Back to Journals » Risk Management and Healthcare Policy » Volume 14
Extended-Spectrum Beta-Lactamase-Producing Gram-Negative Bacteria on Healthcare Workers’ Mobile Phones: Evidence from Tikur Anbessa Specialized Hospital, Addis Ababa, Ethiopia
Authors Araya S , Desta K , Woldeamanuel Y
Received 12 November 2020
Accepted for publication 12 January 2021
Published 26 January 2021 Volume 2021:14 Pages 283—291
DOI https://doi.org/10.2147/RMHP.S291876
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Marco Carotenuto
Shambel Araya,1 Kassu Desta,1 Yimtubezinash Woldeamanuel2
1Department of Medical Laboratory Sciences, College of Health Sciences, Addis Ababa University, Addis Ababa, Ethiopia; 2Department of Medical Microbiology, Immunology and Parasitology, School of Medicine, College of Health Sciences, Addis Ababa University, Addis Ababa, Ethiopia
Correspondence: Shambel Araya Tel: +251939459529
Email [email protected]
Background: Mobile phones are widely used in hospital settings for different purposes. Mobile phones of healthcare workers (HCWs) could be colonized or harbor extended-spectrum beta-lactamase (ESBL) producing gram-negative bacteria and may act as source of infectious agents. The aim of this study was to determine the rate of extended-spectrum beta-lactamase-producing Gram-negative bacteria on mobile phones of healthcare workers, to assess their antimicrobial susceptibility patterns and associated factors.
Methods: A laboratory-based cross-sectional study was conducted involving a total of 572 samples by rubbing swabs of the front screen, back, keypad, and metallic surfaces of mobile phones of healthcare workers using simple random sampling technique. All specimens were screened for ESBL using ESBL CHROME agar and confirmed using double-disk diffusion test (DDDT). Antibiotic susceptibility testing was done by the Kirby–Bauer disk diffusion technique on Mueller–Hinton agar. Data were analyzed using SPSS version 25, odds ratio and p-value was calculated to determine the association among variables.
Results: Overall, the number of mobile phones contaminated by gram-negative bacteria was 454 out of 572 (79.4%). Female sex (OR 0.651, p-value=0.039) and service year (OR 0.468, p-value=0.038) of healthcare workers were found to be the most significant factors associated with healthcare professionals’ mobile phone and bacterial contamination. Nine percent of the isolates were ESBL-producers. K. pneumoniae (27%) was the dominant ESBL-producing isolate followed by Acinetobacter spp. (14.5%) and E.coli (14.5%). ESBL-producers were highly resistant to ampicillin (95.8%), piperacillin (83.3%), cotrimoxazole (70.8%), and chloramphenicol (54.2%), but highly sensitive to meropenem (87.5%), amikacin (85.4%), and piperacillin-tazobactam (81.2%).
Conclusion: ESBL-producing Gram-negative bacteria were isolated from 8.3% of HCWs’ mobile phones. As high as 79.4% of the isolates were multidrug resistant. Mobile phones can lead to bacterial cross-contamination and could be a source of nosocomial infections.
