Back to Journals » International Journal of Nanomedicine » Volume 10 » Issue 1
External beam radiotherapy synergizes 188Re-liposome against human esophageal cancer xenograft and modulates 188Re-liposome pharmacokinetics
Authors Chang C, Liu S, Chi C, Yu H, Chang T, Tsai T, Lee T, Chen Y
Received 4 January 2015
Accepted for publication 18 March 2015
Published 19 May 2015 Volume 2015:10(1) Pages 3641—3649
DOI https://doi.org/10.2147/IJN.S80302
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Thomas Webster
Chih-Hsien Chang,1,2 Shin-Yi Liu,3 Chih-Wen Chi,3 Hsiang-Lin Yu,1 Tsui-Jung Chang,1 Tung-Hu Tsai,4 Te-Wei Lee,1 Yu-Jen Chen3–5
1Isotope Application Division, Institute of Nuclear Energy Research, Taoyuan, Taiwan; 2Department of Biomedical Imaging and Radiological Sciences, National Yang-Ming University, 3Department of Medical Research MacKay Memorial Hospital, 4Institute of Traditional Medicine, National Yang-Ming University, 5Department of Radiation Oncology, MacKay Memorial Hospital, Taipei, Taiwan
Abstract: External beam radiotherapy (EBRT) treats gross tumors and local microscopic diseases. Radionuclide therapy by radioisotopes can eradicate tumors systemically. Rhenium 188 (188Re)-liposome, a nanoparticle undergoing clinical trials, emits gamma rays for imaging validation and beta rays for therapy, with biodistribution profiles preferential to tumors. We designed a combinatory treatment and examined its effects on human esophageal cancer xenografts, a malignancy with potential treatment resistance and poor prognosis. Human esophageal cancer cell lines BE-3 (adenocarcinoma) and CE81T/VGH (squamous cell carcinoma) were implanted and compared. The radiochemical purity of 188Re-liposome exceeded 95%. Molecular imaging by NanoSPECT/CT showed that BE-3, but not CE81T/VGH, xenografts could uptake the 188Re-liposome. The combination of EBRT and 188Re-liposome inhibited tumor regrowth greater than each treatment alone, as the tumor growth inhibition rate was 30% with EBRT, 25% with 188Re-liposome, and 53% with the combination treatment at 21 days postinjection. Combinatory treatment had no additive adverse effects and significant biological toxicities on white blood cell counts, body weight, or liver and renal functions. EBRT significantly enhanced the excretion of 188Re-liposome into feces and urine. In conclusion, the combination of EBRT with 188Re-liposome might be a potential treatment modality for esophageal cancer.
Keywords: Radionuclide therapy, liposome, teletherapy, biodistribution
Introduction
Esophageal cancer is a cancer with poor prognosis, with an average 5-year survival rate less than 25%.1 Locally advanced esophageal carcinoma is known to be refractory to treatment of a single modality. Patients with unresectable or medically inoperable disease are usually treated with concurrent radiotherapy and chemotherapy for preoperative or definitive treatment.2–4 Although various chemotherapy regimens are available, esophageal cancer carries a very poor prognosis, with a mean survival time of less than 8.1 months.5 Currently, neoadjuvant treatment (such as capecitabine and cisplatin with concurrent radiotherapy (RT) followed by esophagectomy) is considered the effective treatment for esophageal cancer,6,7 leading to complete pathologic response at the time of surgical resection and improved local tumor control and eradication of micrometastases.8 Clearly, the development of novel and potent therapeutics in/not in combination with RT to improve both local and distant tumor control in esophageal cancer is an urgent task.
