Back to Journals » International Journal of Nanomedicine » Volume 12
Gold nanoparticles enhance TRAIL sensitivity through Drp1-mediated apoptotic and autophagic mitochondrial fission in NSCLC cells
Authors Ke S, Zhou T, Yang P, Wang Y , Zhang P, Chen K, Ren L, Ye S
Received 2 December 2016
Accepted for publication 28 February 2017
Published 31 March 2017 Volume 2017:12 Pages 2531—2551
DOI https://doi.org/10.2147/IJN.S129274
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Lei Yang
Sunkui Ke,1 Tong Zhou,2 Peiyan Yang,3 Yange Wang,2 Peng Zhang,2 Keman Chen,2 Lei Ren,2 Shefang Ye2
1Department of Thoracic Surgery, Zhongshan Hospital of Xiamen University, 2Department of Biomaterials, College of Materials, Xiamen University, 3Department of Surgery, First Affiliated Hospital of Xiamen University, Xiamen University, Xiamen, People’s Republic of China
Abstract: Although tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) and its agonistic receptors have been identified as highly promising antitumor agents preferentially eliminating cancer cells with minimal damage, the emergence of TRAIL resistance in most cancers may contribute to therapeutic failure. Thus, there is an urgent need for new approaches to overcome TRAIL resistance. Gold nanoparticles (AuNPs) are one of the most promising nanomaterials that show immense antitumor potential via targeting various cellular and molecular processes; however, the effects of AuNPs on TRAIL sensitivity in cancer cells remain unclear. In this study, we found that AuNPs combined with TRAIL exhibited a greater potency in promoting apoptosis in non-small-cell lung cancer (NSCLC) cells compared with TRAIL alone, suggesting that AuNPs sensitize cancer cells to TRAIL. Further experiments demonstrated that the combination of TRAIL and AuNPs was more effective in causing excessive mitochondrial fragmentation in cancer cells accompanied by a dramatic increase in mitochondrial recruitment of dynamin-related protein 1 (Drp1), mitochondrial dysfunctions, and enhancement of autophagy induction. Small interfering RNA (siRNA) silencing of Drp1 or inhibition of autophagy could effectively alleviate apoptosis in cells exposed to TRAIL combined with AuNPs. In vivo studies revealed that AuNPs augmented TRAIL sensitivity in tumor-bearing mice. Our data indicated that AuNPs potentiate apoptotic response to TRAIL in NSCLC cells through Drp1-dependent mitochondrial fission, and TRAIL combined with AuNPs can be a potential chemotherapeutic strategy for the treatment of NSCLC.
Keywords: AuNPs, TRAIL, mitochondrial dynamics, Drp1, autophagy/mitophagy
Introduction
Lung cancer causes the highest rate of cancer-related mortality worldwide. Non-small-cell lung cancer (NSCLC) is by far the most common type of lung cancer, making up ~85% of all diagnosed lung cancers.1 Although intensive efforts have been devoted to developing novel combinational therapeutic options based on molecular targets for NSCLC, the outcome of patients with NSCLC remains poor due to chemoresistance.2 Tumor necrosis factor (TNF)-related apoptosis-inducing ligand (TRAIL), a member of the TNF family of ligands, is capable of initiating apoptosis by interacting with two death-inducing receptors, death receptor 4 (DR4) and death receptor 5 (DR5).3,4 TRAIL binding to its receptors leads to the assembly of death-inducing signaling complex by recruiting Fas-associated death domain and caspase-8, which in turn initiates a cascade of caspase activation events mediating apoptosis.5 Preclinical trials reported that recombinant TRAIL and its receptor agonists have been shown to preferentially eliminate cancer cells while leaving normal cells unaffected. Nevertheless, the fact that tumor cells such as NSCLC can develop resistance to TRAIL-mediated apoptosis remains a major roadblock to clinical utility.6 To maximize the efficacy of TRAIL-based treatments, other pharmacological agents that can sensitize cancer cells to TRAIL may offer a novel therapeutic strategy for the treatment of cancer.7,8
The emergence of nanotechnology provides optimistic expectations for its wide applications in the fields of biology and medicine and offers unique ways to detect and modulate a variety of cellular behaviors and processes at nanoscale.9 Recently, gold nanoparticles (AuNPs) have been shown to hold great promise for future applications because of their distinctive properties, such as small size, unique photophysical features, easy to surface modify, and favorable biocompatibility.9,10 All these properties render AuNPs as a versatile nanoplatform for emerging biomedical applications in the design of biosensors, targeted drug delivery vehicles, photothermal therapy, and diagnostic bioimaging.11 Recently, AuNPs have been extensively employed as emerging therapeutic agents for the treatment of AIDS,12 Parkinson’s disease,13 and diabetes,14 or controlling neural stem/progenitor cell renewal15 and promoting osteogenic differentiation of mesenchymal stem cells.16–18 Furthermore, AuNPs are also exploited as a novel class of antitumor agents in cancer therapy through inhibiting angiogenesis and ablating the tumor microvasculature,19 enhancing chemosensitivity of cancer cells by reversing epithelial–mesenchymal transition,20 preventing tumor growth and metastasis via abrogating growth factor signaling cascades,21 and boosting the antitumor immune response as a vaccine platform.22 These results strongly demonstrate that AuNPs may serve as self-therapeutic nanoparticles in cancer treatment.23
As highly dynamic organelles in a living cell, mitochondria undergo complementary fission and fusion, forming networks of varying size and dimensions.24 Mitochondrial balance of fusion and fission is strictly orchestrated by several large GTP-binding proteins. Fission proteins, including dynamin-related protein 1 (Drp1) and fission protein 1 (Fis1), and fusion proteins, including mitofusins 1 and 2 (Mfn1 and Mfn2) and optic atrophy 1 (Opa1), perform as the main executors in the maintenance of mitochondrial dynamics.25,26 Mitochondrial dynamics is known to be involved in various fundamental biological processes, such as mitochondrial biogenesis, maintenance of mitochondrial DNA, cell metabolism, calcium buffering, and cell survival and death,27 and abnormal mitochondrial dynamics has been linked to neurodegenerative diseases, diabetes, aging, and cancer.28,29 Mitochondrial fission is a critical event leading to mitochondrial outer membrane permeabilization, resulting in the release of cytochrome c that ultimately activates the intrinsic apoptotic pathway, while fusion confers resistance to apoptosis.30 Recent evidence highlighted the importance of mitochondrial fission in quality control and autophagic clearance of damaged mitochondria, the process referring to mitophagy.29 Since the efficiency of the mitochondria-mediated cell death can be greatly influenced by mitochondrial fission, extensive effort has been devoted to developing therapeutic approaches to reverse chemoresistance focusing on the regulation of mitochondrial dynamics.31,32 Indeed, several studies suggested that chemoresistant cancer cells show a higher mitochondrial fusion rate than their chemosensitive counterparts.33 On the other hand, chemosensitivity in cancer cells can be restored by shifting the mitochondria fusion toward fission.33,34 These findings demonstrate that targeting mitochondrial dynamics may aid in the development of novel therapeutic strategies for reversing chemoresistance.35
This study aimed to investigate the involvement of mitochondrial dynamics in chemosensitization effects of AuNPs in response to TRAIL in NSCLC cells. Herein, we showed that AuNPs enhance the apoptotic action of TRAIL by Drp1-mediated mitochondrial fragmentation, mitochondrial dysfunctions, and occurrence of autophagy. Drp1 silencing abrogated the enhancement of TRAIL sensitivity by preventing apoptotic and autophagic cell death, and these findings provide a mechanistic explanation for the combination of AuNPs and TRAIL in NSCLC treatment.
