Back to Journals » International Journal of Nanomedicine » Volume 16
Fixed-Combination Eye Drops Based on Fluorometholone Nanoparticles and Bromfenac/Levofloxacin Solution Improve Drug Corneal Penetration
Authors Otake H, Goto R, Ogata F, Isaka T, Kawasaki N , Kobayakawa S, Matsunaga T, Nagai N
Received 24 April 2021
Accepted for publication 22 July 2021
Published 10 August 2021 Volume 2021:16 Pages 5343—5356
DOI https://doi.org/10.2147/IJN.S317046
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Mian Wang
Hiroko Otake,1,* Ryoka Goto,1,* Fumihiko Ogata,1 Takumi Isaka,1 Naohito Kawasaki,1 Shinichiro Kobayakawa,2 Toru Matsunaga,3 Noriaki Nagai1
1Faculty of Pharmacy, Kindai University, Higashi-Osaka, Osaka, 577-8502, Japan; 2Department of Ophthalmology, Nippon Medical School, Musashi-Kosugi Hospital, Kawasaki, Kanagawa, 211-8533, Japan; 3Design and Development, SEED Co., Ltd., Kounosu-shi, Saitama, 369-0131, Japan
*These authors contributed equally to this work
Correspondence: Noriaki Nagai
Faculty of Pharmacy, Kindai University, 3-4-1 Kowakae, Higashi-Osaka, Osaka, 577-8502, Japan
Tel +81 6 4307 3640
Fax +81 6 6730 1394
Email [email protected]
Purpose: The multi-instillation of three commercially available (CA) eye drops [fluorometholone (FL)-, bromfenac (BF)- and levofloxacin (LV)-eye drops] has been used to manage pain and inflammation post-intraocular surgery. However, the multi-instillation of these three eye drops causes corneal damage, and the FL drops have the disadvantage of low ocular bioavailability. To overcome these problems, we prepared fixed-combination eye drops based on FL nanoparticles (FL-NPs) and BF/LV solution (nFBL-FC), and evaluated the corneal toxicity and transcorneal penetration of the nFBL-FC eye drops.
Methods: FL powder was mixed in 2-hydroxypropyl-β-cyclodextrin solution containing benzalkonium chloride, mannitol and methylcellulose, and milled with a Bead Smash 12 (5500 rpm for 30 s× 30 times). The BF/LV solution was then added to the milled-dispersions to be used as nFBL-FC. The FL, BF and LV concentrations were measured by HPLC methods, and transcorneal penetration was evaluated in rabbits.
Results: The FL particle size in nFBL-FC was 40– 150 nm, with only 0.0018% in liquid form. No aggregation of FL particles in the nFBL-FC was observed for 1 month. The viability of human corneal epithelial cells treated with nFBL-FC was remarkably higher than that of cells subjected to the multi-instillation of the corresponding three CA-eye drops. In addition, the corneal penetrations (AUC) of the FL, BF and LV in nFBL-FC were 4.9-, 1.8-, and 7.1-fold those of the corresponding CA-eye drops, respectively. Moreover, the caveolae-dependent endocytosis (CavME) inhibitor (nystatin) significantly prevented the transcorneal penetration of these drugs.
Conclusion: We prepared fixed-combination eye drops based on FL-NPs and BF/LV solution (nFBL-FC), and show that high levels of FL-NPs and dissolved BF/LV (liquid drugs) can be delivered into the aqueous humor by the instillation of nFBL-FC. Further, we show that CavME is mainly related to the enhancement of transcorneal penetration of both the solid (NPs) and liquid drugs.
Keywords: fluorometholone, bromfenac, levofloxacin, fixed-combination eye drops, corneal permeability, endocytosis
Graphical Abstract:
Plain Language Summary
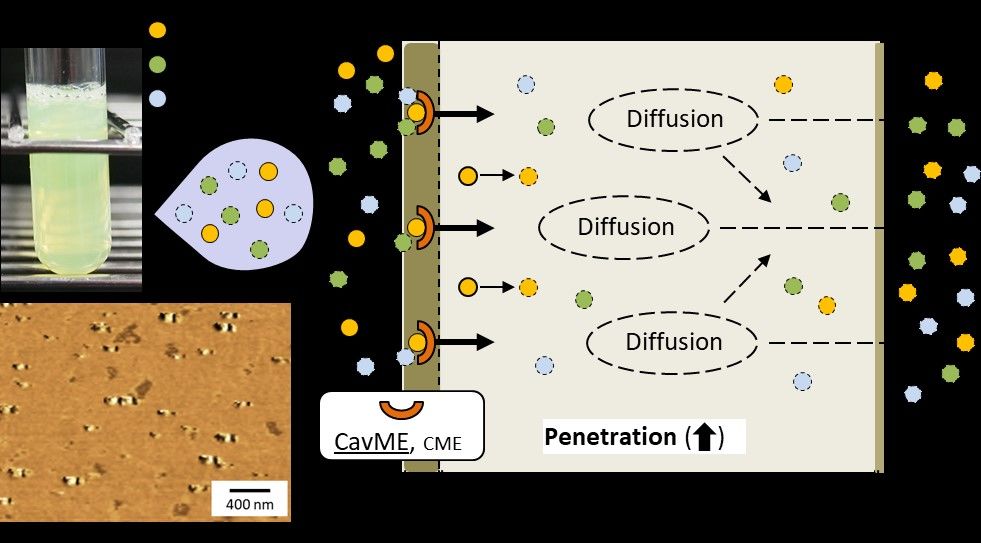
The multi-instillation of commercially available (CA) eye drops [fluorometholone (FL)-, bromfenac (BF)- and levofloxacin (LV)-eye drops] has been used to manage pain and inflammation following intraocular surgery. However, the multi-instillation of these three eye drops causes corneal damage, and FL has the disadvantage of a low ocular bioavailability. To overcome these problems, we prepared fixed-combination eye drops based on FL nanoparticles (FL-NPs) and BF/LV solution (nFBL-FC), and showed the low corneal toxicity and high corneal penetration of nFBL-FC in comparison with multi-treatment with the corresponding three CA-eye drops (CA-FL, CA-BF and CA-LV). Moreover, the FL-NPs are taken up into the cornea by energy-dependent endocytosis (CavME pathways), and dissolve in the stromal side of the cornea, whereas liquid FL is released into the aqueous humor. In addition, CavME inhibitors attenuate the corneal penetration of the liquid drugs (BF and LV). We hypothesize that the activation of CavME by the FL-NPs may also promote the corneal uptake of the BF/LV solution (liquid drugs). Our studies are the first to demonstrate that the corneal penetration of both solid and liquid drugs can be enhanced by the design of a fixed combination of solid NPs and solution.