External beam radiotherapy (EBRT) is the most commonly used modality of RT for local control of cancer. EBRT can deliver high-energy radiation beams to cover both gross tumors and potential microscopic tumor cells in the vicinity of a tumor. Radionuclide therapy (RNT), another form of RT using radioisotopes emitting short-distance radiation, can directly destroy local and systemic gross tumors inside the torso.9 RNT uses radiolabeled molecules referred to as “therapeutic radiopharmaceuticals” and is a unique treatment modality lying between chemotherapy and EBRT. The goal of RNT is to kill tumor cells selectively by delivering high radiation doses to a specific target while minimizing damage to normal cells.9 These carriers include nanoparticles, liposomes, and water-soluble polymers to aid radioisotopes to selectively target and accumulate at cancer sites, thereby improving cancer detection and therapeutic effectiveness.10
Rhenium-188 (188Re) is a radionuclide designed for dual imaging and therapeutic applications because of its short physical half-life of 16.9 hours with 155 keV gamma emission (for molecular imaging) and 2.12 MeV emission (for therapeutic RT) and a maximum tissue penetration range of 11 mm.11 The high efficiency, stability, and convenience of 188Re-liposome have been demonstrated in the treatment of cancer.12 Although 188Re-liposome and radiochemotherapeutic drugs, for example, 188Re-doxorubicin-liposome, have been studied in many experiments, the application potential of 188Re-liposome in cancer therapy remains to be developed.13–17 Our previous studies have investigated the biodistribution, pharmacokinetics, and NanoSPECT/CT imaging after systematic administration of 188Re-liposome by intraperitoneal and intravenous injection methods in colon carcinoma ascites and solid-tumor animal models.13,14,18–22 The 188Re-liposome has been approved for Phase I clinical trial in the treatment of metastatic late-stage patients. The Phase I clinical trial of 188Re-liposome is still ongoing in Taiwan.
Although 188Re-liposome has a comparative therapeutic efficacy with chemotherapeutic drug 5-FU in colon carcinoma ascites, we found that 188Re-liposome possesses a better therapeutic efficacy in both lung-metastatic and solid-tumor animal models.18,20–22 No documents using 188Re-liposome as RNT or RNT combined with EBRT to assess the therapeutic efficacy in esophageal cancer have been published yet. EBRT combined with RNT may enhance the cytotoxicity in tumor cells and reduce normal tissue damage, using the advantages of conformal EBRT and the selective targeting of tumors using RNT. The efficacy and radiation dose of EBRT or RNT alone and EBRT combined with RNT have not been determined yet. It is important to clarify the difference in characteristics between EBRT and RNT, and the impact on the combination of EBRT and RNT.
In the present study, we examined the combinatory effect of EBRT and 188Re-liposome for treatment of esophageal cancer. The impact of EBRT on the biodistribution of 188Re-liposome was also assessed.
Materials and methods
Xenografts of esophageal cancer
Human esophageal cancer cell lines, CE81T/VGH (squamous cell carcinoma) and BE-3 (adenocarcinoma) cells, were kindly provided by Professor Hu (Veteran’s General Hospital, Taipei, Taiwan) and purchased from the American Type Culture Collection (Manassas, VA, USA), respectively. These two esophageal cancer cell lines were maintained in DMEM (Dulbecco’s Modified Eagle’s Medium) (GIBCO BRL, Grand Island, NY, USA) supplemented with NaHCO3 (10 mmol/L), HEPES (20 mmol/L), 10% heat-inactivated fetal bovine serum (Hyclone, Logan, UT, USA), 2 mM L-glutamine, 1 mM nonessential amino acid, and 1 mM sodium pyruvate and incubated at 37°C in 5% CO2. Four-week-old, male BALB/c nude mice were obtained from the National Laboratory Animal Center of Taiwan (Taipei, Taiwan) and bred in a specific pathogen-free animal facility. All experimental protocols involving animals were reviewed and approved by the Institutional Animal Experimentation Committee of Mackay Memorial Hospital and the Institute of Nuclear Energy Research (INER). All animal care and husbandry was conducted in accordance with the A Guidebook for the Care and Use of Laboratory Animals (Version 3 2010). Human esophageal cancer cells were implanted in nude mice by subcutaneous injections of 0.1 mL PBS containing a cell suspension of 5×106 cells into the right hind limb. Fourteen days after inoculation, the mice developed tumors of approximately 150–200 mm3 in size and were subjected to further experiments.