Materials and methods
Cell culture and reagents
Human NSCLC cell lines (Calu-1, A549, and H460) and BEAS-2B normal bronchial epithelial cells were purchased from the Cell Bank of Chinese Academy of Sciences (Shanghai, China) and grown in RPMI-1640 medium (Gibco, Grand Island, NY, USA), supplemented with 10% heat-inactivated fetal bovine serum and 100 U/mL penicillin and 100 μg/mL streptomycin (Invitrogen, Carlsbad, CA, USA) in a humidified atmosphere of 95% air and 5% CO2 at 37°C. Recombinant human TRAIL was purchased from PeproTech (Rocky Hill, NJ, USA). Mouse monoclonal anti-Drp1, anti-phosphorylated (p)-Drp1(Ser616), anti-caspase-3, anti-PARP, anti-LC3, anti-p62, anti-COX IV, anti-PINK1, and anti-Parkin were obtained from Santa Cruz Biotechnology (Santa Cruz, CA, USA). Mouse monoclonal anti-Fis1, anti-Mfn1, anti-Mfn2, anti-Opa1, anti-β-actin, N-acetyl-L-cysteine (NAC), and mitochondrial division inhibitor 1 (Mdivi-1) were purchased from Sigma (St Louis, MO, USA). Mn(III) tetrakis (1-methyl-4-pyridyl) porphyrinpentachloride (MnTMPyP) was obtained from Cayman Chemical (Ann Arbor, MI, USA).
Characterization of AuNPs
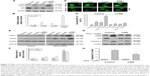
AuNPs (15, 50, and 80 nm diameters) were obtained from Sigma. The morphology and size of AuNPs were observed with a JEOL 2100F transmission electron microscope (TEM; JEOL, Tokyo, Japan) with an operating voltage of 200 kV. The ultraviolet/visible (UV/vis) absorption spectra of AuNPs were recorded with a Genesys 10s UV/vis spectrophotometer (Thermo Scientific). The size distribution and zeta potential (ζ) were conducted on a Zetasizer Nano ZS (Malvern Nano ZS, Malvern, UK). TEM showed that AuNPs with various sizes were homogenous in size and spherical in shape (Figure 1A). The size distribution by dynamic light scattering showed a higher mean diameter for all three sizes of AuNPs (Table 1), which reflected the hydrodynamic diameter of AuNPs in aqueous solution and serum proteins in the media adsorbed on the surface of AuNPs. Figure 1B shows the UV/vis spectra of AuNPs suspension with maximum red shifts with increasing particle diameter, and there was no significant difference in the absorbance peaks of AuNPs in water and culture medium (Table 1). All AuNPs showed lower negative zeta potential values in water than in culture medium, suggesting that the adsorption of serum protein could affect the surface charge of AuNPs.
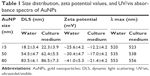  | Table 1 Size distribution, zeta potential values, and UV/vis absorbance spectra of AuNPs |
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay
Cell viability was determined using the MTT assay as previously described.36 Briefly, cells were seeded in a 96-well plate at a density of 5.0×104 cells/well for 24 h and then treated with TRAIL with or without AuNPs for 24 h. For the combination treatment, the cells were pretreated with 50 μg/mL AuNPs for 6 h, followed by incubation with 20 ng/mL TRAIL for an additional 24 h. After incubation with 0.5 mg/mL MTT (Sigma) for 4 h, the MTT-formazan crystals were subsequently dissolved in dimethyl sulfoxide. Absorbance at 570 nm was measured with a Model 680 microplate reader (Bio-Rad, Hercules, CA, USA).
Flow cytometry
Cellular uptake of AuNPs was quantified by flow cytometry, which determined the granularity of the cells based on the side scatter (SSC) light signal intensity following the protocol as reported previously.37 Briefly, Calu-1 cells were seeded (1×105 per well) on to 12-well plates 24 h prior to exposure to 50 μg/mL AuNPs with various sizes for 3, 6, and 12 h. Cells were washed three times with phosphate-buffered saline (PBS) to remove free AuNPs. Samples were then trypsinized and suspended in medium, and the uptake amount of AuNPs was analyzed with FACScan flow cytometry (BD Biosciences). Changes in cellular SSC following treatment with AuNPs reflected the uptake potential of the particles.
Apoptosis assay
After treatment with TRAIL alone or in combination with AuNPs for 24 h, apoptotic cell death of Calu-1 cells was identified using a Dead End Fluorometric TUNEL system kit according to the manufacturer’s instructions (Promega, Madison, WI, USA). Cell nuclei (blue) were counterstained using DAPI. Results were expressed as the number of terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling (TUNEL)-stained nuclei divided by the total number of DAPI-stained nuclei. For cell cycle analysis, Calu-1 cells were harvested, fixed, and incubated with 50 μg/mL propidium iodide (PI) containing 10 μg/mL ribonuclease A in the dark at 37°C for 30 min. The percentage of cells in the sub-G1 phase of the cell cycle was assessed by FACScan flow cytometry (BD Biosciences).
Analysis of mitochondrial morphology and immunocytostaining
To measure the changes in the mitochondrial morphology, Calu-1 cells were stained with 200 nM MitoTracker® Green FM (Molecular Probes, Eugene, OR, USA) for 30 min at 37°C according to the manufacturer’s instructions. After washing with PBS twice, the cells were visualized under a confocal laser scanning microscope (CLSM; Leica TCS SP5). The classification of mitochondria was performed as described previously.38 For the analysis of mitochondrial translocation of Drp1, cells were seeded into 12-well chamber slides and then fixed with 4% paraformaldehyde for 20 min at 37°C, permeabilized with 0.1% Triton X-100 for 20 min, and then blocked with 2% bovine serum albumin in PBS for 30 min at 37°C. Cells were further labeled with anti-Drp1 monoclonal antibody at a dilution of 1:100 for 20 min at 37°C, followed by staining with secondary Alexa Red-conjugated antibodies at a dilution of 1:100 for 2 h at 37°C. The cells were mounted in mounting medium containing 200 nM MitoTracker Green FM and visualized under a CLSM.