Introduction
Ocular surface injury following intraocular surgery causes persistent inflammation and cystoid macular oedema, resulting in patient discomfort and delayed recovery.1 Because of this, multi-treatment with topical ophthalmic corticosteroids, nonsteroidal anti-inflammatory drugs (NSAIDs) and/or antibacterial drugs has been applied to decrease the incidence of persistent inflammation and cystoid macular oedema following intraocular surgery, such as cataract surgery.1–4 In Japan, ophthalmic formulations of fluorometholone (FL) and bromfenac (BF) are often applied for the management of pain, discomfort and inflammation after cataract surgery. In addition, topical ophthalmic formulations of levofloxacin (LV), which is antibacterial, are also widely used in patients undergoing cataract extraction with posterior chamber intraocular lens (IOL) implantation.5,6
FL Ophthalmic Suspension, an ophthalmic fluorinated corticosteroid, is one of the many corticosteroids used as therapy for inflammatory and allergic conditions of the eye,7 and the BF 0.1% ophthalmic solution, which is a brominated once-daily ophthalmic NSAID, is often applied as a treatment for postoperative pain, discomfort and inflammation in patients undergoing cataract surgery in Japan. LV is a pyridone carboxylic acid derivative structurally related to nalidixic acid and newer fluorinated quinolone antibacterial agents; the LV 0.5% ophthalmic solution is approved for perioperative use during intraocular surgery to prevent ocular infections, such as bacterial conjunctivitis.8 The multi-instillation of FL, BF and LV eye drops reduce the incidence of persistent inflammation and cystoid macular edema after cataract surgery. On the other hand, the benzalkonium chloride (BAC), ophthalmic preservative, was contained in the commercially available (CA) eye drops, and the BAC induces oxidative stress or significantly alters precorneal mucins.9 Therefore, the multi-instillation of eye drops is a burden for post-surgical patients, and causes eye-smarting and corneal damage, since the multi-instillation of BAC enhance the risk of corneal toxicity. In addition, it is known that the ocular bioavailability (BA) of FL is low, and that the therapeutic levels reached in the intraocular and posterior area of the eye are ineffective.10 Therefore, the development of a FL/BF/LV fixed-combination (FBL-FC) with high ocular drug BA would be useful for the management of pain, discomfort and inflammation in patients undergoing cataract extraction with posterior chamber IOL implantation.
Recently, it was reported that the aqueous eye drops based on dexamethasone/cyclodextrin complexes in a microsuspension gave significant improvements.11 Thus, to overcome the low BA of traditional eye drops (liquid formulations and ophthalmic suspensions), various colloidal carriers for topical administration, such as liposomes, dendrimers, emulsions, suspensions, solutions, surfactant-based systems, nanospheres, microspheres, nanomicelles, nanostructured lipid carriers, nanosuspensions of polymeric nanoparticles (NPs), solid NPs, in situ gelling systems, and implants have recently emerged.12–18 We have also reported that solid NPs prepared by a breakdown method provide for high ocular BA with low corneal toxicity.19 In addition, we found that the combination of magnesium hydroxide NPs and a dissolved drug (carteolol or timolol maleate) increases the ocular BA to enhance the corneal permeability of solid NPs and dissolved drugs,20,21 and that energy-dependent endocytosis is related to the high corneal penetration by solid NPs.19 Therefore, a transcorneal drug delivery system (DDS) using solid NPs is expected to enhance the ocular BA of both solid and liquid drugs.
In this study, we prepared fixed-combination eye drops based on three drugs, FL-NPs and BF/LV solution (nFBL-FC), by a breakdown method (bead mill method), and evaluated the corneal toxicity and transcorneal penetration of nFBL-FC in comparison with the CA-eye drops.
Materials and Methods
Animals
Male adult rabbits (weight 2.69±0.71 kg) were commercially purchased from Shimizu Laboratory Supplies Co., Ltd. (Kyoto, Japan), and used in a protocol approved by Kindai University (KAPS-31-002, 1 April 2019). The animal experiments were performed in accordance with the Association for Research in Vision and Ophthalmology (ARVO) and Kindai University guidelines.