Preparation and characterization of 188Re-liposome
Pegylated liposomes (Nano-X, Taiwan Liposome Company, Taipei, Taiwan) were prepared according to the method described by Tseng et al.23 The lipid composition of liposomes contains hydrogen soybean phosphatidylchloine (HSPC), cholesterol, polyethylene glycol (1,2-distearoyl-sn-glycero-3-phosphatidylethanolamine, PEG-DSPE; molar ratio 3:2:0.3), and ammonium sulfate solution with 250 mM (NH4)2SO4, pH 5.0 in the inner water phase. Pegylated nanoliposomes have an average particle size of approximately 82.59 nm and contain 13.16 μmol/mL phospholipids. The labeling method of 188Re-liposome was as previously described.13,14 Prior to labeling, 188Re was conjugated with N, N-bis(2-mercapatoethly)-N′, N′-diethylenediamine (BMEDA). Briefly, 188Re was eluted from an alumina-based 188W/188Re generator (IRE Company, Brussels, Belgium) using normal saline to provide solutions of carrier-free 188Re as sodium perrhenate. 188Re-BMEDA was generated by the conjugation of free 188Re and BMEDA (ABX, Radeberg, Germany) in the presence of sodium gluconate (Sigma-Aldrich, Bangalore, India) and stannous chloride dihydrate (Merck, Darmstadt, Germany). Three milligrams of BMEDA was mixed with 0.34 mol/L of sodium gluconate dissolved in a 10% acetate solution; 0.02 mol/L stannous chloride dihydrate was added and incubated at 80°C for 1 hour. The labeling efficiency of 188Re-BMEDA complexes was analyzed by silica gel–impregnated glass fiber sheets using normal saline as the developer (Rf values: 188Re, 0.8–1.0; 188Re-BMEDA, 0.0–0.2). Before labeling with liposomes, 188Re-BMEDA was adjusted to a pH of 7.0 with 2 N NaOH. Nano-X pegylated liposomes were added to 188Re-BMEDA and incubated at 60°C for 30 minutes to form 188Re-BMEDA-liposome complexes. The 188Re-liposomes were separated from free 188Re-BMEDA using a PD-10 column (GE Healthcare, Piscataway, NJ, USA) and eluted with normal saline. The encapsulation efficiency was determined using the activity in pegylated liposomes after separation divided by the total activity before separation.
Delivery of external beam radiotherapy and combinatory regimen
Before irradiation, tumor-bearing mice were anesthetized by intramuscular injections of ketamine (100 mg/kg)/xylazine (10 mg/kg body weight). EBRT with a 6 MeV electron beam was delivered by a linear accelerator (Clinac 1800, Varian Associates, Inc., Palo Alto, CA, USA) with a dose rate of 2.4 Gy/min and, locally, irradiation was performed within tumor region (right hind limb). Before the combination of EBRT and RNT, optimal dosage of each treatment was estimated to suppress less than 50% of tumor growth (3 Gy for ERBT and 13.3 MBq [360 μCi] for RNT, data not shown). After 2 hours of transport time, RNT was performed by intravenous injection of 188Re-liposome.
NanoSPECT/CT analysis
Tumor-bearing mice were intravenously injected with 18.5 MBq (500 μCi) of 188Re-liposome, and images were acquired using the NanoSPECT/CT scanner system (NanoSPECT/CT PLUS, Mediso, Alsotorokvesz, Budapest, Hungary). Before scanning, mice were anesthetized with 1.5% isoflurane, and NanoSPECT/CT imaging was performed at 1, 4, and 24 hours after injection of 188Re-liposome. The imaging acquisition was accomplished using 70 frames at 40 seconds per frame, and the energy windows were 155 keV ±20% and 64 keV ±20%. The SPECT imaging was followed by CT image acquisition with the animal in exactly the same position (X-ray source: 45 kV, 0.1 mA; 180 projections). For image reconstruction, HiSPECT and Nucline software were used for the SPECT and CT images, respectively. The InVivoScrope software was used for the fusion of SPECT and CT images.
