Back to Journals » International Journal of Nanomedicine » Volume 14
G1-S4 or G2-S16 carbosilan dendrimer in combination with Platycodin D as a promising vaginal microbicide candidate with contraceptive activity
Authors Ceña-Diez R, Martin-Moreno A, de la Mata FJ , Gómez-Ramirez R, Muñoz E, Ardoy M, Muñoz-Fernández MA
Received 24 September 2018
Accepted for publication 19 December 2018
Published 2 April 2019 Volume 2019:14 Pages 2371—2381
DOI https://doi.org/10.2147/IJN.S188495
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Thomas Webster
Rafael Ceña-Diez,1,2 Alba Martin-Moreno,1 F Javier de la Mata,2,3 Rafael Gómez-Ramirez,2,3 Eduardo Muñoz,4 Manuel Ardoy,5 Ma Ángeles Muñoz-Fernández1,2
1Immunology Section, Head Inmuno-Biology Molecular Laboratory, Gregorio Marañón University General Hospital, Gregorio Marañón Health Research Institute (IiSGM), Spanish HIV HGM BioBank, Madrid, Spain; 2Networking Research Center on Bioengineering, Biomaterials and Nanomedicine (CIBER-BBN), Madrid, Spain; 3Organic and Inorganic Chemistry Department, Alcalá University, Alcalá de Henares, Spain; 4Department of Cell Biology, Physiology and Immunology, Instituto Maimónides de Investigación Biomédicas de Córdoba (IMIBIC), Reina Sofía University Hospital, University of Córdoba, Córdoba, Spain; 5Human Reproduction Unit, Gregorio Marañon University General Hospital, Madrid, Spain
Purpose: HIV-1 and herpes simplex virus type-2 (HSV-2) represent two of the most relevant sexually transmitted diseases (STDs) worldwide. Moreover, each year there are >200 million pregnancies worldwide, and more than half are unintended. Continued high rates of unintended pregnancies and spread of HIV-1 and HSV-2 require new approaches to address these problems. G1-S4 and G2-S16 dendrimers emerge as potential candidates for the development of a topical microbicide due to their safety and effectivity against HIV-1 and HSV-2 infection, both in vitro and in vivo. Our goal is to develop a dual topical microbicide to prevent the transmission of STDs and unintended pregnancies. Platycodin D (PD) was selected for its great spermicidal activity, topical application, and biocompatibility.
Materials and methods: Toxicology and inhibitory profile of G1-S4/PD and G2-S16/PD were evaluated in vitro and in vivo. Spermicidal activity was assessed by a computer-assisted sperm analysis system (CASA).
Results: G1-S4/PD and G2-S16/PD presented >95% of HIV-1 inhibition in TZM-bl cells and peripheral blood mononuclear cells. CASA assessment determined that 0.25 mM of PD with therapeutic concentrations of G1-S4 or G2-S16 was able to induce 100% immobilization of the sperm in 30 seconds. To evaluate the toxicity in vivo, a vaginal toxicity assay was performed in BALB/c mice. No significant changes or damage to the vaginal epithelium after 7 consecutive days of application were observed.
Conclusion: Our data indicate that G1-S4/PD and G2-S16/PD combinations are promising candidates to be developed for vaginal microbicides with contraceptive activity.
Keywords: topical microbicide, HIV-1, HSV-2, Platycodin D, G1-S4 dendrimer, G2-S16 dendrimer
Introduction
HIV-1 infection remains as one of the most serious health challenges worldwide; heterosexual transmission in women stands out as the fastest growing phase of this pandemic.1 Adolescent girls and young women are particularly at high risk of HIV-1 infection, accounting for 25% of new HIV-1 infections globally. The gender imbalance is more pronounced in geographical zones with high HIV-1 prevalence, such as sub-Saharan Africa, where women account for almost 60% of the total number of people living with HIV-1.2 On the contrary, due to the fact that there are 417 millions of herpes simplex virus type-2 (HSV-2) infected people worldwide and around 19 million new infections every year,3 HSV-2 is the most prevalent sexually transmitted disease (STD). HSV-2 is associated with the appearance of painful ulcers and sores around the genital area or rectum,4 and its viral reservoir is maintained within neurons of the sensory sacral ganglia in latency.5 As HIV-1 infection, HSV is incurable and can be mitigated or modulated through current antiviral treatments, but latent infection cannot be eliminated.6 In addition, no effective human vaccine candidates to date in clinical trials have been licensed for either of them.3,7 Prevention strategies are essential to fight against the STDs, with the development of safe topical microbicides, especially when HSV-2 infection is associated with a 3-fold to 4-fold increased risk of HIV-1 infection.8 Taking into account that globally there are >200 million pregnancies each year, of which ~40% are unintended,9 the development of new compounds with dual microbicidal and spermicide activity, which could prevent HIV-1 and HSV-2 transmission and unintended pregnancies, emerges as a promising alternative.9,10