Measurement of mitochondrial membrane potential and cellular ATP content
Mitochondrial membrane potential (ΔΨm) was measured by flow cytometry using 5,5′,6′6′-tetrachloro-1,1′,3,3′-tetraethylbenzimidazolcarbocyanine iodide (JC-1) dye fluorescent probe (Molecular Probes, Eugene, OR, USA). After treatment with TRAIL alone or in combination with AuNPs for 6 h, Calu-1 cells were harvested and washed with ice-cold PBS, followed by incubation with 5 μg/mL JC-1 at 37°C for 20 min. Subsequently, cells were processed for flow cytometry analysis (FACScan; BD Biosciences) using CellQuest software. An ENLITEN®ATP Assay System Bioluminescence Detection Kit (Promega) was applied to measure cellular ATP levels as per the manufacturer’s protocol.
Measurement of mitochondrial superoxide anion
Calu-1 cells were treated with TRAIL alone and in combination with AuNPs for 6 h in the absence or presence of 20 mM NAC, 20 mM MnTMPyP, or 10 μM Mdivi-1. The mitochondrial production of superoxide anion was detected with MitoSOX Red probe (Invitrogen) as per the manufacturer’s instructions. In brief, after treatment, cells were then incubated with 2.5 mM MitoSOX Red in a serum-free culture medium for 30 min at 37°C. The fluorescence intensity was measured at an excitation wavelength of 510 nm and emission wavelength of 580 nm using flow cytometry.
Autophagy measurement
To assess autophagy, Calu-1 cells were transfected with 0.5 μg of pEGFP-LC3 using Lipofectamin 2000 (Invitrogen) as described previously.36 Forty-eight hours later, stable transfectants were selected in the presence of 500 μg/mL G418 and examined by fluorescence microscope. Autophagic flux was determined by incubating the cells with TRAIL alone and in combination with AuNPs in the absence or presence of 3-methyladenine (3-MA). The percentage of cells with more than three GFP-LC3 punctate spots was counted under a fluorescent microscope.
Small interfering RNA (siRNA) knockdown of Drp1
siRNA knockdown was performed as previously described.36 Calu-1 cells were grown until ~80% confluence in a 24-well plate, then transfected with a siRNA sequence targeting Drp1, with 20 nmol/L siRNA concentration per well, using a Lipofectamine RNAiMAX Transfection Reagent Kit (Life Technologies, Carlsbad, CA, USA) following the protocol provided by the manufacturer. A nonspecific control siRNA (GeneChem, Shanghai, China) was used as scrambled siRNA control. The knockdown efficiency of Drp1 was confirmed by immunoblotting analysis at 48 h post-transfection.
Subcellular fractionation and Western blotting analysis
Cytosolic and nuclear fractions were extracted from cells using NE-PER nuclear and cytoplasmic extraction reagents provided by Thermo Scientific. Mitochondrial fractions were prepared with a mitochondria isolation kit (Thermo Scientific). Protein concentration was quantified using a Bio-Rad DC protein assay kit (Bio-Rad). Equal amounts of protein from each subcellular fraction were separated by electrophoresis on a 12% sodium dodecyl sulfate–polyacrylamide gel electrophoresis and transferred on to PVDF membranes (Millipore, Bedford, MA, USA). After blocking with 5% nonfat milk, the membranes were probed with appropriate dilutions of specific primary antibodies overnight at 4°C. Horseradish peroxidase-conjugated anti-rabbit immunoglobulin G (IgG) and anti-mouse IgG were used as secondary antibodies. Blots were revealed with enhanced chemiluminescent reagents (Amersham Biosciences, Piscataway, NJ, USA).
In vivo antitumor activity study
All animal experiment protocols were conducted according to the National Institutes of Health Guide for the Care and Use of Laboratory Animals, and were approved by the ethics committee for the Use of Experimental Animals of Xiamen University. Calu-1 cells were harvested and resuspended in serum-free media at a density of 1×107 cells/mL. BALB/c female nude mice (7–8 weeks old) were inoculated subcutaneously with 200 μL of cell suspension (2×106) in the front flank region. Tumor growth was monitored daily until they reached a volume of ~50 mm3 before treatment. The mice were randomized into four treatment groups (n=10): PBS only, TRAIL only (10 mg/kg), 50 nm AuNP only (100 μg), and TRAIL + AuNPs (10 mg/kg and 100 μg, respectively). After randomization, mice were administrated via tail vein with 100 μg of 50 nm AuNPs, and the AuNP treatments were thrice/week for a period of 6 weeks. Subsequent TRAIL injections were also performed thrice/week 12 h after AuNP treatments. Tumor growth and body weight in each treatment were monitored daily during the experimental period. Tumor volume (V) calculation was obtained using the formula V =1/2× (length × width2). After the final treatment, the mice were sacrificed, and tumors were excised, fixed, embedded in paraffin wax, sectioned, and examined by hematoxylin and eosin (H&E) staining. For Ki67 staining, immunohistochemistry was performed according to the standard labeled streptavidin–biotin–peroxidase (LSAB kit; Dako, Glostrup, Denmark) method. TUNEL assay was performed using a Dead End Fluorometric TUNEL System (Promega). The slides were assessed by counting the number of Ki-67- or TUNEL-positive cells in five randomly chosen visual fields per sample.
Data analysis
Data are presented as mean ± standard deviation of three independent experiments. A significant difference existing between the various groups was judged by Student’s t-test or one-way analysis of variance. All experiments were carried out at least three times.