The biodistribution of 188Re-liposome
Nude mice bearing esophageal cancer cells were intravenously injected with 2.22 MBq (60 μCi) of 188Re-liposome with and without EBRT. Experimental animals were sacrificed by CO2 asphyxiation and organs of interest were removed, washed, and weighed at 1, 4, 24, and 48 hours after injection. The radioactivity of 188Re-liposome was detected by using the auto-gamma counter (Packard Cobra II, Packard-Canberra, Frankfurt, Germany), and the uptake of 188Re-liposome in each organ was expressed as the percentage of injected dose per gram of tissue (% ID/g).
Tumor growth evaluation
Fourteen days after inoculation, tumor-bearing mice were divided randomly into five groups with five mice per group, and two groups (injected with normal saline or with liposomes) were used as the controls. The other three groups of mice were treated with EBRT (ionizing radiation, 3 Gy), RNT (188Re-liposome, 13.3 MBq [360 μCi]), and EBRT and RNT combinations (IR + 188Re-liposome). Tumor size was recorded twice a week using a caliper to measure tumor growth, and tumor volumes were estimated according to the formula 0.5ab2, where a is the largest and b is the smallest perpendicular diameter.24 The tumor growth delay was defined as the subtraction of days required for three fold tumor volume growth between treated and untreated groups.
Toxicity assessment
Toxicity was monitored twice a week by the changes in mouse weight and immunological and hematological indicators. Immunological toxicity was observed from the counts of white blood cells (WBC) using the retro-orbital blood sampling method in a Hemavet blood analyzer (Drew Scientific, Oxford, CT). Hematological toxicities were examined by the detection of alanine aminotransferase (ALT) and creatinine (CRE), measuring the functions of liver and kidneys, respectively. The level of ALT and CRE were freshly measured by a Fuji Dri-Chem 3500 machine (Fujifilm Medical System, Tokyo, Japan).
Statistics
All values were expressed as mean ± standard deviation. Statistical comparisons were performed using Student’s t-test or one-way analysis of variance (ANOVA). The difference was considered significant for P<0.05. Data analysis was performed using SPSS software version 17.0 (Chicago, IL, USA).
Results
Labeling efficiency of 188Re-liposome
The labeling efficiency of 188Re and BMEDA was measured by instant thin-layer chromatography on silica gel–impregnated glass fiber sheets (ITLC-SG), and the result showed that the purity of 188Re-BMEDA was 96.7%±5.8% (Figure 1), as the radioactivity complex (188Re-BMEDA) remained at the origin (Figure 1B), whereas free 188Re migrated in the strip (Figure 1A). The encapsulation efficiency of 188Re-BMEDA-liposome was 65.7%±1.6%, and the radiochemical purity of 188Re-liposome exceeded 95% after purification with PD-10 columns. The average particle size of 188Re-liposome was similar to that before 188Re-BMEDA encapsulation.
NanoSPECT/CT imaging of 188Re-liposome
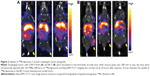
To discriminate the uptake of 188Re-liposome in two major cell types of esophageal cancer, we examined the uptake of 188Re-liposome by NanoSPECT/CT imaging in two human esophageal cancer xenografts. For molecular imaging, NanoSPECT/CT scan results showed that the uptake of 188Re-liposome was present in esophageal adenocarcinoma BE-3-bearing mice, but not in squamous cell carcinoma CE81T/VGH xenografts (Figure 2).
