Back to Journals » International Journal of Nanomedicine » Volume 17
Exosomes as Promising Nanostructures in Diabetes Mellitus: From Insulin Sensitivity to Ameliorating Diabetic Complications
Authors Ashrafizadeh M, Kumar AP , Aref AR, Zarrabi A , Mostafavi E
Received 2 December 2021
Accepted for publication 7 March 2022
Published 19 March 2022 Volume 2022:17 Pages 1229—1253
DOI https://doi.org/10.2147/IJN.S350250
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Mian Wang
Milad Ashrafizadeh,1 Alan Prem Kumar,2,3 Amir Reza Aref,4,5 Ali Zarrabi,6 Ebrahim Mostafavi7,8
1Faculty of Engineering and Natural Sciences, Sabanci University, Tuzla, 34956, Istanbul, Turkey; 2NUS Centre for Cancer Research (N2CR), Yong Loo Lin School of Medicine, National University of Singapore, Singapore, 117600, Singapore; 3Cancer Science Institute of Singapore and Department of Pharmacology, Yong Loo Lin School of Medicine, National University of Singapore, Singapore, 117599, Singapore; 4Belfer Center for Applied Cancer Science, Dana-Farber Cancer Institute, Harvard Medical School, Boston, MA, USA; 5Translational Sciences, Xsphera Biosciences Inc., Boston, MA, 02210, USA; 6Department of Biomedical Engineering, Faculty of Engineering and Natural Sciences, Istinye University, Istanbul, 34396, Turkey; 7Stanford Cardiovascular Institute, Stanford University School of Medicine, Stanford, CA, 94305, USA; 8Department of Medicine, Stanford University School of Medicine, Stanford, CA, 94305, USA
Correspondence: Ebrahim Mostafavi, Stanford Cardiovascular Institute, Stanford University School of Medicine, Stanford, CA, 94305, USA, Email [email protected]; [email protected] Ali Zarrabi, Department of Biomedical Engineering, Faculty of Engineering and Natural Sciences, Istinye University, Istanbul, 34396, Turkey, Email [email protected]
Abstract: Diabetes mellitus (DM) is among the chronic metabolic disorders that its incidence rate has shown an increase in developed and wealthy countries due to lifestyle and obesity. The treatment of DM has always been of interest, and significant effort has been made in this field. Exosomes belong to extracellular vesicles with nanosized features (30– 150 nm) that are involved in cell-to-cell communication and preserving homeostasis. The function of exosomes is different based on their cargo, and they may contain lipids, proteins, and nucleic acids. The present review focuses on the application of exosomes in the treatment of DM; both glucose and lipid levels are significantly affected by exosomes, and these nanostructures enhance lipid metabolism and decrease its deposition. Furthermore, exosomes promote glucose metabolism and affect the level of glycolytic enzymes and glucose transporters in DM. Type I DM results from the destruction of β cells in the pancreas, and exosomes can be employed to ameliorate apoptosis and endoplasmic reticulum (ER) stress in these cells. The exosomes have dual functions in mediating insulin resistance/sensitivity, and M1 macrophage-derived exosomes inhibit insulin secretion. The exosomes may contain miRNAs, and by transferring among cells, they can regulate various molecular pathways such as AMPK, PI3K/Akt, and β-catenin to affect DM progression. Noteworthy, exosomes are present in different body fluids such as blood circulation, and they can be employed as biomarkers for the diagnosis of diabetic patients. Future studies should focus on engineering exosomes derived from sources such as mesenchymal stem cells to treat DM as a novel strategy.
Keywords: diabetes mellitus, insulin resistance, exosome, glucose uptake, lipid metabolism
Corrigendum for this paper has been published
Introduction
The discovery of exosomes returns to the 1980s, when they were considered debris secreted by reticulocytes.1,2 These vesicles with endosomal origin were extensively examined, until ten years after their emergence that, Raposo et al found that exosomes have an antigen-presenting role and they can stimulate T cell response. These exosomes were derived from Epstein-Barr-transformed B lymphocytes.3,4 Then, more investigation was directed towards exosomes until their biological functions in cell-to-cell communication were realized. Overall, mammalian cells secrete three distinct kinds of extracellular vesicles (EVs), including exosomes, microvesicles, and apoptotic vesicles among which exosomes are the most well-known and largely investigated ones.5 EVs differ in their secretion process, molecular content, and biological functions.6 The apoptotic bodies are generated as post-apoptotic remnants of parent cells. The microvesicles emanate from blebbing of cell membrane, and they may contain cytosol parts. The main pathway for the formation of exosomes is endocytosis, and exosomes are vital for preserving homeostasis in cells.7 The multivesicular bodies (MVBs) are considered as the origin of exosomes, and they have a cup shape with particle size of 50 to 150 nm (average size of 100 nm).8,9 In 2018, Zhang et al discovered two subcategories of exosomes, including large exosome vesicles (90–120 nm in size) and small exosome vesicles (60–80 nm in size).10,11
After the release from cells, exosomes can transfer various cargoes among cells, and there are proteins on the surface of exosomes that mediate their interaction with receptors available on the surface of target cells.12,13 Lipids, proteins, and nucleic acids can be found in exosomes. Cholesterol, sphingomyelin, hexosylceramids, phosphatidylserine, and fatty acids are among the lipid particles that can be found in exosomes.14 The proteins include endosomal, plasma, cytosolic, and nuclear proteins.15 It is worth mentioning that two kinds of proteins are found in exosomes. The first category of proteins is related to those involved in membrane transport and fusion, including Rab GTPases and annexins. Other proteins can participate in the biogenesis of exosomes, including ESCRT complex, ALIX, and TSG101. However, there are other proteins in exosomes, including heat shock proteins (HSPs), integrins and tetraspanins, and MHC proteins.14,16–20 As it was mentioned, exosomes can also transfer nucleic acids among cells. Based on experiments, microRNA (miRNA) and messenger RNA (mRNA) are present in exosomes.21–23 Besides, other kinds of RNA molecules, including lncRNAs, circRNAs, viral RNA, and transfer RNA (tRNA) can be enriched in exosomes.23–25
With respect to the role of exosomes in cell-cell communication and maintaining homeostasis, the present review was dedicated to investigating exosome function in diabetes mellitus (DM). For this purpose, we first discuss exosome biogenesis, isolation, and its role in various diseases to prepare readers for other sections of the manuscript. Then, we briefly describe DM, complications, treatments, and challenges. Next, we specifically discuss the role of exosomes in affecting glucose and lipid metabolism in DM, insulin delivery, affecting β-cell function and gene delivery. Such topics will shed more light on the role of exosomes in the effective treatment of DM and pave the way for translating pre-clinical findings to clinical courses. This overview is different from previously published articles provides a unique and new outline about the role of exosomes in DM.26–36
Exosomes and Diseases
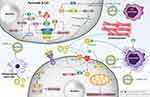
Based on previous statements in the introduction, exosomes are nanostructures (30–150 nm) and can function as communicators among cells.37 The biogenesis of exosomes is still controversial, and despite significant research in this area, this mechanism has not been completely understood yet. The exosome biogenesis begins with the generation of intraluminal vesicles (ILVs) resulting from the elimination of plasma membrane due to inward budding into cells. Therefore, lipid raft domains of plasma membrane can produce endocytic vesicles, which happens very fast, leading to the formation of early endosomes.38–43 The destination of early endosomes is completely different based on cargo. For instance, if the aim is to degrade cargo, multivesicular bodies can fuse with lysosomes.44–46 Then, maturation of early endosome to late endosome occurs that is accompanied by accumulation of ILVs in multivesicular bodies.44,47 Then, multivesicular bodies fuse with the cell membrane to release ILVs as exosomes to extracellular space.44,45,47–49 Different kinds of proteins are involved in the process of exosomes formation. Two major kinds of proteins are SNARE and ESCRT proteins. Although reducing ESCRT levels decreases exosome production, it does not completely interfere with exosome formation, showing that exosomes also depend on other proteins for their biogenesis, known as ESCRT-independent mechanisms.50,51 The lipids and GTPase are involved in exosome generation from membrane deformation, fission, and fusion.52 ALIX, HSPs, and SNAREs participate in exosome formation.53–55 Furthermore, the Golgi apparatus transforms the early endosome to late endosome.56 Figure 1 demonstrates the biogenesis route of exosomes in cells. Table 1 also summarizes various techniques applied in isolation of exosomes.
 |
Table 1 Various Techniques Applied in Isolation of Exosomes |
Exosomes and their function in various diseases have been identified; for instance, in carcinogenesis, the function of exosomes has been investigated in depth. The exosomes containing circ-0000069 function as tumor-promoting factors and mediate the malignant transformation of pancreatic tumor cells via promoting the expression level of STIL.64 Notably, tumor cells also secrete exosomes to promote migration. For instance, a recent experiment has shown the role of exosomes derived from colon cancer cells in increasing migration and invasion of SW480 cells via inducing the Akt/mTOR axis.65 The cancer-associated fibroblasts available in the tumor microenvironment can secrete exosomes to down-regulate miRNA-142-5p expression and increase PD-L1 expression in elevating lung tumor progression.66 In neurological diseases, such as Alzheimer’s disease, exosomes can prevent the accumulation of amyloid-beta (Aβ) plaques and improve cognitive function by inducing the S1P/SphK axis.67 Interestingly, exosomes can deliver therapeutic agents in Alzheimer’s disease therapy. The exosomes containing quercetin prevent phosphorylation of tau proteins and reduce levels of neurofibrillary tangles in the treatment of Alzheimer’s disease.68 In inflammatory bowel disease, exosomes can improve epithelial regeneration and induce repair of the intestinal barrier with TSG-6 overexpression.69,70 Based on these studies, exosomes function as therapeutic agents in various diseases, and this review focuses on DM therapy.
Diabetes Mellitus
DM is a metabolic disease that alters the metabolism of proteins, fats, and carbohydrates.71–73 The incidence rate of DM seems to be increasing in different timelines. In 2019, the global prevalence of DM was 9.3%; it appears to be 10.2% in 2030 and higher in 2045 (10.9%). Interestingly, the incidence rate of DM is different in urban and rural areas. Based on estimates, the prevalence of DM is 10.8% in urban areas, while it is 7.2% in rural areas. Furthermore, DM is more common in wealthy countries compared to low-income countries that are 10.4% and 4%, respectively.74,75 The incidence rate of DM is more common in the USA, India, and China than other countries in the world.76 Various factors can lead to DM development, including lack of insulin secretion, β cell inability to secrete insulin, and reduced insulin distribution in peripheral regions. Type I DM (TIDM) and type II DM (TIDM) are the most common kinds of DM observed in patients in clinical courses.77–79 The TIDM is an autoimmune disease in which the immune system inappropriately affects β cells in the pancreas and removes their insulin secretion capacity and decreases serum glucose levels. The treatment of TIDM seems to be vital, and lack of management can lead to severe complications. The TIDM is the most common type in which there is insulin secretion from β cells. However, cells do not respond to insulin, and hyperglycemia occurs. The occurrence of TIDM is more common in obese patients.80 Another kind of DM, known as gestational DM, happens in pregnant women and causes a significant decrease in sensitivity to insulin.81 The exact reason for DM development has not been completely understood. There are a variety of assumptions and experiments to find underlying causes of DM development.82 Environmental factors such as malfunction, lack of exercise, infections, and toxins can result in DM development. Genetic alterations and family history of DM are other causes of DM development.83,84 The genomic experiments have revealed the role of different gene loci in DM, and most of these genes demonstrate expression in β cells of the pancreas and modulate the response of these cells to apoptosis, inflammation, and immune system.85–87
Various therapeutics have been utilized to treat DM, and each of them has its own advantages and disadvantages. Plant derived-natural products have been used for the treatment of DM due to their capacity in ameliorating diabetic complications. Curcumin is a phytochemical with different pharmacological activities, including antioxidant, anti-inflammatory, and anti-diabetes, among others.88–95 Curcumin administration ameliorates apoptosis and oxidative damage in cardiomyocytes during DM, and for this purpose, curcumin induces PI3K/Akt and SIRT1/FOX1 pathways.96 Clinical trials demonstrated the role of curcumin in decreasing oxidative stress and inflammation scores in diabetic patients.97 Quercetin is another phytochemical that its administration seems to be beneficial in the treatment of different diseases such as Cancer, diabetes, and Cardiovascular diseases.98–102 Quercetin induces AMPK signaling to increase SIRT1 expression in inhibiting NF-κB signaling that is correlated with a decrease in oxidative stress and inflammation and subsequent prevention of DM-mediated atherosclerosis.103 Furthermore, a combination of quercetin and vitamin E prevents mitochondrial dysfunction and inhibits apoptosis to alleviate testicular damage in DM.104 Resveratrol is also a non-flavonoid polyphenol that can decrease oxidative damage and inflammation, and its administration is promising in treating neurological diseases and diabetes.105–110 Resveratrol can prevent infertility during diabetes111 and its co-administration with dapagliflozin as an antidiabetic agent promotes capacity in lowering glucose levels.112 The nucleic acid-based drugs, including small interfering RNA (siRNA) and short hairpin RNA (shRNA) are utilized in DM therapy. The down-regulation of Cyp4a14 and P2X7 by siRNA ameliorates diabetic nephropathy.113,114 Furthermore, CTGF and P2Y12 down-regulation by shRNA ameliorates retinopathy and prevents inflammation in DM treatment.115,116 However, these therapeutics have some problems such as poor bioavailability of drugs and degradation of siRNA in serum that for improving their efficacy, application of nanostructures is recommended.89,117–120 Noteworthy, recent experiments have focused on using nanostructures in DM treatment. The Cyperus rotundus-loaded zinc oxide nanostructures were capable of decreasing inflammation via NLRP3 down-regulation to ameliorate diabetic retinopathy.121 The empagliflozin is an antidiabetic agent that can be delivered by nanostructures, which in turn enhances its ability to alleviate inflammation and oxidative stress in diabetic neuropathy.122 The chitosan-based nanoparticles also deliver metformin for reducing glucose levels.123 The nanoparticles can provide sustained release of drugs in DM treatment.124,125 Hence, the application of nanoparticles appears to be promising in DM therapy, and therefore in the next sections, we focus on exosomes as nanostructures in DM treatment.
Exosomes, Glucose, and Lipid Metabolism
The metabolism and uptake of glucose undergo some abnormal alterations in DM. The glucose metabolism significantly decreases in DM and leads to hyperglycemia.126 The polymorphism in genes related to glucose metabolism, including OCT3 and GCK, has been reported in TIIDM.127 The ghrelin system participates in enhancing glucose metabolism in the treatment of TIDM, and hyperbaric oxygen can affect the ghrelin system, which affects glucose metabolism.128 The clinical trials on DM patients have shown a potential role for GLP-1 in improving glycemia.129 Therefore, studies have focused on glucose metabolism levels in DM.130 The deposition of lipid significantly increases in DM patients, making them prone to the development of other diseases such as non-alcoholic fatty liver disease.131 Enhancing lipid metabolism by cola nitida is beneficial in alleviating hepatic injury in TIIDM.132 Thus, this section focuses on exosome functions in affecting lipid and glucose metabolism to pave the way for the treatment of DM patients.