Results
AuNPs sensitize NSCLC cells to TRAIL
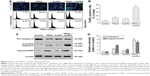
To examine the sensitivity of NSCLC cells to TRAIL, Calu-1, A549, and H460 were exposed to various concentrations of TRAIL. We showed that H460 cells and A549 cells exhibited strong resistance to TRAIL; however, Calu-1 cells were relatively more sensitive to TRAIL with an IC50 of 52.3 ng/mL (Figure 2A). To investigate whether AuNPs could modulate the sensitivity of NSCLC cells to TRAIL, Calu-1 cells were pretreated with AuNPs for 6 h, followed by incubation with 20 ng/mL TRAIL for an additional 24 h. As shown in Figure 2C, a concentration-dependent decrease in the cell viability of Calu-1 cells was observed after treatment with all sizes of AuNPs alone for 24 h; however, when cells were co-treated with TRAIL, AuNPs were found to further accelerate the reduction in the viability of Calu-1 cells. Interestingly, the TRAIL sensitivity was correlated with the sizes of AuNPs. Among the tested AuNPs with various sizes, 50 nm AuNPs showed the highest efficacy, followed by 15 and 80 nm AuNPs. These results indicated that 50 nm AuNPs exert strong effect on the TRAIL sensitivity of cancer cells. Figure S1 shows that TRAIL and AuNPs co-treatment manifested synergistic inhibitory effects in Calu-1 cells according to isobologram analysis. In contrast, TRAIL combined with AuNPs had limited effects on the cell viability in BEAS-2B cells (Figure S2). Figure 2B shows that normal bronchial epithelial cells (BEAS-2B) appeared to tolerate well under AuNPs treatment at the doses <100 μg/mL.
Next, we sought to determine whether the AuNPs-induced TRAIL sensitivity correlate with the amount of intracellular AuNPs. As shown in Figure 2D, AuNPs can be efficiently uptaken by the cells in a time-dependent manner. While the maximum uptake of AuNPs occurs for a size of 50 nm and less uptake was observed for 15 and 80 nm AuNPs, as quantified by SSC analysis (Figure 2D and E), suggesting that the particle sizes greatly affect the cellular uptake of AuNPs.39 Therefore, the therapeutic potential of TRAIL improved by 50 nm AuNPs may be attributed to its greater potential of cellular uptake. Since the 50 nm AuNPs potentiate TRAIL sensitivity in NSCLC cells with the greatest efficacy, we carried out all of our subsequent experiments with 50 nm AuNPs and further focused on defining the molecular mechanisms of their combination treatment.
AuNP-induced TRAIL sensitivity is associated with apoptotic cell death
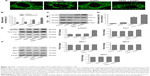
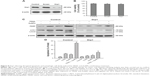
To investigate whether AuNPs overcome the TRAIL resistance due to the apoptotic induction, we analyzed apoptosis in Calu-1 cells exposed to a combination of TRAIL and AuNPs. Analysis of DNA fragmentation using TUNEL assay revealed that single TRAIL treatment slightly induced DNA fragmentation, a biochemical hallmark of apoptosis, and this response was greatly enhanced by the combination of TRAIL and AuNPs (Figure 3A). AuNPs promoted TRAIL-induced apoptosis by 3.3-fold compared with treatment with TRAIL alone (Figure 3B). We further analyzed the sub-G1 peak as an indicator of apoptosis. After different treatments, cell cycle phase distribution analysis was performed on cells using PI staining. As shown in Figure 3A, TRAIL plus AuNPs combination treatment resulted in a significant increase in the sub-G1 apoptotic population in 67.6% of cells, compared with 20.2% in the TRAIL alone group, which was notably abolished by a pan-caspase inhibitor zVAD-fmk, supporting that apoptosis was involved in cell decrease induced by co-treatment with TRAIL and AuNPs. Consistent with these findings, TRAIL and AuNPs combination treatment also caused a dramatic increase in cleaved caspase-3 and cleaved PARP, compared with TRAIL treatment alone (Figure 3C and D). Overall, these results demonstrated that AuNPs sensitize NSCLC cell lines to TRAIL by promoting apoptosis.
TRAIL combined with AuNPs facilitates Drp1-dependent mitochondrial fission
Mitochondria have been implicated in the commitment of a cell to death by triggering intrinsic and extrinsic signaling cascades during apoptosis.40 Recently, emerging evidence showed that imbalance in mitochondria dynamic and mitochondrial impairment are key mechanisms involved in apoptosis and autophagy in various cells.35 To determine whether mitochondria fragmentation facilitates the response of NSCLC cells to chemotherapeutic agent, Calu-1 cells were incubated with TRAIL alone and in combination with AuNPs, and mitochondria were stained with MitoTracker Green FM, then the changes in mitochondrial network morphology were observed under confocal microscopy. As shown in Figure 4A, the morphology of mitochondria in control cells or TRAIL-treated cells displayed an obviously filamentous pattern characteristic of mitochondria. In contrast, treatment with AuNPs significantly caused an obvious mitochondrial fragmentation. Cells with fragmented mitochondria were further increased upon co-treatment with TRAIL and AuNPs (Figure 4A and B). Since the extensive mitochondrial fragmentation occurred in cells co-treated with TRAIL and AuNPs (Figure 4A) resembling mitochondrial fission, we further examined which components of the mitochondrial division machinery contributed to mitochondrial fission by analyzing the expression of mitochondrial dynamic-related proteins. As expected, immunoblotting analysis of subcellular fractions demonstrated that combination treatment with TRAIL and AuNPs caused a remarkable recruitment of mitochondrial Drp1 in cells in comparison to treatment with either agent alone (Figure 4C and D). Moreover, an increase in phosphorylation of Drp1 at Ser616 was also observed by co-treatment with TRAIL and AuNPs (Figure 4E), an event that precedes mitochondrial fission to occur.25 However, other key proteins including Fis1, Mfn1, Mfn2, and Opa1, which involve mitochondrial division machinery, were not significantly affected by TRAIL and AuNPs combined treatment (Figure 4E and F). These findings suggested that mitochondrial recruitment of Drp1 triggered by combination treatment might shift the balance in mitochondrial dynamics favoring fission.
The size effect of AuNPs on mitochondrial fragmentation
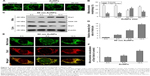
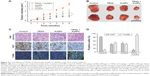
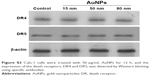
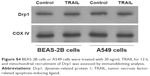
To elucidate the mechanism by which AuNPs overcome TRAIL resistance, we first evaluated the expression of the death receptors, DR4 and DR5. As shown in Figure S3, DR4 and DR5 expression was unchanged in AuNPs-treated Calu-1 cells compared with controls, suggesting that both DR4 and DR5 are not involved in AuNPs-induced TRAIL sensitivity. Next, we examined the size effect of AuNPs on the mitochondria morphology. As shown in Figure 5A and B, only limited fragmented mitochondria (<18%) were found for the control cells. However, after treatment with 50 nm AuNPs, the percentage of cells with fragmented mitochondria was significantly increased to 45%. The changes in mitochondria morphology in cells exposed to either 15 or 80 nm AuNPs were comparable to the untreated control at the same concentrations and exposure intervals. In fractionation experiments, we found that the treatment with 50 nm AuNPs promoted the gradual accumulation of Drp1 in mitochondria in a time-dependent manner (Figure 5C and D). An immunofluorescence study was carried out to further monitor the subcellular localization of Drp1 before and after 50 nm AuNPs treatment. Treatment of Calu-1 cells with 50 nm AuNPs led to an obvious colocalization of Drp1 (red) with MitoTracker Green indicative of mitochondrial recruitment of Drp1 (Figure 5E and F), thus corroborating the results from immunoblotting analysis (Figure 5C). In the same experimental setting, we observed that there was no obvious change in mitochondrial recruitment of Drp1 in either BEAS-2B cells or A549 cells exposed to TRAIL (Figure S4). These results clearly illustrated that the TRAIL sensitivity induced by AuNPs is most likely through its ability to promote mitochondrial fragmentation.
