Back to Journals » International Medical Case Reports Journal » Volume 9
Exceptionally elevated triglyceride in severe lipemia retinalis
Authors Yin HY, Warman R, Suh EH, Cheng AM
Received 1 August 2016
Accepted for publication 22 September 2016
Published 21 October 2016 Volume 2016:9 Pages 333—336
DOI https://doi.org/10.2147/IMCRJ.S118594
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Han Y Yin,1–3 Roberto Warman,2,4 Edward H Suh,2 Anny MS Cheng2,3
1Wayne State University, School of Medicine, Detroit, MI, 2Department of Ophthalmology, Florida International University, Herbert Wertheim College of Medicine, 3Ocular Surface Center, Miami, 4Division of Pediatric Ophthalmology, Nicklaus Children’s Hospital, Miami, FL, USA
Purpose: To report a case of successful treatment for severe lipemia retinalis with extreme severe hypertriglyceridemia (sHTG).
Design: Observational case report.
Observations: A 6-week-old infant with severe lipemia retinalis manifested diffuse creamy retinal vessels complicated with vulvar xanthomas. Extreme sHTG with 185-folds of the normal level was reported. Chromosome microarray and lipid gene sequencing confirmed a homozygous lipoprotein lipase gene coding mutation.
Results: Under strict adherence to a high medium-chain triglycerides formula and discontinuation of breast milk, the lipemia retinalis and vulval lesions resolved along with a stable plasma lipid level throughout the follow-up period of 6 months.
Conclusion: Strict adherence to a low-fat diet without breast milk appears to be effective in treating infants with severe lipemia retinalis associated with exceptionally high triglycerides.
Keywords: hypertriglyceride, infant, lipemia retinalis, lipoprotein lipase gene
Introduction
Lipemia retinalis is a rare creamy white discoloration of the retina due to hyperlipidemia. It is associated with increased blood viscosity and retinal ischemia that may lead to significant visual morbidity.1,2 The severity of the disease is graded anatomically2 according to the area of the affected vessels. Grade III is the most severe and rarest form that affects the periphery toward the optic disc and is characterized by salmon-colored retina with diffuse milky vessel. Despite the different treatment modalities in infantile lipemia retinalis,3–6 there is limited literature regarding the therapeutic efficacy in very severe cases. Herein, we obtained written informed consent from the patient’s parent to illustrate the images and report an effective treatment for the highest ever reported triglyceride (TG) of grade III infantile lipemia retinalis.
Case report
A 6-week-old Hispanic female infant, who was delivered after a full-term uneventful pregnancy, presented with emesis and blood-streaked stools for 3 days. Blood testing disclosed milky, lipemic serum, and abnormal lipid profiles (Figure 1). Laboratory studies reported markedly elevated TG of 316 mmol/L (normal level <1.7 mmol/L) via the dilution technique, as the machine was unable to read the high concentration of lipids in the blood. Her family history did not reveal any lipid disorders. Her sibling and parents were in good health with normal lipid profiles. Other laboratory data, including blood cell counts, electrolytes, liver function, thyroid function, glucose, and lipase tests were normal. In addition, liver and spleen size were also normal.
 | Figure 1 Lipemic serum after centrifugation. |
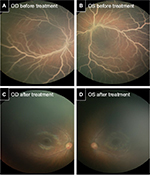
Despite the observed severe hypertriglyceridemia (sHTG), 185 times of normal level, no xanthelasma was observed. The cycloplegic retinoscopy value measured +1.00 in both eyes. Slit-lamp examination revealed the absence of cornea opacity, arcus senilis, crystalline, cataract, and anterior segment xanthoma of either eye. The optical media was clear. Fundus examination of both eyes disclosed a whitish optic disc, diffuse creamy retinal vessels in peripheral and posterior poles, and salmon-pink retina (Figures 2A and B). These retinal findings with involvement of all vessels were classified clinically as severe (grade III) lipemia retinalis. No lipid extravasation, optic disk swelling, vascular sheathing, or exudative hemorrhage was found. Chromosome microarray showed a large segment of homozygosity of ~12 MB in chromosome 8p23.1p21.3, an area where the LPL gene is found. Targeted lipid gene sequencing panel demonstrated one homozygous coding mutation in the LPL gene (c.644G>A, p.G215E) which confirmed type I hyperlipoproteinemia.
On admission, the patient was treated conservatively with breast milk and adequate intravenous hydration combined with Enfaport (Mead Johnson Nutrition, Jersey City, NJ, USA), a milk protein-based powder that is low in fat and high (84%) in medium-chain triglycerides (MCT). On day 8, the TG level decreased to 4.5 mmol/L and the patient was discharged with a total cholesterol level of 6.5 mmol/L, low-density liproprotein 4.3 mmol/L, and high-density liproprotein 0.26 mmol/L. A week later, the patient was readmitted due to a rebound TG level of 56.5 mmol/L, total cholesterol of 43.7 mmol/L, and presentation of eruptive vulvar xanthomas (Figure 3). Monogen (Nutricia, the Netherlands), a formula with similar (80%) MCT content was used to replace the Enfaport formula. In addition, breastfeeding was ceased to maximize the dietary treatment. The vulvar xanthomas self-resolved, and TG and cholesterol levels decreased dramatically to 1.75 and 9.3 mmol/L, respectively, after 1-week treatment with high MCT with no breast milk. Intriguingly, the lipemia retinalis had resolved completely in both eyes (Figures 2C and D) 3 months later. Indeed, the patient remained symptom-free and maintained an average TG level of 4.6 mmol/L with a normal fundus for a follow-up period of 6 months. She eventually established a long-term low-fat semi-solid diet with the high MCT formula.