Keywords: mobile phones, extended-spectrum β-lactamase, Gram-negative bacteria, hospital-acquired infection, Addis Ababa, Ethiopia
Background
Extended-spectrum beta-lactamase (ESBL) producing organisms are a challenge for clinicians, microbiologists, researchers, and infection control professionals.1,2 The prevalence of such pathogens among nosocomial infections has varied over time; some of the changes in this pattern are attributable to healthcare changes, the use of antimicrobial agents, new practices of infection control, and the onset of antimicrobial resistance.3
ESBL-producing Gram-negative bacteria like Acinetobacter, Pseudomonas, and various Enterobacteriaceae including Klebsiella spp., E. coli, Serratia marcescens, and Proteus spp. are a major concern due to concomitant multidrug resistance and their relation to severe and often deadly infections, such as bloodstream infections and pneumonia.1,2,4
Currently, one of the main concerns is the increased occurrence of gram-negative bacteria, including multidrug-resistant non-fermenters (Acinetobacterbaumannii & Pseudomonas species) and Enterobacteriaceae-producing extended-spectrum β-lactamases and carbapenemases in severe healthcare-associated infections for the medical community in recent decades.2,5,6,7
Healthcare-associated infections (HAIs) are associated with significant mortality, morbidity, and high economic burden and they are a major challenge to the healthcare system. The constant handling of mobile phones (MPs) by users in hospitals (patients, visitors, and healthcare workers, etc.) makes it a suitable place for transmission of multidrug-resistant bacteria (MDR), as well as healthcare-associated infections.8,9
Mobile phones are widely used in healthcare facilities, even reaching operating rooms. It has been shown that mobile phones may serve as reservoir of MDR bacteria like ESBL, MRSA, and VRE and even they serve as a reservoir for viruses like COVID-19 which could easily be transmitted from MPs to the healthcare workers hands, therefore facilitating the transmission from one patient to another in different hospital settings.3,9–11 There are very few published data on mobile phones of HCWs in Ethiopia but ESBL focused information is scarce.12,13 Thus, the aim of this study was to determine the rate of ESBL-producing Gram-negative bacteria found on mobile phones of HCWs and to assess antimicrobial susceptibility patterns and associated factors.
Methods
A laboratory-based cross-sectional study was conducted from January to April 2019, involving 572 swab samples of mobile phones from different units of TikurAnbessa Specialized Hospital (TASH) in Addis Ababa, Ethiopia. For the purpose of this study, bacterial isolates from mobile phones of HCWs in the hospital which were collected during an ongoing PhD project (Title of project: Burden of Methicillin Resistant Staphylococcus aureus (MRSA) and associated factors at TASH: Evidence from colonization of patients, healthcare workers, administrative staffs and selected inanimate objects) were used. By considering 99% confidence level and 5% degree of precision the maximum proportion of HCWs’ mobile phones assumed to be contaminated was 50%. The calculated sample size was 622. The study participants were allocated proportionally using simple random sampling technique from the different units of the hospital. From these fifty samples were contaminated and were excluded. The final sample size was 572. One hundred fifty-six swabs came from phones of physicians, 293 from nurses, 36 from medical laboratory personnel, 28 from pharmacists, and 59 from mobile phones of other healthcare workers. The socio-demographic data and associated factors including age, sex, work experience, and mobile phone types of participants were taken from a pre-collected questionnaire from the ongoing PhD project.
Culture and Bacteria Identification
Swab samples from surface of mobile phones were inoculated in MacConkey agar during the primary project. Bacterial Isolates were stored in a medium containing skim milk, tryptic soy broth, and glycerin (STGG) under −40°C. Stored isolates were sub-cultured on MacConkey agar plates (Oxoid Ltd. Bashing store Hampshire, UK) and on CHROMagar ESBL (CHROMagar, Paris, France) by surface streaking and incubated afterwards aerobically at 37°C for 18 to 24 hours. CHROMagar ESBL was used for screening of ESBL-production among Gram-negative bacteria.14 Bacterial identification to species level was done considering growth morphology on culture plates and biochemical characteristics by using biochemical identification tests; fermentation of sugars and production of CO2 and H2S gases in Triple Sugar Iron agar, Sulfide indole motility, Urease production, Citrate utilization, Mannitol fermentation, Malonate utilization, Oxidase tests, and catalase. Control organisms used were E. coli ATCC 25922 (non-ESBL-producer) and E. coli ATCC 35218 (ESBL-producer).
