Back to Journals » Nature and Science of Sleep » Volume 14
Examining the Relationship Between Sleep Quality, Social Functioning, and Behavior Problems in Children with Autism Spectrum Disorder: A Systematic Review
Authors Whelan S , Mannion A, Madden A, Berger F , Costello R, Ghadiri S , Leader G
Received 16 November 2021
Accepted for publication 6 April 2022
Published 14 April 2022 Volume 2022:14 Pages 675—695
DOI https://doi.org/10.2147/NSS.S239622
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Ahmed BaHammam
Sally Whelan, Arlene Mannion, Azeem Madden, Fine Berger, Rachel Costello, Saeid Ghadiri, Geraldine Leader
Irish Center for Autism and Neurodevelopmental Research (ICAN), School of Psychology, National University of Ireland, Galway, Ireland
Correspondence: Sally Whelan, Irish Center for Autism and Neurodevelopmental Research (ICAN), School of Psychology, National University of Ireland, Galway, Ireland, Tel +35 391493434, Email [email protected]
Abstract: Over forty percent of autistic children experience poor sleep quality, and social interaction difficulties are a core characteristic of autism. However, the relationship between sleep quality and social functioning and behavior remains poorly understood. This systematic review examined the evidence concerning the impact of sleep quality on the social functioning and behavior problems in autistic children and adolescents. It also identified key related factors and evaluated how this issue has been researched to date. Seven key journals were hand-searched and five databases were systematically searched, using keywords. Titles and abstracts of 4123 items were screened against eligibility criteria by two researchers. Relevant studies were retained if they were peer-reviewed empirical papers, published in English between 2000 and 2021. Then, the full text of 97 papers was screened and if they met the eligibility criteria, their reference lists were hand-searched. Forty-six studies were included in the final review. Data were systematically extracted and two authors critically appraised the strengths and weaknesses of studies using Critical Appraisal Skills Programme tools. Key themes were identified, because a meta-analysis was not possible due to the studies’ heterogeneity. The review identified that sleep quality and social functioning are associated with one another and there is a small amount of evidence that a bi-directional causal relationship may exist. Evidence suggests that several nights of suboptimal sleep duration and a lack of deep continuous sleep negatively impact externalizing and internalizing behavior. Sleep quality is also reduced by anxiety and sensory sensitivity. However, longitudinal studies with larger samples are needed to establish causality. Future research needs to examine confounding factors and to develop consensus regarding best-practice processes for the objective measurement of sleep with autistic children. Additional research also needs to further examine the consequences of poor sleep quality on internalizing behavior, and the impact of socio-cultural practices.
Keywords: autism spectrum disorder, night waking, insomnia, behavior, anxiety, bedtime resistance
Introduction
Autism is a neurodevelopmental disorder with a prevalence rate of 1 in every 68 children.1,2 Autism is characterized by the core features of repetitive behaviors, restricted interests, affective and social interaction difficulties.3 Social functioning has been defined as “the ability to navigate through the social world and form and maintain relationships that involve one’s home, school, or workplace” (p2).4 It can be challenging for autistic children to satisfy roles within an environment, relationships with family and friends, and social activities.4 Autistic children differ in several ways from neuro-typical (NT) children regarding their social functioning.5 They may be less likely to look towards social stimuli6 or to initiate and share attention jointly with other people,7 and individuals may exhibit less ability to recognize emotions, interpret vocal and visual cues,8 and imitate behavior based on social cues.9 Furthermore, autistic children use different strategies to process facial information, emphasizing features and the mouth rather than focusing on the eyes.10,11 Social functioning is also impacted by motivation, perception, anxiety, social skills, and emotional challenges.4,12
Sleep problems include dyssomnias that disturb the quality, amount, timing of sleep, including difficulty falling or staying asleep, excessive daytime sleepiness.13,14 They also include parasomnias that involve physiologic events or behaviors that interrupt sleep, including arousal and partial arousal disorders or during the transition between sleep stages.14 Sleep problems in children have been operationalized in research in terms of bedtime resistance, sleep onset delay, sleep duration, sleep anxiety, night-time waking, parasomnias, sleep-disordered breathing, and daytime sleepiness.15
