Back to Journals » International Journal of Nanomedicine » Volume 12
Gentamicin coating of nanotubular anodized titanium implant reduces implant-related osteomyelitis and enhances bone biocompatibility in rabbits
Authors Liu D, He C, Liu Z, Xu W
Received 15 March 2017
Accepted for publication 4 July 2017
Published 31 July 2017 Volume 2017:12 Pages 5461—5471
DOI https://doi.org/10.2147/IJN.S137137
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Lei Yang
Denghui Liu,1,* Chongru He,2,* Zhongtang Liu,2 Weidong Xu2
1Department of Orthopedics, the 113 Military Hospital, Ningbo, 2Department of Orthopedics, Changhai Hospital Affiliated to the Second Military Medical University, Shanghai, People’s Republic of China
*These authors contributed equally to this work
Abstract: Titanium and titanium alloy are widely used as orthopedic implants for their favorable mechanical properties and satisfactory biocompatibility. The aim of the present study was to investigate the antibacterial effect and bone cell biocompatibility of a novel implant made with nanotubular anodized titanium coated with gentamicin (NTATi-G) through in vivo study in rabbits. The animals were divided into four groups, each receiving different kinds of implants, that is, NTATi-G, titanium coated with gentamicin (Ti-G), nanotubular anodized titanium uncoated with gentamicin (NTATi) and titanium uncoated with gentamicin (Ti). The results showed that NTATi-G implant prevented implant-related osteomyelitis and enhanced bone biocompatibility in vivo. Moreover, the body temperature of rabbits in NTATi-G and Ti-G groups was lower than those in Ti groups, while the weight of rabbits in NTATi-G and Ti-G groups was heavier than those in NTATi and Ti groups, respectively. White blood cell counts in NTATi-G group were lower than NTATi and Ti groups. Features of myelitis were observed by X-ray films in the NTATi and Ti groups, but not in the NTATi-G and Ti-G groups. The radiographic scores, which assessed pathology and histopathology in bone tissues, were significantly lower in the NTATi-G and Ti-G groups than those in the NTATi and Ti groups, respectively (P<0.05). Meanwhile, explants and bone tissue culture demonstrated significantly less bacterial growth in the NTATi-G and Ti-G groups than in the NTATi and Ti groups, respectively (P<0.01). The bone volume in NTATi-G group was greater than Ti-G group, and little bone formation was seen in NTATi and Ti groups.
Keywords: nanotubular anodized titanium, gentamicin, titanium implant, infection, antibiotic
Corrigendum for this paper has been published
Introduction
Osteoarthritis (OA) is one of the most common causes of chronic joint pain and functional disability, which has become a major public health issue globally.1,2 Since the first report of total knee arthroplasty (TKA) in the 1960s,3 TKA and total hip arthroplasty (THA) have been demonstrated to be the most reliable surgical procedures to relieve pain and improve rehabilitation in patients with serious OA. Infection and aseptic loosening are the two major causes of surgical failure, with reported incidences of 25.2% and 16.1%, respectively, in the US, and 38% and 33%, respectively, in South Korea.4,5 On the other hand, poor biocompatibility of the prosthesis with surrounding bone tissues or stress shielding is the main culprit for aseptic loosening, which may subsequently lead to bacterial contamination and infection. Implant removal and revision are the major therapeutic methods for patients suffering from infection or aseptic loosening after primary TKA or THA. The longevity after revision is shortened than that after primary surgeries.6 Meanwhile, orthopedic surgical-site infection prolongs hospital stay by a median of 2 weeks, doubles readmission rate and triples hospital care costs.7 Therefore, it is important to prevent infection and improve biocompatibility of prosthesis during primary TKA and THA.