Impact of EBRT on the biodistribution of 188Re-liposome
The biodistribution profile of 188Re-liposome in mice bearing esophageal xenografts showed high uptake levels in the tissues of the liver, spleen, kidney, and bone marrow. The 188Re-liposome distribution patterns with and without EBRT displayed no significant differences in tumors, major organs, and the majority of normal tissues as demonstrated in Table 1. Our data indicated that EBRT significantly enhanced the excretion of 188Re-liposome into urine at 24 and 48 hours and into feces at 1 hour. To validate the effect of IR on 188Re-liposome excretion, dose-dependent irradiation (3, 6, 9 Gy) was delivered to BE-3 tumor xenografts followed by 188Re-liposome intravenous injection (Figure 3). The result confirmed that the fecal excretion of 188Re-liposome was significantly enhanced by irradiation without significant influence on the uptake of 188Re-liposome by tumor and liver.
Therapeutic effects and toxicity of EBRT and 188Re-liposome
EBRT and 188Re-liposome treatment exhibited moderate inhibitory effects on esophageal BE-3 xenografts. The combination of EBRT and 188Re-liposome inhibited tumor growth greater than each treatment alone. The tumor growth inhibition rate was 30% with EBRT, 25% with 188Re-liposome, and 53% with the combination of the two at 21 days postinjection (Figure 4). The three fold increase in tumor size was delayed 8.3 days with EBRT, 8.3 days with 188Re-liposome, and 12.4 days with the combination of the two. Both tumor growth inhibition and delayed days of three fold increase in tumor size in the combination group were significantly different from single treatment or control groups at 21 days postinjection (P<0.05). The combinatory treatment had no additive adverse effects on WBC counts, body weight, and liver and renal function (Figure 5). The values of detected marker in the combination or single treatment groups all fell within the normal range of WBC (normal range in mouse: 6 K–15 K/μL), ALT (normal range in mouse: 17–77 U/L), and CRE (normal range in mouse: 0.2–0.9 mg/dL). In mice that received EBRT and EBRT plus 188Re-liposome at the experimental dosage, no significant abnormality in the hemogram and biochemistry profile were noted, indicating that the combination of EBRT and 188Re-liposome had the potential of being a safe therapeutic strategy for human esophageal cancer.
Discussion
The current standard of treatment for locally advanced or unresectable esophageal cancer is concurrent chemoradiation therapy (CCRT). The long-term results of RTOG 85-01 trials show a survival benefit of CCRT in comparison with RT alone.25 Van Hagen et al26 found neoadjuvant chemoradiotherapy improved overall survival of patients with resectable esophageal cancer compared to the group with surgery alone. The most common toxic effects after CCRT were leukopenia (6%), neutropenia (2%), anorexia (5%), and fatigue (3%). Bass et al27 revealed that neoadjuvant chemoradiotherapy significantly provided survival advantage and risk reduction in lymph-node metastasis over surgical monotherapy for patients with esophageal adenocarcinoma. These reports supported that CCRT could be the definitive treatment or a neoadjuvant treatment prior to major surgery. However, the 5-year survival rate of locally advanced esophageal cancer remains less than 30% after aggressive treatment by CCRT.28 The persistence of local disease was the greatest cause of treatment failures.2 To address this clinical issue in a translational research model, we demonstrated better tumor control by using a combination of EBRT and 188Re-liposome against primary tumors. The major pattern of failure for treatment of esophageal cancer is local recurrence and distant metastasis. Thus, the next clinical issue to be examined is the effect of this combination regimen on the distant metastasis of esophageal cancer in experimental animal models.