AuNPs combined with TRAIL induce mitochondrial dysfunction and reactive oxygen species (ROS) generation
To assess whether the occurrence of mitochondrial fission is correlated with mitochondrial dysfunction, we determined Δψm, total cellular ATP content, and mitochondrial ROS following treatment of Calu-1 cells with TRAIL and/or AuNPs. Δψm was estimated by using the lipophilic cationic dye JC-1. As shown in Figure 6A, TRAIL combined with AuNPs resulted in a 3.5-fold loss of Δψm compared with TRAIL-alone group. Consistently, the total ATP content, which is crucial for cellular energy metabolism, was decreased in the presence of TRAIL and AuNPs (Figure 6). Using MitoSOX Red that specifically labeled mitochondria, our findings showed that the mitochondrial ROS production was obviously increased in the cells treated with AuNPs combined with TRAIL compared with either agent alone (Figure 6A and B). To verify the specificity of MitoSOX assay for superoxide production, Calu-1 was pretreated with either a superoxide radical scavenger MnTMPyP or H2O2 scavenger NAC. As expected, both MnTMPyP and NAC effectively quenched mitochondrial ROS production in cells exposed to TRAIL combined with AuNPs (Figure 6E). Likewise, we obtained similar results in cells by inhibiting mitochondrial fission using Mdivi-1 (Figure 6E), which supported the notion that mitochondrial fragmentation constitutes a critical upstream event that mediates mitochondrial dysfunction such as Δψm loss, ATP depletion, and mitochondrial ROS generation, as observed previously.36
AuNP-induced TRAIL sensitivity is associated with autophagy activation
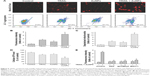
We observed a pronounced mitochondrial fragmentation in cells treated with a combination of TRAIL and AuNPs, which hints the possibility that autophagic pathway could be involved in the removal of damaged mitochondria. Western blotting analysis revealed that compared with TRAIL alone, TRAIL combined with AuNPs resulted in an increase in the ratio of LC3-II/LC3-I, which confirmed accumulation of autophagosome (Figure 7A and B). Consistently, the p62 protein levels, another feature indicative of autophagosome formation, was found to be much lower in the cells simultaneously treated with TRAIL and AuNPs than TRAIL alone. The p62 expression was inversely correlated with an increase in the conversion of LC3-I to LC3-II (Figure 7A and B). Using GFP-tagged LC3 to monitor the autophagosome formation, we found that TRAIL and AuNPs combination treatment strongly triggered the formation of GFP-LC3 puncta compared with other treatment groups, and autophagic flux was calculated by scoring the percentage of cells with GFP-LC3 puncta (Figure 7C and D). As shown in Figure 7C and D, treatment with a pharmacologic inhibitor of autophagy 3-MA significantly prevented the formation of GFP-LC3 fluorescent puncta. From these results, we concluded that TRAIL combined with AuNPs indeed triggers an enhanced autophagic response in Calu-1 cells.
Mitophagy, the predominant mechanism to selectively remove mitochondria by autophagy, often occurs to impaired mitochondria following stress or damage.41 We therefore examine the expression of PINK1 and Parkin that were required for mitophagy.42 As expected, we found that both PINK1 and Parkin were recruited into mitochondrial fractions of cells following combined treatment with TRAIL and AuNPs, demonstrating that the PINK1/Parkin pathway promotes mitophagy, allowing selective removal of mitochondrial fragmentation (Figure 7E and G). Since autophagy or mitophagy is required for the maintenance of cellular homeostasis probably by facilitating or inhibiting apoptosis,43 we further explore the relationship if any between autophagy and apoptosis in Calu-1 cells in response to TRAIL and AuNPs combination treatment. As shown in Figure 7F and H, pharmacological inhibition of autophagy by 3-MA results in a dramatic decrease in apoptotic cell death as reflected by a decrease in cleaved caspase-3. Similar results were shown in Calu-1 cells by siRNA silencing of autophagy-related genes ATG6 (Figure S5). These results suggested that the autophagic activation in response to TRAIL combined with AuNPs may serve as a pro-death factor.
Mitochondrial fission mediates AuNP-induced TRAIL sensitivity
To investigate whether mitochondrial fission is a requirement for AuNPs-induced TRAIL sensitivity, we knocked down Drp1 expression using specific siRNA to assess the role of Drp1 in apoptosis and autophagy. Immunoblotting analysis confirmed that the expression of Drp1 was abrogated in Drp1-siRNA-transfected cells, demonstrating the effectiveness of Drp1 silencing (Figure 8A). As shown in Figure 8B, transfection alone did not induce a loss of cell viability at 48 h posttransfection. Drp1 silencing significantly abrogated both apoptosis and autophagy in Calu-1 cells upon co-treatment with TRAIL and AuNPs compared with scrambled siRNA-treated cells, as evidenced by a dramatic reduction in the expression of cleaved PARP and LC3-I to LC3-II conversion (Figure 8C). Likewise, similar results were obtained in Calu-1 cells by assessment of apoptotic cell death (Figure 8D). Taken together, these findings suggested that Drp1-dependent mitochondrial fission plays a critical role in the enhancement of TRAIL sensitivity mediated by AuNPs in NSCLC cells.
Combination treatment with TRAIL and AuNPs attenuates tumor growth in vivo
To demonstrate the inhibitory effects of TRAIL combined with AuNPs on tumor growth in vivo, we performed an antitumor study using athymic nude mice bearing Calu-1 cells. We found that co-treatment with TRAIL (10 mg/kg) and AuNPs (100 μg) was more effective in suppressing tumor growth than that achieved with TRAIL or AuNPs alone over the course of 6 weeks (Figure 9A). The regression in tumor growth was further confirmed by measuring the tumor mass (Figure 9B). Note that experimental treatments do not affect the body weights of mice, suggesting that nontoxic response was observed for the dose schedule (data not shown). Moreover, the group treated with TRAIL and AuNPs showed more prominent necrosis than single-agent alone (Figure 9C, top panel). In addition, growth regression was further confirmed by analyzing the immunoexpression of general tumor biomarkers. Treatment with TRAIL and AuNPs significantly reduced the percentage of Ki67-positive proliferating cells (Figure 9C, middle panel) and increased the percentage of TUNEL-positive apoptotic cells (Figure 9C, bottom panel), as compared with single-agent alone (Figure 9D). Collectively, our data demonstrated that the AuNPs enhance the therapeutic potential of TRAIL in a xenograft model of NSCLC.