Confirmation of ESBL-Production
ESBL-production was confirmed using double-disk diffusion test (DDDT) according to CLSI 2018 recommendations. Ceftazidime 30 μg, ceftazidime-clavulanic acid 30/10 μg, cefotaxime 30 μg, and cefotaxime-clavulanic acid 30/10 μg discs were used. A ≥5 mm increase in the inhibition zone diameter for either antimicrobial agent tested in combination with clavulanate vs the inhibition zone diameter of the agent when tested alone confirmed ESBL-producers. E. coli ATCC 25922 (non-ESBL-producer) and E. coli ATCC 35218 (ESBL-producer) were used for quality control.15
Antibiotic Susceptibility Testing (AST)
Antimicrobial susceptibility testing was done for all bacterial isolates using the disk diffusion method according to Clinical Laboratory Standards Institute 2018 guidelines. A standard inoculum adjusted to 0.5 McFarland standards was swabbed on to Mueller-Hinton agar (Oxoid Ltd. Bashing store Hampshire, UK); the antibiotic disc was dispensed after drying the plate for 3–5 minutes and incubated at 37°C for 24 hours. The susceptibility of the isolated Gram-negative bacteria was tested against 14 antibiotics from 8 different families using the Kirby–Bauer disc diffusion technique. Quality control was assured by concurrent testing with the American Type Culture Collection (ATCC) strains including E. coli ATCC 25922 and P. aeruginosa ATCC 27852.15 MDR was defined as acquired non-susceptibility to at least one agent in three or more antimicrobial categories.16
Statistical Analysis
Data was collected, double entered, cleaned, and analyzed using SPSS version 21 (IBM Corp. Released 2012: IBM SPSS Statistics for Windows, Version 21.0. Armonk, NY: IBM Corp.) according to the study objectives. Frequency, percentage, chi-square of outcome, and bi-variate analysis were used to see association. A p-value < 0.05 was considered as significant. Categorical data were also compared, using the Chi-Square test.
Results
Socio-Demographic Characteristics of HCWs
A total of 572 HCWs were included from different units of the TikurAnbessa Specialized Hospital (TASH). Of those HCWs, 334 (58.4%) were females and 238 (41.6%) males. Age of study participants ranged from 20 to 57 years old and the majority (72%) was between 21 and 30 years old. The mean age of the study participants was 29± 6.9 years. Majority of the participants were nurses (51.2%), followed by medical doctors (27.3%) (Table 1).
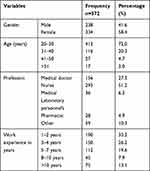 |
Table 1 Socio-Demographic Characteristics of Study Participants Among TASH HCWs, 2019 |
Associated Factors for Bacterial Contamination in Mobile Phones of HCWs
The Gram-negative bacteria contamination rate on mobile phones in this study was 454/572 (79.4%). Females’ mobile phones were more often (82.3% or 275 mobile phones) contaminated than males’ mobile phones 75.2% (179 mobile phones); the difference is significant with a p-value = 0.039. Of all investigated phones, 465 (81.3%) were touchscreen mobile phones (TMP) and 107 (18.7%) were keypad mobile phones (KMP). Of all contaminated phones, 83.2% (89/107) of keypad mobile phones was contaminated with Gram-negative bacteria as compared to 78.5% (365/465) of the touch screen mobile phones; the difference, however, was not statistically significant (p-value = 0.282).
Gram-negative bacteria contamination rate was higher among mobile phones used by the older age group (>51years old) than among phones used by younger (20–30years old) HCWs: 94.1% versus 76.9%.
About 91.7% (33/36) of the mobile phones screened from medical laboratory personnel showed growth of at least one Gram-negative bacterium, followed by 82.4% from the mobile phones screened from nurses (Table 2), and the difference between these results was found to be statistically significant (p-value = 0.044).
 |
Table 2 Associated Factors of Bacterial Contamination for Mobile Phones of HCWs |
In relation to years of service of HCWs in the hospital, those who served 1–2 years showed a lower degree of contamination (73.1%) compared to those who served for more than 10 years (85%) followed by 84% who had served more than 5–7 years. There was a significant association between the service years of HCWs and mobile phones contaminated with one or more Gram-negative bacteria (p = 0.038, CI 0.229–0.958) (Table 2).