Various strategies have been developed to minimize the risk of infection in prosthetic joint surgery, such as ultraclean operation rooms and standardized surgical techniques, and prosthetic devices with improved designs are adopted to shorten operation time.8–11 However, infection remains to be prevalent with adverse impacts on the prognosis of patients undergoing joint replacement surgeries. Systemic antibiotic treatment has been the mainstay strategy to prevent and cure infections despite its various drawbacks such as poor penetration into the surgical site due to ischemic and necrotic nature of the posttraumatic and postoperative tissue, and adverse effects such as liver- and nephrotoxicity.12,13 Meanwhile, topical antibiotic administration by alternative local delivery may be a potential approach to maintain high local concentration without systemic toxicity.14 Bacterial adhesion plays a critical role in biofilm formation at the implantation site, which may subsequently lead to local infection. Various antibiotic-eluting devices and materials have been reported in joint arthroplasty, such as nondegradable materials like spacer beads and polymethylmethacrylate bone cements, titanium implants,15–17 biodegradable materials such as poly-(lactide-co-glycolide) copolymers,18–20 polycaprolactone,21,22 polyanhydrides,23,24 polyhydroxybutyrate-co-hydroxyvalerate25,26 and polyhydroxyalkanoates27 and natural polymers such as collagen28,29 and chitosan.30,31
Titanium and titanium alloys are widely used as orthopedic implants due to their ideal mechanical properties and satisfactory biocompatibility. The adoption of nanotechnology has promoted the development of novel orthopedic implant materials with ideal properties, such as better cytocompatibility and the ability to be used as specific drug delivery platforms.32 In vitro studies on nanotubular anodized titanium uncoated with gentamicin (NTATi) were performed to examine its biocompatibility and antibacterial effects using physical drug-loading method, and results indicated that NTATi surface could improve osteogenic activity, and gentamicin coating could lead to significantly improved antibacterial ability. To prolong the duration of continuous antibiotic delivery from NTATi, simulated body fluid (SBF) was used to mix the drug molecules to facilitate their precipitation with calcium phosphate crystals on the surface of NTATi. Results showed that such coprecipitation method could promote continuous drug release for up to 3 weeks.32 To our knowledge, no in vivo study has been performed previously to demonstrate the cytocompatibility properties of NTATi and the effect of NTATi loaded with antibiotics on the prevention of implantation-site infection. The present study aimed to investigate using a coprecipitation drug-loading approach the effects of nanotubular anodized titanium coated with gentamicin (NTATi-G) on infection prevention and bone cell biocompatibility in a rabbit model with Staphylococcus aureus inoculation in the tibial metaphysis.
Materials and methods
Preparation of implant materials
Two implant materials, pure titanium and NTATi, were used in this study. All implant materials were provided by Shanghai Institute of Ceramics, Chinese Academy of Sciences. Pure titanium uncoated with gentamicin (Ti) of 99.5% purity (0.25×0.25 cm) was sonicated with acetone to remove surface oil pollution, and the nanotube arrays were prepared by an electrochemical anodization method. The anodization process was performed employing a two-electrode system; pure Ti served as the anode, and Pt sheet served as the cathode. NH4F (0.09 M) in ethylene glycol solution containing 10 vol% water was used as the electrolyte solution.33 After anodization for 30 min at a constant voltage of 60 V, an NTATi was obtained with a length of 1.05 μm (Figure S1B), an inner diameter of 125 nm and an outside diameter of 170 nm (Figure S1A). An energy dispersive spectrometer attached to the scanning electron microscopy system was used to determine the surface chemistry of the nanotubular anodized titanium. The energy dispersive spectrum of NTATi samples revealed the existence of Ti, O and Na; atom% of Ti:O was about 1:2 (Figure S1C). The characterization of the morphology and chemical composition of the anodized titanium are presented in Figure S1. Gentamicin sulfate (Sangon Biotech A100304-0001) of 10 mg/mL was loaded on implant materials by coprecipitation approach as previously described.32 After anodization, NaOH soaking and heat treatment of the implant materials, the samples were soaked into 1.5× SBF mixed with gentamicin sulfate (10 mg/mL) and incubated for 3 days.32
Preparation of bacteria
S. aureus subsp. aureus Rosenbach (ATCC 25923; American Type Culture Collection, Manassas, VA, USA) was used in this study. The strain is sensitive to gentamicin, erythromycin, ampicillin and cephalexin.
S. aureus was cultured overnight in tryptic soy broth (TSB) (9 mL, caseinpepton–soybean flour–pepton solution; Oxoid Ltd., Basingstoke, UK). The suspension of 100 μL was then transferred into 3-mL sterile tubes and incubated for 3 hours in 37°C incubator to obtain log-phase growth. After incubation, these tubes were centrifuged for 10 min at 3,000 rpm, and the supernatant was disposed. The bacterial sediment was washed three times with phosphate-buffered saline (PBS) and resuspended in PBS. The concentration of the bacteria was adjusted to 3×109 colony-forming units (CFU)/mL with TSB according to the McFarland method34 for the use in the following procedures.
Animals and surgical procedures
All animal experiments were approved by the Institutional Animal Care and Use Committee of the Second Military Medical University. All operations in this study were performed according to the Guide for Care and Use of Laboratory Animals published by the National Institutes of Health of the US.
A total of 36 healthy New Zealand White rabbits with body weight ranging between 2.2 and 2.8 kg were selected for the present study. Each rabbit was kept in separate cage and fed with commercial pelleted diet.