As mentioned before, obesity is one of the factors responsible for insulin resistance and DM development. A recent experiment has focused on the role of exosomes in high-fat diet (HFD) mice. The exosomes were derived from brown adipose tissue, and they could diminish serum glucose levels and body weight. Furthermore, these exosomes decreased lipid deposition in HFD mice. Exosomes promoted oxygen consumption in cells and improved cardiac function. These exosomes were enriched in mitochondrial components and stimulated catalytic mechanisms. Hence, they are promising agents in improving glucose and lipid metabolism and uptake in HFD mice as well as preventing the development of related diseases, including DM.133 On the other hand, based on the cargo of exosomes, they can negatively affect glucose metabolism. High glucose levels induce exosome secretion from macrophages, and miRNA-210 was enriched in these exosomes. For instance, the miRNA-210-enriched exosomes interfere with glucose uptake and for this purpose, miRNA-210 decreases the expression level of NDUFA4. Silencing miRNA-210 expression can promote glucose uptake and prevents diabetic obesity development.134 As pathways involved in exosome biogenesis have been identified, future experiments can focus on targeting ESCRT or SNARE proteins in impairing exosome biogenesis and interfering with miRNA-210 delivery to cells to elevate glucose uptake. The exosomes derived from macrophages may not affect insulin-dependent glucose uptake. The stimulation of macrophages by lipopolysaccharide (LPS) mediates exosome secretion from macrophages, and they can induce carbohydrate catabolism. The miRNA-530, chr9_22532, and chr16_34840 show high levels in exosomes derived from LPS-induced macrophages.135
The mesenchymal stem cells are another source of exosomes.136 These exosomes induce glucose and lipid metabolism and uptake, and significantly diminish gluconeogenesis. Notably, autophagy inhibition impairs the therapeutic impact of mesenchymal stem cell-derived exosomes in DM. More investigation revealed that exosomes induced AMP-activated protein kinase (AMPK) to stimulate autophagy via upregulating LC3-II and Beclin-1 for promoting glucose uptake and lipid metabolism.121 It is worth mentioning that AMPK has interactions with mammalian target of rapamycin (mTOR) signaling in autophagy regulation,137 and future experiments can show how AMPK/mTOR axis regulation by exosomes affects autophagy and subsequently, lipid and glucose metabolism in cells for DM treatment. The exosomes are also able to affect transporters involved in glucose uptake and enzymes. The cardiomyocytes demonstrate capacity in the synthesis and secretion of exosomes that promote the levels of GLUT1 and GLUT4 to elevate glucose uptake. Furthermore, exosomes secreted by cardiomyocytes promote lactate dehydrogenase (LDH) activity to induce the glycolytic process for reducing glucose levels.138 Another experiment revealed the role of exosomes derived from hepatic stellate cells in enhancing GLUT1 levels and promoting glucose uptake139 that important in DM treatment. These statements highlight the fact that exosomes are potential modulators of lipid and glucose metabolism and uptake in cells. However, one of the limitations of current experiments is the lack of attention to underlying molecular pathways. For instance, it has been shown that exosomes regulate GLUTs in enhancing glucose uptake. However, there is no trace of related molecular pathways that can be the focus of future studies.
Exosomes and β-Cell Function
There are high numbers of Langerhans islets in the pancreas that is estimated to be 1 million, and each islet is composed of approximately 1000 β cells and an overall weight of 0.9 g.140 The β cells respond to various conditions such as DM and obesity. The interesting point is the β cell mass in obese individuals. The increased levels of insulin in plasma of obese patients, called hyperinsulinemia, is a compensatory process for preventing insulin resistance. Therefore, the mass of β cells enhances in obese patients to induce hyperinsulinemia.141 An obese model in rodents demonstrated enhanced β cell mass up to three times due to obesity.142 The β cell destruction or their lack of capacity in insulin secretion can be observed in TIDM, and based on an experiment, the mass of β cells in TIDM patients over long period of time decreases to near 100%.143–145 However, there are some insights in TIIDM that as insulin levels are high in TIIDM patients, the mass of β cells is unaltered in TIID or enhanced. Therefore, β cell function and its mass are important for preventing DM.
An experiment has utilized exosomes derived from mesenchymal stem cells to treat TIDM. The exosomes were administered to animal models via intravenous rate, and after 6 weeks, the animals were examined in terms of insulin and glucose levels. Based on results, exosomes induce regeneration of β cells in the pancreas and promote their mass that is of importance for promoting insulin secretion in TIDM. In addition to promoting insulin levels, exosomes derived from mesenchymal stem cells significantly diminished glucose levels in serum. Mechanistically, exosomes induce Pdx-1 expression to mediate β cell regeneration and promote insulin secretion. This study provided insights into the biodistribution of exosomes in different organs. The mesenchymal stem cell-derived exosomes show high accumulation in the liver and kidney, and their accumulation is lesser in the lungs and pancreas. Noteworthy, the distribution of these exosomes significantly enhances in the pancreas after 48 h.146 One of the factors responsible for the onset of DM is cancer, in a way that 30% of patients with pancreatic cancer develop DM. The pancreatic stellate cells (PSCs) are present around the islets and neoplastic lesions. The pancreatic tumor cells and PSCs are able to secrete exosomes, which subsequently impair the normal function of β cells and induces insulin resistance, leading to DM development.147
As mentioned previously, the expression level of Pdx-1 can induce regeneration of β cells in DM treatment. Exosomes were isolated from mesenchymal stem cells via a centrifugation process, which was then administered to rats via the intraperitoneal route (200 µg/mL). Noteworthy, these exosomes induced regeneration of β cells and increased insulin secretion. The expression analysis revealed upregulation of Pdx-1, TGF-β, Smad2, and Smad3 in this process.148 Therefore, based on experiments, exosomes can be beneficial in the treatment of TIDM by preventing the impairment and destruction of β cells. Furthermore, exosomes can be advantageous in treating TIIDM via suppressing insulin resistance.149 The exosome transplantation to diabetic mice is correlated with a decrease in macrophage infiltration in β cell islets and decreases CD31 expression as a marker of endothelial cells that together lead to an increase in insulin levels.150
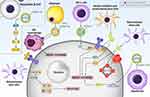
One of the strongest points of any set of experiments in this regard is the attention towards underlying molecular pathways involved in the therapeutic impacts of exosomes in DM. The hypoxia or low levels of oxygen can lead to β cell apoptosis and their inability in insulin secretion via triggering endoplasmic reticulum (ER) stress. For inducing apoptosis, hypoxia promotes BIP and Grp94 expression levels. Then, an increase occurs in phosphorylation of elF2a, leading to CHOP/p38 axis induction. At the next step, p38 reduces survivin expression while it promotes caspase-3 expression to upregulate PARP, resulting in apoptosis induction. Notably, exosomes derived from mesenchymal stem cells can deliver miRNA-21 that inhibits Grp34 expression. Furthermore, exosomes prevent p38 upregulation to suppress apoptosis in β cells.151 However, the story is not always in favor of β cell function. The M1 polarized macrophages can secrete exosomes. Then, exosomes interfere with normal function of β cells and suppress insulin resistance to develop TIDM. The exosomes derived from M1 macrophages contain high level of miRNA-212-5p that decreases sirtuin 2 (SIRT2) expression. Then, Akt down-regulation and subsequent inhibition of GSK-3β occurs. Then, nuclear translocation of β-catenin occurs, and it induces F-actin depolymerization. This activity prevents the secretion of insulin vesicles produced in cells and leads to the development of TIDM.152 Figure 2 summarizes the role of exosomes in modulating β cell function and improving glucose and lipid metabolism.
Exosomes and Insulin Resistance
Insulin resistance is an increasing challenge in the treatment of DM patients, and it means that a shift in insulin concentration-effect curve occurs and it tends to higher concentration levels to exert a similar effect.153 The irresponsive of cells to insulin and inducing insulin resistance can be observed commonly in TIIDM and some cases, in obese people. The insulin is composed of 54 amino acids, and this peptide is generated in β cells. The insulin function is to significantly promote glucose consumption in cells, and it suppresses hepatic glucose generation. Besides, insulin enhances glucose uptake by cells via upregulating expression and activity of GLUTs such as GLUT4.154 However, there are some reasons responsible for insulin resistance. For instance, in obese people, fat accumulation results in pro-inflammatory cytokine release, an increase in lipolysis, and reduced adipokine release. These impacts lead to enhanced levels of fatty acid and free glycerol associated with insulin resistance.155 Recent experiments have focused on the application of nanostructures in reversing insulin resistance. The MgO nanostructures have the capacity of decreasing insulin resistance up to 29.5%.156 Another study reveals the potential of gadofullerene nanostructures in improving the function of the pancreas and suppressing insulin resistance via inducing the IRS-2/PI3K/Akt axis.157 This section focuses on the dual role of exosomes in preventing or inducing insulin resistance.
Depending on their cargo the exosomes can dually suppress/induce insulin resistance in DM. The exosomes containing Sonic Hedgehog (Shh) can induce insulin resistance and significantly promote glucose levels in TIIDM. For exerting such impact, Shh-loaded exosomes induce M1 polarization of macrophages to enhance their pro-inflammatory activity. In this way, Shh-loaded exosomes promote the expression level of Ptch to induce PI3K signaling, leading to M1 polarization of macrophages. More investigation revealed that M1 polarized macrophages and their secreted exosomes reduce expression levels of IRS-1 and HSL in adipocytes. All these interactions and signaling networks lead to insulin resistance and hyperglycemia in TIIDM.158 Previously, it was mentioned that cancer cells could secrete exosomes in triggering DM development and negatively affecting β cells. Noteworthy, exosomes derived from tumor cells can also stimulate insulin resistance in cells. The pancreatic tumor cells have the capacity of exosome secretion, and these nanostructures penetrate into C2C12 myotubes to reduce glucose uptake and mediate lipidosis. The exosomes suppress PI3K/Akt signaling to promote the expression level of FoxO1 and its nuclear translocation. This signaling network diminishes the expression level of GLUT4 to interfere with glucose uptake in cells. Therefore, inhibiting exosome secretion from pancreatic tumor cells or upregulating GLUT4 expression can be beneficial in reversing insulin resistance.159 Adipose tissue is another source of exosome secretion.160 The exosome-like vesicles derived from adipocyte tissue can participate in triggering insulin resistance. In this case, exosomes function as a communicator between macrophages and adipose tissue. Macrophages can absorb the exosome-like vesicles after secretion through adipocyte tissue. Then, exosome-like vesicles mediate the differentiation of monocytes to macrophages and induce secretion of IL-6 and TNF-α, resulting in insulin resistance. Silencing TLR4/TRIF axis prevents insulin resistance by these exosomes.161
However, some experiments show that exosomes can function as therapeutic agents in promoting insulin sensitivity, which is important in DM treatment. The consumption of fatty acids (high consumption) can lead to insulin resistance, and palmitic acid (PA) is among them. The hepatocytes obtain the feature of insulin resistance due to high PA consumption. The PA can induce apoptosis in INS-1 cells, and it diminishes IRS-2 phosphorylation and prevents glucose uptake as a result of insulin induction. The neutral ceramidase (NCDase)-enriched exosomes prevent the generation of reactive oxygen species (maybe in favor of decreasing apoptosis in INS-1 cells) and prevent insulin resistance resulted from high consumption of PA.162 Another experiment uses umbilical cord mesenchymal stem cells to isolate exosomes and their application in TIIDM treatment. These exosomes can promote insulin-induced glucose uptake, and this impact appears to be persistent for at least 48 h. Besides, exosomes decrease levels of leptin, and the expression level of SIRT1 and IRS-1 significantly enhances at mRNA level, showing the capacity of exosomes in elevating insulin sensitivity of adipocytes.163
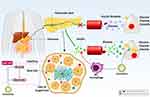
As previously mentioned in the introduction, exosomes are capable of transferring proteins, lipids, and nucleic acids among cells to affect biological events.164,165 The miRNAs are among the most well-known cargoes of exosomes and based on the activity of miRNAs, the impact of exosomes on target cells will be different.166–168 The miRNA-3075 is suggested to possess anti-diabetic activity via enhancing insulin sensitivity. The exosomes derived from hepatocytes show capacity in inducing insulin sensitivity. These exosomes are enriched with miRNA-3075, and its function is decreasing the expression level of FA2H. The process of insulin resistance is related to the pro-inflammatory function of macrophages. The miRNA-3075-loaded exosomes and their capacity in down-regulating FA2H can pave the way for reversing insulin resistance.169 Overall, these studies highlight that exosomes can carry various cargoes to affect the insulin sensitivity of cells. Exosomes possess both functions in triggering and inhibiting insulin resistance.170,171 However, more experiments are required to evaluate other pathways involved in insulin resistance and their regulation by exosomes. Furthermore, other sources of exosomes from stem cells should be utilized in affecting the insulin sensitivity of cells (Figure 3 and Table 2).
 |
Table 2 Exosomes in Regulating Insulin Resistance, β Cell Function, Glucose, and Lipid Metabolism |
Exosomes and Diabetic Complications
Wound Healing
The wound healing process is delayed in DM, and the patients are prone to chronic non-healing wounds.174 In diabetic wounds, there are high levels of pro-inflammatory cytokines and proteases as well as low levels of growth factors.175–177 More than 70% of DM patients receive diabetic medications, and there is little understanding about the impact of these medications on the process of wound healing. The wound healing process is complicated, and various cellular and molecular mechanisms, including inflammation, proliferation, angiogenesis, collagen deposition and re-epithelization occur.178,179 The first step of wound healing is the recruitment of immune cells to eliminate pathogens, and at the next step, wound-associated genes undergo overexpression by cutting epidermal edges to mediate cell migration. The next step is proliferation and migration of fibroblasts to generate wound granulation tissue, and a number of fibroblasts may undergo differentiation to improve wound contraction. Then, angiogenesis occurs, and the wound healing process ends.180 Disturbance in each of these processes can lead to delayed wound healing. The purpose of the current section is to evaluate the role of exosomes in affecting wound healing in DM.