Five hundred thirty-eight bacterial isolates were found out of the 454contaminated mobile phones, 83.2% of KMPs were contaminated with at least one Gram-negative bacterium, 9.1% of KMPs was contaminated with two Gram-negative bacteria and only one mobile phone was contaminated with three Gram-negative bacteria. The number of isolates per specimen was high among KMP compared to TMP. However, there was no significant difference in microbial contamination between the types of mobile phone (p = 0.282) (Figure 1).
 |
Figure 1 Distribution of bacterial isolates per specimen across different phone types, 2019. |
Distribution of ESBL-Producing Gram-Negative Bacteria from Mobile Phones of HCWs
Out of the total 538 isolates screened for ESBL-production, 10.6% (57/538) of them were positive for ESBL using ESBL CHROME agar as a screening method. From these; 9% (48/538) of the species were confirmed to be ESBL-producers using a confirmatory test method (double-disk diffusion test).
K. pneumoniae was the most common 27.1% (13/48) species found among the 48 confirmed ESBL-producing Gram-negative bacteria, followed by E.coli14.6% (7/48) and Acinetobacter spp.14.6% (7/48) (Figure 2).
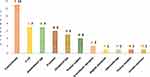 |
Figure 2 Distribution of ESBL-producing Gram-negative bacteria from mobile phones of HCWs of TASH, 2019. |
Antimicrobial Susceptibility Pattern of ESBL-Producing Gram-Negative Bacteria from Mobile Phones of HCWs
A total of 48 species were available for antimicrobial testing. Most of these isolates indicated widespread resistance to multiple drugs. High-level resistance to ampicillin was observed in 100% of all isolated species except for Citrobacter (80%) and K. pneumoniae (92%). K. pneumoniae and E. coli also exhibited substantial resistance to ampicillin (92% and 100%, respectively), chloramphenicol (44%), and cotrimoxazole (28%). K. pneumoniae isolates were resistant for meropenem (30.4%) and imipenem (46%) (Table 3).
 |
Table 3 Antimicrobial Resistance Pattern of ESBL-Producing Gram-Negative Bacteria from Mobile Phones of HCW, 2019 |
MDR Pattern of ESBL-Producing Bacterial Isolates from Mobile Phones
In our study, 79.2% of the ESBL-producing isolates showed multidrug resistance. Amongst all the bacterial isolates, K. oxytoca, Salmonella spp., P. vulgaris, and P. mirabilis showed 100% MDR characteristics, whereas Klebsiella spp. and Citrobacter spp. showed MDR characteristics with 77.1% and 75%, respectively. Seventy-one percent of the Acinetobacter spp exhibited resistance to more than nine tested antibiotics. In K. oxytoca, 50% of the isolates were resistant to nine and more tested antibiotics, and 16.7% was resistant to three antibiotics, 16.7% to four antibiotics, and 16.7% to six antibiotics (Table 4).