After anesthesia with mainline pentobarbital (30 mg/kg) induction and sterilization, an incision was made at the proximal tibia of the left hind leg. A 2-mm soteotomy was introduced into the intramedullary cavity after a 3- to 5-mm bone slot was made. Bone marrow (1–1.5 mL) was aspirated to obtain sufficient space to insert the implant material. S. aureus (0.1 mL PBS with 3×109 CFU/mL) was then injected into the intramedullary cavity, and bone wax was used to close the bone slot. The fascia and skin were closed with suture after soft tissues were irrigated with entoiodine.
Animals were divided into the following groups, with each group having eight animals:
Group I NTATi-G +3×109 CFU/mL S. aureus 0.1 mL |
Measurements of body temperature, weight and white blood cell (WBC)
Body temperature, body weight and WBC were measured on days 0, 3, 7, 14, 21, 28, 35 and 42 postoperatively. Digital thermometer (Terumo, Zhejiang, People’s Republic of China) and precision scale (TCS, Shanghai, People’s Republic of China) were used to measure the body temperature and weight, respectively. Blood was drawn from the ear veins, and WBC count was measured. Operated rabbits were inspected for clinical signs of infection (appearance of the wound, status of the soft tissues at the entry site of implants, and presence of joint effusion or loss of mobility).
Radiographic evaluation
Anterior–posterior- and lateral-view X-rays of the operative limbs were taken on days 0, 7, 14, 21, 28, 35 and 42 after surgery, using a Mobilett Plus X-ray unit (Siemens AG, Munich, Germany) and digital films (DLR Cassette, Digiscan 2H/2C; Siemens).
Three senior radiologists blinded of the grouping status independently evaluated radiographs taken on days 0, 21 and 42, and assessed the severity of infection according to a modified scoring criteria by An and Friedman:35 1) general impression, 2) periosteal reaction, 3) soft tissue swelling, 4) osteolysis, 5) deformity, 6) sequestrum formation and 7) spontaneous fracture. Criteria 1–5 were judged as 0 (absent), 1 (mild), 2 (moderate) or 3 (severe), whereas criteria 6 and 7 were judged with 0 (absent) or 1 (present). The maximum score was 17.
Sacrifice of rabbits
Rabbits were sacrificed 6 weeks postoperatively through air injection. The proximal end of tibias containing implant materials was harvested and weighed using a precision scale under sterile conditions, which was subsequently used for microbiological and histological evaluations.
Microbiological evaluation
One tibia with implants was randomly chosen from animals in each group to assess the microbiological status. The implant materials were explanted and rolled over in the agar (Columbia® full blood; Becton Dickinson Microbiology Europe, Meylan Cedex, France) before being placed into the 2.5-mL sterile TSB. Both agar plates were incubated with TSB for 24 h in the 37°C incubator. Colonies of bacteria formed on the agar plates were counted, and bacterial growth in TSB was evaluated (cloudy appearance represents positive growth, while clear appearance represents no growth) after 24 h of incubation.
Tibial bone tissues were frozen with liquid nitrogen and pulverized with a bone mill (Tissuelyser-48; Jingxin Tech, Shanghai, People’s Republic of China) under sterile conditions. Powder of bone tissue (500 mg) was added into sterile PBS (6 mL) and agitated for 2 min by vortex mixing. The suspension was centrifuged for 10 s at 10,000 rpm, and supernatant of 100 μL was withdrawn and serially diluted to 10-fold. The supernatants were plated in triplicate on agar and incubated at 37°C for 24 h. The CFU on agar was counted, and the ratio between bacterial level and sample weight (CFU/g bone tissue) was calculated.
Histological and histopathological evaluation
All the remaining tibial tissues were split into two parts for histological and histopathological evaluations, respectively.
The samples were fixed for 24 h in 4% formaldehyde, dehydrated in alcohol solution of different concentration gradient, transparentized in xylene and embedded in methylmethacrylate (Technovit 9100; Heraeus, Hanau, Germany) for histological evaluation. Longitudinal sections perpendicular to the implants were cut at 300-μm thickness by a Leica SM 2500S microtome (Leica, Wetzlar, Germany) with a 40° stainless steel knife before toluidine blue staining. Stained slices were then used for histological evaluation according to the criteria described by Petty et al:44 1) abscess formation, 2) sequestrum formation, 3) enlargement of corticalis, 4) destruction of corticalis and 5) general impression. Parameters 1–4 were judged as absent (0) or present (1), while parameter 5 was judged as absent (0), mild (1) or severe (2). Scoring was done by three independent observers in a blinded manner with the maximum score of 6.