Chemicals
FL powder, LV powder, cytochalasin D, isoflurane, mannitol (D-mannitol) and butyl p-hydroxybenzoate were purchased from Wako Pure Chemical Industries, Ltd (Osaka, Japan). 2-hydroxypropyl-β-cyclodextrin (HPβCD) was obtained from Nihon Shokuhin Kako Co., Ltd (Tokyo, Japan), and nystatin was provided by Sigma-Aldrich Japan (Tokyo, Japan). Methylcellulose (MC) was obtained from Shin-Etsu Chemical Co., Ltd (Tokyo, Japan). BF, dimethyl sulfoxide (DMSO), dynasore, rottlerin and Cell Count Reagent SF were purchased from Nacalai Tesque (Kyoto, Japan), and BAC was provided by Kanto Chemical Co., Inc. (Tokyo, Japan). Fetal bovine serum, Dulbecco’s modified Eagle’s medium/Ham’s F12 (DMEM/F12), streptomycin and penicillin were provided by GIBCO (Tokyo, Japan). CA-FL, CA-LV and CA-oxybuprocaine hydrochloride eye drops (Benoxil® ophthalmic solution 0.4%) were obtained from Santen Pharmaceutical Co., Ltd. (Osaka, Japan), and CA-BF was purchased from Nitto Medic Co., Ltd. (Toyama, Japan). All other chemicals used were of the highest purity commercially available.
Preparation of nFBL-FC
FL-NPs were prepared following our previous reports using zirconia beads (diameter: 0.1 mm) and Bead Smash 12 (a bead mill, Wakenyaku Co. Ltd, Kyoto, Japan).19,22 The 0.2 g of sterilized FL powder (MPs) was mixed in 0.2 µm-filtrated solvent [5% (w/v) HPβCD solution (100 mL) containing 0.001 g BAC, 0.1 g mannitol and 0.5 g MC], and milled with the Bead Smash 12 (5500 rpm for 30 s×30 times, 4°C). Then, 0.2 µm-filtrated solvent containing 0.2% BF and 1% LV solutions were added to the milled-dispersion (1:1), and used as nFBL-FC in this study. In addition, sterilized FL-MPs were mixed in 0.2 µm-filtrated solvent containing BF, LV, and used as mFBL-FC in this study. The pH of mFBL-FC and nFBL-FC was adjusted to 6.5, since LV uptake is pH-dependent, and maximal corneal penetration occurs at pH 6.5.23 The compositions of mFBL-FC and nFBL-FC are as follows: 0.1% FL, 0.1% BF, 0.5% LV, 0.001% BAC, 0.1% mannitol, 5% HPβCD, 0.5% MC. The concentration of FL, BF and LV were determined according to the concentration of each CA-eye drops.
Characterization of nFBL-FC
A laser diffraction particle size analyzer SALD-7100 (Shimadzu Corp.) was used to measure particle size with the refractive index set at 1.60–0.010i. Particle distribution and the number of FL-NPs were determined by Nanosight LM10 (QuantumDesign Japan, Tokyo, Japan) with the time, wavelength and viscosity set at 60 s, 405 nm (blue), and 1.27 mPa⋅s, respectively. An SPM-9700 (Shimadzu Corp., Kyoto, Japan) was used to obtain atomic force microscopy (AFM) images created by combining phase and height images. The viscosity at 20 °C and the zeta potential of the eye drops were measured by a SV-1A (A&D Company, Limited, Tokyo, Japan), a micro-electrophoresis zeta potential analyzer model 502 (Nihon Rufuto Co., Ltd, Tokyo, Japan), respectively.24 The solubilized and non-solubilized FL in the eye drops were separated by centrifugation at 100,000 g using an OptimaTM MAX-XP Ultracentrifuge (Beckman coulter, Osaka, Japan), and the levels of solubilized and non-solubilized FL were measured by HPLC.
Measurement of FL by an HPLC Method
FL concentrations were measured on an LC-20AT system (HPLC, Shimadzu Corp., Kyoto, Japan). Ten microliters of FL sample and 50 μL of 2.5 μg/mL butyl p-hydroxybenzoate in methanol used as an internal standard were mixed, and 10 μL of the mixture was injected using an auto sampler SIL-10AF (Shimadzu Corp., Kyoto, Japan). A TSKgel ODS-100V column (4.6×150 mm, Tosoh Corporation, Tokyo, Japan) was used at 35°C, and the wavelength for detection was 254 nm. The mobile phase consisted of 70% methanol at a flow rate of 0.8 mL/min. The FL concentration was analyzed from calibration curve (range 0–10 µg/mL, y=0.0794x+0.0109), and the R level was 0.9987.
Measurement of BF and LV by an HPLC Method
The simultaneous analysis method was used to determine the BF and LV concentration, and the BF and LV concentrations were measured on an LC-20AT system (HPLC, Shimadzu Corp.). Ten microliters of BF or LV sample was mixed with 50 μL of 2.5 μg/mL gatifloxacin in methanol used as an internal standard, and 10 μL of the mixtures was injected using an auto sampler SIL-10AF (Shimadzu Corp., Kyoto, Japan). An Inertsil® ODS-3 column (2.1×50 mm, GL Science Co., Inc., Tokyo, Japan) was used at 35°C, and the wavelength for detection was 280 nm. The mobile phase consisted of 25 mM citric acid solution containing 10 mM Sodium Dodecyl Sulfate and 10 mM tert-butyl acetate/acetonitrile (57/43 v/v%) at a flow rate of 0.25 mL/min. The BF and LV concentration was analyzed from calibration curve in BF (range 0–10 µg/mL, y=0.0655x+0.0003) and LV (range 0–10 µg/mL, y=0.3134x-0.0917), and the R level was 0.9993 and 0.9988, respectively.