Nanocarrier delivery systems have been shown to have enhanced imaging and therapeutic efficacy by targeted delivery of drugs to the tumor site and by reducing their toxic side effects.29–31 Major advantages of nanocarriers are that they can be prepared in sizes <100 nm and ensuring minimal drug or radionuclide leaking out from the carriers during circulation to enhance passive targeting and to increase the localization of drugs and radionuclides in the tumor through enhanced permeability and retention (EPR) effect to the leaky tumor tissues, and nanoliposomes have been widely studied as an important carrier in controlling the localization and concentration of drugs to improve the pharmaceutical and therapeutic properties of drug administration by EPR effect.32,33 Pegylated liposomes can evade the reticuloendothelial system and remain in the circulatory system for prolonged periods, resulting in sufficient tumor targeting and efficacy in vivo.34,35
External beam radiation has been reported as a method that enhances nanoliposome uptake in the tumor region due to radiation-induced inflammation.36 The increased uptake of nanoliposomes was detected at 24 hours following external beam radiation. The result suggests that combining modality therapy with external beam radiation and nanoliposome chemotherapy may be more effective for the treatment of tumors than administering these therapies sequentially.36 In our study, the effect of EBRT on the accumulation of 188Re-liposome in tumors was not found to be statistically significant (Table 1). It might be due to the deficiency of the dose of EBRT (3 Gy) as well as the time interval (2 hours) between the application of EBRT and 188Re-liposome, which may be too short to enhance permeability and retention. Since the combination of EBRT and 188Re-liposome increased therapeutic efficacy compared to EBRT and 188Re-liposome treatment alone, the uptake of 188Re-liposome on esophageal adenocarcinoma BE-3 cells is likely to play a role in enhancing radiation-induced toxicity. Further investigation will need to be done to confirm or refute these findings. The effect of EBRT on the biodistribution of 188Re-liposome in this study, especially on the secretion of isotopes into urine and feces, is unique. It may indicate a better secretion profile after treatment. Whether this effect would damage the lower urinary tract or the lower gastrointestinal tract remains to be elucidated.
To control local tumors, regional lymphatics, and distant metastasis simultaneously, our design was to combine EBRT and 188Re-liposome for the treatment of esophageal cancer, a malignancy with a high risk of recurrence and metastasis. Our results indicate that the combinatory regimen may have additive effects against the growth of human esophageal adenocarcinoma xenografts without an increase in toxicity (Figure 5).
Conclusion
In this study, it was found that human esophageal adenocarcinoma BE-3 cells, but not squamous cell carcinoma CE81T/VGH, showed specific uptake of 188Re-liposome. The 188Re-liposome combined with EBRT could have therapeutic efficacy on tumor growth control in BE-3 cells and might modulate the fecal and urinary excretions of 188Re-liposome. The combination of EBRT with 188Re-liposome might be a potential treatment modality for esophageal cancer by enabling molecular imaging, image-guided treatment, and comprehensive radiation therapy coverage simultaneously.
Acknowledgments
This study was supported by grants NSC 100-2623-E-195-NU, 101-2623-E-195-NU and 102-2623-E-195-NU from the National Science Council, Taiwan, and grant MMH-E-102-13 from Mackay Memorial Hospital, Taipei, Taiwan. We presented an earlier version of the abstract in a poster at the 23rd Biennial Congress of the European Association for Cancer Research in Munich (Germany) in 2014.
Disclosure
The authors report no conflicts of interest in this work.