Contraceptive pills are formulated as a combination of progesterone and estrogens, which induces imbalance in the hypothalamic–pituitary–ovarian axis, which leads to a failure in ovulation. This contraceptive is not free of important risks and side effects, such as thromboembolism, hypertension, hyperlipidemia, cardiovascular diseases, or cancer of the breast and cervix.11 In addition, continued exposure over a long period of time to these hormones has shown certain disorganization in the vaginal mucosa, which increases susceptibility to HIV-1 infection, since vaginal mucosa is the first defense barrier against pathogens and that HIV-1 present in the semen of an infected male could infect the woman after a sexual encounter.12 In this sense, a spermicide gel carries much less risk for a woman, because it is based on impeding the sperm training process. Spermicides are considered low-cost drugs, widely available, and easy to acquire and administer. A compound is considered as spermicide if it has the ability to inhibit sperm motility present in the seminal fluid in 20 seconds,11 although the drug is indeed considered effective whenever it acts before 120 seconds. The fertilization potentiality of a spermatozoon is based on a number of parameters such as mobility, morphology, concentration, or total number of cells in the ejaculation, among others. However, sperm will only be able to fertilize an egg once it has passed through the aforementioned process of sperm training. A series of physiological and biochemical changes that cause the sperm to be attached to the egg membrane at the level of the glycoproteins present in the pellucid area and to penetrate into the cytoplasm of the cell occur physiologically in the reproductive tract of a woman at the level of the fallopian tubes. The effectiveness of spermatozoa to pass through the first barrier involving the cervical mucus, endocervix, and cervix depends on their status as progressive mobile spermatozoa, ie, their ability to advance at an average velocity greater than 25 μm/s.13 The process of sperm training encompasses a series of phenomena such as the acquisition of sperm hypermotility and the loss of its disabling factor, after which the acrosomal reaction begins, which ends at the moment of contact between the inner membranes of the spermatozoid and the acrosome and oocyte plasma membrane, at which time fertilization occurs.14,15 Based on this, it is logical to be interested in a spermicide administered vaginally to prevent this process of sperm training, either by acting on sperm motility in situ acrosome reaction or by repressing the union with the pellucid zone of the ovum.
Briefly, we are working in the development of a microbicidal agent to achieve a compound or combination of compounds that not only reduces the number of new STDs but also has a contraceptive activity, which could be an additional advantage for those women who also want to avoid conception. G1-S2 and G2-S16 polyanionic carbosilane dendrimers are highly branched nanocompounds with potent activity against HIV-1 and HSV-2 as microbicides. The aim of our work was to evaluate the efficiency of Platycodin D (PD), a promising contraceptive, in combination with G1-S2 or G2-S16 dendrimer to develop a prophylactic strategy with dual activity.16–18
Materials and methods
Dendrimers and compounds
Stable and water-soluble polyanionic carbosilane dendrimers of first-generation G1-S4, with four sulfated end groups (C52H101N12Na4O16S4Si5, molecular weight [MW]: 1,509 g/mol), and second-generation sulfonate-functionalized G2-S16 (C112H244N8Na16O48S16Si13, MW: 3,717.15 g/mol) were synthesized according to methods reported by the Dendrimers for Biomedical Applications Group (BioInDen) of the University of Alcalá (UAH, Alcalá de Henares, Madrid, Spain). The batches of these dendrimers were dissolved in distilled water, and stock was generated to a final concentration of 1 mM. Working dilutions (μM) were obtained from the stock of dendrimers by diluting them in phosphate buffered saline (PBS; Lonza, Walkersville, MD, USA) or nuclease-free water (Promega, Madison, WI, USA).
PD (C57H92O28; MW: 1,225.335 g/mol; Biomol, Hamburg, Germany) is a triterpenoid saponin-like compound extracted from the root of the Platycodon grandiflorus plant, widely used in traditional Eastern medicine. PD has been studied primarily as a chemotherapeutic, anti-inflammatory, and spermicidal agent.19–23
Cell culture and viral strains
Blood samples were obtained from buffy coats of healthy anonymous donors from the transfusion center of Madrid following national guidelines. Peripheral blood mononuclear cells (PBMCs) were isolated on a Ficoll-Hypaque density gradient (Rafer, Madrid, Spain) following the current procedures of Spanish HIV HGM BioBank.24 TZM-bl cell line (NIH-AIDS Research and Reference Reagent Program [ARRRP]) was cultured as previously described.25 Viral stocks of CCR5-tropic R5-HIV-1NL(AD8) laboratory strain and CXCR4-tropic X4-HIV-1NL4.3 were obtained by transient transfection of pNL(AD8) and pNL(4.3) plasmids, respectively (NIH-ARRRP) in 293 T cells (ATCC, Manassas, VA, USA). Viral stocks were clarified by centrifugation prior to evaluating the viral titer by using HIV-1 p24gag ELISA kit (INNOTEST® Innogenetics, Ghent, Belgium).