Dispersibility in Ophthalmic Formulations
Three milliliter samples of mFBL-FC and nFBL-FC were added to 5 mL test tubes (total length 4 cm), and incubated in the dark at 20°C for 1 M. Images were obtained using a digital camera at the indicated time intervals. Fifty microliter samples of mFBL-FC and nFBL-FC were collected from the upper 90% of the solution,19,22 and the dispersibility of the samples was evaluated by measuring the concentration, particle size and NPs number by the HPLC and NANOSIGHT LM10 methods described above.
Corneal Toxicity of Ophthalmic Formulations
The immortalized human corneal epithelial cell line HCE-T cells were used in this study. The HCE-T cells was developed by Araki-Sasaki et al25 and the commercially HCE-T cell was purchased by Professor Araki-Sasaki. HCE-T cells were cultured in DMEM/F12 with heat-inactivated fetal bovine serum (5%), streptomycin (0.1 mg/mL), and penicillin (1000 IU/mL). 1×104 HCE-T cells were seeded in 96-well microplates (IWAKI, Chiba, Japan), and incubated for 24 h. Eye drops (100 µL) were added to the cell cultures, and the cells were stimulated for 30–120 s/eye drop.22 Then, the cells were washed with phosphate buffer and incubated in DMEM/F12 for 1 h. Following incubation, Cell Count Reagent SF (10 µL) was added DMEM/F12, the cells were incubated for an additional 1 h, and the absorbance at 490 nm was measured. Cell viability (%) was calculated in relation to the non-treatment group. In the in vivo evaluation using rabbits (n=6), the eye drops (30 µL) were repetitively instilled twice a day for 14 days (10:00 and 19:00). The damaged corneal area was dyed by the instillation of 1% fluorescein, and the corneal damage was observed by a TRC-50X (Topcon, Tokyo, Japan).
In vitro Transcorneal Penetration of Ophthalmic Formulations
The rabbits were euthanized by injecting a lethal dose of pentobarbital into the marginal ear vein, the removed corneas were removed, and set on a transcorneal cell methacrylate cell.19,22 The eye drop formulations and 10 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES) buffer (pH 7.4) consisting of 1 mM K2HPO4, 5.5 mM glucose, 136.2 mM NaCl, 1.7 mM CaCl2, and 5.3 mM KCl were added to the donor and reservoir chambers, respectively. The transcorneal penetration experiments were performed at 4°C (energy-dependent endocytosis-inhibited conditions26) and 37°C (normal conditions) for 6 h, and samples of the solution in the reservoir chamber were collected over time. FL, BF and LV concentrations in the samples were measured by the HPLC methods described above. The area under the drug concentration–time curve in the in vitro study (AUCpenetration) was determined according to the trapezoidal rule up to the last indomethacin concentration measurement point (90 min).19,22
In vivo Corneal Permeation of Ophthalmic Formulations
Rabbits were anesthetized with isoflurane and a topical anesthetic (Benoxil® ophthalmic solution 0.4%), and a 29 gauge injection needle connected to silicon tubing joined to a 25 µL microsyringe was inserted, and left in place to stabilize for 30 min. Following stabilization, the ophthalmic formulation (30 µL) was instilled into the eyes of the rabbits. After that, the samples of the aqueous humor (5 μL) were collected through the microsyringe over time, and the drug concentrations in the aqueous humor were determined by HPLC as described above. The area under the drug concentration–time curve in the aqueous humor (AUCAH) was determined according to the trapezoidal rule up to the last indomethacin concentration measurement point (90 min).19,22 In this study, 54 μM nystatin,27 40 μM dynasore,28 2 μM rottlerin,29 or 10 μM cytochalasinD27 were pre-instilled to inhibit caveolae-dependent endocytosis (CavME), clathrin-dependent endocytosis (CME, macropinocytosis (MP), or phagocytosis, respectively. All endocytosis inhibitors were dissolved in 0.5% DMSO, and 30 μL was instilled 3 times at 5 min intervals.
Statistical Analysis
Differences between mean values were analyzed with ANOVA followed by the Student’s t-test and Dunnett’s multiple comparison, with P<0.05 considered to be significant. The data are expressed as mean ± standard error (S.E.).
Results
Preparation of FL/BF/LV-Fixed Combination Eye Drops (FBL-FC) and Their Characteristics
First, we prepared nFBL-FC. The particle sizes of FL in mFBL-FC and CA-FL were 0.4–30 µm (mean particle size 3.98 µm) and 0.1–20 µm (mean particle size 2.13 µm), respectively. On the other hand, the particle size of FL in nFBL-FC was decreased by the bead mill treatment, so that the particle size of the FL in nFBL-FC was reduced to 40–150 nm from 0.4 to 30 µm (Figure 1). Next, we investigated the solubility, viscosity and zeta potential of FL in nFBL-FC (Figure 2). The solubility of the FL in nFBL-FC was 1.12-fold higher than that in mFBL-FC (Figure 2A). While no difference in viscosity was observed between nFBL-FC and mFBL-FC, the level was higher than that of CA-FL (Figure 2B). The zeta potential of FL in nFBL-FC was 1.13-fold that in mFBL-FC, and also significantly higher than that in CA-FL (Figure 2C). Obtaining a high dispersibility is an important factor in the development of nanodispersions, so we also evaluated the dispersibility of the FL-NPs in nFBL-FC (Figure 3). In the case of the mFBL-FC and CA-FL formulations, precipitation was observed immediately after the start of the experiment, while no precipitation or aggregation of FL-NPs was observed for 1 M in the case of the nFBL-FC formulation as no changes in particle size or amount were found (Figure 3).