Discussion
Despite the obvious benefits of TRAIL-based treatment for NSCLC, the majority of patients eventually relapses and dies from metastatic progression due to intrinsic or acquired TRAIL resistance.7 Accordingly, tremendous efforts have been made to clarify the mechanisms responsible for the resistance to TRAIL, and development of TRAIL-based combinatorial approaches is considered to be one of the most promising therapeutic strategies for cancer treatment.8,33,34 In this study, we investigated whether AuNPs could modulate TRAIL sensitivity in NSCLC in vitro and in vivo toward the goal of providing new therapeutic options to treat refractory NSCLC. Here, we demonstrated that AuNPs enhance TRAIL-induced apoptosis in NSCLC by modulating mitochondrial dynamics, and TRAIL combined with AuNPs performs as an effective chemotherapeutic strategy for NSCLC.
The application of nanotechnology for cancer therapy has received intense attention in recent years. Owing to their remarkable properties, considerable success with the use of AuNPs as self-therapeutic nanoparticles has been achieved in the treatment of cancer.20–23 Therefore, we proposed that AuNPs could act as a potent sensitizer for TRAIL-induced apoptosis. Previous studies indicated that the size of AuNPs is one of the most predominant properties affecting cellular uptake and other bioreactivities.39 To optimize the features of AuNPs for antitumor study, AuNPs with representative diameters around 15, 50, and 80 nm were used. We compared the effects of AuNPs alone and in combination with TRAIL on the viability of several well-established NSCLC cells in vitro and found that NSCLC cells exhibit more sensitivity toward TRAIL when combined with 50 nm AuNPs as compared with either 15 or 80 nm AuNPs, suggesting that the size of AuNPs could be an important factor affecting sensitivity to TRAIL (Figure 2A–C). Recent research shows that internalized AuNPs may alter intracellular processes, which is greatly dependent upon particle size. In line with previous findings,10,44 our results revealed that AuNPs with a size of ~50 nm show the best efficiency of cellular uptake (Figure 2D and E). Following a close interaction between AuNPs and various intracellular compartments including organelles and protein kinases, AuNPs may interfere with certain cellular signaling pathways, thus leading to the desired antiproliferative effect in various cancer cells. The efficient apoptotic induction by a combination of TRAIL with other classical agents has been critical consideration in cancer therapy.7,33,34 Consistent with these results, our study demonstrated that the major mechanism by which AuNPs sensitize the cells to TRAIL might be through driving TRAIL-resistant cells to undergo apoptosis (Figure 3A–D).
Mitochondria have been shown to constantly undergo remodeling via fusion and fission, and the well-orchestrated mitochondrial dynamic processes are controlled by a family of GTPases, some of which mediate fission (Drp1), while some favor mitochondrial fusion (Mfn1, Mfn2, and Opa1).25,26 The role of mitochondrial fission in the regulation of apoptosis has long been recognized, and most reports have suggested that the occurrence of mitochondrial fragmentation is an upstream event that correlated with mitochondrial dysfunction in early stage, leading to the loss of Δψm, cytochrome c release, and activation of caspase cascades during apoptotic process.30 Furthermore, accumulating evidence has further demonstrated that mitochondria dynamics are involved in the regulation of chemoresistance.31,32 In this study, our findings showed that the 50 nm AuNPs exhibited the most potency in promoting mitochondrial fragmentation by recruiting Drp1 to mitochondria (Figures 4 and 5), which correlated well with its higher abundance of cell uptake compared with other sizes (Figure 2D and E). Previous study demonstrated that AuNP shows preferential binding of cysteine/lysine-rich proteins,19 which may lead to Drp1 phosphorylation. These results were in line with previous findings showing an increase in the p38 phosphorylation level upon the treatment of AuNPs in mesenchymal stem cells.9
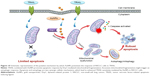
In addition, when cells were simultaneously treated with TRAIL and AuNPs, a more extensive mitochondrial fragmentation was observed (Figure 4A), followed by the loss of Δψm, depletion of ATP content, and an increase in mitochondrial ROS production compared with TRAIL alone (Figure 6A–E). In agreement with previous reports,45 our findings suggested the involvement of mitochondrial fission and subsequent mitochondrial dysfunction during apoptosis. Mitochondrial fission may facilitate the segregation of mitochondria from the networks and is emerging as a critical step that selectively removes defective mitochondria by autophagy.46 For instance, Drp1 silencing by siRNA or Drp1 inhibition by Mdivi-1 preserves reticular mitochondrial networks and inhibited autophagy.47 When considering that pretreatment with zVAD-fmk did not totally block apoptotic cell death in response to TRAIL and AuNPs co-treatment as observed in Figure 3A, it is reasonable to speculate that autophagic cell death could also favor the reduction in cell viability induced by a combination of TRAIL and AuNPs. As expected, TRAIL combined with AuNPs led to a more effective autophagy induction in Calu-1 cells compared with either agent alone. Furthermore, our results further demonstrated that Calu-1 cells treated with TRAIL and AuNPs exhibited an enhanced accumulation of PINK1 and Parkin in mitochondria (Figure 7E and G), thus indicating that mitophagy may participate in the selective autophagic degradation of mitochondrial fragmentation. At present, the functional role of autophagy in cellular homeostasis remains controversial, and an extensive literature documents that autophagy appears to be able to function in both a pro-survival and a pro-death manner.48 Previously, autophagy was found to play a protective role in the acquisition of cisplatin resistance of ovarian cancer cells.49 Conversely, autophagy induction was also shown to promote cell death rather than cellular survival in response to external stress or some anticancer agents,50,51 suggesting that autophagy performs as the main cytotoxic mechanism by promoting apoptotic cell death. Thus, it would seem that the role of autophagy may depend on the type of cancer cell and drug. To confirm that mitochondrial fission and autophagy affect AuNPs-induced TRAIL sensitivity in NSCLC cells, a specific autophagy inhibitor or siRNA targeted to Drp1 was employed. Our results showed that either Drp1 silencing by siRNA or inhibition of autophagy by 3-MA could effectively alleviate apoptotic and autophagic cell death stimulated by the combination treatment (Figures 7 and 8). These findings suggested that mitochondrial fission and subsequent autophagy serve a pro-death role in Calu-1 cells upon co-treatment with TRAIL and AuNPs. Similar results using either ABT737, an inhibitor of Bcl-2,33 or fullerene C60 to enhance chemosensitization in cancer have been previously reported.52,53 Thus, we predicted that apoptosis and autophagy are two synergic cellular mechanisms, which simultaneously regulate TRAIL sensitivity by Drp1-dependent mitochondrial fission when cells were exposed to co-treatment with TRAIL and AuNPs (Figure 10).