The viral strain HSV-2(333) used in this study was donated by Dr A Alcamí and grown in Vero cells (ATCC® CCL-81™) purchased from ATCC as previously described.17 Virus titers were determined by plaque assay and expressed as plaque forming units (PFU/mL) as was already described.26
Human spermatozoa
Semen samples were collected after masturbation in a sterile vial from ten healthy, young, and fertile donors, after a period of abstinence of at least 3 days. In collaboration with the Human Reproduction Unit (Gregorio Marañon University General Hospital, Madrid, Spain), a basic initial seminal assessment of the collected samples was carried out. Experiments were approved by the local Ethics Committee at Gregorio Marañon University General Hospital (SemDem-15-02), and written informed consents were signed by all patients. The manual of laboratory for the examination and processing of the human semen published by the WHO in 2010 was used. Only those samples that surpassed the criteria of inclusion: 1) the volume of the sample should be at least 2 mL; 2) the total number of spermatozoa should be >65×106 sperm/mL; and 3) the nature of these (vitality and >70% motility) were accepted to be used later in spermicidal trials.
For other experiments, the semen was maintained at room temperature (RT) between 30 and 120 minutes for liquefaction, as the density of the semen after ejaculation is too high. Semen pools were kept in various aliquots of 500 μL at −20°C. Samples were quickly used after thawing, and surplus were ruled out.
Cell proliferation assays
The PBMCs treated with IL-2 60 IU/mL were passed to a 96-well round bottom plate (2×105 cells/well), and thereafter, the corresponding stimuli were added. After 72-hour incubation period, the experiment was developed using a Millipore® proliferation kit, based on the incorporation of bromodeoxyuridine (BrdU), a thymidine analog, into the DNA of proliferating cells. Briefly, PBMCs were washed and fresh medium was added with 1× BrdU solution. After 6–8 hours at 37°C, PBMCs were centrifuged at 300× g for 10 minutes and medium was removed. BrdU assay began with the addition of 100 μL/well of the fixing/denaturing solution for 30 minutes at RT. Thereafter, solution was removed and 100 μL/well of 1× detection antibody solution was added. After 1 hour at RT, three extensive washes were performed and samples incubated with 100 μL/well of horseradish peroxidase-conjugated secondary antibody solution for 1 hour at RT. Subsequently, PBMCs were washed and 100 μL/well of 3,3′,5,5″-tetramethylbenzidine substrate was added for 30 minutes at RT followed by 100 μL/well of Stop solution (if color change was too drastic, it may be necessary to stop the reaction prior to the standard 30 minutes). Absorbance was measured at 450 nm.
Time-of-addition experiment
TZM-bl cells were infected with 15 ng of HIV-1 and compounds and their combinations were added at 0, 1, 2, 4, 6, and 8 hours postinfection. After 2 hours postinfection, cells were washed. Infection values were evaluated by measuring luciferase in luminescence-based assay 48 hours after infection. All experiments were carried out in triplicate and analyzed statistically. Maraviroc, enfuvirtide, and tenofovir disopropil fumarate were used as controls of the HIV-1 infection.
Sperm training: evaluation of the sperm cell count
The sperm training determines the mobile sperm counted after the treatment of semen samples with the combinations of G1-S4 or G2-S16 dendrimer, with the spermicide PD. After collection and processing of the seminal samples, they were incubated for 20 minutes at RT to allow their liquefaction. After this period, a seminal training was performed by density gradient selection on SpermGrad™ medium in two 45% and 90% solutions. Subsequently, the protocol was followed according to the kit 1069 Sperm Preparation Medium (ORIGIO MediCult, Barcelona, Spain) to process and wash the sediment of displaced spermatozoa to the bottom of the tube, in order to collect the final sample of trained spermatozoids to be used in the experiment. An initial evaluation of relative mobile spermatozoa was performed to select aliquots with a final concentration of progressive mobile spermatozoa of 5×106/mL. Spermatozoa were treated with combinations of the compounds for which their spermicidal capacity was tested: G1-S4 or G2-S16 at 10 μM in combination with PD at 0.25 mM. Previously, Lu et al have demonstrated that the effect on sperm motility and viability depends on the administered dose of PD, observing the maximum spermicidal effect for PD at the concentration of 0.25 mM.23 The calculation of sperm survival and the percentage of progressive spermatozoa were performed at 0, 20, 30, 40, 50, 60, 70, 80, 90, 100 seconds and 3 hours post-treatment.
BALB/c vaginal irritation assay
We studied whether G2-S16/PD and G1-S4/PD combinations were toxic in vagina or not, after 7 days of daily application. Eight female BALB/c mice 8 weeks old, 20±3 g (Charles River Laboratories, Wilmington, MA, USA), were purchased and housed in the animal facility at the Centro de Biología Molecular Severo Ochoa (CBMSO), Madrid, Spain. Animal studies were conducted and approved by the CBMSO Institutional Animal Care and the Animal Ethic Committee (AEC-CBMSO, Madrid, Spain). All experiments were performed following AEC-CBMSO and National Guidelines and Regulations (Real Decreto 1201/2005). Mice were injected subcutaneously with 2 mg of medroxyprogesterone acetate (Depo-Provera [depo]) (Pfizer, New York, NY, USA). Five days later, BALB/c mice were randomized into four groups. Control group was treated with 30 μL 1% hidroxyetilcelulose (HEC) (NIH-ARRRP) gel vaginally, G2-S16/PD group was treated with 1% HEC gel with 3% w/v of G2-S16 and 250 μM of PD, G1-S4/PD group was treated with 1% HEC gel with 3% w/v of G1-S4 and 250 μM of PD, and irritation control group was treated with 6% Nonoxinol-9 in PBS. Treatment was applied daily for 7 consecutive days in BALB/c mice previously anesthetized with isoflurane (Forane; Abbott, Madrid, Spain). On the eighth day, mice were sacrificed and vaginas were extracted and conserved in 3.7%–4% formaldehyde w/v (Panreac AppliChem, Darmstadt, Germany).