Ocular BA of Three Drugs After the Instillation of nFBL-FC
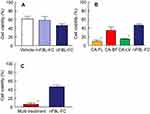
We evaluated the corneal toxicity of nFBL-FC (Figure 4). Although the viability of HCE-T cells multi-treated with three drugs (CA-FL, CA-BF and CA-LV) was 6.1% that of non-treated HCE-T cells, the viability of HCE-T cells treated with nFBL-FC was 46.4% that of non-treated HCE-T cells, indicating a significantly lower toxicity of nFBL-FC than the traditional multi-drug treatment. Moreover, the viability of HCE-T cells treated with nFBL-FC was 5.2-, 1.3-, or 3.2-fold those of HCE-T cells treated singly with CA-FL, CA-BF or CA-LV, respectively. In addition, no corneal wounds were observed in rabbits following the repetitive instillation of nFBL-FC twice a day for 14 days. Next, we evaluated the ocular BA of nFBL-FC using rabbits. Figure 5 shows the in vitro transcorneal penetration of nFBL-FC under normal conditions (37 °C). The transcorneal penetration of FL-NPs in corneas treated with nFBL-FC was significantly enhanced in comparison with CA-FL. In addition, the transcorneal penetrations of the BF and LV in nFBL-FC were also increased, with the AUCpenetration values of the BF and LV in nFBL-FC being 3.9- and 2.2-fold those of the corresponding CA-eye drops, respectively. Furthermore, we measured the FL, BF and LV concentrations in the aqueous humor of rabbits instilled with nFBL-FC or CA-eye drops (Figure 6). The AUCAH values for FL, BF and LV in the aqueous humor of rabbits after the instillation of the corresponding CA-eye drops were 222, 118, and 93 µM∙h, respectively, while the values in the aqueous humor of rabbits instilled with nFBL-CF were significantly higher at 4.9-, 1.8-, and 7.1-fold those of the CA-eye drops, respectively. We also measured the state of the FL in the aqueous humor of rabbits instilled with nFBL-CF by the Nanosight LM10. The FL detected in the aqueous humor existed only in the liquid state.
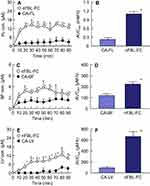
Effect of Energy-Dependent Endocytosis on the Corneal Penetration of nFBL-FC
It is important to elucidate the mechanism for the high ocular BA of nFBL-FC. In this study, we investigated whether the enhanced corneal permeation of the FL, BF and LV in nFBL-FC would change by the inhibition of energy-dependent endocytosis (Figs. 7 and 8). In the in vitro transcorneal penetration study using isolated rabbit corneas, the inhibition of energy-dependent endocytosis was induced by incubation at a cold temperature (4 °C).26 The penetration levels of FL, BF and LV in were attenuated under the 4 °C conditions to levels similar to those of the corresponding CA-eye drops (Figure 7). Moreover, we identified the endocytosis pathways involved in the increased BA in an in vivo study using endocytosis inhibitors (Figure 8). The AUCAH values of rabbits treated with rottlerin (MP inhibitor) or cytochalasin D (phagocytosis inhibitor) and nFBL-FC were similar to those of the control-treated group, while there was a tendency toward a decrease in the AUCAH of all three drugs in the rabbits treated with dynasore (CME inhibitor) and nFBL-FC. On the other hand, in rabbits treated with nystatin (CavME inhibitor) and nFBL-FC, there was a significant attenuation of the enhanced AUCAH of all three drugs; the AUCAH values for FL, BF and LV in the rabbits treated with nystatin and nFBL-FC were 24.7%, 55.0% and 50.0% of the control-treated group, respectively (Figure 8).
Discussion
The multi-instillation of three drugs (FL, BF and LV eye drops) has been used to manage the pain, discomfort and inflammation that occurs post-intraocular surgery. However, the multi-instillation of three eye drops causes corneal damage, and FL has the disadvantage of having a low ocular BA.10 Therefore, the design of new ophthalmic formulations of these three drugs has been anticipated. In this study, we designed nFBL-FC by a bead mill method, and compared the corneal toxicity and transcorneal penetration of nFBL-FC in comparison with the traditional CA-eye drops.