A recent experiment has revealed the potential of exosomes derived from human amniotic epithelial cells in improving the wound healing process in DM. The exosomes had a cup or spherical shape, and their particle size was 105.89 nm. They were positive for CD63 and TSG101 and their uptake occurs by HFBs and HUVECs. These exosomes were able to elevate the growth and migration of HFBs, while they induced angiogenesis in HUVECs. These exosomes stimulated PI3K/Akt/mTOR axis to induce angiogenesis and improve fibroblast function in enhancing wound healing in DM.181 Therefore, inducing angiogenesis appears to be beneficial in improving the wound healing process in DM. Different molecular pathways have been examined in this case. The overexpression of ERK1/2 results in angiogenesis. The exosomes derived from endothelial progenitor cells enhance the expression level of ERK1/2 to induce angiogenesis and facilitate the wound healing process in DM.182 Furthermore, vascular endothelial cells are affected by exosomes in modulating the wound healing process in DM. The exosomes derived from endothelial progenitor cells stimulate tube formation in vascular endothelial cells and promote their growth and migration. More investigation revealed the potential of exosomes in promoting expression levels of FGF-1, VEGFA, VEGFR-2, ANG-1, E-selectin, CXCL-16, eNOS, and IL-8 for improving the wound healing process in DM.183 Hence, Engineering exosomes is advantageous in enhancing wound healing in DM. The expression level of miRNA-31-5p significantly decreases in chronic diabetic wounds, and restoring miRNA-31-5p expression is correlated with the growth and migration of cells in wounds. Loading miRNA-31-5p in exosomes stimulates angiogenesis, fibrogenesis, and reepithelization to promote the wound healing process in DM that are mediated via down-regulating HIF-1 and EMP-1.184 Furthermore, miRNA-221-3p-enriched exosomes show potential in enhancing wound healing in DM and increasing the expression level of VEGF and CD31 at the protein level and promoting proliferation of cells. Furthermore, miRNA-221-3p-enriched exosomes induce the AGE/RAGE axis in improving the wound healing process in DM.185 Based on these experiments, exosomes are mainly involved in improving the wound healing process in DM, and different cells, including endothelial and fibroblast cells are affected. Furthermore, various molecular pathways related to proliferation, migration, and angiogenesis are affected, and most of the experiments have focused on the role of miRNAs.
Neuropathy
Approximately 50% of DM patients demonstrate neuropathy due to diffuse and focal nervous system damages.186 Neuropathy is prevalent among diabetic patients and leads to falls, pain, and reduced quality of life.187 Diabetic neuropathy is responsible for 10 billion costs annually in the USA.188 Therefore, efforts should be made in preventing and management of diabetic neuropathy. Notably, anti-diabetic medications have shown potential in ameliorating DM-related brain damages. Anti-diabetic drugs can significantly diminish neuroinflammation and oxidative damage to prevent the development of neurological diseases such as Alzheimer’s and Parkinson’s diseases. Furthermore, anti-diabetic drugs such as GLP-1R and DPP-4i, among others, can improve neurogenesis and cognitive function.189 This section is focused on the role of exosomes in diabetic neuropathy.
The apoptosis in neurons can be regulated by exosomes. The administration of paeoniflorin improves the therapeutic impacts of exosomes derived from Schwann cells and promotes homeostasis and integrity of the endoplasmic reticulum. Furthermore, paeoniflorin intervention improves the capacity of exosomes in preventing apoptosis in neurons via down-regulating GRP78 and IRE1α. These interactions are vital for preventing neuropathy in DM.190 The exosomes isolated from mesenchymal stem cells demonstrate potential in ameliorating diabetic peripheral neuropathy.191 The underlying molecular pathways affected by these exosomes are of importance. The exosomes derived from mesenchymal stem cells have the capacity of carrying miRNA-146a. The exosomes enriched with miRNA-146a enhance nerve conduction velocity while they can diminish thermal and mechanical stimuli threshold. These exosomes prevent inflammation by monocytes and inhibit endothelial cell activation to ameliorate diabetic neuropathy. The miRNA-146a-enriched exosomes can suppress TLR-4/NF-κB axis in alleviating diabetic neuropathy.192 The inhibition of TLR4/NF-kB axis by exosomes prevents inflammation in ameliorating diabetic neuropathy. More importantly, exosomes derived from mesenchymal stromal cells can decrease M1 polarization of macrophages and promote levels of M2 polarized macrophages in exerting anti-inflammatory activity.193 The neurite outgrowth of neurons and migration of Schwann cells are significantly enhanced by exosomes. In this case, exosomes isolated from Schwann cells are able to promote the maturation of miRNAs, including miRNA-21, −27a, and −146a, while they decrease expression levels of NF-κB, PTEN, SEMA6A, and RhoA to ameliorate diabetic neuropathy.194 The miRNA-126-enriched exosomes have demonstrated potential in decreasing infarct size, promoting neurogenesis, and inducing angiogenesis to prevent brain injury in DM. For exerting such protective impacts, miRNA-126-loaded exosomes decrease caspase-3 expression on day 2 and elevate VEGFR2 expression at day 14.194 The exosomes derived from bone marrow mesenchymal stem cells decrease miRNA-9 expression to provide upregulation of ABCA1, which is important for exerting anti-inflammatory activity and increasing white matter remodeling.195 Based on these experiments, exosomes are potential therapeutic nanostructures for ameliorating diabetic neuropathy, and more experiments are required to evaluate their impact on molecular pathways.
Nephropathy
Nephropathy is another complication of DM that results in end-stage renal disease.196 The incidence rate of nephropathy appears to be 25%. Various mechanisms are responsible for the development of diabetic nephropathy. Hyperglycemia can result in diabetic nephropathy via generating Amadori products and advanced glycation end products. Furthermore, hyperglycemia significantly enhances levels of ROS via inducing the electron transport chain to mediate diabetic nephropathy.197 The treatment of diabetic nephropathy is still challenging, and there is a need for the development of animal models to understand its pathogenesis and drug targets effectively. The nanoparticles can provide selective targeting of renal cells and improve the efficacy of anti-diabetic drugs such as Rhein.198 Furthermore, gold nanostructures can ameliorate podocyte injury and alleviate albuminuria for the treatment of diabetic nephropathy.199
An experiment has cultured urine-derived stem cells, and exosomes with a particle size of 50–100 nm have been isolated that are positive for CD29, CD73, CD90, and CD44 markers, while they are negative for CD34, CD45, and HLA-DR. The intravenous administration of these exosomes seems to be beneficial in decreasing urine volume and urinary microalbumin secretion. Furthermore, exosomes inhibited apoptosis in podocytes and tubular epithelial cells and enhanced endothelial cell growth in diabetic rats. More investigation revealed that exosomes carried high levels of TGF-β1, angiogenin, and BMP-7 that are advantageous in enhancing the viability of renal cells and inducing vascular regeneration in DM.200 There have been efforts to enhance podocytes’ survival rate to prevent nephropathy in DM. The mesangial cells exposed to high glucose levels can secrete exosomes in triggering podocyte injury and mediating diabetic nephropathy. For inducing podocyte apoptosis, the exosomes transfer TGF-β1 among cells. The berberine administration is beneficial in decreasing TGF-β1 transportation by exosomes and reducing its levels to protect podocytes against apoptosis and promote adhesion.201 However, exosomes can possess therapeutic impact in diabetic nephropathy. The exosomes derived from mesenchymal stem cells have high levels of miRNA-125b that down-regulates TRAF6 to induce Akt signaling, resulting in autophagy induction and decreasing apoptosis that are of importance for alleviating diabetic nephropathy.202 One of the important molecular mechanisms is autophagy as a programmed cell death process. Overall, autophagy mediates the degradation of organelles and macromolecules that are aged and toxic to main cell homeostasis. Autophagy has both pro-death and pro-survival functions, and its function is context-dependent.101,189,203–206 Notably, autophagy with pro-survival function can prevent apoptosis in a cell.207 The exosomes derived from mesenchymal stem cells inhibit mTOR signaling as a negative regulator of autophagy to significantly enhance levels of Beclin-1 and LC3 to induce autophagy and prevent apoptosis in alleviating diabetic nephropathy.208 Based on these two experiments, autophagy induction is beneficial in treating diabetic nephropathy. However, autophagy also has a pro-death role, and this dual function should be considered in future experiments.
Endothelial Dysfunction
Most of the sections of this review focused on the TIDM and TIIDM. However, gestational DM also occurs in the second or third trimester of pregnancy, and cells are not responsive to insulin.209,210 The gestational DM can occur in 5–20% of pregnancies worldwide. One of the pathologies occurring in gestational DM is endothelial dysfunction.211 The foetoplacental endothelial dysfunction results from an increase in nitric oxide (NO) synthesis and enhanced transportation of L-arginine.212,213 The exosomes derived from the umbilical vein endothelium could induce endothelial dysfunction and provide a gestational DM phenotype. The transportation of L-arginine is significantly enhanced by using exosomes, and they also enhanced the hCAT-1 and eNOS expression levels. Furthermore, these exosomes induced MAPK signaling to promote uptake of L-arginine in cells. Therefore, suppressing MAPK signaling can prevent the impacts of exosomes in aggravating gestational DM and inducing endothelial dysfunction.214 Notably, exosomes are a way of communication between endothelial cells and monocytes. Exposing both monocytes and endothelial cells to high glucose induces exosome secretion. These exosomes can enhance ICAM-1 expression that is a marker of endothelial cell activation.215 The importance of this experiment is that ICAM-1 upregulation provides a condition for adhesion of immune cells to vascular wall216 and as inflammation occurs in DM, further experiments can focus on reducing ICAM-1 expression in the treatment of DM-mediated inflammation. Another experiment provides more information about the role of exosomes in alleviating endothelial dysfunction and the role of ICAM-1. The endothelial progenitor cells are able to secrete exosomes, and these nanostructures decrease levels of various factors, including malondialdehyde (MDA), superoxide dismutase (SOD), IL-8, and more importantly, ICAM-1 for preventing endothelial dysfunction in DM.217 It is worth mentioning that exosomes carrying arginase 1 are able to stimulate endothelial dysfunction in DM.218 Based on these experiments, exosomes can effectively modulate endothelial function in DM, and their targeting can be considered as a therapeutic strategy.219
Erectile Dysfunction
Erectile dysfunction is defined as the inability to maintain penile erection for at least 6 months.220 DM is considered as a risk factor for erectile dysfunction.221 This disease decreases men's quality of life and self-esteem, and its incidence rate is at the range of 19–90% and can occur 10 years before the start of DM.222–224 Erectile dysfunction is a multifactorial disease. In addition to DM, smoking, cardiovascular diseases, dyslipidemia, hypertension, and obesity are among other factors involved in erectile dysfunction development.225 The most common factors responsible for erectile dysfunction are vasculogenic factors that mediate arterial inflow or venous outflow disorders.226
Exosomes were derived from adipose stem cells and administered via the intracavernous route to promote intracavernous pressure and enhance smooth muscle level compared to collagen, revealed by CD31 overexpression. Furthermore, exosomes decrease the expression level of caspase-3, while they promote Bcl-2 expression to prevent apoptosis. Based on results, exosomes derived from adipose stem cells are therapeutic agents for alleviating erectile dysfunction in DM.227 The adipose stem cell-derived exosomes can induce angiogenesis, increase the proliferation rate of endothelial cells, and prevent erectile dysfunction. Furthermore, these exosomes reduced corpus cavernosum fibrosis. More investigation revealed that exosomes derived from adipose stem cells carried high levels of miRNA-126, −130a, and −132 as proangiogenic factors and miRNA-let7b and -let7c as anti-fibrotic factors.228 Based on the experiments, preventing fibrosis is the best option in ameliorating DM-induced erectile dysfunction. The exosomes derived from smooth muscle cells enhance smooth muscle content and diminish collagen accumulation to exert anti-fibrotic activity. For this purpose, exosomes upregulate expression levels of eNOS and nNOS to induce NO/cGMP axis in improving erectile dysfunction in DM.229
Cardiovascular Diseases
The DM can negatively affect the heart and may develop various cardiovascular diseases such as atherosclerosis.230,231 The polydatin shows potential in decreasing myocardial injury via down-regulating NF-κB and inhibition of NADPH oxidase.232 As NF-κB signaling is involved in inflammatory processes,233 more experiments are required to reveal its role in DM-related cardiovascular diseases. The PLGA-GLP-1 nanocomposites prevent apoptosis and DNA damage and enhance antioxidant activity to ameliorate myocardial injury in DM.234 Furthermore, endothelium ET-1 enhances inflammation and oxidative damage via NOX1 overexpression to aggravate atherosclerosis in DM.235
Apoptosis and necrosis are two types of cell death that commonly occur in cardiomyocytes during DM. Hyperglycemia is involved in triggering cell death in diabetic cardiomyopathy, and there have been efforts in using therapeutic nanostructures in alleviating this condition. A recent experiment has shown that exosomes can be secreted and isolated from parasympathetic ganglionic neurons (PGN), and they show capacities in treating diabetic cardiomyopathy. High glucose levels decrease the survival rate of H9C2 cells and induce apoptosis via down-regulation of Bcl-2, and upregulation of Bax and caspase-3. The exosomes derived from PGN ameliorated diabetic cardiomyopathy and prevented apoptosis.236 Another negative impact in TIIDM is vasa vasorum (VV) angiogenesis, which promotes the possibility of plaque rupture. The insulin resistance adipocytes have the capacity of exosome secretion. These exosomes can be easily taken up by endothelial cells, and in vivo experiment on mice has shown their capacity in penetrating endothelial cells after intravenous administration. Noteworthy, these exosomes contain Sonic Hedgehog, and they can induce VV angiogenesis in aortic plaques. The Gli1 is the downstream target of Sonic Hedgehog, and these exosomes can promote plaque burden in diabetic mice.237 DM patients are prone to atherosclerosis as one of the cardiovascular complications. The macrophages exposed to high glucose secrete exosomes and promote hematopoiesis, atherosclerotic lesions, and several myeloid cells. Furthermore, these exosomes enhanced cell growth rate and induced changes in metabolic profile via upregulating glycolysis. Further investigations revealed that exosomes derived from macrophages have high levels of miRNA-486-5p that mediate their potential in aggravating atherosclerosis in DM.238 Myocardial fibrosis is also another challenge in DM. Notably, TGF-β/Smad signaling is involved in fibrosis.90 The exosomes isolated from mesenchymal stem cells can reduce the expression level of Smad2 via suppressing TGF-β signaling to ameliorate myocardial fibrosis in DM.239 Based on these experiments, exosomes can dually induce or inhibit heart injury by carrying various cargoes such as miRNAs in this case. Engineering novel exosomes can be beneficial in preventing DM-mediated cardiac injury.