Histological studies in BALB/c mice
The presence of histological lesions in the mice vaginas was evaluated with hematoxylin-eosin staining. Samples were embedded in paraffin by passage in increasing degree of alcohols (Rectapur [VWR]), two baths of xylene (Analar [VWR]) and other of paraffin, before being placed in paraffin mold. Subsequently, they were cut by using a microtome semimotorized RM2145 Leica and processed for staining. For dewaxing, samples were submitted to two baths of xylene (10 minutes) and three baths in descending order of alcohols (100%, 90%, and 70%) (5 minutes), before being stained with hematoxylin (Merck, Madrid, Spain) for 5 minutes and eosin (Merck) for another 5 minutes. Post-eosin staining dehydration was performed with passage in increasing degree of alcohols (70%, 90%, and 100%) and bath of xylene solution. Finally, they were mounted with DPX (Prolabo, Obregón, Mexico). The existence of injury in vaginal epithelium, inflammatory infiltrate, vascular congestion, and/or edema in the submucosa was evaluated in each histological sample. The score assigned for each of these lesions was as follows: 0 (no change) when no injury or the observed changes were within normal range; 1 (minimum) when changes were sparse but exceeded those considered normal; 2 (light) when injuries were identifiable but with no severity; 3 (moderate) for significant injury that could increase in severity; 4 (very serious) for injuries that occupy most of the analyzed tissue. These values were added up and determined the level of vaginal irritation as minimum 1–3, average 4–6, moderate 7–9, and severe 9+.27
In vivo HSV-2 vaginal challenge assay
The G1-S4 and G2-S16 carbosilane dendrimers and their combinations with PD were selected for analyzing their ability to inhibit the HSV-2 infection in BALB/c mice. Female BALB/c mice (20±2 g) that were 6–8 weeks old were purchased from Charles River Laboratories (Barcelona, Spain) and housed at the CBMSO. Animal studies were conducted and approved by the CBMSO Institutional Animal Care and Use Committee (CEEA-CBMSO, Madrid, Spain). Prior to vaginal HSV-2 challenge, BALB/c mice received a single 2 mg subcutaneous injection of medroxyprogesterone acetate to increase susceptibility to HSV-2 infection.28 Five days later, BALB/c mice were anesthetized with isoflurane (2-chloro-2-(difluoromethoxy)-1,1,1-trifluoro-ethane) for the gel applications and HSV-2 infection. Then, BALB/c mice were randomized into four groups of ten mice per group; placebo group only received 50 μL 1% HEC gel vaginally, G2-S16/PD group received 1% HEC gel with 3% G2-S16 with 0.25 mM of PD, G1-S4 group received 1% HEC with 3% G1-S4 with 0.25 mM of PD, and control group did not receive treatment at all. One hour later, the 40 BALB/c mice were inoculated intravaginally with 105 PFU/20 μL of HSV-2(333) strain and maintained in a supine position with slight elevation of the pelvis for 15 minutes postapplication.
BALB/c mice were examined daily for body weight and genital pathology over 16 days. Disease score was graded according to a 4-point scale: 0, no apparent infection; 1, genital erythema; 2, moderate genital infection; 3, purulent genital ulceration and hair loss, generally poor condition; 4, severe ulceration of genital and surrounding tissue and hind limb paralysis (leading to euthanasia).
Results
Biosafety of G1-S4 or G2-S16 dendrimer in combination with Platycodin D
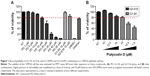
A cellular toxicity assay was performed to evaluate the biocompatibility of G1-S4, G2-S16, PD, and their G1-S4/PD and G2-S16/PD combinations in TZM-bl epithelial cell line (Figure 1). Concentrations at which the viability of treated cells was over 80% of the viability of untreated controls were established as nontoxic concentrations. As demonstrated, PD was nontoxic at 15 μM and G1-S4 and G2-S16 dendrimers were nontoxic at 10 μM (Figure 1A). G1-S4/PD combination was nontoxic at 10/15 μM, and G2-S16/PD was nontoxic at 10/10 μM (Figure 1B).
The G1-S4/PD combination was nontoxic up to 10/15 μM and the G2-S16/PD combination was nontoxic up to 10/10 μM. It means that the second-generation G2-S16 needed lower concentration of PD not to be toxic in comparison with the first-generation G1-S4. Due to the fact that the second-generation G2-S16 dendrimer is bigger than the first-generation G1-S4 dendrimer, the concentration of PD is higher in G2-S16 dendrimer as it has a bigger size.