References
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ. Cancer statistics, 2009. CA Cancer J Clin. 2009;59(4):225–249. | ||
Cooper JS, Guo MD, Herskovic A, et al. Chemoradiotherapy of locally advanced esophageal cancer: long-term follow-up of a prospective randomized trial (RTOG 85-01). Radiation Therapy Oncology Group. JAMA. 1999;281(17):1623–1627. | ||
Tepper J, Krasna MJ, Niedzwiecki D, et al. Phase III trial of trimodality therapy with cisplatin, fluorouracil, radiotherapy, and surgery compared with surgery alone for esophageal cancer: CALGB 9781. J Clin Oncol. 2008;26(7):1086–1092. | ||
Liu HC, Hung SK, Huang CJ, et al. Esophagectomy for locally advanced esophageal cancer, followed by chemoradiotherapy and adjuvant chemotherapy. World J Gastroenterol. 2005;11(34):5367–5372. | ||
Chiarion-Sileni V, Corti L, Ruol A, et al. Phase II trial of docetaxel, cisplatin and fluorouracil followed by carboplatin and radiotherapy in locally advanced oesophageal cancer. Br J Cancer. 2007;96(3): 432–438. | ||
Wahba HA, El-Hadaad HA, Abd-Ellatif EA. Neoadjuvant concurrent chemoradiotherapy with capecitabine and oxaliplatin in patients with locally advanced esophegeal cancer. Med Oncol. 2012;29(3): 1693–1698. | ||
Koo DH, Park SI, Kim YH, et al. Phase II study of use of a single cycle of induction chemotherapy and concurrent chemoradiotherapy containing capecitabine/cisplatin followed by surgery for patients with resectable esophageal squamous cell carcinoma: long-term follow-up data. Cancer Chemother Pharmacol. 2012;69(3):655–663. | ||
Swisher SG, Hofstetter W, Wu TT, et al. Proposed revision of the esophageal cancer staging system to accommodate pathologic response (pP) following preoperative chemoradiation (CRT). Ann Surg. 2005;241(5):810–817; discussion 817–820. | ||
Dash A, Knapp FF, Pillai MR. Targeted radionuclide therapy – an overview. Curr Radiopharm. 2013;6(3):152–180. | ||
Mitra A, Nan A, Line BR, Ghandehari H. Nanocarriers for nuclear imaging and radiotherapy of cancer. Curr Pharm Des. 2006;12(36): 4729–4749. | ||
Hafeli U, Tiefenauer LX, Schbiger PA, Weder HG. A lipophilic complex with 186Re/188Re incorporated in liposomes suitable for radiotherapy. Int J Rad Appl Instrum B. 1991;18(5):449–454. | ||
Bao A, Goins B, Klipper R, Negrete G, Phillips WT. 186Re-liposome labeling using 186Re-SNS/S complexes: in vitro stability, imaging, and biodistribution in rats. J Nucl Med. 2003;44(12):1992–1999. | ||
Chen LC, Chang CH, Yu CY, et al. Biodistribution, pharmacokinetics and imaging of (188)Re-BMEDA-labeled pegylated liposomes after intraperitoneal injection in a C26 colon carcinoma ascites mouse model. Nucl Med Biol. 2007;34(4):415–423. | ||
Chang YJ, Chang CH, Chang TJ, et al. Biodistribution, pharmacokinetics and microSPECT/CT imaging of 188Re-bMEDA-liposome in a C26 murine colon carcinoma solid tumor animal model. Anticancer Res. 2007;27(4B):2217–2225. | ||
Wang SX, Bao A, Herrera SJ, et al. Intraoperative 186Re-liposome radionuclide therapy in a head and neck squamous cell carcinoma xenograft positive surgical margin model. Clin Cancer Res. 2008; 14(12):3975–3983. | ||
Chang YJ, Chang CH, Yu CY, et al. Therapeutic efficacy and micro SPECT/CT imaging of 188Re-DXR-liposome in a C26 murine colon carcinoma solid tumor model. Nucl Med Biol. 2010;37(1):95–104. | ||
Chen MH, Chang CH, Chang YJ, et al. MicroSPECT/CT imaging and pharmacokinetics of 188Re-(DXR)-liposome in human colorectal adenocarcinoma-bearing mice. Anticancer Res. 2010;30(1):65–72. | ||