Samples taken for histopathology were fixed for 24 h with 4% formaldehyde and decalcified in EDTA for 28 days.36 The bone tissues were then embedded in paraffin and cut into 5-μm longitudinal sections with a microtome (CUT 6062; SLEE Medical, Mainz, Germany). The sections were then stained with hematoxylin and eosin and evaluated for histopathological changes according to the scoring method described by Smeltzer et al.37 The main factors taken into account were intraosseous chronic inflammation (ICI), intraosseous acute inflammation (IAI), periosteal inflammation (PI) and bone necrosis (BN).37 The criteria for histological parameters and scoring system are shown in detail in the research by Smeltzer et al.37
Statistical analysis
Statistical analysis was conducted by SPSS 16.0 software (SPSS for Windows, version 16.0; SPSS Inc., Chicago, IL, USA). All data were expressed as mean ± standard deviation. Differences among four groups in body temperature, body weight, WBC count and radiographic and histological scores were analyzed by one-way analysis of variance. Differences between two groups were compared by the LSD-t test when the data were normally distributed. Differences in microbiological outcomes among four groups were analyzed using Mann–Whitney U test with balancing with Bonferroni–Holm test if two or more groups were compared. P<0.05 was considered statistically significant.
Results
Clinical evaluation
Two rabbits died during anesthesia on day 0, and another two in Ti group died on days 5 and 13 due to diarrhea and severe infection, respectively. Dead rabbits were replaced. The remaining rabbits tolerated the operation without life-threatening conditions. A total of five rabbits in NTATi group and four rabbits in Ti group had surgical-site infection, while animals in NTATi-G and Ti-G groups did not present signs of local infection.
The body temperature in all four groups followed a similar pattern: It gradually increased from day 1 to day 3, followed by gradual decrease from day 3 to day 14 and maintained at a stable level from day 14 to day 42. The body temperature of rabbits in NTATi-G and Ti-G groups was lower than those in Ti groups on day 3 (P<0.05), and there was no difference among four groups from day 3 to day 42 (Figure 1A). The body weights of the rabbits were the lowest on day 3, and rabbits in NTATi-G and Ti-G groups were heavier than those in NTATi and Ti groups on day 3 (P<0.05). From day 3 to day 42, the body weights increased gradually, and there was no difference among four groups (Figure 1B). WBC of rabbits in NTATi and Ti group exhibited a gradually increasing pattern from day 1 to day 3 followed by a gradual decrease from day 3 to day 42, while in NTATi-G and Ti-G groups, WBC increased gradually from day 1 to day 7 and decreased gradually from day 7 to day 42. In general, the WBC count in NTATi-G group was less than that in NTATi and Ti groups on days 3, 35 and 42, while WBC in Ti-G group was less than that in NTATi and Ti groups on days 3, 21 and 42 (P<0.05) (Figure 1C).
Radiograph evaluation
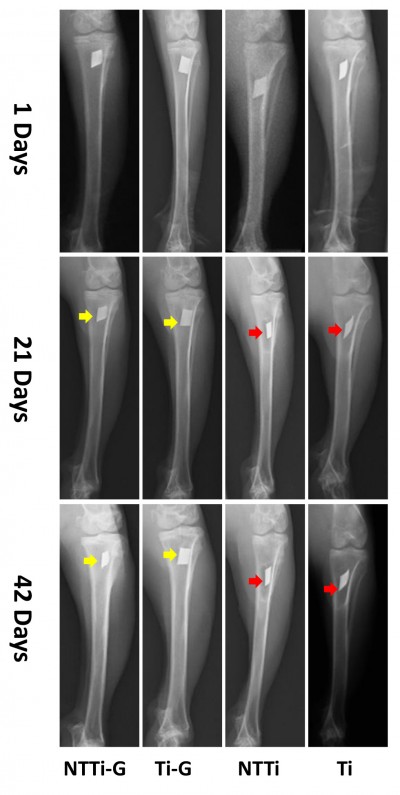
Abnormal radiographic signs such as osteolysis, periosteal reaction, soft tissue swelling and deformity were observed in NTATi and Ti groups on day 21, and these signs became more obvious on day 42 after surgery. In contrast, spontaneous fracture and sequestrum formation were not observed in these groups (Figure 2). No apparent signs of osteomyelitis were noticed on day 21, while slight periosteal reaction, osteolysis and soft tissue swelling were observed in some rabbits on day 42 in NTATi-G and Ti-G groups after surgery (Figure 2).
