Back to Journals » International Journal of Nanomedicine » Volume 13 » T-NANO 2014 Abstracts
Fluorescent gold nanoclusters for efficient cancer cell targeting
Received 18 October 2016
Accepted for publication 31 October 2016
Published 15 March 2018 Volume 2018:13(T-NANO 2014 Abstracts) Pages 15—17
DOI https://doi.org/10.2147/IJN.S125003
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Lei Yang
Rahul Purohit, Sanjay Singh
Institute of Life Sciences, School of Science and Technology, Ahmedabad University, Ahmedabad, Gujarat, India
Abstract: Well-known surface properties of gold nanoparticles (AuNPs) offer easy surface modification with desired biomolecule, thus enabling them to be used for targeting and imaging of cancer cells/tissues. However, targeting and imaging capability come through after synthesis coating of AuNPs’ surface with targeting or imaging molecules. Attempts have been made to conjugate both imaging and targeting molecules over the AuNPs, but have seen limited success. Hence, exploiting the fluorescence properties of gold nanoclusters (AuNCs), we have synthesized glucose-coated AuNCs for exhibiting both the imaging and targeting properties. These clusters have shown rapid and selective uptake in cancerous (A549) cells when compared with bovine serum albumin-coated AuNCs.
Keywords: gold nanoclusters, fluorescent AuNCs, cancer cell imaging and targeting
Introduction
Unique properties of bovine serum albumin (BSA)-coated gold nanoclusters (AuNCs) such as small size, high surface to volume ratio, in situ fluorescence, and biocompatibility (imparted due to the BSA coating)1 make them ideal candidates for studying the interaction of NCs with living systems (cells, tissues, organisms). The fluorescence properties exhibited by AuNCs can be used to track them easily within the biological system. AuNCs are nontoxic and stabile in the body fluids. Owing to their small size (2–3 nm), they interact with living system differently and do not elicit any immune response. It has been shown that almost all cancerous cells have a large number of glucose receptors that help in glucose uptake.2 The high glucose uptake is required for the high energy need of cancerous cells to meet their extra physiological requirements such as uncontrolled growth, cell division, metastasis, and angiogenesis. Exploiting this striking difference, we have synthesized novel glucose-coated AuNCs (Glu-AuNCs) to target the cancer cells, and BSA-AuNCs were synthesized as control.
Experimental methods
AuNCs synthesis
BSA-AuNCs were synthesized using BSA solution (50 mg/mL, 5 mL) and HAuCl4 (10 mM, 5 mL) solution. Both solutions were mixed for 10 minutes followed by the addition of NaOH (1 M, 0.5 mL). Then, the solution was kept overnight with constant stirring.3 For the synthesis of Glu-AuNCs, glucose solution (50 mg/mL) was added to HAuCl4 solution (10 mM/mL), and total volume was made up to 5 mL. The solution was stirred for 30 minutes followed by the addition of BSA solution. After mixing for 20 minutes, NaOH solution was added (1 M, 0.75 mL), and the solution was stirred overnight. Finally, AuNCs were dialyzed using 12.4 kDa cutoff dialysis membranes to remove the free and unbounded glucose, NaOH, and unreduced gold ions. Samples were stored at 4°C until use.
Characterization
AuNCs were characterized by ultraviolet–visible spectroscopy (Synergy HT, BioTek Instruments, Winooski, VT, USA), Fourier transform infrared spectroscopy (Shimadzu FTIR 8400S, Kyoto, Japan), and fluorescence spectroscopy (NanoLog Spectrofluorometer, Horiba Scientific, Kyoto, Japan).
Uptake assay
A549 cells were exposed to BSA-AuNCs and Glu-AuNCs for 6 hours for the uptake study and assessed by flow cytometer and fluorescent microscopy. A549 cells were commercially purchased from National Centre for Cell Sciences, Pune, India. For fluorescent microscopic imaging, cells (~5,000) were grown on coverslip for 24 hours. Subsequently, cells were washed and exposed to AuNCs for 6 hours followed by washing with phosphate-buffered saline and fixation with formaldehyde. Next, the cells were mounted on the glass slide and used for imaging.