Between 25% and 40% of NT children experience sleep disturbances and these have been associated with decreased social functioning in middle childhood and preadolescents16 and in children aged 18–42 months.17 A systematic review of studies that involved children aged 5–12 years found that shorter sleep duration was associated with internalizing and externalizing behavioral problems and having less complex cognitive skills and executive functioning ability.18 Positive relationships between sleep quality/quantity, and better cognitive and/or behavioral outcomes have also been found for children aged 2–5 years.19 Sleep disturbances occur in 40–83% of autistic children.20–23 A recent meta-analysis examined studies that had measured sleep objectively using polysomnography (PSG) or actigraphy. This study found small but significant differences between autistic children and NT children regarding their sleep parameters. These differences included shorter total sleep time (TST), longer sleep onset latency (SOL), and reduced sleep efficiency (SE). However, there was no significant difference between the total sleep time (TST) in autistic children without intellectual disability (ID) and their NT peers.24 Díaz-Roman et al25 also conducted a meta-analysis of studies that included both subjective and objective sleep measures. They found that when sleep was subjectively measured, autistic children had significantly higher sleep anxiety, sleep onset delay, sleep onset latency, bedtime resistance, night awakenings, sleep-disordered breathing, parasomnias, and daytime sleepiness. The results determined using objective measures revealed autistic children had lower TST, a lower percentage of rapid eye movement (REM) sleep, longer SOL, lower SE, than the NT group.
In autistic children, poor sleep quality is negatively associated with physical and psychosocial wellbeing and quality of life,26 and it is associated with behavior problems27 and higher levels of hyperactivity, social withdrawal, and irritability.28 Higher rates of externalizing and internalizing behavior are also associated with lower levels of social functioning in autistic children and adolescents29 and NT children.30 However, the impact of sleep quality on social functioning and behaviour problems, and the factors that moderate these relationships remain poorly understood. Further examination of this topic is warranted because sleep quality is a potentially modifiable predictor of social functioning and problematic behavior. Detailed knowledge about these relationships could inform caregivers, therapists, and clinicians who seek to improve the well-being of autistic children.
This systematic review aimed to increase understanding of the relationships between sleep quality and the social functioning and behavior problems of autistic children and adolescents. Its objectives were to examine the current evidence and identify the factors that underlie and mediate this relationship, and evaluate how this relationship has been examined to date.
Methods
Search Strategy
The key search terms listed in Table 1 were identified using the patient, intervention, comparison, outcome search strategy tool.31 Keywords and MeSH terms were used and adapted according to the requirements of the following databases and as they were searched: Medline, PubMed, Scopus, Psych Info, and the Web of Science. In addition, the following journals were
 |
Table 1 Key Word Search Terms |
hand-searched for articles that met the eligibility criteria which is summarized in Table 2: Sleep, Sleep Medicine, Behavioral Sleep Medicine, Nature and Science of Sleep Journal; Journal of Autism and Developmental Disorders, Autism, Autism and Research. Initial searches were limited to papers published between the years 2000–2021, and older articles were identified through hand searching the reference lists of articles that met the inclusion criteria. Papers were included if they were peer-reviewed empirical studies, published in English. They also needed to involve children diagnosed with any type of autism and to focus on sleep quality and social functioning. These searches identified 5270 items that were imported into an Endnote database. After duplicates were removed there were 4119 items.
 |
Table 2 Eligibility Criteria |
Study Selection
Firstly, two researchers worked independently (FB, RC), screening the title and abstract of each item against the eligibility criteria. This resulted in the retention of 97 papers, with 87% inter-rater reliability between researchers. Then, researchers (AM, SW) reviewed the papers on which FB and RC disagreed, and decided upon their eligibility. Following this, FB and RC independently screened the full-text of 97 papers, which resulted in an inter-rater reliability of 88%. SW and AM reviewed and discussed any papers on which there was disagreement and determined their eligibility. Finally, the reference lists of relevant papers were hand-searched for additional papers. The PRISMA flow diagram below (Figure 1) summarizes the results of the paper selection processes.