Evaluation of G1-S4/PD or G2-S16/PD combination against HIV-1 infection
G1-S4 and G2-S16 dendrimers were selected for their high antiviral activity demonstrated in in vitro and in vivo assays against R5 and X4 HIV-1 and HSV-2 viral isolates.29,30
Both G1-S2 and G2-S16 dendrimers tested significantly inhibited HIV-1 infection in TZM-bl cells (Figure 2A). Therefore, when TZM-bl cells were pretreated with G1-S4 or G2-S16 dendrimers at 10 μM, inhibition percentages >95% and 99%, respectively, were obtained vs both R5-HIV-1NL.AD8 and X4-HIV-1NL.4.3 viral isolates. In addition, the combination of the dendrimers with PD did not modify the results obtained. It is noteworthy that PD has anti-HIV-1 activity per se, reaching inhibition rates of 86% and 81% against both R5 and X4 HIV-1 isolates. Moreover, in PBMCs, G1-S4, G2-S16, and PD significantly inhibited HIV-1 infection (Figure 2B). When the PBMCs were pretreated with G1-S4 or G2-S16 dendrimer at the concentration of 10 μM, the inhibition against the isolate R5-HIV-1NL.AD8 was >93% with G1-S4 and >91% with G2-S16 dendrimer. In contrast to the isolate X4-HIV-1NL.4.3, it was >98% in both cases.
Again, the combination of the dendrimers with PD did not modify the results. On PBMC, PD also shows significant antiviral activity per se over both viral isolates, reaching a percentage of inhibition >85% with the isolate R5-HIV-1NL.AD8 and >74% with the isolate X4-HIV-1NL.4.3.
To evaluate the stage of the HIV-1 viral cycle where the PD acts, a “time of addition assay” was performed (Figure 3). G1-S4 and G2-S16 have previously shown to inhibit HIV-1 when added up to 2 hours postinfection. Only a percentage inhibition of HIV-1 infection is observed when PD is added up to 1 hour postinfection, which leads to the conclusion that PD acts in the early stages of the viral cycle. The combinations of PD with G1-S4 and G2-S16 inhibited HIV-1 infection up to 2 hours postinfection, suggesting that the dendrimers and PD are acting at the same stages of the viral cycle as they did alone.
Effect of G1-S4/PD or G2-S16/PD combination in induction of PBMC proliferation
The safety of the combination of the G1-S4/PD or G2-S16/PD was assessed by studying their ability to deregulate the proliferative activity of PBMC. Our results suggest that G1-S4 and G2-S16 dendrimers did not induce cell proliferation in PBMC (Figure 4). PBMC treated with 2 μg/mL phytohemagglutinin was used as positive control of proliferation. G2-S16 did not induce any kind of cell proliferation. The combination of G1-S4/PD or G2-S16/PD did not significantly affect the proliferative capacity of PBMC. Although the combination of the 10 μM G1-S4 dendrimer with PD at 7.5 μM induced 127% cell proliferation compared with untreated 100% PBMC, this value was not significant.
Evaluation of PD spermicidal activity in combination with G1-S4 or G2-S16 dendrimer
To demonstrate whether PD maintains spermicidal activity in combination with G1-S4 or G2-S16 dendrimer, a sperm training experiment was performed on human spermatozoa (Figure 5). Based on the training phenomenon and its ability to move, human spermatozoa can be differentiated into four different types: type A with rapid progressive motility (>20 μm/s), type B with slow progressive motility (5–20 μm/S), type C nonprogressive motility (0–5 μm/s), and type D immotile (0 μm/s). The sperm cells type A+type B are referred to as progressive mobile spermatozoa and represent the spermatozoa that have been able to carry out the sperm training process.13 Thus, a standard semen analysis that has the ability to fertilize should contain at least 50% of progressive mobile spermatozoa.31
The results obtained show that in 20 seconds PD induces a significant decrease in sperm motility, reaching values close to 93% inhibition of sperm motility compared with untreated control spermatozoa. The values obtained for REM in the G1-S4/PD or G2-S16/PD combination were also significant. The combination G1-S4/PD at 40 seconds totally reduced sperm motility, whereas the combination G2-S16/PD completely reduced sperm motility after 60 seconds. As already shown, G1-S4 and G2-S16 dendrimers did not affect sperm motility individually because even after 100 seconds post-treatment the percentage of sperm motility did not fall from 70% with either of the two dendrimers. At 3 hours post-treatment, the results remained stable without significant variations. After 3 hours, PD inhibited all sperm motility unlike dendrimers that did not affect sperm viability (Figure 5).
BALB/c vaginal irritation assay in the presence of PD
With the objective of knowing if the combination of the polyanionic carbosilane dendrimers with PD was causing vaginal irritation to the mice, a vaginal irritation assay was performed in BALB/c mice for 7 days with daily application of the compounds. Eight female BALB/c mice of 7 weeks old with the weight of 20±3 g were purchased. G1-S4/PD or G2-S16/PD was added to 1% HEC placebo gel to a final concentration of 3% w/v of dendrimer and 0.25 mM of PD. BALB/c mice were randomized into four groups of two mice per group. Group A (control) was treated vaginally only with 30 μL of 1% HEC gel, group B (irritation group) was treated vaginally with 4.5% Nonoxinol-9 in PBS, group C was treated with 1% HEC gel with G2-S16/PD, and group D was treated with 1% HEC gel with G1-S4/PD. The four conditions were applied intravaginally in female BALB/c mice previously anaesthetized with isoflurane. After 20 hours, BALB/c mice were sacrificed and vaginas were extracted and conserved in 4% formaldehyde w/v. This histological study in BALB/c vaginal tissue showed that when mice were treated with G2-S16/PD or with G1-S4/PD combinations, after 7 days of exposure all mice had close to the same level of irritation and inflammation of their vaginal epithelium (total score =4) as nonirritation control (Table 1).
