158/1541-7786.MCR-19-0391.
39. Takayama G, Ohtani M, Minowa A, Matsuda S, Koyasu S. Class I PI3K-mediated Akt and ERK signals play a critical role in FcεRI-induced degranulation in mast cells. Int Immunol. 2013;25(4):215–220. doi:10.1093/intimm/dxs105
40. Li Z, Zhang Y, Kim WJ, et al. PGE2 promotes renal carcinoma cell invasion through activated RalA. Oncogene. 2013;32(11):1408–1415. doi:10.1038/onc.2012.161
41. Takahashi T, Uehara H, Ogawa H, et al. Inhibition of EP2/EP4 signaling abrogates IGF-1R-mediated cancer cell growth: involvement of protein kinase C-θ activation. Oncotarget. 2015;6(7):4829–4844. doi:10.18632/oncotarget.3104
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.