Chang YJ, Hsu CW, Chang CH, Lan KL, Ting G, Lee TW. Therapeutic efficacy of 188Re-liposome in a C26 murine colon carcinoma solid tumor model. Invest New Drugs. 2013;31(4):801–811. | ||
Chen LC, Chang CH, Yu CY, et al. Pharmacokinetics, micro-SPECT/CT imaging and therapeutic efficacy of (188)Re-DXR-liposome in C26 colon carcinoma ascites mice model. Nucl Med Biol. 2008;35(8):883–893. | ||
Chen LC, Wu YH, Liu IH, et al. Pharmacokinetics, dosimetry and comparative efficacy of 188Re-liposome and 5-FU in a CT26-luc lung-metastatic mice model. Nucl Med Biol. 2012;39(1):35–43. | ||
Hsu CW, Chang YJ, Chang CH, et al. Comparative therapeutic efficacy of rhenium-188 radiolabeled-liposome and 5-fluorouracil in LS-174T human colon carcinoma solid tumor xenografts. Cancer Biother Radiopharm. 2012;27(8):481–489. | ||
Tsai CC, Chang CH, Chen LC, et al. Biodistribution and pharmacokinetics of 188Re-liposomes and their comparative therapeutic efficacy with 5-fluorouracil in C26 colonic peritoneal carcinomatosis mice. Int J Nanomedicine. 2011;6:2607–2619. | ||
Tseng YL, Hong RL, Tao MH, Chang FH. Sterically stabilized anti-idiotype immunoliposomes improve the therapeutic efficacy of doxorubicin in a murine B-cell lymphoma model. Int J Cancer. 1999;80(5):723–730. | ||
Mazumdar M, Smith A, Schwartz LH. A statistical simulation study finds discordance between WHO criteria and RECIST guideline. J Clin Epidemiol. 2004;57(4):358–365. | ||
Herskovic A, Martz K, al-Sarraf M, et al. Combined chemotherapy and radiotherapy compared with radiotherapy alone in patients with cancer of the esophagus. N Engl J Med. 1992;326(24):1593–1598. | ||
van Hagen P, Hulshof MC, van Lanschot JJ, et al. Preoperative chemoradiotherapy for esophageal or junctional cancer. N Engl J Med. 2012;366(22):2074–2084. | ||
Bass GA, Furlong H, O’Sullivan KE, Hennessy TP, Walsh TN. Chemoradiotherapy, with adjuvant surgery for local control, confers a durable survival advantage in adenocarcinoma and squamous cell carcinoma of the oesophagus. Eur J Cancer. 2014;50(6):1065–1075. | ||
Minsky BD, Pajak TF, Ginsberg RJ, et al. INT 0123 (Radiation Therapy Oncology Group 94-05) phase III trial of combined-modality therapy for esophageal cancer: high-dose versus standard-dose radiation therapy. J Clin Oncol. 2002;20(5):1167–1174. | ||
Allen TM, Cullis PR. Drug delivery systems: entering the mainstream. Science. 2004;303(5665):1818–1822. | ||
Lammers T, Hennink WE, Storm G. Tumour-targeted nanomedicines: principles and practice. Br J Cancer. 2008;99(3):392–397. | ||
Sun Q, Radosz M, Shen Y. Challenges in design of translational nanocarriers. J Control Release. 2012;164(2):156–169. | ||
Oude Blenke E, Mastrobattista E, Schiffelers RM. Strategies for triggered drug release from tumor targeted liposomes. Expert Opin Drug Deliv. 2013;10(10):1399–1410. | ||
Greish K. Enhanced permeability and retention (EPR) effect for anticancer nanomedicine drug targeting. Methods Mol Biol. 2010;624: 25–37. | ||
MacPherson KH, Carr PD, Verger D, Kwok T, Davidson BE, Ollis DL. Crystallization of the N-terminal domain of the Escherichia coli regulatory protein TyrR. Acta Crystallogr D Biol Crystallogr. 1999;55(Pt 11):1923–1924. | ||
Torchilin VP. Recent advances with liposomes as pharmaceutical carriers. Nat Rev Drug Discov. 2005;4(2):145–160. | ||
Goins BA, Phillips WT. Nanoimaging. Pan Stanford Series on Biomedical Nanotechnology. London, England: CRC Press; 2011. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.