The existence of an injury in the vaginal epithelium was evaluated in each biological sample: 0 (no change) when no injury or the observed changes were within normal range; 1 (minimum) when changes were sparse but exceeded those considered normal; 2 (light) when injuries were identifiable but with no severity; 3 (moderate) for significant injury that could increase in severity; 4 (very serious) for very serious injuries that occupy most of the analyzed tissue. These values were added up and determined the level of vaginal irritation as minimum 1–3, average 4–6, moderate 7–9, and severe >9+.
Evaluation of G1-S4/PD or G2-S16/PD combination against HSV-2 in vivo vaginal challenge assay
To demonstrate whether G1-S4 or G2-S16 maintains anti-HSV-2 activity in combination with PD, an in vivo vaginal challenge assay was performed. G1-S4 and G2-S16 were able to retain their antiherpetic activity in the presence of PD, being capable to halt the infection in 100% of the female mice (Figure 6). When mice were exposed to a lethal dose of HSV-2, G1-S4/PD and G2-S16/PD combinations showed significant differences when compared with the control group (P<0.001). In the control of infection group, the mice showed significant weight loss after day 7 postinfection. The first signs of redness and inflammation appeared on day 5 postinfection and the disease score increased until the moment of sacrifice between days 9 and 16 postinfection. No female mice treated with G1-S4/PD or G2-S16/PD showed signs of HSV-2 infection.
Discussion
Although the STDs stand out as the greatest problem of unprotected sex, the possibility of unintended pregnancies should also be taken into consideration. Annually there are about 200 million pregnancies, of which 40% are unintended. Therefore, a topical vaginal compound with dual activity as microbicide and contraceptive, which could prevent both the transmission of HIV-1 and HSV-2 as well as unintended pregnancies, emerges as a promising alternative. Throughout our work, the potentiality of combining the G1-S4 or G2-S16 dendrimer with the PD spermicide has been evaluated to know if topical microbicide gels for the prevention of STDs and unintended pregnancies can be developed. It is of great importance that all the properties already demonstrated for the G1-S4 and G2-S16 dendrimers were not modified in the presence of PD and vice versa.
In vitro HIV-1 inhibition experiments with G1-S4 or G2-S16 dendrimer and in combination with PD in TZM-bl epithelial cell line and PBMCs show that the percentage of inhibition in both R5-HIV-1NL.AD8 and X4-HIV-1NL.4.3 isolate is practically the same compared with results obtained in the absence of PD (Figure 2). In TZM-bl cells, in both presence and absence of PD, the percentage of inhibition is >95% and >99% when the cells are pretreated with G1-S4 or G2-S16 dendrimer. The presence of PD does not modify the inhibition rates in PBMC. Differences in inhibition rates with both viral isolates were not significant, although a higher percentage of inhibition of R5-HIV-1NL.AD8 was observed in TZM-bl epithelia cell line and in PBMC compared with the X4-HIV-1NL.4.3 isolate. Several theories have been proposed as a possible explanation. Polyanionic carbosilane dendrimers act as antiviral nanocompounds because they bind to the V3 region of the HIV-1 gp120. In the case of X4-HIV-1NL.4.3 isolates, the V3 region of gp120 is more exposed at the viral surface.32 Thus, the availability of positive charges with which the dendrimers can interact is greater than in the case of the R5-HIV-1NL.AD8 isolate, which use CCR5 as coreceptor and in which V3 region appears to have a more internal arrangement in the virus membrane (Figure 2).
PD is a plant-derived spermicide that induces its contraceptive action by disrupting the sperm plasma membrane.23 One of the hypotheses that could justify the antiviral activity per se presented by PD, which was studied by carrying out a time of addition assay, is the disruption of HIV-1 envelope glycoprotein (Figure 3). This conclusion can justify PD performance on the early stages of the HIV-1 cycle.
The combination of G1-S4/PD or G2-S16/PD does not induce PBMC proliferation (Figure 4). This property is of great relevance to the development of a topical microbicide due to the fact that an increase in the proliferation of PBMC by the combination of G1-S4/PD or G2-S16/PD favors PBMC recruitment, alters the profile of cytokine secretion, and increases HIV-1 infection in CD4+ T lymphocytes and HSV-2 infection.