Results and discussion
Ultraviolet–visible spectra recorded for BSA-AuNCs show absorption edge at ~530 nm,4 whereas Glu-AuNCs showed absorption edge at ~500 nm (Figure 1A). This shift in absorbance maxima suggests that AuNCs are coated with glucose. The typical color of suspension of BSA-AuNCs and Glu-AuNCs is displayed in figure 1B. Both the suspensions look similar suggesting that the optical behavior of BSA-AuNCs does not change substantially after coating with glucose. This observation is in accordance with UV-vis spectra pattern of BSA-AuNCs and Glu-AuNCs (Figure 1A). It is well known that AuNCs <3 nm do not show well-defined surface plasmon resonance in the visible region, rather show absorption edge around ~500 nm.
BSA-AuNCs show maximum fluorescent emission intensity at 624 nm, whereas Glu-AuNCs show a shift of 5 nm with the emission maxima being recorded at 629 nm (Figure 1D). BSA-AuNCs show a clean and narrow spectrum, whereas Glu-AuNCs show a slightly broader spectrum. For further confirmation of florescence nature of AuNCs after synthesis, we exposed AuNCs to ultraviolet light wavelength of 280 nm. Clean bright fluorescence light was observed for both BSA-AuNCs and Glu-AuNCs (Figure 1C). When BSA-AuNCs and Glu-BSA-AuNCs were exposed to the visible light, no fluorescence was observed (Figure 1A).
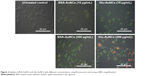
Human lung carcinoma cells (A549) are known to express many glucose receptors. Therefore, to check the selective uptake of Glu-AuNCs, we incubated the particles with A549 cells and compared the results with the uptake of BSA-AuNCs. To our surprise, the Glu-AuNCs uptake was more than BSA-AuNCs, which is clearly evident by intense green florescence from A549 cells when exposed to Glu-AuNCs (Figure 2). However BSA-AuNCs-treated cells showed meager fluorescence, which could be due to the endocytosis of particles. Therefore, the swift internalization of Glu-AuNCs in cancerous cells further confirms that these nanocomplexes could be used as a targeting molecule for cancerous cells. From our results, it can be concluded that Glu-AuNCs could be used for selective targeting (due to glucose) and imaging (due to AuNCs) of cancer cells.
Conclusion
Our study shows the synthesis of stable and fluorescent BSA-AuNCs and Glu-AuNCs. Glu-AuNCs show more uptake than BSA-AuNCs in cancerous cells at any given time. Therefore, these AuNCs can be used for selective targeting and imaging of cancerous cells.
Acknowledgment
The authors gratefully acknowledge the financial assistance from the Department of Chemistry, Indian Institute of Technology Madras, IITM (Grant no TTR1314010IITMTPRA), and the provision of laboratory and chemical facilities by the Centre for Nanotechnology Research and Applications (CENTRA) supported by The Gujarat Institute for Chemical Technology (GICT).
Disclosure
The authors report no conflicts of interest in this work.
References
Purohit R, Vallabani S, Shukla R, Kumar A, Dhawan A, Singh S. BSA coated gold nanoparticles exhibit size dependent interaction with lung cancer (A549) cells. Mol Cytogenet. 2014;7(1):83. | ||
Jones RG, Thompson CB. Tumor suppressors and cell metabolism: a recipe for cancer growth. Genes Dev. 2009;23(5):537–548. | ||
Liu JM, Jiang SL, Wang XX, et al. BSA-protected gold nanoclusters as fluorescent sensor for selective and sensitive detection of pyrophosphate. Anal Methods. 2013;5(16):3942–3947. | ||
Li H, Guo Y, Xiao L, Chen B. Selective and sensitive detection of acetylcholinesterase activity using denatured protein-protected gold nanoclusters as a label-free probe. Analyst. 2014;139(1):285–290. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.