Eye Disorders
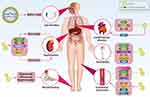
The administration route of exosomes appears to be important in their protective impact on the retina. The intravenous administration of exosomes derived from adipose mesenchymal stem cells enhances the thickness of layers and induces irregularity in the ganglionic layer. The subconjunctival administration of exosomes leads to better results compared to intravenous administration and organizes cellular components of the retina in a well manner. The intraocular administration is considered as the best route and improves retinal layers similar to the normal retina. It has been reported that hyperglycemia reduces miRNA-222 expression. The exosomes derived from mesenchymal stem cells promote miRNA-222 expression to ameliorate retinal degeneration in DM.240 Next experiments will shed more light on the function of exosomes in alleviating retinal degeneration and revealing underlying molecular pathways in DM. Based on these experiments, various diabetic complications occur, including neuropathy, nephropathy, endothelial dysfunction, erectile dysfunction, delayed wound healing, cardiovascular diseases, and eye disorders that can be improved or aggravated by exosomes based on the cargo. Various molecular pathways, including VEGF, IRE1α, Bax, caspase-3, and NLRP3, are affected by the exosomes derived from different sources such as adipose stem cells, mesenchymal cells, and macrophages.190,194,200,202,217,219,241–249 Table 3 provides a summary of exosomes and their application in alleviation of diabetic complications. Figure 4 demonstrates the potential of exosomes in alleviating diabetic complications. Figure 5 is also a summary of exosomes in the treatment of DM.
 |
Table 3 The Role of Exosomes in Improving Diabetic Complications |
Clinical Application
Exosomes can be considered as biomarkers, and they are isolated from body fluids. A recent experiment isolated placental exosomes from urine at the first, second, and third trimesters of gestation. Five miRNAs, including miRNA-516-5p, −517-3p, −518-5p, −16-5p, and −222-3p, were examined in different periods of pregnancy. The expression level of these miRNA significantly enhanced in healthy women during gestation except miRNA-516-5p. Notably, the expression level of these miRNAs underwent down-regulation in gestational DM and at the third trimester of pregnancy. Therefore, these exosomes can be considered valuable sources for diagnosing gestational DM.256 Noteworthy, the size of exosomes significantly enhances in gestational DM patients, and their concentration is also higher. Various circRNAs undergo down-regulation and upregulation in these exosomes, and most of them are related to affecting miRNA expression, which is important for the diagnosis of gestational DM.257 Another source for diagnosis of gestational DM is the umbilical cord, and the presence of lncRNAs and their interaction with miRNA can be a determining factor for initiation and development of gestational DM.258 Besides, exosomes can be derived from adipocytes. The visceral fat thickness is a factor responsible for the development of gestational DM. The exosomes derived from adipocytes carrying the miRNA-148 family affect gestational DM development, and visceral fat thickness modulates exosomes containing the miRNA-148 family.259 The miRNA-125 and miRNA-144 are two miRNAs with differences in expression level in pregnancy and gestational DM. The expression level of miRNA-125 decreases in circulating exosomes and placenta, mediating gestational DM development. The miRNA-144 shows upregulation in circulating exosomes in the third trimester of pregnancy and induces gestational DM development.244 The exosomes can deliver free fatty acids from the bloodstream to cardiac tissue that may result in metabolic disorders such as DM.260 The expression level of lncRNAs H19 and MALAT1 undergoes abnormality in exosomes. The exosomes derived from TIIDM patients confirm the aforementioned statement. The expression level of lncRNA MALAT1 decreases in serum exosomes of TIIDM, while lncRNA H19 shows an increase in expression level.261 Based on these experiments, exosomes can be considered as non-invasive or minimally invasive diagnosis of DM.262–264
Conclusion and Remarks
In recent years, therapeutics for the treatment of DM have been developed in a growing trend and the reason of high incidence rate of this metabolic disorder and altered lifestyle. The DM can occur in both developing and developed countries, and it is more common in urban areas than rural areas due to the lifestyle. In DM, hyperglycemia occurs, which can lead to complications and negative impacts on cells, such as cell death and reduced viability. The present review aimed to investigate the role of exosomes as nanostructures in the treatment of DM.
The glucose uptake and metabolism, as well as lipid metabolism, undergo changes during DM. Exosomes can enhance the metabolism of glucose and lipid in DM. The impact of exosomes will be different based on cargo. For instance, miRNA-210-enriched exosomes inhibit glucose uptake, while exosomes triggering AMPK signaling can induce autophagy which enhances glucose and lipid metabolism. The interesting point is that exosomes can affect β cells in DM. Exosomes derived from pancreatic cancer cells impair β cell function and result in DM development. Noteworthy, exosomes can also promote regeneration and function of β cells via upregulating expression levels of Pdx-1, TGF-β, Smad2, and Smad3. Therefore, exosomes possess both pro-diabetic and anti-diabetic activities. The same story occurs in insulin resistance. The miRNA-3075-enriched exosomes induce insulin sensitivity, while exosomes decreasing expression levels of IRS-1, HSL, and PI3K/Akt, can stimulate insulin resistance. The diabetic complications, including retinal degeneration, osteoporosis, diabetic wound healing, atherosclerosis, neuropathy, and nephropathy, can be dually regulated by exosomes. The interesting point is the clinical trials evaluating the function of exosomes in DM patients, and based on these studies exosomes can be derived from various sources such as urine to detect gestational DM in the third trimester of pregnancy. The enrichment of miRNAs, lncRNAs, and circRNAs in exosomes determines the function of exosomes in DM and their biomarker role. Although different experiments have explored the role of exosomes in DM, some limitations remain that should be the focus of future studies. Most of the studies have disregarded the underlying molecular pathways affected by exosomes in DM. Furthermore, surface modification of exosomes or loading cargo in exosomes can promote their therapeutic activity in DM treatment that has been overlooked by current experiments.
Abbreviations
EVs, extracellular vesicles; MVBs, multivesicular bodies; HSPs, heat shock proteins; mRNA, messenger RNA; miRNA, microRNA; tRNA, transfer RNA; lncRNA, long non-coding RNA; circRNA, circular RNA; DM, diabetes mellitus; ILVs, intraluminal vesicles; Aβ, amyloid-beta; TIDM, type I DM; TIIDM, type II DM; NF-κB, nuclear factor-kappaB; Akt, protein kinase-B; PI3K, phosphoinositide 3-kinase; siRNA, small interfering RNA; shRNA, short hairpin RNA; HFD, high-fat diet; LPS, lipopolysaccharide; AMPK, AMP-activated protein kinase; mTOR, mammalian target of rapamycin; LDH, lactate dehydrogenase; GLUT, glucose transporter; PSCs, pancreatic stellate cells; ER, endoplasmic reticulum; SIRT2, sirtuin 2; Shh, Sonic Hedgehog; NCDase, neutral ceramidase; PA, palmitic acid; NO, nitric oxide; SOD, superoxide dismutase; MDA, malondialdehyde; PGN, parasympathetic ganglionic neurons; VV, vasa vasorum.
Acknowledgments
E.M. would like to acknowledge the support from the National Institute of Biomedical Imaging and Bioengineering (5T32EB009035). The work was supported by a grant from the Singapore Ministry of Education Tier 2 (MOE-T2EP30120-0016) to A.P.K. A.P.K. is also supported by the National Medical Research Council of Singapore and the Singapore Ministry of Education under its Research Centers of Excellence initiative to Cancer Science Institute of Singapore, National University of Singapore.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Disclosure
Amir Reza Aref is the vice president at Translational Sciences, Xsphera Biosciences Inc. The authors declare no other potential conflicts of interest for this work.
References
1. Fu M, Gu J, Jiang P, Qian H, Xu W, Zhang X. Exosomes in gastric cancer: roles, mechanisms, and applications. Mol Cancer. 2019;18(1):1–12.
2. Syn N, Wang L, Sethi G, et al. Exosome-mediated metastasis: from epithelial-mesenchymal transition to escape from immunosurveillance. Trends Pharmacol Sci. 2016;37(7):606–617. doi:10.1016/j.tips.2016.04.006
3. Raposo G, Nijman HW, Stoorvogel W, et al. B lymphocytes secrete antigen-presenting vesicles. J Exp Med. 1996;183(3):1161–1172. doi:10.1084/jem.183.3.1161
4. Wee I, Syn N, Sethi G, et al. Role of tumor-derived exosomes in cancer metastasis. Biochim Biophys Acta Rev Cancer. 2019;1871(1):12–19. doi:10.1016/j.bbcan.2018.10.004
5. Zhang L, Yu DJ. Exosomes in cancer development, metastasis, and immunity. Biochimica Et Biophysica Acta. 2019;1871(2):455–468. doi:10.1016/j.bbcan.2019.04.004
6. Whiteside TL. Tumor-derived exosomes and their role in cancer progression. Adv Clin Chem. 2016;74:103–141. doi:10.1016/bs.acc.2015.12.005
7. Wilson DH, Rissin DM, Kan CW, et al. The simoa HD-1 analyzer: a novel fully automated digital immunoassay analyzer with single-molecule sensitivity and multiplexing. J Lab Autom. 2016;21(4):533–547. doi:10.1177/2211068215589580
8. Théry C, Ostrowski M, Segura E. Membrane vesicles as conveyors of immune responses. Nat Rev Immunol. 2009;9(8):581–593. doi:10.1038/nri2567
9. Weng J, Xiang X, Ding L, et al. Extracellular vesicles, the cornerstone of next-generation cancer diagnosis? Semin Cancer Biol. 2021;74:105–120. doi:10.1016/j.semcancer.2021.05.011
10. Zhang H, Freitas D, Kim HS, et al. Identification of distinct nanoparticles and subsets of extracellular vesicles by asymmetric flow field-flow fractionation. Nat Cell Biol. 2018;20(3):332–343. doi:10.1038/s41556-018-0040-4
11. Jayasinghe MK, Tan M, Peng B, et al. New approaches in extracellular vesicle engineering for improving the efficacy of anti-cancer therapies. Semin Cancer Biol. 2021;74:62–78. doi:10.1016/j.semcancer.2021.02.010
12. Zhang X, Yuan X, Shi H, et al. Exosomes in cancer: small particle, big player. J Hematol Oncol. 2015;8:83. doi:10.1186/s13045-015-0181-x
13. Mincheva-Nilsson L, Baranov V. Cancer exosomes and NKG2D receptor-ligand interactions: impairing NKG2D-mediated cytotoxicity and anti-tumour immune surveillance. Semin Cancer Biol. 2014;28:24–30. doi:10.1016/j.semcancer.2014.02.010
14. Colombo M, Raposo G, Théry C. Biogenesis, secretion, and intercellular interactions of exosomes and other extracellular vesicles. Annu Rev Cell Dev Biol. 2014;30:255–289. doi:10.1146/annurev-cellbio-101512-122326
15. Kowal J, Tkach M, Théry C. Biogenesis and secretion of exosomes. Curr Opin Cell Biol. 2014;29:116–125. doi:10.1016/j.ceb.2014.05.004
16. Théry C, Zitvogel L, Amigorena S. Exosomes: composition, biogenesis and function. Nat Rev Immunol. 2002;2(8):569–579. doi:10.1038/nri855
17. Simons M, Raposo G. Exosomes–vesicular carriers for intercellular communication. Curr Opin Cell Biol. 2009;21(4):575–581. doi:10.1016/j.ceb.2009.03.007
18. Runz S, Keller S, Rupp C, et al. Malignant ascites-derived exosomes of ovarian carcinoma patients contain CD24 and EpCAM. Gynecol Oncol. 2007;107(3):563–571. doi:10.1016/j.ygyno.2007.08.064
19. Baran J, Baj-Krzyworzeka M, Weglarczyk K, et al. Circulating tumour-derived microvesicles in plasma of gastric cancer patients. Cancer Immunol Immunother. 2010;59(6):841–850. doi:10.1007/s00262-009-0808-2
20. Ciravolo V, Huber V, Ghedini GC, et al. Potential role of HER2-overexpressing exosomes in countering trastuzumab-based therapy. J Cell Physiol. 2012;227(2):658–667. doi:10.1002/jcp.22773
21. Valadi H, Ekström K, Bossios A, et al. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol. 2007;9(6):654–659. doi:10.1038/ncb1596
22. Ratajczak J, Miekus K, Kucia M, et al. Embryonic stem cell-derived microvesicles reprogram hematopoietic progenitors: evidence for horizontal transfer of mRNA and protein delivery. Leukemia. 2006;20(5):847–856. doi:10.1038/sj.leu.2404132
23. Gusachenko ON, Zenkova MA, Vlassov VV. Nucleic acids in exosomes: disease markers and intercellular communication molecules. Biochemistry (Mosc). 2013;78(1):1–7. doi:10.1134/S000629791301001X
24. Ashrafizaveh S, Ashrafizadeh M, Zarrabi A, et al. Long non-coding RNAs in the doxorubicin resistance of cancer cells. Cancer Lett. 2021;508:104–114. doi:10.1016/j.canlet.2021.03.018
25. Bullock MD, Silva AM, Kanlikilicer-Unaldi P, et al. Exosomal non-coding RNAs: diagnostic, prognostic and therapeutic applications in cancer. Noncoding RNA. 2015;1(1):53–68. doi:10.3390/ncrna1010053
26. Sun Y, Sun Y, Li C, et al. The utility of exosomes in diagnosis and therapy of diabetes mellitus and associated complications. Optics Express. 2021;29:1360. doi:10.1364/OE.410603
27. Lei LM, Lin X, Xu F, et al. Exosomes and obesity-related insulin resistance. Front Cell Dev Biol. 2021;9:651996. doi:10.3389/fcell.2021.651996
28. Cui X, Zhu L, Zhai R, Zhang B, Zhang F. Mesenchymal stem cell-derived exosomes: a promising vector in treatment for diabetes and its microvascular complications. Am J Transl Res. 2021;13(5):3942–3953.
29. Chen J, Zhang Q, Liu D, et al. Exosomes: advances, development and potential therapeutic strategies in diabetic nephropathy. Metabolism. 2021;122:154834. doi:10.1016/j.metabol.2021.154834
30. Zhang B, Yang Y, Xiang L, et al. Adipose-derived exosomes: a novel adipokine in obesity-associated diabetes. J Cell Physiol. 2019;234(10):16692–16702. doi:10.1002/jcp.28354
31. Castaño C, Novials A, Párrizas M. Exosomes and diabetes. Diabetes Metab Res Rev. 2019;35(3):e3107. doi:10.1002/dmrr.3107
32. Chang W, Wang J. Exosomes and their noncoding RNA cargo are emerging as new modulators for diabetes mellitus. Cells. 2019;8(8):853.
33. Beuzelin D, Kaeffer B. Exosomes and miRNA-loaded biomimetic nanovehicles, a focus on their potentials preventing type-2 diabetes linked to metabolic syndrome. Front Immunol. 2018;9:2711. doi:10.3389/fimmu.2018.02711
34. Garcia-Contreras M, Brooks RW, Boccuzzi L, Robbins PD, Ricordi C. Exosomes as biomarkers and therapeutic tools for type 1 diabetes mellitus. Eur Rev Med Pharmacol Sci. 2017;21(12):2940–2956.