Moreover, on samples of human spermatozoa, it has been shown that the combination of PD at the concentration in which the compound produced its 0.25 mM maximum spermicidal effect, together with G1-S4 or G2-S16 dendrimer, was able to induce a total decrease in sperm motility in <40 and 60 seconds, respectively. The maximum spermicidal effect of PD occurs at 20 seconds (Figure 5). After copulation, the spermatozoa may take minutes to reach the vaginal canal and about 10 minutes if they travel at an average velocity of ~25 μm/s, traversing the uterine cavity.13,14 Although it seems clear that the presence of the G1-S4 and G2-S16 dendrimers somewhat retard the spermicidal action of saponin, it remains effective over a relatively short period. For this reason, 60 seconds are sufficient to consider the spermicidal action of the combination of the two dendrimers analyzed and PD to be completely effective.
A histopathological experiment was also performed on BALB/c mice, where it is shown that the daily administration for 7 days of a single dose of 3% gel of G1-S4 or G2-S16 in combination with the 0.25 mM of PD does not generate alteration of epithelial cells, inflammation, or damage to the vaginal mucosa of BALB/c mice (Table 1). Although it had previously been shown that the administration of one, two, or seven doses of 3% gel of G1-S14 or G2-S16 in BALB/c mice did not cause any irritation or alteration on the epithelial barrier of the urogenital tract or the vaginal mucosa.16 This experiment consolidates the use of the combination of PD as a biocompatible and safe potential spermicide in the genital tract.
Following in vitro HIV-1 assays and in vivo toxicity studies, we proceeded to evaluate whether the antiherpetic ability of the dendrimers remained intact in the presence of PD. As can be seen in Figure 6, both G1-S4/PD and G2-S16/PD combinations are capable of 100% halting of the vaginal transmission of HSV-2 after prophylactic application.
Summing up, the results presented in this work seem promising, assuming an incentive to continue investigating with new studies in vivo models, opening a window to the future of the development of a topical administration microbicide with dual activity based on nanotechnology and PD in order to develop a powerful prevention strategy in the fight against the STD in the world and unwanted pregnancies.
Conclusion
Combination of G1-S4 or G2-S16 polyanionic carbosilane dendrimer with PD showed that PD does not affect the antiviral activity of the dendrimers and the dendrimers do not affect the spermicide activity of PD. It was shown that the combination of PD with G1-S4 or G2-S16 has a spermicide effect over human semen in <30 seconds.
Acknowledgments
This work was partially funded by the RD16/0025/0019 projects as part of Acción Estratégica en Salud, Plan Nacional de Investigación Científica, and Desarrollo e Innovación Tecnológica (2013–2016), and cofinanced by Instituto de Salud Carlos III (Subdirección General de Evaluación) and Fondo Europeo de Desarrollo Regional (FEDER), RETIC PT17/0015/0042, Fondo de Investigacion Sanitaria (FIS) (grant number PI16/01863), and the EPIICAL Project. CIBER-BBN is an initiative funded by the VI National R&D&i Plan 2008–2011, Iniciativa Ingenio 2010, the Consolider Program, and CIBER Actions, and financed by the Instituto de Salud Carlos III with assistance from the European Regional Development Fund. In addition, The European Cooperation in Science and Technology (COST), CA17140 – Cancer Nanomedicine – From the Bench to the Bedside, partially funded this work.
Disclosure
The authors report no conflicts of interest in this work.
References
UNAIDS. Report on the global AIDS epidemic/2016; 2016. Available from: http://aidsinfo.unaids.org/. Accessed September 30, 2018. | ||
Carter AJ, Bourgeois S, O’Brien N, et al. Women-specific HIV/AIDS services: identifying and defining the components of holistic service delivery for women living with HIV/AIDS. J Int AIDS Soc. 2013;16(1):17433. | ||
Looker KJ, Magaret AS, Turner KM, Vickerman P, Gottlieb SL, Newman LM. Global estimates of prevalent and incident herpes simplex virus type 2 infections in 2012. PLoS One. 2015;10(1):e114989. | ||
Gupta R, Warren T, Wald A. Genital herpes. Lancet. 2007;370(9605):2127–2137. | ||
Wagner EK, Flanagan WM, Devi-Rao G, et al. The herpes simplex virus latency-associated transcript is spliced during the latent phase of infection. J Virol. 1988;62(12):4577–4585. | ||
Johnston C, Saracino M, Kuntz S, et al. Standard-dose and high-dose daily antiviral therapy for short episodes of genital HSV-2 reactivation: three randomised, open-label, cross-over trials. Lancet. 2012;379(9816):641–647. | ||
Johnston C, Koelle DM, Wald A. Current status and prospects for development of an HSV vaccine. Vaccine. 2014;32(14):1553–1560. | ||