Conclusion
Taken together, our findings demonstrated that AuNPs exhibit potent activity in increasing the sensitivity of NSCLC cells to TRAIL-induced apoptosis both in vitro and in vivo by promoting excessive mitochondrial fragmentation with a concomitant increase in Drp1 mitochondrial translocation, dysfunction in mitochondria, and the occurrence of autophagy. These findings are of potential clinical relevance and provide mechanistic insights into the development of combination therapies using AuNPs to prevent resistance to TRAIL in NSCLC and suggested that targeting mitochondrial dynamics may present an opportunity to regulate TRAIL sensitivity in the future treatment of NSCLC.
Acknowledgment
This study was financially supported by the National Natural Science Foundation of China (31271071, 31371012, and U1505228) and the Medical Scientific Innovation Program of Fujian (2014-CXB-39).
Disclosure
The authors report no conflicts of interest in this work.
References
Tan WL, Jain A, Takano A, et al. Novel therapeutic targets on the horizon for lung cancer. Lancet Oncol. 2016;17(8):e347–e362. | ||
Joshi P, Jeon YJ, Laganà A, et al. MicroRNA-148a reduces tumorigenesis and increases TRAIL-induced apoptosis in NSCLC. Proc Natl Acad Sci U S A. 2015;112(28):8650–8655. | ||
Dimberg LY, Anderson CK, Camidge R, et al. On the TRAIL to successful cancer therapy: predicting and counteracting resistance against TRAIL-based therapeutics. Oncogene. 2013;32(11):1341–1350. | ||
Hellwig CT, Rehm M. TRAIL signaling and synergy mechanisms used in TRAIL-based combination therapies. Mol Cancer Ther. 2012;11(1):3–13. | ||
Huang K, Zhang J, O’Neill KL, et al. Cleavage by Caspase 8 and mitochondrial membrane association activate the BH3-only protein bid during TRAIL-induced apoptosis. J Biol Chem. 2016;291(22):11843–11851. | ||
O’Leary L, van der Sloot AM, Reis CR, et al. Decoy receptors block TRAIL sensitivity at a supracellular level: the role of stromal cells in controlling tumour TRAIL sensitivity. Oncogene. 2016;35(10):1261–1270. | ||
Azijli K, Weyhenmeyer B, Peters GJ, de Jong S, Kruyt FA. Non-canonical kinase signaling by the death ligand TRAIL in cancer cells: discord in the death receptor family. Cell Death Differ. 2013;20(7):858–868. | ||
Trivedi R, Mishra DP. Trailing TRAIL resistance: novel targets for TRAIL sensitization in cancer cells. Front Oncol. 2015;5:69. | ||
Yi C, Liu D, Fong CC, Zhang J, Yang M. Gold nanoparticles promote osteogenic differentiation of mesenchymal stem cells through p38 MAPK pathway. ACS Nano. 2010;4(11):6439–6448. | ||
Ma X, Wu Y, Jin S, et al. Gold nanoparticles induce autophagosome accumulation through size-dependent nanoparticle uptake and lysosome impairment. ACS Nano. 2011;5(11):8629–8639. | ||
Yang X, Yang M, Pang B, Vara M, Xia Y. Gold nanomaterials at work in biomedicine. Chem Rev. 2015;115(19):10410–10488. | ||
Muthu MS, Singh S. Targeted nanomedicines: effective treatment modalities for cancer, AIDS and brain disorders. Nanomedicine (Lond). 2009;4(1):105–118. | ||
Alvarez YD, Fauerbach JA, Pellegrotti JV, Jovin TM, Jares-Erijman EA, Stefani FD. Influence of gold nanoparticles on the kinetics of α-synuclein aggregation. Nano Lett. 2013;13(12):6156–6163. | ||
Selim ME, Abd-Elhakim YM, Al-Ayadhi LY. Pancreatic response to gold nanoparticles includes decrease of oxidative stress and inflammation in autistic diabetic model. Cell Physiol Biochem. 2015;35(2):586–600. | ||
Baranes K, Shevach M, Shefi O, Dvir T. Gold nanoparticle-decorated Scaffolds promote neuronal differentiation and maturation. Nano Lett. 2016;16(5):2916–2120. | ||
Li JJ, Kawazoe N, Chen G. Gold nanoparticles with different charge and moiety induce differential cell response on mesenchymal stem cell osteogenesis. Biomaterials. 2015;54:226–236. | ||
Li J, Li JJ, Zhang J, Wang X, Kawazoe N, Chen G. Gold nanoparticle size and shape influence on osteogenesis of mesenchymal stem cells. Nanoscale. 2016;8(15):7992–8007. | ||
Zhang D, Liu D, Zhang J, Fong C, Yang M. Gold nanoparticles stimulate differentiation and mineralization of primary osteoblasts through the ERK/MAPK signaling pathway. Mater Sci Eng C Mater Biol. 2014;42:70–77. | ||
Arvizo RR, Saha S, Wang E, Robertson JD, Bhattacharya R, Mukherjee P. Inhibition of tumor growth and metastasis by a self-therapeutic nanoparticle. Proc Natl Acad Sci U S A. 2013;110(17):6700–6705. | ||
Xiong X, Arvizo RR, Saha S, et al. Sensitization of ovarian cancer cells to cisplatin by gold nanoparticles. Oncotarget. 2014;5(15):6453–6465. | ||
Arvizo RR, Rana S, Miranda OR, Bhattacharya R, Rotello VM, Mukherjee P. Mechanism of anti-angiogenic property of gold nanoparticles: role of nanoparticle size and surface charge. Nanomedicine. 2011;7(5):580–587. | ||
Niikura K, Matsunaga T, Suzuki T, et al. Gold nanoparticles as a vaccine platform: influence of size and shape on immunological responses in vitro and in vivo. ACS Nano. 2013;7(5):3926–3938. | ||
Misra R, Acharya S, Sahoo SK. Cancer nanotechnology: application of nanotechnology in cancer therapy. Drug Discov Today. 2010;15(19–20):842–850. | ||
Yin F, Cadenas E. Mitochondria: the cellular hub of the dynamic coordinated network. Antioxid Redox Signal. 2015;22(12):961–964. | ||
Losón OC, Song Z, Chen H, Chan DC. Fis1, Mff, MiD49, and MiD51 mediate Drp1 recruitment in mitochondrial fission. Mol Biol Cell. 2013;24(5):659–667. | ||