35. Li X, Shi S, Jing D, et al. Signal transduction mechanism of exosomes in diabetic complications (Review). Exp Ther Med. 2022;23(2):155. doi:10.3892/etm.2021.11078
36. Chang W, Wang JJC. Exosomes and their noncoding RNA cargo are emerging as new modulators for diabetes mellitus. Cells. 2019;8(8):853.
37. Ahmadi M, Rezaie JJ. Ageing and mesenchymal stem cells derived exosomes: molecular insight and challenges. Cell Biochem Funct. 2021;39(1):60–66. doi:10.1002/cbf.3602
38. Hamdan Y, Mazini L, Malka GJB. Exosomes and micro-RNAs in aging process. Biomedicines. 2021;9(8):968. doi:10.3390/biomedicines9080968
39. Liu Q, Piao H, Wang Y, et al. Circulating exosomes in cardiovascular disease: novel carriers of biological information. BioMed Pharmacother. 2021;135:111148. doi:10.1016/j.biopha.2020.111148
40. Nikfarjam S, Rezaie J, Kashanchi F, Jafari R. Dexosomes as a cell-free vaccine for cancer immunotherapy. J Exp Clin Cancer Res. 2020;39(1):1–20.
41. Hassanpour M, Rezabakhsh A, Rezaie J, Nouri M, Rahbarghazi R. Exosomal cargos modulate autophagy in recipient cells via different signaling pathways. Cell Biosci. 2020;10(1):1–16.
42. Rezaie J, Aslan C, Ahmadi M, Zolbanin NM, Kashanchi F, Jafari R. The versatile role of exosomes in human retroviral infections: from immunopathogenesis to clinical application. Cell Biosci. 2021;11(1):1–15.
43. Babaei M, Rezaie JJ. Application of stem cell-derived exosomes in ischemic diseases: opportunity and limitations. J Transl Med. 2021;19(1):1–11.
44. Abels ER, Breakefield XO. Introduction to extracellular vesicles: biogenesis, RNA cargo selection, content, release, and uptake. Cell Mol Neurobiol. 2016;36(3):301–312. doi:10.1007/s10571-016-0366-z
45. Grant BD, Donaldson JG. Pathways and mechanisms of endocytic recycling. Nat Rev Mol Cell Biol. 2009;10(9):597–608. doi:10.1038/nrm2755
46. Zhao C, Zhang G, Liu J, et al. Exosomal cargoes in OSCC: current findings and potential functions. PeerJ. 2020;8:e10062. doi:10.7717/peerj.10062
47. Ghossoub R, Lembo F, Rubio A, et al. Syntenin-ALIX exosome biogenesis and budding into multivesicular bodies are controlled by ARF6 and PLD2. Nat Commun. 2014;5:3477. doi:10.1038/ncomms4477
48. Saheera S, Potnuri AG, Krishnamurthy P. Nano-vesicle (Mis)communication in senescence-related pathologies. Cells. 2020;9(9):1974. doi:10.3390/cells9091974
49. Liao W, Du Y, Zhang C, et al. Exosomes: the next generation of endogenous nanomaterials for advanced drug delivery and therapy. Acta Biomater. 2019;86:1–14. doi:10.1016/j.actbio.2018.12.045
50. Henne WM, Stenmark H, Emr SD. Molecular mechanisms of the membrane sculpting ESCRT pathway. Cold Spring Harbor Perspect Biol. 2013;5(9):a016766. doi:10.1101/cshperspect.a016766
51. Stuffers S, Sem Wegner C, Stenmark H, et al. Multivesicular endosome biogenesis in the absence of ESCRTs. Traffic (Copenhagen, Denmark). 2009;10(7):925–937. doi:10.1111/j.1600-0854.2009.00920.x
52. McMahon HT, Boucrot EJ. Membrane curvature at a glance. J Cell Sci. 2015;128(6):1065–1070.
53. Conde-Vancells J, Rodriguez-Suarez E, Embade N, et al. Characterization and comprehensive proteome profiling of exosomes secreted by hepatocytes. J Proteome Res. 2008;7(12):5157–5166. doi:10.1021/pr8004887
54. Subra C, Grand D, Laulagnier K, et al. Exosomes account for vesicle-mediated transcellular transport of activatable phospholipases and prostaglandins [S]. J Lipid Res. 2010;51(8):2105–2120. doi:10.1194/jlr.M003657
55. Reddy VS, Madala SK, Trinath J, Reddy GB. Extracellular small heat shock proteins: exosomal biogenesis and function. Cell Stress Chaperones. 2018;23(3):441–454.
56. Möbius W, van Donselaar E, Ohno-Iwashita Y, et al. Recycling compartments and the internal vesicles of multivesicular bodies harbor most of the cholesterol found in the endocytic pathway. Traffic. 2003;4(4):222–231. doi:10.1034/j.1600-0854.2003.00072.x
57. Bu H, He D, He X, et al. Exosomes: isolation, analysis, and applications in cancer detection and therapy. Chembiochem. 2019;20(4):451–461. doi:10.1002/cbic.201800470
58. Shao H, Chung J, Balaj L, et al. Protein typing of circulating microvesicles allows real-time monitoring of glioblastoma therapy. Nat Med. 2012;18(12):1835–1840. doi:10.1038/nm.2994
59. Witwer KW, Buzás EI, Bemis LT, et al. Standardization of sample collection, isolation and analysis methods in extracellular vesicle research. J Extracell Vesic. 2013;2(1):20360.
60. Liu C, Guo J, Tian F, et al. Field-free isolation of exosomes from extracellular vesicles by microfluidic viscoelastic flows. ACS Nano. 2017;11(7):6968–6976. doi:10.1021/acsnano.7b02277
61. Batrakova EV, Kim MS. Using exosomes, Naturally-equipped nanocarriers, for drug delivery. J Control Release. 2015;219:396–405. doi:10.1016/j.jconrel.2015.07.030
62. Zhu L, Sun HT, Wang S, et al. Isolation and characterization of exosomes for cancer research. J Hematol Oncol. 2020;13(1):1–24.
63. Gurunathan S, Kang MH, Jeyaraj M, Qasim M, Kim JH. Review of the isolation, characterization, biological function, and multifarious therapeutic approaches of exosomes. Cells. 2019;8(4):307.
64. Ye Z, Zhu Z, Xie J, et al. Hsa_circ_0000069 knockdown inhibits tumorigenesis and exosomes with downregulated hsa_circ_0000069 suppress malignant transformation via inhibition of STIL in pancreatic cancer. Int J NanoMedicine. 2020;15:9859–9873. doi:10.2147/IJN.S279258
65. Pang H, Liu L, Sun X, et al. Exosomes derived from colon cancer cells and plasma of colon cancer patients promote migration of SW480 cells through Akt/mTOR pathway. Pathol Res Pract. 2021;222:153454. doi:10.1016/j.prp.2021.153454
66. Jiang Y, Wang K, Lu X, et al. Cancer-associated fibroblasts-derived exosomes promote lung cancer progression by OIP5-AS1/ miR-142-5p/ PD-L1 axis. Mol Immunol. 2021;140:47–58. doi:10.1016/j.molimm.2021.10.002
67. Wang X, Yang G. Bone marrow mesenchymal stem cells-derived exosomes reduce Aβ deposition and improve cognitive function recovery in mice with Alzheimer’s disease by activating sphingosine kinase/sphingosine-1-phosphate signaling pathway. Cell Biol Int. 2021;45(4):775–784. doi:10.1002/cbin.11522
68. Qi Y, Guo L, Jiang Y, et al. Brain delivery of quercetin-loaded exosomes improved cognitive function in AD mice by inhibiting phosphorylated tau-mediated neurofibrillary tangles. Drug Deliv. 2020;27(1):745–755. doi:10.1080/10717544.2020.1762262
69. Yu H, Yang X, Xiao X, et al. Human adipose mesenchymal stem cell-derived exosomes protect mice from DSS-induced inflammatory bowel disease by promoting intestinal-stem-cell and epithelial regeneration. Aging Dis. 2021;12(6):1423–1437. doi:10.14336/AD.2021.0601
70. Yang S, Liang X, Song J, et al. A novel therapeutic approach for inflammatory bowel disease by exosomes derived from human umbilical cord mesenchymal stem cells to repair intestinal barrier via TSG-6. Stem Cell Res Ther. 2021;12(1):315. doi:10.1186/s13287-021-02404-8
71. Kumar S, Behl T, Sachdeva M, et al. Implicating the effect of ketogenic diet as a preventive measure to obesity and diabetes mellitus. Life Sci. 2021;264:118661. doi:10.1016/j.lfs.2020.118661
72. Mandal N, Grambergs R, Mondal K, et al. Role of ceramides in the pathogenesis of diabetes mellitus and its complications. J Diabetes Complicat. 2021;35(2):107734. doi:10.1016/j.jdiacomp.2020.107734
73. Behl T, Arora A, Sehgal A, et al. Molecular and biochemical pathways encompassing diabetes mellitus and dementia. CNS Neurol Disord Drug Targets. 2021;20. doi:10.2174/1871527320666211110115257
74. Ashish A, Shah A, Pandey SJ. Interaction between oxidative stress and diabetes: a mini-review. J Diabetes Metab Disord Control. 2020;7(2):58–61.
75. Bekele H, Asefa A, Getachew B, Belete AM. Barriers and strategies to lifestyle and dietary pattern interventions for prevention and management of TYPE-2 diabetes in Africa, systematic review. J Diabetes Res. 2020;2020:7948712.
76. Khan MAB, Hashim MJ, King JK, et al. Epidemiology of type 2 diabetes–global burden of disease and forecasted trends. J Epidemiol Glob Health. 2020;10(1):107.
77. Wilcox GJ. Insulin and insulin resistance. Clin Biochem Rev. 2005;26(2):19.
78. Gosmanov AR, Gosmanova EO, Dillard-Cannon E. Management of adult diabetic ketoacidosis. Diabetes Metabol Syndr Obes. 2014;7:255.
79. Wang HJB. MicroRNA, diabetes mellitus and colorectal cancer. Biomedicines. 2020;8(12):530.
80. American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care. 2013;36(Suppl 1):S67–S74. doi:10.2337/dc13-S067
81. Wang J, Pan Y, Dai F, et al. Serum miR-195-5p is upregulated in gestational diabetes mellitus. J Clin Lab Anal. 2020;34(8):e23325. doi:10.1002/jcla.23325
82. Resanović I, Zarić B, Radovanović J, et al. Hyperbaric oxygen therapy and vascular complications in diabetes mellitus. Angiology. 2020;71(10):876–885. doi:10.1177/0003319720936925
83. Atkinson MA, Maclaren NK. The pathogenesis of insulin-dependent diabetes mellitus. N Engl J Med. 1994;331(21):1428–1436. doi:10.1056/NEJM199411243312107
84. Pociot FJC. Type 1 diabetes genome‐wide association studies: not to be lost in translation. Immunology. 2017;6(12):e162.
85. Bradfield JP, Qu HQ, Wang K, et al. A genome-wide meta-analysis of six type 1 diabetes cohorts identifies multiple associated loci. PLoS genet. 2011;7(9):e1002293.
86. Larsen CM, Faulenbach M, Vaag A, et al. Interleukin-1–receptor antagonist in type 2 diabetes mellitus. N Engl J Med. 2007;356(15):1517–1526. doi:10.1056/NEJMoa065213
87. Cleland S, Fisher BM, Colhoun HM, et al. Insulin resistance in type 1 diabetes: what is ‘double diabetes’ and what are the risks? Diabetologia. 2013;56(7):1462–1470. doi:10.1007/s00125-013-2904-2
88. Pourbagher-Shahri AM, Farkhondeh T, Ashrafizadeh M, et al. Curcumin and cardiovascular diseases: focus on cellular targets and cascades. Biomed Pharmacother. 2021;136:111214. doi:10.1016/j.biopha.2020.111214
89. Farkhondeh T, Ashrafizadeh M, Azimi-Nezhad M, et al. Curcumin efficacy in a serum/glucose deprivation-induced neuronal PC12 injury model. Curr Mol Pharmacol. 2021;14:1146–1155. doi:10.2174/1874467214666210203211312
90. Ashrafizadeh M, Zarrabi A, Hushmandi K, et al. Toward regulatory effects of curcumin on transforming growth factor-beta across different diseases: a review. Front Pharmacol. 2020;11:1785.
91. Ashrafizadeh M, Najafi M, Makvandi P, et al. Versatile role of curcumin and its derivatives in lung cancer therapy. J Cell Physiol. 2020;235(12):9241–9268. doi:10.1002/jcp.29819
92. Ashrafizadeh M, Zarrabi A, Hashemi F, et al. Polychemotherapy with curcumin and doxorubicin via biological nanoplatforms: enhancing antitumor activity. Pharmaceutics. 2020;12(11):1084.
93. Ashrafizadeh M, Zarrabi A, Hashemipour M, et al. Sensing the scent of death: modulation of microRNAs by curcumin in gastrointestinal cancers. Pharmacol Res. 2020;160:105199. doi:10.1016/j.phrs.2020.105199
94. Ashrafizadeh M, Zarrabi A, Hashemi F, et al. Curcumin in cancer therapy: a novel adjunct for combination chemotherapy with paclitaxel and alleviation of its adverse effects. Life Sci. 2020;256:117984. doi:10.1016/j.lfs.2020.117984
95. Mohammadi Z, Sharif Zak M, Majdi H, et al. The effect of chrysin–curcumin-loaded nanofibres on the wound-healing process in male rats. Artif Cells Nanomed Biotechnol. 2019;47(1):1642–1652. doi:10.1080/21691401.2019.1594855
96. Ren BC, Zhang Y-F, Liu -S-S, et al. Curcumin alleviates oxidative stress and inhibits apoptosis in diabetic cardiomyopathy via Sirt1-Foxo1 and PI3K-Akt signalling pathways. J Cell Mol Med. 2020;24(21):12355–12367. doi:10.1111/jcmm.15725
97. Shafabakhsh R, Mobini M, Raygan F, et al. Curcumin administration and the effects on psychological status and markers of inflammation and oxidative damage in patients with type 2 diabetes and coronary heart disease. Clin Nutr ESPEN. 2020;40:77–82. doi:10.1016/j.clnesp.2020.09.029
98. Hussain Y, Mirzaei S, Ashrafizadeh M, et al. Quercetin and its nano-scale delivery systems in prostate cancer therapy: paving the way for cancer elimination and reversing chemoresistance. Cancers. 2021;13(7):1602.
99. Najafi M, Tavakol S, Zarrabi A, Ashrafizadeh M. Dual role of quercetin in enhancing the efficacy of cisplatin in chemotherapy and protection against its side effects: a review. Archiv Physiol Biochem. 2020;2020:1–15.