First, we evaluated the characteristics of the FL in nFBL-FC, including particle size, dispersibility, solubility, viscosity and zeta potential. The size of the FL particles in nFBL-FC was measured by the laser scattering method, dynamic scattering method and AFM, which revealed a particle size of 40 −150 nm (Figure 1). Further, the solubility of the FL in nFBL-FC was enhanced by bead mill treatment (Figure 2A) in comparison with the solubility of MPs. HPβCD shows an inclusion ability for drugs,24 and the inclusion ability for HPβCD and NPs was higher than for HPβCD and MPs.24 These factors may have enhanced the solubility of FL in this study. Only 0.0018% of the FL in nFBL-FC existed in liquid form, with the remainder existing as solid NPs (Figure 2A). The results show that preparation by bead mill treatment can produce fixed-combination eye drops containing three drugs starting with FL-NPs and a BF/LV solution (nFBL-FC). We found that the dispersibility of FL-NPs in nFBL-FC was enhanced in comparison with FL-MPs in mFBL-FC, and no precipitation or aggregation of the FL-NPs was observed for 1 M (Figure 3). It is known that a high viscosity and zeta potential are related to the dispersibility in dispersions containing NPs. The viscosity of nFBL-FC was 1.7 mPa∙s at 20 °C (Figure 2B), higher than that in water (0.9 mPa∙s) (Figure 2C). We considered whether the enhanced viscosity might be due to the addition of 0.5% MC, since the viscosity in 0.5% MC solution is approximately 1.4 mPa∙s. However, the viscosities and zeta potentials of nFBL-FC were similar to those of mFBL-FC (Figure 2B and C), suggesting that the decrease in particle size contributed most to the high dispersibility of nFBL-FC (Figure 3). Mori et al30 showed that adsorption to the surface of cyclodextrin prevents the cohesion of solid NPs, and we have also reported that the adsorption to the surface of HPβCD enhances the dispersibility of NPs, such as indomethacin.22 Therefore, we examined the dispersibility of nFBL-FC prepared without 5% HPβCD, and found it to be 63.1±3.5% (n=8) 1 M after bead mill treatment. These results show that nFBL-FC dispersions are protected against FL-NPs aggregation by HPβCD, resulting in high dispersibility.
Next, we investigated the corneal toxicity of nFBL-FC using HCE-T cells (Figure 4). No significant difference viability between HCE-T cells treated with nFBL-FC and CA-BF, and the viability was higher than that of cells treated with CA-FL or CA-LV (Figure 4B). The viability of cells treated with nFBL-FC was remarkably higher (7.7-fold) in comparison with cells multi-treated with the three CA-eye drops (FL, BF and LV). These results show that nFBL-FC may reduce corneal damage in post-surgical patients as well as the burden of instilling three types of eye drops. We also investigated whether the corneal penetration of drugs in nFBL-FC was increased in comparison with the individual CA-eye drops (Figs. 5 and 6). The corneal penetration of FL in nFBL-FC was significantly increased in comparison to CA-FL in both the in vitro and in vivo studies (Figures 5 and 6). It was reported that the drug/cyclodextrin complexes enhanced the ocular BA.11 Otherwise, our previous study using rabbit cornea showed that three energy-dependent endocytosis pathways (CavME, CME and MP) are related to the transcorneal penetration of 35–200 nm sized solid indomethacin NPs. In particular, the CavME is strongly involved.19 Moreover, the solid NPs of indomethacin taken up into the corneal epithelium are released on the aqueous humor side, resulting in an increase in corneal penetration.19 In this study, the enhanced FL transcorneal penetration was induced by energy-dependent endocytosis, since the corneal penetration of the FL in nFBL-FC was attenuated under 4 °C conditions,26 a temperature at which energy-dependent endocytosis is inhibited (Figure 7). These results support the previous reports for indomethacin NPs used as ocular DDS,19 and suggested that the enhanced corneal penetration of FL may due to energy-dependent endocytosis rather than drug/cyclodextrin complex. On the other hand, the penetrations of the BF and LV in nFBL-FC were also enhanced in both the in vitro (Figure 7) and in vivo (Figure 8) studies, and cold temperature treatment (4 °C) prevented their enhanced transcorneal penetrations as well, with no differences observed between nFBL-FC and the corresponding CA-eye drops (Figure 7). These results suggest that the corneal penetration of FL-NPs via energy-dependent endocytosis may promote the corneal uptake of the liquid drugs as well.
It is important to clarify the relationship between of high rates of corneal permeation and energy-dependent endocytosis. Therefore, we demonstrated the mechanism of corneal permeation using various endocytosis inhibitors. CavME, CME, MP and phagocytosis are the known energy-dependent forms endocytosis,31,32 and can be individually inhibited by nystatin, dynasore, rottlerin and cytochalasin D, respectively.33–36 In this study, these inhibitors were used to investigate the relationship between corneal permeation and energy-dependent endocytosis (Figure 8). The corneal permeation of the FL, BF and LV in nFBL-FC was attenuated by treatment with nystatin, with AUCAH of the drugs in rabbits treated with nystatin similar to those of the corresponding CA-eye drops (Figures 6 and 8). It is known that the particle size corresponding to CavME is about 80 nm,31,32 and the particle size of the FL in nFBL-FC is 40–150 nm. In addition, FL-NPs are dissolved as they permeate the cornea, since no solid FL NPs are detected in the aqueous humor (only liquid FL is detected). Taken together, we hypothesize that CavME is responsible for the enhanced uptake of FL-NPs into the cornea, that the FL-NPs are dissolved in the cornea, and only liquid FL is released into the aqueous humor. In addition, the activation of CavME by solid NPs may induce the increase in the corneal permeation of the liquid BF and LV in the nFBL-FC as well (Figure 8). Further studies are needed to clarify the mechanism for the activation of energy-dependent endocytosis in corneas instilled with FL-NPs. In addition, it is important to evaluate whether nFBL-FC inhibits the persistent inflammation and cystoid macular edema that occur after cataract extraction with posterior chamber IOL implantation. In a future work, we plan to investigate the therapeutic effect of nFBL-FC using rabbits undergoing cataract extraction with posterior chamber IOL implantation.