Palmer CS, Elgass KD, Parton RG, Osellam LD, Stojanovski D, Ryan MT. Adaptor proteins MiD49 and MiD51 can act independently of Mff and Fis1 in Drp1 recruitment and are specific for mitochondrial fission. J Biol Chem. 2013;288(38):27584–27593. | ||
Liesa M, Palacín M, Zorzano A. Mitochondrial dynamics in mammalian health and disease. Physiol Rev. 2009;89(3):799–845. | ||
Cho DH, Nakamura T, Lipton SA. Mitochondrial dynamics in cell death and neurodegeneration. Cell Mol Life Sci. 2010;67(20):3435–3447. | ||
Stephan J, Franke J, Ehrenhofer-Murray AE. Chemical genetic screen in fission yeast reveals roles for vacuolar acidification, mitochondrial fission, and cellular GMP levels in lifespan extension. Aging Cell. 2013;12(4):574–583. | ||
Youle RJ. Morphology of mitochondria during apoptosis: worms-to-beetles in worms. Dev Cell. 2005;8(3):298–299. | ||
Kong B, Tsuyoshi H, Orisaka M, Shieh DB, Yoshida Y, Tsang BK. Mitochondrial dynamics regulating chemoresistance in gynecological cancers. Ann N Y Acad Sci. 2015;1350:1–16. | ||
Zhao J, Zhang J, Yu M, et al. Mitochondrial dynamics regulates migration and invasion of breast cancer cells. Oncogene. 2013;32(40):4814–4824. | ||
Fan Z, Yu H, Cui N, et al. ABT737 enhances cholangiocarcinoma sensitivity to cisplatin through regulation of mitochondrial dynamics. Exp Cell Res. 2015;335(1):68–81. | ||
Farrand L, Byun S, Kim JY, et al. Piceatannol enhances cisplatin sensitivity in ovarian cancer via modulation of p53, X-linked inhibitor of apoptosis protein, XIAP, and mitochondrial fission. J Biol Chem. 2013;228(33):23740–23750. | ||
Lennon FE, Salgia R. Mitochondrial dynamics: biology and therapy in lung cancer. Expert Opin Investing Drugs. 2014;23(5):675–692. | ||
Ye S, Yang P, Cheng K, et al. Drp1-dependent mitochondrial fission mediates toxicity of positively charged graphene in microglia. ACS Biomater Sci Eng. 2016;2(5):722–733. | ||
Toduka Y, Toyooka T, Ibuki Y. Flow cytometric evaluation of nanoparticles using side-scattered light and reactive oxygen species-mediated fluorescence-correlation with genotoxicity. Environ Sci Technol. 2012;46(14):7629–7636. | ||
Jahani-Asl A, Pilon-Larose K, Xu W, et al. The mitochondrial inner membrane GTPase, optic atrophy 1, Opa1), restores mitochondrial morphology and promotes neuronal survival following excitotoxicity. J Biol Chem. 2011;286(6):4772–4782. | ||
Dykman LA, Khlebtsov NG. Uptake of engineered gold nanoparticles into mammalian cells. Chem Rev. 2014;114(2):1258–1288. | ||
Kasahara A, Scorrano L. Mitochondria: from cell death executioners to regulators of cell differentiation. Trends Cell Biol. 2014;24(12):761–770. | ||
Durcan TM, Fon EA. The three ‘P’s of mitophagy: PARKIN, PINK1, and post-translational modifications. Genes Dev. 2015;29(10):989–999. | ||
Chen Y, Dorn GW. PINK1-phosphorylated mitofusin 2 is a Parkin receptor for culling damaged mitochondria. Science. 2013;340(6131):471–475. | ||
Goodall ML, Fitzwalter BE, Zahedi S, et al. The autophagy machinery controls cell death switching between apoptosis and necroptosis. Dev Cell. 2016;37(4):337–349. | ||
Wang SH, Lee CW, Chiou A, Wei PK. Size-dependent endocytosis of gold nanoparticles studied by three-dimensional mapping of plasmonic scattering images. J Nanobiotechnology. 2010;8:33. | ||
Brooks C, Cho SG, Wang CY, Yang T, Dong Z. Fragmented mitochondria are sensitized to Bax insertion and activation during apoptosis. Am J Physiol Cell Physiol. 2011;300(3):C447–C455. | ||
Ordureau A, Heo JM, Duda DM, et al. Defining roles of PARKIN and ubiquitin phosphorylation by PINK1 in mitochondrial quality control using a ubiquitin replacement strategy. Proc Natl Acad Sci U S A. 2015;112(21):6637–6642. | ||
Stewart JB, Chinnery PF. The dynamics of mitochondrial DNA heteroplasmy: implications for human health and disease. Nat Rev Genet. 2015;16(9):530–542. | ||
Mariño G, Niso-Santano M, Baehrecke EH, Kroemer G. Self-consumption: the interplay of autophagy and apoptosis. Nat Rev Mol Cell Biol. 2014;15(2):81–94. | ||
Wang J, Wu GS. Role of autophagy in cisplatin resistance in ovarian cancer cells. J Biol Chem. 2014;289(24):17163–17173. | ||
Kondo Y, Kanzawa T, Sawaya R, Kondo S. The role of autophagy in cancer development and response to therapy. Nat Rev Cancer. 2005;5(9):726–734. | ||
Gargini R, García-Escudero V, Izquierdo M. Therapy mediated by mitophagy abrogates tumor progression. Autophagy. 2011;7(5):466–476. | ||
Wei P, Zhang L, Lu Y, Man N, Wen L. C60(Nd) nanoparticles enhance chemotherapeutic susceptibility of cancer cells by modulation of autophagy. Nanotechnology. 2010;21(49):495101. | ||
Zhang Q, Yang W, Man N, et al. Autophagy-mediated chemosensitization in cancer cells by fullerene C60 nanocrystal. Autophagy. 2009;5(8):1107–1117. |
Supplementary materials
Methods
Small interfering RNA (siRNA) silencing of ATG6
Calu-1 cells were grown until ~80% confluence in a 24-well plate and washed once with phosphate-buffered saline (PBS) and replaced with 500 μL of fresh growth medium. Then cells were transfected with an siRNA oligonucleotide against ATG6 and scrambled siRNA (Dharmacon, Lafayette, CO, USA), with 20 nmol/L siRNA concentration per well, using a Lipofectamine RNAiMAX Transfection Reagent Kit (Life Technologies, Carlsbad, CA, USA) following the protocol provided by the manufacturer. The knockdown efficiency of ATG6 was confirmed by immunoblotting analysis at 48 h post-transfection.
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.