Radiographic features in all groups were quantified and analyzed according to a modified score by An and Friedman.35 The radiographic scores increased gradually with time after surgery in all four groups. The mean scores of NTATi-G, Ti-G, NTATi and Ti were 1.88±0.99, 2.13±0.64, 5±1.78 and 5.62±2 on day 21, respectively, and the mean scores in NTATi-G and Ti-G groups were lower than that in NTATi and Ti groups (P<0.01). The mean scores of NTATi-G, Ti-G, NTATi and Ti were 2.12±0.51, 2.5±0.7, 7.75±0.98 and 8±2.33 on day 42, respectively. Specifically, the mean scores of NTATi-G and Ti-G groups were lower than NTATi and Ti groups (P<0.01). Relevant data are shown in Table 1.
Microbiological evaluation
Cultures of implants
After being detached and rolled over on agar plates, implants were cultured in TSB. The CFU on each agar plate were counted after 24 h of incubation. Bacterial colony formation was noticed on agar plates of all four groups. The average CFU counts in NTATi-G and Ti-G groups were 40.5±12.36 and 73.75±10.69, respectively. The implant cultures in NTATi and Ti groups showed substantial bacterial growth with average CFU counts of more than 1,000, which was significantly higher than that in NTATi-G and Ti-G groups. All these results were corresponding to the implants immersed in TSB; all implants TSB cultures were positive (cloudy appearance) (Figure 3 and Table 2).
CFU/g bone
The average CFU/g ratios in NTATi-G, Ti-G, NTATi and Ti groups were 7.08±2.5×103, 1.95±0.21×103, 3.15±0.82×106 and 3.48±0.49×106, respectively. Bacterial loads in the NTATi-G and Ti-G groups were significantly lower than those in NTATi and Ti groups (Table 2).
Histological and histopathological results
Histological images from different groups are shown in Figure 4. Signs of infections (development of abscesses, cortical bone destruction, cancellous bone and periosteal new bone formation) were evaluated in NTATi and Ti groups, while NTATi-G and Ti-G groups showed no signs of apparent bone infection. The bone volume was greater in NTATi-G group compared to Ti-G group, whereas little bone formation was seen in NTATi and Ti groups. The quantitative histological scores of NTATi-G, Ti-G, NTATi and Ti groups were 2.25±0.5, 2.5±0.58, 3.5±0.58 and 3.25±0.5, respectively. The histological scores of NTATi-G and Ti-G groups were significantly lower than NTATi and Ti groups, respectively (P<0.05). No significant difference was found between NTATi-G and Ti-G groups, as well as between NTATi and Ti groups.
The histopathological results of all groups are shown in Figure 5. Quantitative pathological scores are presented in Table 3. The mean total scores were 3.5±1, 4.5±1.3, 11.25±2.06 and 11±0.82 in the NTATi-G, Ti-G, NTATi and Ti groups, respectively. Specifically, the mean IAI scores of NTATi-G and Ti-G groups (1±0.82 and 1.25±0.25, respectively) were significantly lower than those in NTATi and Ti groups (2.75±0.96 and 2.5±0.58, respectively, P<0.05); Similarly, the mean ICI scores of NTATi-G and Ti-G groups (1.25±0.5 and 1.25±0.96, respectively) were significantly lower than those in NTATi and Ti groups (3±0.7 and 2.75±0.5, respectively, P<0.05). The mean PI scores of NTATi-G and Ti-G groups (0.75±0.5 and 1.25±0.96, respectively) were significantly lower than those in NTATi and Ti groups (2.75±0.96 and 2.75±0.96, respectively, P<0.05). The mean BN scores of NTATi-G and Ti-G groups (0.50±0.58 and 0.75±0.5, respectively) were significantly lower than those in NTATi and Ti groups (2.75±0.5 and 3.00±0.82, respectively, P<0.001). No significant difference was found between NTATi-G and Ti-G groups, as well as between NTATi and Ti groups.
Discussion
Infection and aseptic loosening are two of the major causes of orthopedic surgical failure. Six hours after the implant operation is a critical time window to prevent organism adhesion on implant surface, which is often referred to as a “decisive period”. The decision and intervention at this time point is vital to the long-term success of an implant.38 Infection would become difficult to control if biofilm had already been formed on the surface of implants or sequestrum, since bacteria within the biofilm are protected from host defenses and antibacterial agents. Studies have demonstrated that once biofilm occurred, implants must be removed so that the infection can be resolved.39 Costs of revision would increase four to six times if infection occurred after initial joint replacement.40 Therefore, timely prevention of infection during initial joint replacement is important in decreasing medical cost and reducing adverse outcomes. In addition, biocompatibility is another important issue for orthopedic-related devices, as both mechanical and biological factors are the key to success for orthopedic surgeries. Stress shielding and insufficient bone formation surrounding the implants may occur if there is an elastic mismatch between the implants and bone tissue. Therefore, appropriate choosing and the modification of surface topography of materials are critical to refine the mechanical and/or biological properties of the implants.