100. Ashrafizadeh M, Samarghandian S, Hushmandi K, et al. Quercetin in attenuation of ischemic/reperfusion injury: a review. Curr Mol Pharmacol. 2020;14(4):537–558.
101. Ashrafizadeh M, Ahmadi Z, Farkhondeh T, Samarghandian S. Autophagy as a molecular target of quercetin underlying its protective effects in human diseases. Archiv Physiol Biochem. 2019;2019:1–9.
102. Soltantabar P, Calubaquib EL, Mostafavi E, et al. Enhancement of loading efficiency by coloading of doxorubicin and quercetin in thermoresponsive polymeric micelles. Biomacromolecules. 2020;21(4):1427–1436. doi:10.1021/acs.biomac.9b01742
103. Zhang F, Feng J, Zhang J, et al. Quercetin modulates AMPK/SIRT1/NF‑κB signaling to inhibit inflammatory/oxidative stress responses in diabetic high fat diet‑induced atherosclerosis in the rat carotid artery. Exp Ther Med. 2020;20(6):280. doi:10.3892/etm.2020.9410
104. Ojo OO, Olorunsogo OO. Quercetin and vitamin E attenuate diabetes-induced testicular anomaly in Wistar rats via the mitochondrial-mediated apoptotic pathway. Andrologia. 2021;53(10):e14185. doi:10.1111/and.14185
105. Ashrafizadeh M, Rafiei H, Mohammadinejad R, Farkhondeh T, Samarghandian S. Anti-tumor activity of resveratrol against gastric cancer: a review of recent advances with an emphasis on molecular pathways. Cancer Cell Int. 2021;21(1):1–10.
106. Amini P, Nodooshan SJ, Ashrafizadeh M, et al. Resveratrol induces apoptosis and attenuates proliferation of MCF-7 cells in combination with radiation and hyperthermia. Curr Mol Med. 2021;21(2):142–150. doi:10.2174/1566524020666200521080953
107. Ashrafizadeh M, Zarrabi A, Najafi M, et al. Resveratrol targeting tau proteins, amyloid‐beta aggregations, and their adverse effects: an updated review. Phytother Res. 2020;34(11):2867–2888. doi:10.1002/ptr.6732
108. Ashrafizadeh M, Najafi M, Orouei S, et al. Resveratrol modulates transforming growth factor-beta (tgf-β) signaling pathway for disease therapy: a new insight into its pharmacological activities. Biomedicines. 2020;8(8):261.
109. Farkhondeh T, Folgado SL, Pourbagher-Shahri AM, et al. The therapeutic effect of resveratrol: focusing on the Nrf2 signaling pathway. Biomed Pharmacother. 2020;127:110234. doi:10.1016/j.biopha.2020.110234
110. Ashrafizadeh M, Ahmadi Z, Farkhondeh T, et al. Resveratrol targeting the Wnt signaling pathway: a focus on therapeutic activities. Journal of Cellular Physiology. 2020;235(5):4135–4145. doi:10.1002/jcp.29327
111. Simas JN, Mendes TB, Fischer LW, et al. Resveratrol improves sperm DNA quality and reproductive capacity in type 1 diabetes. Andrology. 2021;9(1):384–399. doi:10.1111/andr.12891
112. Sun X, Cao Z, Ma Y, et al. Resveratrol attenuates dapagliflozin-induced renal gluconeogenesis via activating the PI3K/Akt pathway and suppressing the FoxO1 pathway in type 2 diabetes. Food Funct. 2021;12(3):1207–1218. doi:10.1039/D0FO02387F
113. Wang S, Chen X, Wang M, et al. SiRNA-Cyp4a14 and diabetic nephropathy: silencing of Cyp4a14 by siRNA inhibits proliferation and fibrosis of mesangial cells. Int J Clin Exp Pathol. 2017;10(12):11909–11917.
114. Rodrigues AM, Serralha RS, Lima DY, et al. P2X7 siRNA targeted to the kidneys increases klotho and delays the progression of experimental diabetic nephropathy. Purinergic Signal. 2020;16(2):175–185. doi:10.1007/s11302-020-09695-1
115. Hu B, Zhang Y, Zeng Q, et al. Intravitreal injection of ranibizumab and CTGF shRNA improves retinal gene expression and microvessel ultrastructure in a rodent model of diabetes. Int J Mol Sci. 2014;15(1):1606–1624. doi:10.3390/ijms15011606
116. Li L, Yang J, Liu B, et al. P2Y12 shRNA normalizes inflammatory dysfunctional hepatic glucokinase activity in type 2 diabetic rats. Biomed Pharmacother. 2020;132:110803. doi:10.1016/j.biopha.2020.110803
117. Mirzaei S, Mahabady MK, Zabolian A, et al. Small interfering RNA (siRNA) to target genes and molecular pathways in glioblastoma therapy: current status with an emphasis on delivery systems. Life Sci. 2021;275:119368. doi:10.1016/j.lfs.2021.119368
118. Mirzaei S, Gholami MH, Hashemi F, et al. Employing siRNA tool and its delivery platforms in suppressing cisplatin resistance: approaching to a new era of cancer chemotherapy. Life Sci. 2021;277:119430. doi:10.1016/j.lfs.2021.119430
119. Mirzaei S, Gholami MH, Zabolian A, et al. Caffeic acid and its derivatives as potential modulators of oncogenic molecular pathways: new hope in the fight against cancer. Pharmacol Res. 2021;171:105759. doi:10.1016/j.phrs.2021.105759
120. Moghadam ER, Ang HL, Asnaf SE, et al. Broad-spectrum preclinical antitumor activity of chrysin: current trends and future perspectives. Biomolecules. 2020;10(10):1374.
121. Zhang L, Chu W, Zheng L, et al. Zinc oxide nanoparticles from Cyperus rotundus attenuates diabetic retinopathy by inhibiting NLRP3 inflammasome activation in STZ-induced diabetic rats. J Biochem Mol Toxicol. 2020;34(12):e22583. doi:10.1002/jbt.22583
122. Khan T, Khan S, Akhtar M, et al. Empagliflozin nanoparticles attenuates type2 diabetes induced cognitive impairment via oxidative stress and inflammatory pathway in high fructose diet induced hyperglycemic mice. Neurochem Int. 2021;150:105158. doi:10.1016/j.neuint.2021.105158
123. Lari AS, Zahedi P, Ghourchian H, et al. Microfluidic-based synthesized carboxymethyl chitosan nanoparticles containing metformin for diabetes therapy: in vitro and in vivo assessments. Carbohydr Polym. 2021;261:117889. doi:10.1016/j.carbpol.2021.117889
124. Cesur S, Cam ME, Sayın FS, et al. Metformin-loaded polymer-based microbubbles/nanoparticles generated for the treatment of type 2 diabetes mellitus. Langmuir. 2021. doi:10.1021/acs.langmuir.1c00587
125. Mostafavi E, Soltantabar P, Webster TJ. Nanotechnology and picotechnology: a new arena for translational medicine. In: Biomaterials in Translational Medicine. Elsevier; 2019:191–212.
126. Smith SM, Boppana A, Traupman JA, et al. Impaired glucose metabolism in patients with diabetes, prediabetes, and obesity is associated with severe COVID-19. J Med Virol. 2021;93(1):409–415. doi:10.1002/jmv.26227
127. Li C, Yang Y, Liu X, et al. Glucose metabolism-related gene polymorphisms as the risk predictors of type 2 diabetes. Diabetol Metab Syndr. 2020;12(1):97. doi:10.1186/s13098-020-00604-5
128. Song L, Yuan J, Liu Y, et al. Ghrelin system is involved in improvements in glucose metabolism mediated by hyperbaric oxygen treatment in a streptozotocin‑induced type 1 diabetes mouse model. Mol Med Rep. 2020;22(5):3767–3776. doi:10.3892/mmr.2020.11481
129. Miras AD, Kamocka A, Pérez-Pevida B, et al. The effect of standard versus longer intestinal bypass on GLP-1 regulation and glucose metabolism in patients with type 2 diabetes undergoing Roux-en-Y gastric bypass: the long-limb study. Diabetes Care. 2021;44(5):1082–1090. doi:10.2337/dc20-0762
130. Tanaka T, Wada T, Uno K, et al. Oestrogen receptor α in T cells controls the T cell immune profile and glucose metabolism in mouse models of gestational diabetes mellitus. Diabetologia. 2021;64(7):1660–1673. doi:10.1007/s00125-021-05447-x
131. Fernandes GW, Bocco B. Hepatic mediators of lipid metabolism and ketogenesis: focus on fatty liver and diabetes. Curr Diabetes Rev. 2021;17(7):e110320187539. doi:10.2174/1573399816999201103141216
132. Erukainure OL, Sanni O, Salau VF, et al. Cola nitida (kola nuts) attenuates hepatic injury in type 2 diabetes by improving antioxidant and cholinergic dysfunctions and dysregulated lipid metabolism. Endocr Metab Immune Disord Drug Targets. 2021;21(4):688–699. doi:10.2174/1871530320666200628030138
133. Zhou X, Li Z, Qi M, et al. Brown adipose tissue-derived exosomes mitigate the metabolic syndrome in high fat diet mice. Theranostics. 2020;10(18):8197–8210. doi:10.7150/thno.43968
134. Tian F, Tang P, Sun Z, et al. miR-210 in exosomes derived from macrophages under high glucose promotes mouse diabetic obesity pathogenesis by suppressing NDUFA4 expression. J Diabetes Res. 2020;2020:6894684. doi:10.1155/2020/6894684
135. De silva N, Samblas M, Martínez JA, et al. Effects of exosomes from LPS-activated macrophages on adipocyte gene expression, differentiation, and insulin-dependent glucose uptake. J Physiol Biochem. 2018;74(4):559–568. doi:10.1007/s13105-018-0622-4
136. Mandal S, Arfuso F, Sethi G, et al. Encapsulated human mesenchymal stem cells (eMSCs) as a novel anti-cancer agent targeting breast cancer stem cells: development of 3D primed therapeutic MSCs. Int J Biochem Cell Biol. 2019;110:59–69. doi:10.1016/j.biocel.2019.02.001
137. Ashrafizadeh M, Mirzaei S, Hushmandi K, et al. Therapeutic potential of AMPK signaling targeting in lung cancer: advances, challenges and future prospects. Life Sci. 2021;278:119649. doi:10.1016/j.lfs.2021.119649
138. Garcia NA, Moncayo-Arlandi J, Sepulveda P, et al. Cardiomyocyte exosomes regulate glycolytic flux in endothelium by direct transfer of GLUT transporters and glycolytic enzymes. Cardiovasc Res. 2016;109(3):397–408. doi:10.1093/cvr/cvv260
139. Wan L, Xia T, Du Y, et al. Exosomes from activated hepatic stellate cells contain GLUT1 and PKM2: a role for exosomes in metabolic switch of liver nonparenchymal cells. FASEB J. 2019;33(7):8530–8542. doi:10.1096/fj.201802675R
140. Wysham C, Shubrook JJ. Beta-cell failure in type 2 diabetes: mechanisms, markers, and clinical implications. Postgrad Med. 2020;132(8):676–686. doi:10.1080/00325481.2020.1771047
141. Inaishi J, Saisho YJN, Zhao Y. Beta-cell mass in obesity and type 2 diabetes, and its relation to pancreas fat: a mini-review. Nanoscale. 2020;12(12):3846. doi:10.1039/c9nr09869k
142. Tschen SI, Dhawan S, Gurlo T, et al. Age-dependent decline in beta-cell proliferation restricts the capacity of beta-cell regeneration in mice. Diabetes. 2009;58(6):1312–1320. doi:10.2337/db08-1651
143. Atkinson MA, Eisenbarth GS, Michels AW. Type 1 diabetes. Lancet. 2014;383(9911):69–82. doi:10.1016/S0140-6736(13)60591-7
144. Löhr M, Klöppel G. Residual insulin positivity and pancreatic atrophy in relation to duration of chronic type 1 (insulin-dependent) diabetes mellitus and microangiopathy. Diabetologia. 1987;30(10):757–762. doi:10.1007/BF00275740
145. Meier JJ, Bhushan A, Butler AE, et al. Sustained beta cell apoptosis in patients with long-standing type 1 diabetes: indirect evidence for islet regeneration? Diabetologia. 2005;48(11):2221–2228. doi:10.1007/s00125-005-1949-2
146. Mahdipour E, Salmasi Z, Sabeti N. Potential of stem cell-derived exosomes to regenerate β islets through Pdx-1 dependent mechanism in a rat model of type 1 diabetes. J Cell Physiol. 2019;234(11):20310–20321. doi:10.1002/jcp.28631
147. Perera CJ, Falasca M, Chari ST, et al. Role of pancreatic stellate cell-derived exosomes in pancreatic cancer-related diabetes: a novel hypothesis. Cancers (Basel). 2021;13(20):5224. doi:10.3390/cancers13205224
148. Sabry D, Marzouk S, Zakaria R, et al. The effect of exosomes derived from mesenchymal stem cells in the treatment of induced type 1 diabetes mellitus in rats. Biotechnol Lett. 2020;42(8):1597–1610. doi:10.1007/s10529-020-02908-y
149. Sun Y, Shi H, Yin S, et al. Human mesenchymal stem cell derived exosomes alleviate type 2 diabetes mellitus by reversing peripheral insulin resistance and relieving β-cell destruction. ACS Nano. 2018;12(8):7613–7628. doi:10.1021/acsnano.7b07643
150. Sun Y, Mao Q, Shen C, et al. Exosomes from β-cells alleviated hyperglycemia and enhanced angiogenesis in islets of streptozotocin-induced diabetic mice. Diabetes Metab Syndr Obes. 2019;12:2053–2064. doi:10.2147/DMSO.S213400
151. Chen J, Chen J, Cheng Y, et al. Mesenchymal stem cell-derived exosomes protect beta cells against hypoxia-induced apoptosis via miR-21 by alleviating ER stress and inhibiting p38 MAPK phosphorylation. Stem Cell Res Ther. 2020;11(1):97. doi:10.1186/s13287-020-01610-0
152. Qian B, Yang Y, Tang N, et al. M1 macrophage-derived exosomes impair beta cell insulin secretion via miR-212-5p by targeting SIRT2 and inhibiting Akt/GSK-3β/β-catenin pathway in mice. Diabetologia. 2021;64(9):2037–2051. doi:10.1007/s00125-021-05489-1