 |
Table 4 MDR Pattern of ESBL-Producing Gram-Negative Bacteria Isolates from Mobile Phones of HCWs, 2019 |
Discussion
Mobile phones are one of the essential portable devices used for communication in daily life and are used everywhere.17–19 However, the most common concern regarding the use of mobile devices in the healthcare setting is that they have the potential to act as a reservoir for pathogenic bacteria and vehicle to transmit them.9,20,21
In the current study, the percentage of bacterial contamination with at least one bacterial pathogen on the tested cell phones was 79.4%. Even though there were no previous studies specifically done for Gram-negative bacteria, this percentage is relatively high compared with previously reported numbers in Ethiopia.12,13 Different studies had reported the prevalence of bacterial contamination of mobile phones across the globe; Nigeria (76–94%), Egypt (61–100%), Ghana (100%), India (56%-98.5), Australia (50%), United Kingdom (85%).22–30 Non-compliance of hospital standards for infection prevention may also contribute to the finding of high bacterial contamination. Female HCWs’ mobile phones were more often (82.3%) contaminated than mobile phones of male HCWs (75.2%), which is statistically significant. A study on female HCWs showed that their handbags could play a role in the bacterial transmission and that mobile phones can be further contaminated by other materials carried inside handbags.31–33 Microbial contamination rate was significantly different between mobile phones of the investigated medical professions. Similar results were found in a study in Egypt.34
Even though it was not significantly associated, the contamination rate of MPs with at least one gram-negative bacterium among HCWs who served for more than 10 years was higher (85%; 64/75) compared to those who served less than five years. Interestingly, we found a higher contamination percentage of mobile phones among the age group of >50 years old (Table 2). However, this was incongruous with a previous study conducted in Harar, eastern Ethiopia, which revealed that the age group between 25 and 49 years had a higher contamination rate.33
Among 107 keypad mobile phones, 83.2% was found to be contaminated, while 78.5% of the 465 touch screen mobile phones were contaminated. Even though there is no statistically significant difference this higher contamination level in KMP compared to TMP might be due to the more complex surface structure of the KMP.34 On the other hand, other studies have shown that touch screen mobile phones are more contaminated by micro-organisms than keypad mobile phones, and suggested that this may be caused by the wider screen and more intense usage pattern of TMP.35,36
In the current study, 79.4% (454/572) of the mobile phones were contaminated with one or more Gram-negative bacteria. Among these, 9% were ESBL-producing bacterial species. This frequency was lower than from a study conducted in Peru (33.3%).3 A study conducted by A. Nieto-Carhuamaca et al. showed that there was a statistical association between presence of ESBL-type bacterial resistance and cell phone disinfection.37 Periodic cleaning of mobile phones with disinfectants as well as frequent hand-washing should be encouraged as a means of curtailing any potential bacterial contamination.38 In this current study, the most dominant ESBL-producing isolate was K. pneumoniae (28%) followed by E. coli (14.6%) and Acinetobacter spp. (14.6%) (Figure 2). Even though this frequency is high compared to other studies but different studies had reported the presence of ESBL-producing Gram-negative bacteria in their studies.20,21,34,37,39 The prevalence of ESBL-producing Gram-negative bacteria from mobile phones observed in our study was higher compared to previous studies conducted in Ethiopia.13,33 This could be due to the study area since earlier studies were conducted in health centers but this study was done in tertiary hospital which is the largest hospital in the country and it could be also due to sample size variation. In our study, 79.2% of ESBL-producing isolates showed multidrug resistance (Table 4). This was similar to a study conducted by Asmari et al in Saudi Arabia; they revealed that 71.8% of the isolates were MDR bacteria,39 Selim et al. in Egypt and by Bodena et al. in Ethiopia: a high degree of resistance was shown for ampicillin (95.8%), piperacillin (83.3%), and cotrimoxazole (79.2%)25,33 but in contrast to Gashaw et al.12 In the present study, most of the ESBL-isolates were sensitive to meropenem (87.5%), amikacin (85.6%), piperacillin-tazobactam (81.2%). These findings were also in agreement with other studies.40–42 Bacterial species like Acinetobacter, Pseudomonas, and E. coli had shown slight resistance to carbapenem and β-lactam inhibitors in the current study.
The presence of Shigella and Salmonella spp. suggests fecal contamination of these phones, which can result in disease outbreaks and community-acquired infections. These isolates were also reported from mobile phones by Tagoe et al. in Ghana and Chitlange et al. in India.26,43 Taking into account the shortcomings, further research is warranted, focusing on the molecular identification of bacteria and other microorganisms like viruses on the surface of MPs, in order to understand whether and to what extent they remain viable and virulent after lying on the devices.
Conclusion
ESBL-producing Gram-negative bacteria were isolated from mobile phones of healthcare workers in Tikur Anbesa Specialized hospital, Addis Ababa, Ethiopia. The majority of ESBL-producing isolates were multidrug resistant. Mobile phones can be contaminated and serve as reservoir of ESBL-producing Gram-negative bacteria.
Data Sharing Statement
The data sets used or analyzed during the current study are included in the manuscript.