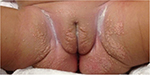  | Figure 3 Eruptive vulvar xanthomas. |
Discussion
Infantile lipemia retinalis is a rare disease, as most reported cases were found in adults in association with hyperlipoproteinemia.7,8 Currently, there is no clear consensus for a standard treatment of infants presenting with sHTG. In this report, we were able to reduce the serum TG using high MCT without breast milk in a rare case of extreme sHTG. As previously reported, high MCT formula with9 or without5,6 breastfeeding was used only to control infants with TG level <170 mmol/L. In this case, the extremely high serum TG was maintained within asymptomatic ranges after the discontinuation of breastfeeding. We thus speculated that the high level of long-chain fatty acids in breast milk may contribute to the elevation of serum TG in patients with extreme sHTG.10 Future studies are needed to support our new findings and confirm that using high MCT formula without breastfeeding is an effective treatment in infants with TG levels >300 mmol/L.
Various studies have demonstrated that lipemia retinalis is associated with high TG levels in hyperlipoproteinemias types 1, 3, 4, and 5 with different underlying causes.11 Specifically, the genetic mutation in the primary LPL gene produced the type 1 hyperlipoproteinemia seen in this case. However, genotype–phenotype correlations have not been established, as there is no strong association between the nature or location of the mutation, age at diagnosis, lipid levels, and severity of symptoms.12 While some studies have suggested that the genetic basis of childhood HTG is not confined to a particular type of hyperlipoproteinemia,13 other studies12 have suggested that TG levels were higher in compound heterozygotes compared to homozygotes, which was not in our case. Therefore, we speculated that there may be other factors, not only the LPL polymorphism for determining the TG levels.
Markedly elevated TG in lipemia retinalis might predispose patients to develop potential ocular complications caused by the inner retinal ischemia.14 Although the literature has shown that fundus lesions of lipemia retinalis might be resolved after appropriate treatment,4–6,9 it remains unknown whether future retinal function and vision development may be impaired in sHTG infants. Despite the chronic lipid-lowering therapy used to reverse the clinical manifestation of sHTG, maintenance of TG levels with dietary fat restriction or combinational treatment when our patient grows through her childhood will be challenging. Further studies are needed to evaluate the long-term effect, not only on the restoration of normal retina vessel morphology but also on the development of vision.
Acknowledgments
This study was not supported by any grant. The genetic work-up was performed by the Department of Genetic at Nicklaus Children’s Hospital. This study was approved by the Institutional Review Board of Nicklaus Children’s Hospital.
Disclosure
The authors report no conflicts of interest in this work.
References
Heyl AG. Intra-ocular lipaemia. Trans Am Ophthalmol Soc. 1880;3:54–66. | ||
Vinger PF, Sachs BA. Ocular manifestations of hyperlipoproteinemia. Am J Ophthalmol. 1970;70(4):563–573. | ||
Chaurasiya OS, Kumar L, Sethi RS. An infant with milky blood: an unusual but treatable case of familial hyperlipidemia. Indian J Clin Biochem. 2013;28(2):206–209. | ||
Cermakova I, Pettenazzo A, Perrone S, Burlina A, Zannin ME. Grade III lipaemia retinalis in a newborn. Acta Ophthalmol. 2010;88(4):e141–e142. | ||
Ikesugi K, Doi M, Nishi A, Uji Y. Lipemia retinalis of prematurity. Arch Ophthalmol. 1996;114(10):1283–1284. | ||
Hayasaka S, Fukuyo T, Kitaoka M, Suzuki H, Omura K, Kondo Y, Nakagawa M. Lipaemia retinalis in a 29-day-old infant with type 1 hyperlipoproteinaemia. Br J Ophthalmol. 1985;69(4):280–282. | ||
Sassa Y, Matsui K, Yoshikawa N, Kajiwara I, Hata Y. Lipemia retinalis: low-density lipoprotein apheresis improved the appearance of retinal vessels in a patient with type 5 hyperlipoproteinemia. Retina. 2005;25(6):803–804. | ||
Kurz GH, Shakib M, Sohmer KK, Friedman AH. The retina in type 5 hyperlipoproteinemia. Am J Ophthalmol. 1976;82(1):32–43. | ||
Rotchford AP, Newman DK, Moore AT, Flanagan DW, Miles R. Lipaemia retinalis in a premature infant with type I hyperlipoproteinaemia. Eye (Lond). 1997;11(Pt 6):940–941. | ||
Ballard O, Morrow AL. Human milk composition: nutrients and bioactive factors. Pediatr Clin North Am. 2013;60(1):49–74. | ||
Thomas PK, Smith EB. Ocular manifestations in idiopathic hyperlipaemia and xanthomatosis. Br J Ophthalmol. 1958;42(8):501–506. | ||
Mailly F, Palmen J, Muller DP, et al. Familial lipoprotein lipase (LPL) deficiency: a catalogue of LPL gene mutations identified in 20 patients from the UK, Sweden, and Italy. Hum Mutat. 1997;10(6):465–473. | ||
Kwiterovich PO Jr, Farah JR, Brown WV, Bachorik PS, Baylin SB, Neill CA. The clinical, biochemical, and familial presentation of type V hyperlipoproteinemia in childhood. Pediatrics. 1977;59(4):513–525. | ||
Nagra PK, Ho AC, Dugan JD Jr. Lipemia retinalis associated with branch retinal vein occlusion. Am J Ophthalmol. 2003;135(4):539–542. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.