153. Mastrototaro L, Roden MJM. Insulin resistance and insulin sensitizing agents. Metabol Clin Exp. 2021;125:154892. doi:10.1016/j.metabol.2021.154892
154. Sędzikowska A, Szablewski LJ. Insulin and insulin resistance in Alzheimer’s disease. Int J Mol Sci. 2021;22(18):9987. doi:10.3390/ijms22189987
155. Galli M, Hameed A, Żbikowski A, et al. Aquaporins in insulin resistance and diabetes: more than channels! Redox Biol. 2021;44:102027. doi:10.1016/j.redox.2021.102027
156. Jeevanandam J, Chan YS, Danquah MK, et al. Cytotoxicity analysis of morphologically different sol-gel-synthesized MgO Nanoparticles and their in vitro insulin resistance reversal ability in adipose cells. Appl Biochem Biotechnol. 2020;190(4):1385–1410. doi:10.1007/s12010-019-03166-z
157. Li X, Zhen M, Zhou C, et al. Gadofullerene nanoparticles reverse dysfunctions of pancreas and improve hepatic insulin resistance for type 2 diabetes mellitus treatment. ACS Nano. 2019;13(8):8597–8608. doi:10.1021/acsnano.9b02050
158. Song M, Han L, Chen -F-F, et al. Adipocyte-derived exosomes carrying sonic hedgehog mediate M1 macrophage polarization-induced insulin resistance via ptch and PI3K pathways. Cell Physiol Biochem. 2018;48(4):1416–1432. doi:10.1159/000492252
159. Wang L, Zhang B, Zheng W, et al. Exosomes derived from pancreatic cancer cells induce insulin resistance in C2C12 myotube cells through the PI3K/Akt/FoxO1 pathway. Sci Rep. 2017;7(1):5384. doi:10.1038/s41598-017-05541-4
160. Żbikowski A, Błachnio-Zabielska A, Galli M, et al. Adipose-derived exosomes as possible players in the development of insulin resistance. Int J Mol Sci. 2021;22(14):7427. doi:10.3390/ijms22147427
161. Deng ZB, Poliakov A, Hardy RW, et al. Adipose tissue exosome-like vesicles mediate activation of macrophage-induced insulin resistance. Diabetes. 2009;58(11):2498–2505. doi:10.2337/db09-0216
162. Zhu Q, Zhu R, Jin J. Neutral ceramidase-enriched exosomes prevent palmitic acid-induced insulin resistance in H4IIEC3 hepatocytes. FEBS Open Bio. 2016;6(11):1078–1084. doi:10.1002/2211-5463.12125
163. Chen MT, Zhao Y-T, Zhou L-Y, et al. Exosomes derived from human umbilical cord mesenchymal stem cells enhance insulin sensitivity in insulin resistant human adipocytes. Curr Med Sci. 2021;41(1):87–93. doi:10.1007/s11596-021-2323-4
164. Liu J, Jiang F, Jiang Y, et al. Roles of exosomes in ocular diseases. Int J Nanomedicine. 2020;15:10519–10538. doi:10.2147/IJN.S277190
165. Tellez-Gabriel M, Knutsen E, Perander M. Current status of circulating tumor cells, circulating tumor DNA, and exosomes in breast cancer liquid biopsies. Int J Mol Sci. 2020;21(24):9457. doi:10.3390/ijms21249457
166. Xie L, Chen Z, Liu M, et al. MSC-derived exosomes protect vertebral endplate chondrocytes against apoptosis and calcification via the miR-31-5p/ATF6 axis. Mol Ther Nucleic Acids. 2020;22:601–614. doi:10.1016/j.omtn.2020.09.026
167. Zhao Y, Gan Y, Xu G, et al. Exosomes from MSCs overexpressing microRNA-223-3p attenuate cerebral ischemia through inhibiting microglial M1 polarization mediated inflammation. Life Sci. 2020;260:118403. doi:10.1016/j.lfs.2020.118403
168. Qiu L, Chen W, Wu C, et al. Exosomes of oral squamous cell carcinoma cells containing miR-181a-3p induce muscle cell atrophy and apoptosis by transmissible endoplasmic reticulum stress signaling. Biochem Biophys Res Commun. 2020;533(4):831–837. doi:10.1016/j.bbrc.2020.09.066
169. Ji Y, Luo Z, Gao H, et al. Hepatocyte-derived exosomes from early onset obese mice promote insulin sensitivity through miR-3075. Nat Metab. 2021;3(9):1163–1174. doi:10.1038/s42255-021-00444-1
170. Nair S, Jayabalan N, Guanzon D, et al. Human placental exosomes in gestational diabetes mellitus carry a specific set of miRNAs associated with skeletal muscle insulin sensitivity. Clin Sci (Lond). 2018;132(22):2451–2467. doi:10.1042/CS20180487
171. Apostolopoulou M, Mastrototaro L, Hartwig S, et al. Metabolic responsiveness to training depends on insulin sensitivity and protein content of exosomes in insulin-resistant males. Sci Adv. 2021;7(41):eabi9551. doi:10.1126/sciadv.abi9551
172. Li F, Li H, Jin X, et al. Adipose-specific knockdown of Sirt1 results in obesity and insulin resistance by promoting exosomes release. Cell Cycle. 2019;18(17):2067–2082. doi:10.1080/15384101.2019.1638694
173. He Q, Wang L, Zhao R, et al. Mesenchymal stem cell-derived exosomes exert ameliorative effects in type 2 diabetes by improving hepatic glucose and lipid metabolism via enhancing autophagy. Stem Cell Res Ther. 2020;11(1):223. doi:10.1186/s13287-020-01731-6
174. Salazar JJ, Ennis WJ, Koh TJ. Diabetes medications: impact on inflammation and wound healing. J Diabetes Complicat. 2016;30(4):746–752.
175. Eming SA, Martin P, Tomic-Canic M. Wound repair and regeneration: mechanisms, signaling, and translation. Sci Transl Med. 2014;6(265):265sr6. doi:10.1126/scitranslmed.3009337
176. Mirza RE, Fang MM, Weinheimer-Haus EM, et al. Sustained inflammasome activity in macrophages impairs wound healing in type 2 diabetic humans and mice. Diabetes. 2014;63(3):1103–1114. doi:10.2337/db13-0927
177. Kalantari K, Mostafavi E, Afifi AM, et al. Wound dressings functionalized with silver nanoparticles: promises and pitfalls. Nanoscale. 2020;12(4):2268–2291. doi:10.1039/C9NR08234D
178. Falanga V. Wound healing and its impairment in the diabetic foot. Lancet. 2005;366(9498):1736–1743. doi:10.1016/S0140-6736(05)67700-8
179. Reiber GE, Raugi GJ. Preventing foot ulcers and amputations in diabetes. Lancet. 2005;366(9498):1676–1677. doi:10.1016/S0140-6736(05)67674-X
180. Martin P, Nunan RJ. Cellular and molecular mechanisms of repair in acute and chronic wound healing. Br J Dermatol. 2015;173(2):370–378. doi:10.1111/bjd.13954
181. Wei P, Zhong C, Yang X, et al. Exosomes derived from human amniotic epithelial cells accelerate diabetic wound healing via PI3K-AKT-mTOR-mediated promotion in angiogenesis and fibroblast function. Burns Trauma. 2020;8:tkaa020. doi:10.1093/burnst/tkaa020
182. Zhang J, Chen C, Hu B, et al. Exosomes derived from human endothelial progenitor cells accelerate cutaneous wound healing by promoting angiogenesis through Erk1/2 signaling. Int J Biol Sci. 2016;12(12):1472–1487. doi:10.7150/ijbs.15514
183. Li X, Jiang C, Zhao J. Human endothelial progenitor cells-derived exosomes accelerate cutaneous wound healing in diabetic rats by promoting endothelial function. J Diabetes Complications. 2016;30(6):986–992. doi:10.1016/j.jdiacomp.2016.05.009
184. Huang J, Yu M, Yin W, et al. Development of a novel RNAi therapy: engineered miR-31 exosomes promoted the healing of diabetic wounds. Bioact Mater. 2021;6(9):2841–2853. doi:10.1016/j.bioactmat.2021.02.007
185. Xu J, Bai S, Cao Y, et al. miRNA-221-3p in endothelial progenitor cell-derived exosomes accelerates skin wound healing in diabetic mice. Diabetes Metab Syndr Obes. 2020;13:1259–1270. doi:10.2147/DMSO.S243549
186. Feldman EL, Callaghan BC, Pop-Busui R, et al. Diabetic neuropathy. Nat Rev Dis Primers. 2019;5(1):41. doi:10.1038/s41572-019-0092-1
187. Pop-Busui R, Boulton AJM, Feldman EL, et al. Diabetic neuropathy: a position statement by the American Diabetes Association. Diabetes Care. 2017;40(1):136–154. doi:10.2337/dc16-2042
188. Gordois A, Scuffham P, Shearer A, et al. The health care costs of diabetic peripheral neuropathy in the US. Diabetes Care. 2003;26(6):1790–1795. doi:10.2337/diacare.26.6.1790
189. Yaribeygi H, Ashrafizadeh M, Henney NC, et al. Neuromodulatory effects of anti-diabetes medications: a mechanistic review. Pharmacol Res. 2020;152:104611. doi:10.1016/j.phrs.2019.104611
190. Zhu Y, Han S, Li X, et al. Paeoniflorin effect of Schwann cell-derived exosomes ameliorates dorsal root ganglion neurons apoptosis through IRE1α pathway. Evid Based Complement Alternat Med. 2021;2021:6079305. doi:10.1155/2021/6079305
191. Singh A, Raghav A, Shiekh PA, et al. Transplantation of engineered exosomes derived from bone marrow mesenchymal stromal cells ameliorate diabetic peripheral neuropathy under electrical stimulation. Bioact Mater. 2021;6(8):2231–2249. doi:10.1016/j.bioactmat.2021.01.008
192. Fan B, Chopp M, Zhang ZG, et al. Treatment of diabetic peripheral neuropathy with engineered mesenchymal stromal cell-derived exosomes enriched with microRNA-146a provide amplified therapeutic efficacy. Exp Neurol. 2021;341:113694. doi:10.1016/j.expneurol.2021.113694
193. Fan B, Li C, Szalad A, et al. Mesenchymal stromal cell-derived exosomes ameliorate peripheral neuropathy in a mouse model of diabetes. Diabetologia. 2020;63(2):431–443. doi:10.1007/s00125-019-05043-0
194. Wang J, Chen S, Zhang W, et al. Exosomes from miRNA-126-modified endothelial progenitor cells alleviate brain injury and promote functional recovery after stroke. CNS Neurosci Ther. 2020;26(12):1255–1265. doi:10.1111/cns.13455
195. Venkat P, Zacharek A, Landschoot-Ward J, et al. Exosomes derived from bone marrow mesenchymal stem cells harvested from type two diabetes rats promotes neurorestorative effects after stroke in type two diabetes rats. Exp Neurol. 2020;334:113456. doi:10.1016/j.expneurol.2020.113456
196. Azushima K, Gurley SB, Coffman TM. Modelling diabetic nephropathy in mice. Nat Rev Nephrol. 2018;14(1):48–56. doi:10.1038/nrneph.2017.142
197. Nagib AM, Elsayed Matter Y, Gheith OA, et al. Diabetic nephropathy following posttransplant diabetes mellitus. Exp Clin Transplant. 2019;17(2):138–146.
198. Chen D, Han S, Zhu Y, et al. Kidney-targeted drug delivery via rhein-loaded polyethyleneglycol-co-polycaprolactone-co-polyethylenimine nanoparticles for diabetic nephropathy therapy. Int J Nanomedicine. 2018;13:3507–3527. doi:10.2147/IJN.S166445
199. Alomari G, Al-Trad B, Hamdan S, et al. Gold nanoparticles attenuate albuminuria by inhibiting podocyte injury in a rat model of diabetic nephropathy. Drug Deliv Transl Res. 2020;10(1):216–226. doi:10.1007/s13346-019-00675-6
200. Jiang ZZ, Liu Y-M, Niu X, et al. Exosomes secreted by human urine-derived stem cells could prevent kidney complications from type I diabetes in rats. Stem Cell Res Ther. 2016;7:24. doi:10.1186/s13287-016-0287-2
201. Wang YY, Tang LQ, Wei W. Berberine attenuates podocytes injury caused by exosomes derived from high glucose-induced mesangial cells through TGFβ1-PI3K/AKT pathway. Eur J Pharmacol. 2018;824:185–192. doi:10.1016/j.ejphar.2018.01.034
202. Cai X, Zou F, Xuan R, et al. Exosomes from mesenchymal stem cells expressing microribonucleic acid-125b inhibit the progression of diabetic nephropathy via the tumour necrosis factor receptor-associated factor 6/Akt axis. Endocr J. 2021;68(7):817–828. doi:10.1507/endocrj.EJ20-0619
203. Ashrafizadeh M, Zarrabi A, Orouei S, et al. MicroRNA-mediated autophagy regulation in cancer therapy: the role in chemoresistance/chemosensitivity. Eur J Pharmacol. 2021;892:173660. doi:10.1016/j.ejphar.2020.173660
204. Ashrafizadeh M, Ahmadi Z, Farkhondeh T, et al. Modulatory effects of statins on the autophagy: a therapeutic perspective. J Cell Physiol. 2020;235(4):3157–3168. doi:10.1002/jcp.29227
205. Ashrafizadeh M, Tavakol S, Ahmadi Z, et al. Therapeutic effects of kaempferol affecting autophagy and endoplasmic reticulum stress. Phytother Res. 2020;34(5):911–923. doi:10.1002/ptr.6577
206. Mohammadinejad R, Ahmadi Z, Tavakol S, Ashrafizadeh M. Berberine as a potential autophagy modulator. J Cell Physiol. 2019;234(9):14914–14926.
207. Wang D, He X, Wang D, et al. Quercetin suppresses apoptosis and attenuates intervertebral disc degeneration via the SIRT1-autophagy pathway. Front Cell Dev Biol. 2020;8:613006. doi:10.3389/fcell.2020.613006
208. Ebrahim N, Ahmed I, Hussien N, et al. Mesenchymal stem cell-derived exosomes ameliorated diabetic nephropathy by autophagy induction through the mTOR signaling pathway. Cells. 2018;7(12):226. doi:10.3390/cells7120226
209. Kerner W, Brückel J, et al. Definition, classification and diagnosis of diabetes mellitus. Exp Clin Endocrinol Diabetes. 2014;122(7):384–386. doi:10.1055/s-0034-1366278
210. National Diabetes Data Group. Classification and diagnosis of diabetes mellitus and other categories of glucose intolerance. Diabetes. 1979;28(12):1039–1057.