Ethical Approval and Consent to Participate
Ethical clearance for this study was provided from Addis Ababa University, College of Health Science, Department of Medical Microbiology, Immunology, and Parasitology ethical review committee and it was in accordance with the principles of the Helsinki II declaration. Written informed consent was also obtained from the participants by the ongoing PhD project.
Consent for Publication
Not applicable as details like videos or images related to study subjects were not recorded for this study.
Acknowledgments
We are very grateful to all HCWs for participating in this study. Mr. Ayelign Deribe, Mequanenet Mitiku, Simegen Terefe and Bekelu Belema are acknowledged for their kind support during laboratory work. Addis Ababa University, College of Health Sciences is duly acknowledged for financial support.
Author Contributions
SA; [MSc in Medical Microbiology and Lecturer of Microbiology]. KD [MSc in Medical Microbiology, Associate professor of Medical Microbiology and Laboratory Sciences, PhD candidate]. Dr. YW; [M.D, M.Sc., Ph.D., Associate professor of Medical Microbiology].
Funding
The study was supported by Addis Ababa University College of Health Science department of Medical Microbiology, Immunology and parasitology. The funder had no role in data collection, study design, data analysis and interpretation.
Disclosure
The authors declare that they have no financial or non-financial conflicts of interest for this work.
References
1. Ndugulile F, Jureen R, Harthug S, Urassa W, Langeland N. Extended spectrum β-lactamases among gram-negative bacteria of nosocomial origin from an intensive care unit of a tertiary health facility in Tanzania. BMC Infect Dis. 2005;5(1):86. doi:10.1186/1471-2334-5-86
2. Mischnik A, Baumert P, Hamprecht A, et al. In vitro susceptibility to 19 agents other than beta-lactams among third-generation cephalosporin-resistant Enterobacteriaceae recovered on hospital admission. J Antimicrob Chemother. 2017;72(5):1359–1363. doi:10.1093/jac/dkw577
3. Loyola S, Gutierrez LR, Horna G, et al. Extended-spectrum β-lactamase–producing Enterobacteriaceae in cell phones of health care workers from Peruvian pediatric and neonatal intensive care units. Am J Infect Control. 2016;44(8):910–916. doi:10.1016/j.ajic.2016.02.020
4. Shaikh S, Fatima J, Shakil S, Rizvi SMD, Kamal MA. Antibiotic resistance and extended spectrum beta-lactamases: types, epidemiology and treatment. Saudi J Biol Sci. 2015;22(1):90–101. doi:10.1016/j.sjbs.2014.08.002
5. Worthington JR, Melander C. Overcoming resistance to β-lactam antibiotics. J Org Chem. 2013;78(9):4207–4213. doi:10.1021/jo400236f
6. Parajuli PN, Parajuli K. High burden of antimicrobial resistance among gram negative bacteria causing healthcare associated infections in a critical care unit of Nepal. ARIC. 2017;6:67. doi:10.1186/s13756-017-0222-z
7. Mehrad B, Clark NM, Zhanel GG, Lynch JP. Antimicrobial resistance in hospital-acquired gram-negative bacterial infections. Chest. 2015;147(5):1413–1421. doi:10.1378/chest.14-2171
8. Mir-shekari T, Sepehri E. Bacterial contamination and resistance to commonly used antimicrobials of healthcare workers’ mobile phones in teaching hospitals, Kerman, Iran. Am J Appl Sci. 2009;6(5):806–810. doi:10.3844/ajassp.2009.806.810
9. Zakai S, Mashat A, Abumohssin A, et al. Bacterial contamination of cell phones of medical students at King Abdulaziz University, Jeddah, Saudi Arabia. J Microsc Ultrastruct. 2016;4:143–146.