The aim of this study is to investigate the cytocompatibility properties and calcium phosphate crystals coating of NTATi orthopedic implant, which may serve as a local antibiotic delivery device for the prevention of implant-related infections. We used an animal model of New Zealand White rabbits.36 Other models inducing chronic myelitis with local osteonecrosis by sodium morrhuate and bacterial inoculation into the bone marrow cavity have been described previously.35 However, in order to better mimic the clinical setting of postoperative infection, no artificial manipulations were conducted to promote the onset of infection in this study. S. aureus was chosen in this study because it is an organism predominantly associated with postoperative infection in the orthopedic department, which is characterized by its high affinity to the skin and bones, resorption of bone matrix and induction of osteonecrosis.41 Gentamicin was used due to its wide antibacterial spectrum and thermostability, and S. aureus strain used in this study was sensitive to gentamicin.34,42
We compared the effectiveness of NTATi-G in the prophylaxis of infection in the rabbit tibial metaphysis with that of Ti-G, NTATi and Ti according to the physical and laboratorial examination, microbiological analysis, radiographic evaluation and histological and histopathological evaluations. Results also indicated fluctuations of body weight, body temperature and WBC in 7 days after operation, and the extent of fluctuation in NTATi and Ti groups was greater than that in NTATi-G and Ti-G groups. These parameters remained stable afterward from days 7 to day 42. Radiographic analysis did not reveal signs of apparent bone infections in the NTATi-G and Ti-G groups, whereas bone infections were obvious in Ti and NTATi groups on days 21 and 42. Significantly lower radiological, histological and histopathological scores were observed in the NTATi-G and Ti-G groups compared with the NTATi and Ti groups. Average CFU counts on agar plates and CFU/g bone ratio in NTATi-G and Ti-G groups were significantly lower than those in NTATi and Ti groups. Histological evaluation results showed better cytocompatibility in NTATi-G group than in Ti-G group. These results further demonstrate that NTATi-G and Ti-G with coprecipitation drug-loading approach can prevent the development of bone infections and that NTATi-G implant has better bone cell responses than Ti-G implant.
Previous studies have reported that orthopedic implant materials processed with advanced nanotechnology can provide optimal bone cell compatibility properties and be used as drug delivery platforms.32 In the present study, we used anodized titanium with nanotubular surface structures. Gentamicin was loaded on pure titanium and NTATi using a coprecipitation drug-loading approach in which gentamicin was mixed with SBF before it was precipitated with calcium phosphate crystals. An in vitro research has demonstrated that such coprecipitated coatings on NTATi could continuously release antibiotics for up to 3 weeks,32 while a previous study has demonstrated that antibiotics coated with physical adsorption can only release for 150 min.43 The study also demonstrated that NTATi-G through physical absorption could significantly improve the antibacterial ability and bone cell activity of orthopedic implants.43 The present study further demonstrated that NTATi-G through a coprecipitation drug-loading method could alleviate infections after bacterial inoculation in the medullary cavity. Notably, the bacteria was not completely eradicated in animals in the NTATi-G and Ti-G groups, which may partly be attributed to the high bacterial dose (109 CFU) inoculated in the medullary cavity of the surgical site; however, in real-life practice, the amount of contaminating bacteria in joint replacement surgeries is much lower. In addition, Ti-G implant could also inhibit bacterial adhesion in rabbit models; however, its biocompatibility was somewhat lower than the NTATi-G implant. Consistent with the previous studies, we have also demonstrated that anodized nanutubular titanium could improve osteoblast compatibility.43
Conclusion
The present study using in vivo rabbit model demonstrated that NTATi-G had significantly better antibacterial effect than NTATi alone or conventional titanium implants. This novel implant also had more optimal biocompatibility compared to conventional titanium implant loaded with gentamicin. This study introduced NTATi-G as a new kind of implant with the potential of preventing local infections for joint replacement surgeries.
Acknowledgments
The authors thank the colleagues of Shanghai Institute of Ceramics, Chinese Academy of Sciences, for technical support. This study was supported by the National Natural Science Fund (81371936, 14JC1493102). No benefit in any form has been received or will be received from a commercial party related directly or indirectly to the subject of this article.
Disclosure
The authors report no conflicts of interest in this work.