211. Sultan SA, Liu W, Peng Y, et al. The role of maternal gestational diabetes in inducing fetal endothelial dysfunction. J Cell Physiol. 2015;230(11):2695–2705. doi:10.1002/jcp.24993
212. Sobrevia L, Salsoso R, Sáez T, et al. Insulin therapy and fetoplacental vascular function in gestational diabetes mellitus. Exp Physiol. 2015;100(3):231–238. doi:10.1113/expphysiol.2014.082743
213. Leiva A, Fuenzalida B, Barros E, et al. Nitric oxide is a central common metabolite in vascular dysfunction associated with diseases of human pregnancy. Curr Vasc Pharmacol. 2016;14(3):237–259. doi:10.2174/1570161114666160222115158
214. Sáez T, Salsoso R, Leiva A, et al. Human umbilical vein endothelium-derived exosomes play a role in foetoplacental endothelial dysfunction in gestational diabetes mellitus. Biochim Biophys Acta Mol Basis Dis. 2018;1864(2):499–508. doi:10.1016/j.bbadis.2017.11.010
215. Sáez T, de Vos P, Kuipers J, et al. Exosomes derived from monocytes and from endothelial cells mediate monocyte and endothelial cell activation under high d-glucose conditions. Immunobiology. 2019;224(2):325–333. doi:10.1016/j.imbio.2019.02.004
216. Karimi Z, Kahe F, Jamil A, et al. Intercellular adhesion molecule-1 in diabetic patients with and without microalbuminuria. Diabetes Metabol Syndr. 2018;12(3):365–368. doi:10.1016/j.dsx.2017.12.028
217. Bai S, Yin Q, Dong T, et al. Endothelial progenitor cell-derived exosomes ameliorate endothelial dysfunction in a mouse model of diabetes. Biomed Pharmacother. 2020;131:110756. doi:10.1016/j.biopha.2020.110756
218. Zhang H, Liu J, Qu D, et al. Serum exosomes mediate delivery of arginase 1 as a novel mechanism for endothelial dysfunction in diabetes. Proc Natl Acad Sci U S A. 2018;115(29):E6927–E6936. doi:10.1073/pnas.1721521115
219. Nie Y, Sato Y, Garner RT, et al. Skeletal muscle-derived exosomes regulate endothelial cell functions via reactive oxygen species-activated nuclear factor-κB signalling. Exp Physiol. 2019;104(8):1262–1273. doi:10.1113/EP087396
220. Ma JX, Wang B, Li HS, et al. Uncovering the mechanisms of leech and centipede granules in the treatment of diabetes mellitus-induced erectile dysfunction utilising network pharmacology. J Ethnopharmacol. 2021;265:113358. doi:10.1016/j.jep.2020.113358
221. Al-Oanzi ZH. Erectile dysfunction attenuation by naringenin in streptozotocin‐induced diabetic rats. J Food Biochem. 2019;43(7):e12885. doi:10.1111/jfbc.12885
222. Giugliano F, Maiorino M, Bellastella G, et al. Determinants of erectile dysfunction in type 2 diabetes. Int J Impotence Res. 2010;22(3):204–209. doi:10.1038/ijir.2010.1
223. Zhang H, Tong W-T, Zhang C-R, et al. Gross saponin of Tribulus terrestris improves erectile dysfunction in type 2 diabetic rats by repairing the endothelial function of the penile corpus cavernosum. Diabetes Metabol Syndr Obes. 2019;12:1705. doi:10.2147/DMSO.S205722
224. Chen S, Zhu J, Wang M, et al. Comparison of the therapeutic effects of adipose‑derived and bone marrow mesenchymal stem cells on erectile dysfunction in diabetic rats. Int J Mol Med. 2019;44(3):1006–1014. doi:10.3892/ijmm.2019.4254
225. Defeudis G, Mazzilli R, Tenuta M, et al. Erectile dysfunction and diabetes: a melting pot of circumstances and treatments. Diabetes Metabol Res Rev. 2021;38:e3494. doi:10.1002/dmrr.3494
226. Gandaglia G, Briganti A, Jackson G, et al. A systematic review of the association between erectile dysfunction and cardiovascular disease. Eur Urol. 2014;65(5):968–978. doi:10.1016/j.eururo.2013.08.023
227. Chen F, Zhang H, Wang Z, et al. Adipose-derived stem cell-derived exosomes ameliorate erectile dysfunction in a rat model of type 2 diabetes. J Sex Med. 2017;14(9):1084–1094. doi:10.1016/j.jsxm.2017.07.005
228. Zhu LL, Huang X, Yu W, et al. Transplantation of adipose tissue-derived stem cell-derived exosomes ameliorates erectile function in diabetic rats. Andrologia. 2018;50(2):e12871. doi:10.1111/and.12871
229. Song J, Sun T, Tang Z, et al. Exosomes derived from smooth muscle cells ameliorate diabetes-induced erectile dysfunction by inhibiting fibrosis and modulating the NO/cGMP pathway. J Cell Mol Med. 2020;24(22):13289–13302. doi:10.1111/jcmm.15946
230. Di Bartolo BA, Cartland SP, Genner S, et al. HDL improves cholesterol and glucose homeostasis and reduces atherosclerosis in diabetes-associated atherosclerosis. J Diabetes Res. 2021;2021:6668506. doi:10.1155/2021/6668506
231. Laddha AP, Kulkarni YA. Daidzein mitigates myocardial injury in streptozotocin-induced diabetes in rats. Life Sci. 2021;284:119664. doi:10.1016/j.lfs.2021.119664
232. Tan YY, Chen L-X, Fang L, et al. Cardioprotective effects of polydatin against myocardial injury in diabetic rats via inhibition of NADPH oxidase and NF-κB activities. BMC Complement Med Ther. 2020;20(1):378. doi:10.1186/s12906-020-03177-y
233. Mirzaei S, Zarrabi A, Hashemi F, et al. Regulation of Nuclear Factor-KappaB (NF-κB) signaling pathway by non-coding RNAs in cancer: inhibiting or promoting carcinogenesis? Cancer Lett. 2021;509:63–80. doi:10.1016/j.canlet.2021.03.025
234. Zhang Y, Zhuang S, Jiang S, et al. Preparation of nanocomposite peptide and its inhibitory effect on myocardial injury in type-II diabetic rats. J Nanosci Nanotechnol. 2021;21(2):1378–1384. doi:10.1166/jnn.2021.18652
235. Ouerd S, Idris-Khodja N, Trindade M, et al. Endothelium-restricted endothelin-1 overexpression in type 1 diabetes worsens atherosclerosis and immune cell infiltration via NOX1. Cardiovasc Res. 2021;117(4):1144–1153. doi:10.1093/cvr/cvaa168
236. Singla R, Garner KH, Samsam M, et al. Exosomes derived from cardiac parasympathetic ganglionic neurons inhibit apoptosis in hyperglycemic cardiomyoblasts. Mol Cell Biochem. 2019;462(1–2):1–10. doi:10.1007/s11010-019-03604-w
237. Wang F, Chen -F-F, Shang -Y-Y, et al. Insulin resistance adipocyte-derived exosomes aggravate atherosclerosis by increasing vasa vasorum angiogenesis in diabetic ApoE(-/-) mice. Int J Cardiol. 2018;265:181–187. doi:10.1016/j.ijcard.2018.04.028
238. Bouchareychas L, Duong P, Phu TA, et al. High glucose macrophage exosomes enhance atherosclerosis by driving cellular proliferation & hematopoiesis. iScience. 2021;24(8):102847. doi:10.1016/j.isci.2021.102847
239. Lin Y, Zhang F, Lian X-F, et al. Mesenchymal stem cell-derived exosomes improve diabetes mellitus-induced myocardial injury and fibrosis via inhibition of TGF-β1/Smad2 signaling pathway. Cell Mol Biol (Noisy-le-Grand). 2019;65(7):123–126. doi:10.14715/cmb/2019.65.7.21
240. Safwat A, Sabry D, Ragiae A, et al. Adipose mesenchymal stem cells-derived exosomes attenuate retina degeneration of streptozotocin-induced diabetes in rabbits. J Circ Biomark. 2018;7:1849454418807827. doi:10.1177/1849454418807827
241. Sáez T, de Vos P, Kuipers J, et al. Fetoplacental endothelial exosomes modulate high d-glucose-induced endothelial dysfunction. Placenta. 2018;66:26–35. doi:10.1016/j.placenta.2018.04.010
242. Wang Y, Ma W-Q, Zhu Y, et al. Exosomes derived from mesenchymal stromal cells pretreated with advanced glycation end product-bovine serum albumin inhibit calcification of vascular smooth muscle cells. Front Endocrinol (Lausanne). 2018;9:524. doi:10.3389/fendo.2018.00524
243. Maisto R, Oltra M, Vidal-Gil L, et al. ARPE-19-derived VEGF-containing exosomes promote neovascularization in HUVEC: the role of the melanocortin receptor 5. Cell Cycle. 2019;18(4):413–424. doi:10.1080/15384101.2019.1568745
244. Zhang L, Wang Q, Su H, et al. Exosomes from adipose derived mesenchymal stem cells alleviate diabetic osteoporosis in rats through suppressing NLRP3 inflammasome activation in osteoclasts. J Biosci Bioeng. 2021;131(6):671–678. doi:10.1016/j.jbiosc.2021.02.007
245. Xu N, Wang L, Guan J, et al. Wound healing effects of a Curcuma zedoaria polysaccharide with platelet-rich plasma exosomes assembled on chitosan/silk hydrogel sponge in a diabetic rat model. Int J Biol Macromol. 2018;117:102–107. doi:10.1016/j.ijbiomac.2018.05.066
246. Afrisham R, Sadegh-Nejadi S, Meshkani R, et al. Effect of circulating exosomes derived from normal-weight and obese women on gluconeogenesis, glycogenesis, lipogenesis and secretion of FGF21 and fetuin A in HepG2 cells. Diabetol Metab Syndr. 2020;12:32. doi:10.1186/s13098-020-00540-4
247. Zhao H, Shang Q, Pan Z, et al. Exosomes from adipose-derived stem cells attenuate adipose inflammation and obesity through polarizing M2 macrophages and beiging in white adipose tissue. Diabetes. 2018;67(2):235–247. doi:10.2337/db17-0356
248. Yang J, Chen Z, Pan D, et al. Umbilical cord-derived mesenchymal stem cell-derived exosomes combined pluronic F127 hydrogel promote chronic diabetic wound healing and complete skin regeneration. Int J Nanomedicine. 2020;15:5911–5926. doi:10.2147/IJN.S249129
249. Qiu J, Shu C, Li X, et al. Exosomes from linc00511-overexpressing ADSCs accelerates angiogenesis in diabetic foot ulcers healing by suppressing PAQR3-induced Twist1 degradation. Diabetes Res Clin Pract. 2021;180:109032. doi:10.1016/j.diabres.2021.109032
250. Han X, Wu P, Li L, et al. Exosomes derived from autologous dermal fibroblasts promote diabetic cutaneous wound healing through the Akt/β-catenin pathway. Cell Cycle. 2021;20(5–6):616–629. doi:10.1080/15384101.2021.1894813
251. Li X, Xie X, Lian W, et al. Exosomes from adipose-derived stem cells overexpressing Nrf2 accelerate cutaneous wound healing by promoting vascularization in a diabetic foot ulcer rat model. Exp Mol Med. 2018;50(4):1–14.
252. Yu M, Liu W, Li J, et al. Exosomes derived from atorvastatin-pretreated MSC accelerate diabetic wound repair by enhancing angiogenesis via AKT/eNOS pathway. Stem Cell Res Ther. 2020;11(1):350. doi:10.1186/s13287-020-01824-2
253. Li M, Wang T, Tian H, et al. Macrophage-derived exosomes accelerate wound healing through their anti-inflammation effects in a diabetic rat model. Artif Cells Nanomed Biotechnol. 2019;47(1):3793–3803. doi:10.1080/21691401.2019.1669617
254. Shi R, Jin Y, Hu W, et al. Exosomes derived from mmu_circ_0000250-modified adipose-derived mesenchymal stem cells promote wound healing in diabetic mice by inducing miR-128-3p/SIRT1-mediated autophagy. Am J Physiol Cell Physiol. 2020;318(5):C848–c856. doi:10.1152/ajpcell.00041.2020
255. Wang J, Xia Y, Li J, et al. miR-129-5p in exosomes inhibits diabetes-associated osteogenesis in the jaw via targeting FZD4. Biochem Biophys Res Commun. 2021;566:87–93. doi:10.1016/j.bbrc.2021.05.072
256. Herrera-van Oostdam AS, Toro‑Ortíz J, López J, et al. Placental exosomes isolated from urine of patients with gestational diabetes exhibit a differential profile expression of microRNAs across gestation. Int J Mol Med. 2020;46(2):546–560. doi:10.3892/ijmm.2020.4626
257. Cao M, Zhang L, Lin Y, et al. Circular RNA expression profiles in umbilical cord blood exosomes from normal and gestational diabetes mellitus patients. Biosci Rep. 2020;40(11). doi:10.1042/BSR20201946
258. Cao M, Zhang L, Lin Y, et al. Differential mRNA and Long Noncoding RNA Expression Profiles in Umbilical Cord Blood Exosomes from Gestational Diabetes Mellitus Patients. DNA Cell Biol. 2020;39(11):2005–2016. doi:10.1089/dna.2020.5783
259. Zhang Z, Xu Q, Chen Y, et al. The possible role of visceral fat in early pregnancy as a predictor of gestational diabetes mellitus by regulating adipose-derived exosomes miRNA-148 family: protocol for a nested case-control study in a cohort study. BMC Pregnancy Childbirth. 2021;21(1):262. doi:10.1186/s12884-021-03737-1
260. Garcia NA, González-King H, Grueso E, et al. Circulating exosomes deliver free fatty acids from the bloodstream to cardiac cells: possible role of CD36. PLoS One. 2019;14(5):e0217546. doi:10.1371/journal.pone.0217546
261. Tello-Flores VA, Valladares-Salgado A, Ramírez-Vargas MA, et al. Altered levels of MALAT1 and H19 derived from serum or serum exosomes associated with type-2 diabetes. Noncoding RNA Res. 2020;5(2):71–76. doi:10.1016/j.ncrna.2020.03.001
262. Mirza AH, Kaur S, Nielsen LB, et al. Breast Milk-Derived Extracellular Vesicles Enriched in Exosomes From Mothers With Type 1 Diabetes Contain Aberrant Levels of microRNAs. Front Immunol. 2019;10:2543. doi:10.3389/fimmu.2019.02543
263. Zhang J, Chi H, Wang T, et al. Altered Amyloid-β and Tau Proteins in Neural-Derived Plasma Exosomes of Type 2 Diabetes Patients with Orthostatic Hypotension. J Alzheimers Dis. 2021;82(1):261–272. doi:10.3233/JAD-210216
264. Shah KB, Chernausek SD, Teague AM, et al. Maternal diabetes alters microRNA expression in fetal exosomes, human umbilical vein endothelial cells and placenta. Pediatr Res. 2021;89(5):1157–1163. doi:10.1038/s41390-020-1060-x
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.