Conclusion
We prepared fixed-combination eye drops based on FL-NPs and BF/LV solution (nFBL-FC), and demonstrated their low corneal toxicity and high corneal penetration in comparison with traditional treatments using CA-eye drops. Moreover, the FL-NPs are taken up into the cornea by energy-dependent endocytosis (CavME pathway), dissolve in the corneal stromal side, and are released into the aqueous humor as liquid FL. In addition, a CavME inhibitor also attenuates the corneal penetration of the liquid drugs (BF and LV) as well. We hypothesize that the activation of CavME by FL-NPs may also promote the corneal uptake of BF and LV solution (liquid drugs). Our studies provide the first report that the corneal penetration of both of solid- and liquid-drugs is enhanced by the design of a fixed combination of solid NPs and solution. In conclusion, we showed that nFBL-FC improved drug corneal penetration and ocular BA in the both of NPs and solution.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Dua HS, Attre R. Treatment of post-operative inflammation following cataract surgery-a review. Eur Ophth Rev. 2012;6(02):98–103. doi:10.17925/EOR.2012.06.02.98.
2. El-Harazi SM, Feldman RM. Control of intra-ocular inflammation associated with cataract surgery. Curr Opin Ophthalmol. 2001;12(1):4–8. doi:10.1097/00055735-200102000-00002.
3. Kessel L, Tendal B, Jorgensen KJ, et al. Post-cataract prevention of inflammation and macular edema by steroid and nonsteroidal anti-inflammatory eye drops: a systematic review. Ophthalmology. 2014;121(10):1915–1924. doi:10.1016/j.ophtha.2014.04.035.
4. Wielders LHP, Lambermont VA, Schouten JSAG, et al. Prevention of cystoid macular edema after cataract surgery in nondiabetic and diabetic patients: a systematic review and meta-analysis. Am J Ophthalmol. 2015;160(5):968–981. doi:10.1016/j.ajo.2015.07.032.
5. Rose P. Management strategies for acute infective conjunctivitis in primary care: a systematic review. Expert Opin Pharmacother. 2007;8(12):1903–1921. doi:10.1517/14656566.8.12.1903.
6. DeCroos FC, Afshari NA. Perioperative antibiotics and anti-inflammatory agents in cataract surgery. Curr Opin Ophthalmol. 2008;19(1):22–26. doi:10.1097/ICU.0b013e3282f30577.
7. Chen PQ, Han XM, Zhu YN, Xu J. Comparison of the anti-inflammatory effects of fluorometholone 0.1% combined with levofloxacin 0.5% and tobramycin/dexamethasone eye drops after cataract surgery. Int J Ophthalmol. 2016;9:1619–1623. doi:10.18240/ijo.2016.11.13.
8. Keating GM. Levofloxacin 0.5% ophthalmic solution: a review of its use in the treatment of external ocular infections and in intraocular surgery. Drugs. 2009;69(9):1267–1286. doi:10.2165/00003495-200969090-00009.
9. Nagai N, Yoshioka C, Tanino T, Ito Y, Okamoto N, Shimomura Y. Decrease in corneal damage due to benzalkonium chloride by the addition of mannitol into timolol maleate eye drops. J Oleo Sci. 2015;64(7):743–750. doi:10.5650/jos.ess14275
10. Awan MA, Agarwal PK, Watson DG, McGhee CNJ, Dutton GN. Penetration of topical and subconjunctival corticosteroids into human aqueous humour and its therapeutic significance. Br J Ophthalmol. 2009;93(6):708–713. doi:10.1136/bjo.2008.154906.
11. Loftsson T, Stefansson E. Cyclodextrins and topical drug delivery to the anterior and posterior segments of the eye. Int J Pharm. 2017;531(2):413–423. doi:10.1016/j.iipharm.2017.04.010
12. Fangueiro JF, Veiga F, Silva AM, Souto EB. Ocular drug delivery-new strategies for targeting anterior and posterior segments of the eye. Curr Pharm Des. 2016;22(9):1135–1146. doi:10.2174/1381612822666151216145900.
13. Hagigit T, Abdulrazik M, Orucov F, et al. Topical and intravitreous administration of cationic nanoemulsions to deliver antisense oligonucleotides directed towards VEGF KDR receptors to the eye. J Control Release. 2010;145(3):297–305. doi:10.1016/j.jconrel.2010.04.013.
14. Hironaka K, Inokuchi Y, Fujisawa T, et al. Edaravone-loaded liposomes for retinal protection against oxidative stress-induced retinal damage. Eur J Pharm Biopharm. 2011;79(1):119–125. doi:10.1016/j.ejpb.2011.01.019.
15. Cholkar K, Patel A, Vadlapudi AD, Mitra AK. Novel nanomicellar formulation approaches for anterior and posterior segment ocular drug delivery. Recent Pat Nanomed. 2012;2(2):82–95. doi:10.2174/1877912311202020082.
16. Sun D, Maeno H, Gujrati M, et al. Self-assembly of a multifunctional lipid with core-shell dendrimer DNA nanoparticles enhanced efficient gene delivery at low charge ratios into RPE cells. Macromol Biosci. 2015;15(12):1663–1672. doi:10.1002/mabi.201500192.