10. Panigrahi SK, Pathak VK, Kumar MM, et al. Covid-19 and mobile phone hygiene in healthcare settings. BMJ Global Health. 2020;5(4):e002505. doi:10.1136/bmjgh-2020-002505
11. D’Alò GL, CicciarellaModica D, Maurici M, et al. Microbial contamination of the surface of mobile phones and implications for the containment of the Covid-19 pandemic. Travel Med Infect Dis. 2020;37:101870. doi:10.1016/j.tmaid.2020.101870
12. Gashaw M, Abtew D, Addis Z. Prevalence and antimicrobial susceptibility pattern of bacteria isolated from mobile phones of health care professionals working in gondar town health centers. ISRN Public Health. 2014;2014(1):1–6. doi:10.1155/2014/205074
13. Misgana MG, Abdissa K, Abebe G. Bacterial contamination of mobile phones of healthcare workers at Jimma University Specialized Hospital, Jimma, South West Ethiopia. Int J Infect Control. 2014;1:2–8.
14. Saito R, Koyano S, Nagai R, Okamura N, Moriya K, Koike K. Evaluation of a chromogenic agar medium for the detection of extended-spectrum -lactamase-producing Enterobacteriaceae. Lett. Appl Microbiol. 2010;51(6):704–706. doi:10.1111/j.1472-765X.2010.02945.x
15. CLSI. Performance Standards for Antimicrobial Susceptibility Testing. CLSI Supplement M100. Wayne, PA: Clinical and Laboratory Standards Institute; 2018.
16. Magiorakos AP, Srinivasan A, Carey RB, Carmeli Y, Falagas ME. Multidrug-resistant, extensively drug-resistant and pan-drug-resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clin Microbiol Infect. 2012;18(3):268–281. doi:10.1111/j.1469-0691.2011.03570.x
17. Vilella A, Bayas J, Diaz MT, Guinovart C, Simo D, Munoz A. The role of mobile phones in improving vaccination rates in travelers. PrevMed. 2004;38:503–509.
18. Were J, Hayford K, Moulton LH, Levine SO, Odhiambo F. Mobile phone delivered reminders and incentives to improve childhood immunization coverage and timeliness in Kenya (M-SIMU): a cluster randomized controlled trial. Lancet Glob Health. 2017;5(4):e428–38. doi:10.1016/S2214-109X(17)30072-4
19. Ferrer-Roca O, Cardenas A, Diaz-Cardama A, Pulido P. Mobile phone text messaging in the management of diabetes. J Telemed Telecare. 2004;10(5):282–286. doi:10.1258/1357633042026341
20. Sedighi I, Alikhani MY, Ramezani S, Nazari M, Nejad MS. Bacterial contamination of mobile phones of health care providers in a teaching hospital in Hamadan Province, Iran. Arch Clin Infect Dis. 2015;10(2):e22104. doi:10.5812/archcid.10(2)2015.22104
21. Ustun C, Cihangiroglu M. Health care workers’ mobile phones: a potential cause of microbial cross-contamination between hospitals and community. J Occup Environ Hyg. 2012;9(9):538–542. doi:10.1080/15459624.2012.697419
22. Akinyemi KO, Atapu AD, Adetona OO, Coker AO. The potential role of mobile phones in the spread of bacterial infections. J Infect Dev Ctries. 2009;3(08):628. doi:10.3855/jidc.556
23. Amadi EC, Nwagu TN, Emenuga V. Mobile phones of health care workers are potential vectors of nosocomial agents. Afr J Microbiol Res. 2013;7(22):2776–2781. doi:10.5897/AJMR12.2353
24. Hassan MM, Ismail IA. Isolation and molecular characterization of some pathogenic mobile phone bacteria. Int J Biochem Biotechnol. 2014;3.
25. Selim HS, Abaza AF. Microbial contamination of mobile phones in a health care setting in Alexandria, Egypt. GMS Hyg Infect Control. 2015;10:Doc03. doi:10.3205/dgkh000246
26. Tagoe D, Gyande K, Ansah O. Bacterial contamination of mobile phones: when your mobile phone could transmit more than just a call. WebmedCentral. 2011;2:1–9.