Freeman EE, Weiss HA, Glynn JR, Cross PL, Whitworth JA, Hayes RJ. Herpes simplex virus 2 infection increases HIV acquisition in men and women: systematic review and meta-analysis of longitudinal studies. AIDS. 2006;20(1):73–83. | ||
Sedgh G, Singh S, Hussain R. Intended and unintended pregnancies worldwide in 2012 and recent trends. Stud Fam Plann. 2014;45(3):301–314. | ||
Yang M, Zhi R, Lu L, et al. A CCR5 antagonist-based HIV entry inhibitor exhibited potent spermicidal activity: potential application for contraception and prevention of HIV sexual transmission. Eur J Pharm Sci. 2018;117:313–320. | ||
Tanphaichitr N, Srakaew N, Alonzi R, et al. Potential use of antimicrobial peptides as vaginal spermicides/Microbicides. Pharmaceuticals. 2016;9(1):13. | ||
Haase AT. Early events in sexual transmission of HIV and SIV and opportunities for interventions. Annu Rev Med. 2011;62(1):127–139. | ||
Vasan SS. Semen analysis and sperm function tests: how much to test? Indian J Urol. 2011;27(1):41–48. | ||
Suarez SS, Pacey AA. Sperm transport in the female reproductive tract. Hum Reprod Update. 2006;12(1):23–37. | ||
Okabe M. The cell biology of mammalian fertilization. Development. 2013;140(22):4471–4479. | ||
Ceña-Diez R, García-Broncano P, Javier de La Mata F, Gómez R, Resino S, Muñoz-Fernández M. G2-S16 dendrimer as a candidate for a microbicide to prevent HIV-1 infection in women. Nanoscale. 2017;9(27):9732–9742. | ||
Ceña-Diez R, Vacas-Córdoba E, García-Broncano P, et al. Prevention of vaginal and rectal herpes simplex virus type 2 transmission in mice: mechanism of antiviral action. Int J Nanomedicine. 2016;11:2147–2162. | ||
Guerrero-Beltrán C, Ceña-Diez R, Sepúlveda-Crespo D, et al. Carbosilane dendrons with fatty acids at the core as a new potential microbicide against HSV-2/HIV-1 co-infection. Nanoscale. 2017;9(44):17263–17273. | ||
Wu JT, Yang GW, Qi CH, Zhou L, Hu JG, Wang MS. Anti-inflammatory activity of platycodin D on alcohol-induced fatty liver rats via TLR4-MYD88-NF-κB signal path. Afr J Tradit Complement Altern Med. 2016;13(4):176–183. | ||
Zhu CG, Zhang LS. [Effect of platycodin D on radiosensitivity of human hepatoma cell line and related mechanisms of action]. Zhonghua Gan Zang Bing Za Zhi. 2017;25(6):458–462. Chinese. | ||
Zhang Z, Zhao M, Zheng W, Liu Y. Platycodin D, a triterpenoid saponin from Platycodon grandiflorum, suppresses the growth and invasion of human oral squamous cell carcinoma cells via the NF-κB pathway. J Biochem Mol Toxicol. 2017;31(9):e21934. | ||
Zare-Zardini H, Fesahat F, Anbari F, Halvaei I, Ebrahimi L. Assessment of spermicidal activity of the antimicrobial peptide sarcotoxin PD: a potent contraceptive agent. Eur J Contracept Reprod Health Care. 2016;21(1):15–21. | ||
Lu Z, Wang L, Zhou R, et al. Evaluation of the spermicidal and contraceptive activity of platycodin D, a saponin from Platycodon grandiflorum. PLoS One. 2013;8(11):e82068. | ||
García-Merino I, de Las Cuevas N, Jiménez JL, et al. The Spanish HIV Biobank: a model of cooperative HIV research. Retrovirology. 2009;6(1):27. | ||
Vacas Córdoba E, Arnaiz E, Relloso M, et al. Development of sulphated and naphthylsulphonated carbosilane dendrimers as topical microbicides to prevent HIV-1 sexual transmission. AIDS. 2013;27(8):1219–1229. | ||
Shippee RL, Koppenheffer T, Watiwat SR, Burleson DG, Mason AD. The interaction of burn injury and zinc nutriture as assessed by the humoral response to sheep red blood cells in a burn rat model. Burns. 1992;18(1):45Jr–48Jr. | ||
Chonco L, Pion M, Vacas E, et al. Carbosilane dendrimer nanotechnology outlines of the broad HIV blocker profile. J Control Release. 2012;161(3):949–958. | ||
Parr MB, Kepple L, Mcdermott MR, Drew MD, Bozzola JJ, Parr EL. A mouse model for studies of mucosal immunity to vaginal infection by herpes simplex virus type 2. Lab Invest. 1994;70(3):369–380. | ||
Ceña-Diez R, García-Broncano P, de La Mata FJ, Gómez R, Muñoz-Fernández MÁ. Efficacy of HIV antiviral polyanionic carbosilane dendrimer G2-S16 in the presence of semen. Int J Nanomedicine. 2016;11:2443–2450. | ||
Sepúlveda-Crespo D, Serramía MJ, Tager AM, et al. Prevention vaginally of HIV-1 transmission in humanized BLT mice and mode of antiviral action of polyanionic carbosilane dendrimer G2-S16. Nanomedicine. 2015;11(6):1299–1308. | ||
Aitken RJ, Sutton M, Warner P, Richardson DW. Relationship between the movement characteristics of human spermatozoa and their ability to penetrate cervical mucus and zona-free hamster oocytes. Reproduction. 1985;73(2):441–449. | ||
Jiang X, Feyertag F, Robertson DL. Protein structural disorder of the envelope V3 loop contributes to the switch in human immunodeficiency virus type 1 cell tropism. PLoS One. 2017;12(10):e0185790. |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.