17. Patel A, Cholkar K, Agrahari V. Mitra AK Ocular drug delivery systems: an overview. World J Pharmacol. 2013;2(2):47–64. doi:10.5497/wjp.v2.i2.47.
18. Shen J, Wang Y, Ping Q, Xiao Y, Huang X. Mucoadhesive effect of thiolated PEG stearate and its modified NLC for ocular drug delivery. J Control Release. 2009;137(3):217–223. doi:10.1016/j.jconrel.2009.04.021.
19. Nagai N, Ogata F, Otake H, Nakazawa Y, Kawasaki N. Energy-dependent endocytosis is responsible for drug transcorneal penetration following the instillation of ophthalmic formulations containing indomethacin nanoparticles. Int J Nanomedicine. 2019;14:1213–1227. doi:10.2147/IJN.S196681.
20. Nagai N, Yamaoka S, Fukuoka Y, et al. Enhancement in corneal permeability of dissolved carteolol by its combination with magnesium hydroxide nanoparticles. Int J Mol Sci. 2018;19(1):282. doi:10.3390/ijms19010282.
21. Nagai N, Ogata F, Otake H, et al. Co-instillation of nano-solid magnesium hydroxide enhances corneal permeability of dissolved timolol. Exp Eye Res. 2017;165:118–124. doi:10.1016/j.exer.2017.10.002.
22. Nagai N, Ito Y, Okamoto N, Shimomura Y. A nanoparticle formulation reduces the corneal toxicity of indomethacin eye drops and enhances its corneal permeability. Toxicology. 2014;319:53–62. doi:10.1016/j.tox.2014.02.012.
23. Kawazu K, Midori Y, Ota A. Cultured rabbit corneal epithelium elicits levofloxacin absorption and secretion. J Pharm Pharmacol. 1999;51(7):791–796. doi:10.1211/0022357991773159.
24. Nagai N, Minami M, Deguchi S, Otake H, Sasaki H, Yamamoto N. An in situ gelling system based on methylcellulose and tranilast solid nanoparticles enhances ocular residence time and drug absorption into the cornea and conjunctiva. Front Bioeng Biotechnol. 2020;8:764. doi:10.3389/fbioe.2020.00764.
25. Araki-Sasaki K, Ohashi Y, Sasabe T, et al. An SV40-immortalized human corneal epithelial cell line and its characterization. Invest Ophthalmol Vis Sci. 1995;36:614–621.
26. He Z, Liu K, Manaloto E, et al. Cold atmospheric plasma induces ATP-dependent endocytosis of nanoparticles and synergistic U373MG cancer cell death. Sci Rep. 2018;8(1):5298. doi:10.1038/s41598-018-23262-0.
27. Mäger I, Langel K, Lehto T, Eiríksdóttir E, Langel U. The role of endocytosis on the uptake kinetics of luciferin-conjugated cell-penetrating peptides. Biochim Biophys Acta. 2012;1818(3):502–511. doi:10.1016/j.bbamem.2011.11.020.
28. Malomouzh AI, Mukhitov AR, Proskurina SE, Vyskocil F, Nikolsky EE. The effect of dynasore, a blocker of dynamin-dependent endocytosis, on spontaneous quantal and non-quantal release of acetylcholine in murine neuromuscular junctions. Dokl Biol Sci. 2014;459(1):330–333. doi:10.1134/S0012496614060052.
29. Hufnagel H, Hakim P, Lima A, Hollfelder F. Fluid phase endocytosis contributes to transfection of DNA by PEI-25. Mol Ther. 2009;17(8):1411–1417. doi:10.1038/mt.2009.121.
30. Mori K, Yoshioka N, Kondo Y, Takeuchi T, Yamashita H. Catalytically active, magnetically separable, and water-soluble FePt nanoparticles modified with cyclodextrin for aqueous hydrogenation reactions. Green Chem. 2009;11(9):1337–1342. doi:10.1039/B905331J.
31. Wang J, Byrne JD, Napier ME, Desimone JM. More effective nanomedicines through particle design. Small. 2011;7(14):1919–1931. doi:10.1002/smll.201100442.
32. Rappoport JZ. Focusing on clathrin-mediated endocytosis. Biochem J. 2008;412(3):415–423. doi:10.1042/BJ20080474.
33. Raghu H, Sharma-Walia N, Veettil MV, Sadagopan S, Chandran B. Kaposi’s sarcoma-associated herpesvirus utilizes an actin polymerization-dependent macropinocytic pathway to enter human dermal microvascular endothelial and human umbilical vein endothelial cells. J Virol. 2009;83(10):4895–4911. doi:10.1128/JVI.02498-08.
34. Dausend J, Musyanovych A, Dass M, et al. Uptake mechanism of oppositely charged fluorescent nanoparticles in HeLa cells. Macromol Biosci. 2008;8(12):1135–1143. doi:10.1002/mabi.200800123.
35. Vercauteren D, Vandenbroucke RE, Jones AT, et al. The use of inhibitors to study endocytic pathways of gene carriers: optimization and pitfalls. Mol Ther. 2010;18(3):561–569. doi:10.1038/mt.2009.281.
36. Qualmann B, Kessels MM, Kelly RB. Molecular links between endocytosis and the actin cytoskeleton. J Cell Biol. 2000;150(5):F111–F116. doi:10.1083/jcb.150.5.f111.
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.