References
Benz T, Angst F, Lehmann S, Aeschlimann A. Association of the sense of coherence with physical and psychosocial health in the rehabilitation of osteoarthritis of the hip and knee: a prospective cohort study. BMC Musculoskelet Disord. 2013;14:159. | ||
Sun Y, Stürmer T, Günther KP, Brenner H. Reliability and validity of clinical outcome measurements of osteoarthritis of the hip and knee – a review of the literature. Clin Rheumatol. 1997;16(2):185–198. | ||
Mota RE, Tarricone R, Ciani O, Bridges JF, Drummond M. Determinants of demand for total hip and knee arthroplasty: a systematic literature review. BMC Health Serv Res. 2012;12:225. | ||
Bozic KJ, Kurtz SM, Lau E, et al. The epidemiology of revision total knee arthroplasty in the United States. Clin Orthop Relat Res. 2010;468(1):45–51. | ||
Koh IJ, Cho WS, Choi NY, Kim TK. Causes, risk factors, and trends in failures after TKA in Korea over the past 5 years: a multicenter study. Clin Orthop Relat Res. 2013;472(1):316–326. | ||
Lie SA, Havelin LI, Furnes ON, Engesaeter LB, Vollset SE. Failure rates for 4762 revision total hip arthroplasties in the Norwegian Arthroplasty Register. J Bone Joint Surg Br. 2004;86(4):504–509. | ||
Whitehouse JD, Friedman ND, Kirkland KB, Richardson WJ, Sexton DJ. The impact of surgical-site infections following orthopedic surgery at a community hospital and a university hospital: adverse quality of life, excess length of stay, and extra cost. Infect Control Hosp Epidemiol. 2002;23(4):183–189. | ||
Bengtson S, Borgquist L, Lidgren L. Cost analysis of prophylaxis with antibiotics to prevent infected knee arthroplasty. BMJ. 1989;299(6701):719–720. | ||
Blackburn WD Jr, Alarcon GS. Prosthetic joint infections. A role for prophylaxis. Arthritis Rheum. 1991;34(1):110–117. | ||
Ching DW, Gould IM, Rennie JA, Gibson PH. Prevention of late haematogenous infection in major prosthetic joints. J Antimicrob Chemother. 1989;23(5):676–680. | ||
Young EJ, Sugarman B. Infections in prosthetic devices. Surg Clin North Am. 1988;68(1):167–180. | ||
Price JS, Tencer AF, Arm DM, Bohach GA. Controlled release of antibiotics from coated orthopedic implants. J Biomed Mater Res. 1996;30(3):281–286. | ||
Ruszczak Z, Friess W. Collagen as a carrier for on-site delivery of antibacterial drugs. Adv Drug Deliv Rev. 2003;55(12):1679–1698. | ||
Zalavras CG, Patzakis MJ, Holtom P. Local antibiotic therapy in the treatment of open fractures and osteomyelitis. Clin Orthop Relat Res. 2004;(427):86–93. | ||
Wininger DA, Fass RJ. Antibiotic-impregnated cement and beads for orthopedic infections. Antimicrob Agents Chemother. 1996;40(12):2675–2679. | ||
Walenkamp GH, Kleijn LL, de Leeuw M. Osteomyelitis treated with gentamicin-PMMA beads: 100 patients followed for 1–12 years. Acta Orthop Scand. 1998;69(5):518–522. | ||
Zhang L, Yan J, Yin Z, et al. Electrospun vancomycin-loaded coating on titanium implants for the prevention of implant-associated infections. Int J Nanomedicine. 2014;9:3027–3036. | ||
Garvin KL, Miyano JA, Robinson D, Giger D, Novak J, Radio S. Polylactide/polyglycolide antibiotic implants in the treatment of osteomyelitis. A canine model. J Bone Joint Surg Am. 1994;76(10):1500–1506. | ||
Overbeck JP, Winckler ST, Meffert R, Töormälä P, Spiegel HU, Brug E. Penetration of ciprofloxacin into bone: a new bioabsorbable implant. J Invest Surg. 1995;8(3):155–162. | ||
Nie L, Nicolau DP, Nightingale CH, Browner BD, Quintiliani R. In vitro elution of ofloxacin from a bioabsorbable polymer. Acta Orthop Scand. 1995;66(4):365–368. | ||
Rutledge B, Huyette D, Day D, Anglen J. Treatment of osteomyelitis with local antibiotics delivered via bioabsorbable polymer. Clin Orthop Relat Res. 2003;(411):280–287. | ||
Hendricks KJ, Lane D, Burd TA, et al. Elution characteristics of tobramycin from polycaprolactone in a rabbit model. Clin Orthop Relat Res. 2001;(392):418–426. | ||
Nelson CL, Hickmon SG, Skinner RA. Treatment of experimental osteomyelitis by surgical debridement and the implantation of bioerodable, polyanhydride-gentamicin beads. J Orthop Res. 1997;15(2): 249–255. | ||