27. Rahangdale V, Kokate S, Surpam R. Cell phones- homes for microbes. IOSR J Dent Med Sci. 2014;13(12):97–100. doi:10.9790/0853-1312597100
28. Singh S. Mobile phone hygiene: potential risks posed by use in the clinics of an Indian dental school. J Dent Educ. 2010;74:1153.
29. Foong YC, Green M, Zargari A, Siddique R, Tan V, Brain T. Mobile phones as a potential vehicle of infection in a hospital setting. J Occup Environ Hyg. 2015;12(10):D232–D235. doi:10.1080/15459624.2015.1060330
30. Al-Harmoosh RA, Mutlaq NH, Alabassi MM, Al-Shamari AM, Al-khafaji HM. Surface of mobile phone: as a carrier of pathogenic bacteria. Res J Pharm Technol. 2017;10(10):3461–3464. doi:10.5958/0974-360X.2017.00618.7
31. Feldman J, Feldman J, Feldman M. Women doctors’ purses as an unrecognized fomite. Del Med J. 2012;84(9):277–280.
32. Dorost A, Safari Y, Akhlaghi M, Soleimani M, Yoosefpour N. Microbial contamination data of keypad and touch screen of cell phones among hospital and non-hospital staffs - a case study: Iran. Data Brief. 2018;20:80–84. doi:10.1016/j.dib.2018.07.041
33. Bodena D, Teklemariam Z, Balakrishnan S, Tesfa T. Bacterial contamination of mobile phones of health professionals in Eastern Ethiopia: antimicrobial susceptibility and associated factors. Trop Med Health. 2019;47(1):15. doi:10.1186/s41182-019-0144-y
34. Pal P, Roy A, Moore G, et al. Keypad mobile phones are associated with a significant increased risk of microbial contamination compared to touch screen phones. JInfectPrevent. 2013;14(2):65–68.
35. Lee YJ, Yoo CG, Lee CT. Contamination rates between smart cell phones and non-smart cell phones of healthcare workers. J Hosp Med. 2013;8:144–147. doi:10.1002/jhm.2011
36. Koroglu M, Gunal S, Yildiz F, Savas M, Ozer A, Altindis M. Comparison of keypads and touch-screen mobile phones/devices as potential risk for microbial contamination. J Infect Dev Ctries. 2015;9(12).
37. Nieto-Carhuamaca A, Castañeda-Japan J, Dámaso-Mata B, Panduro-Correa V, Arteaga-Livias K. Bacterial resistance in cell phone cultures of medical students. Infez Med. 2019;27(4):374–379.
38. Cicciarella Modica D, Maurici M, D’Alò GL, et al. Taking screenshots of the invisible: a study on bacterial contamination of mobile phones from university students of healthcare professions in Rome, Italy. Microorganisms. 2020;8(7):1075. doi:10.3390/microorganisms8071075
39. Al- Asmari A, Mahfoud M, Barrak AA, Babair Y, Noura E, Al- Oman S. antimicrobial resistance pattern of pathogens isolated from mobile phone at tertiary care hospital in Saudi Arabia. World J Pharm Res. 2015;11(4):1744–1761.
40. Uneke JC, Ogbonna A, Oyibo PG, Ekuma U. Bacteriological assessment of stethoscopes used by medical students in nigeria: implications for nosocomial infection control. World Health Popul. 2009;10(4):37–45. doi:10.12927/whp.2009.20696
41. Wattal C, Goel N, Oberoi JK, Raveendran RS, Datta S, Prasad KJ. Surveillance of multidrug resistant organisms in a tertiary care hospital in Delhi, India. J Assoc Physicians India. 2010;58:32–36.
42. Umadevi S, Kandhakumari G, Joseph NM, et al. Prevalence and antimicrobial susceptibility pattern of ESBL producing gram negative bacilli. J Clin Diagn Res. 2011;5(2):236–239.
43. Chitlange RP. Contamination of cell phones by pathogenic microorganisms: comparison between hospital staff and college students. Nusantara Biosci. 2014;6(2):203–206.
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