Li LC, Deng J, Stephens D. Polyanhydride implant for antibiotic delivery – from the bench to the clinic. Adv Drug Deliv Rev. 2002;54(7):963–986. | ||
Yagmurlu MF, Korkusuz F, Gürsel I, Korkusuz P, Ors U, Hasirci V. Sulbactam-cefoperazone polyhydroxybutyrate-co-hydroxyvalerate (PHBV) local antibiotic delivery system: in vivo effectiveness and biocompatibility in the treatment of implant-related experimental osteomyelitis. J Biomed Mater Res. 1999;46(4):494–503. | ||
Rossi S, Azghani AO, Omri A. Antimicrobial efficacy of a new antibiotic-loaded poly(hydroxybutyric-co-hydroxyvaleric acid) controlled release system. J Antimicrob Chemother. 2004;54(6):1013–1018. | ||
Türesin F, Gürsel I, Hasirci V. Biodegradable polyhydroxyalkanoate implants for osteomyelitis therapy: in vitro antibiotic release. J Biomater Sci Polym Ed. 2001;12(2):195–207. | ||
Lee JE, Park JC, Lee KH, Oh SH, Kim JG, Suh H. An infection-preventing bilayered collagen membrane containing antibiotic-loaded hyaluronan microparticles: physical and biological properties. Artif Organs. 2002;26(7):636–646. | ||
Park SN, Kim JK, Suh H. Evaluation of antibiotic-loaded collagen-hyaluronic acid matrix as a skin substitute. Biomaterials. 2004;25(17):3689–3698. | ||
Aoyagi S, Onishi H, Machida Y. Novel chitosan wound dressing loaded with minocycline for the treatment of severe burn wounds. Int J Pharm. 2007;330(1–2):138–145. | ||
Rossi S, Marciello M, Sandri G, et al. Wound dressings based on chitosans and hyaluronic acid for the release of chlorhexidine diacetate in skin ulcer therapy. Pharm Dev Technol. 2007;12(4):415–422. | ||
Yao C, Webster TJ. Prolonged antibiotic delivery from nanotubular anodized titanium using a co-precipitation drug loading method. J Biomed Mater Res B Appl Biomater. 2009;91(2):587–595. | ||
Wang P, Li H, Zhang Y, et al. Morphology of nanotube arrays grown on Ti–35Nb–2Ta–3Zr alloys with different deformations. Appl Surf Sci. 2014;290:308–312. | ||
Lucke M, Schmidmaier G, Sadoni S, et al. Gentamicin coating of metallic implants reduces implant-related osteomyelitis in rats. Bone. 2003;32(5):521–531. | ||
An YH, Friedman RJ. Animal models of orthopedic implant infection. J Invest Surg. 1998;11(2):139–146. | ||
Tan HL, Ao HY, Ma R, Lin WT, Tang TT. In vivo effect of quaternized chitosan-loaded polymethylmethacrylate bone cement on methicillin-resistant Staphylococcus epidermidis infection of the tibial metaphysis in a rabbit model. Antimicrob Agents Chemother. 2014;58(10):6016–6023. | ||
Smeltzer MS, Thomas JR, Hickmon SG, et al. Characterization of a rabbit model of staphylococcal osteomyelitis. J Orthop Res. 1997;15(3):414–421. | ||
Hetrick EM, Schoenfisch MH. Reducing implant-related infections: active release strategies. Chem Soc Rev. 2006;35(9):780–789. | ||
Costerton JW. Biofilm theory can guide the treatment of device-related orthopaedic infections. Clin Orthop Relat Res. 2005;(437):7–11. | ||
Sugarman B, Young EJ. Infections associated with prosthetic devices: magnitude of the problem. Infect Dis Clin North Am. 1989;3(2):187–198. | ||
Cioffi GA, Terezhalmy GT, Taybos GM. Total joint replacement: a consideration for antimicrobial prophylaxis. Oral Surg Oral Med Oral Pathol. 1988;66(1):124–129. | ||
Dunne N, Hill J, McAfee P, et al. In vitro study of the efficacy of acrylic bone cement loaded with supplementary amounts of gentamicin: effect on mechanical properties, antibiotic release, and biofilm formation. Acta Orthop. 2007;78(6):774–785. | ||
Lin WT, Tan HL, Duan ZL, et al. Inhibited bacterial biofilm formation and improved osteogenic activity on gentamicin-loaded titania nanotubes with various diameters. Int J Nanomedicine. 2014;9:1215–1230. | ||
Petty W, Spanier S, Shuster JJ, Silverthorne C. The influence of skeletal implants on incidence of infection. Experiments in a canine model. J Bone Joint Surg Am. 1985;67(8):1236–1244. |
Supplementary material
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.