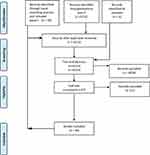 |
Figure 1 PRISMA Flow Diagram. Notes: Adapted from: Liberati A, Altman D, Tetzlaff J et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. Journal of Clinical Epidemiology. 2009;62(10)e1-e34. Creative Commons86. |
Data Extraction and Quality Assessment
Two researchers (AM, SW) collaborated to extract details from the included studies, using a bespoke template table designed according to the review aims. Due to the heterogeneity of the studies, it was not possible to conduct a meta-analysis. Instead, the study contents were repeatedly examined and themes were identified.
The quality of the studies included in the review was assessed by researchers (RC, SG), who worked independently using the Critical Appraisal Skills Programme (CASP-uk.net) checklists, as recommended by World Health Organisation guidelines.32 The CASP checklists are designed to assess the quality of research against criteria relevant to the design of the study under assessment. The CASP checklist for randomized controlled trials (RCTs) and the checklist for quantitative cohort studies include 12 points and the checklist for studies using qualitative methodology includes 10 points.
Results
Table 3 summarizes the content of the 46 studies that were included in the review and their key findings in relation to how sleep quality relates to social functioning and behavior in autistic children. The critical appraisal of the studies revealed that the methodological quality of these peer-reviewed studies ranged between moderate and high ratings on the CASP checklists. Due to the design of the studies, this review applied the RCT, Quantitative Cohort, and Qualitative checklists. The one qualitative study included in the review was rated 9 on the CASP Qualitative checklist; the four RCT studies were rated as between 10 and 12 using the CASP RCT checklist; the remaining 41 studies were rated as between 7 and 12 on the CASP Quantitative Cohort studies checklist. The inter-rater reliability between researchers’ ratings during the quality appraisal was initially high at 86% and after discussions between researchers that also involved SW, 100% agreement was achieved.
 |
 |
 |
 |
 |
 |
 |
|
Table 3 Study Details (Abbreviations Key Below) |
Relationships Between Sleep Quality, Social Functioning and Behavior Problems
Externalizing Behavior Problems
There is substantial evidence that externalizing behavior problems can be associated with sleep problems67 and multiple hierarchical regressions have been used to show that they can be statistically predicted by sleep quality.59 Fadini et al,39 using parental reports, found sleep disorders, particularly arousal disorder and excessive daytime sleepiness, correlated with behavioral problems. This finding concurs with Aathira et al33 which measured sleep prospectively with a small sample, using one night of PSG. This study found that poor sleepers had a mean Child Behavior Checklist Score of 63.8 (60.8 to 66.8) whereas good sleepers scored a mean of 57.8 (53.0 to 62.5)(P ¼ 0.004). Malhi et al52 also found a significant relationship between sleep dysfunction and daytime behavior difficulties (r = 0.53, P = 0.01), and autistic children with sleep problems (t = 2.42, P = 0.018) had significantly more behavioral difficulties (M = 15.67, SD = 6.10) than those without sleep problems (M = 9.50, SD = 3.01). Several studies have found that sleep problems are significantly associated with irritability, hyperactivity, inattentiveness,20,41,69,71 and aggression, which parents attributed to tiredness.48 Sleep disturbance has also been identified as a predictor of attention deficit, hyperactivity, and oppositional defiant behavior.47 Allik et al27 found that 31.2% of their sample of autistic children, without ID, had insomnia. These children had more teacher-reported hyperactivity symptoms than those without insomnia. Goldman et al42 also found that the poor sleeper group within their sample showed more inattention and hyperactivity, and they had higher scores on compulsive and ritualistic scales in comparison to the good sleeper group. Goldman et al43 also found that 50% of the poor sleeper group had problems with attention, social interaction, language, hyperactivity, sensory issues, eating behaviors, and self-stimulatory behavior, in comparison 50% of the good sleeper group who only reported problems in language, attention, and social interaction. The characteristics of good sleepers versus poor sleepers have also been examined in an RCT.28 In this RCT, the poor sleeper group had significantly higher daytime behavioral problems including, hyperactivity, stereotypical behaviors, and more irritability. However, Lambert et al4 found that SWS positively correlated with the score of the ADI-R subscale for repetitive behavior (r = 0.714, p = 0.02), a positive association was difficult to explain.
Internalizing Behavior Problems
Sleep disturbances have been associated with anxiety43 and emotional problems.27,46,47,69 Lambert et al50 used data collected through parent-reported questionnaires, daily sleep diaries, and laboratory PSG (2 nights). Their results showed that the amount of slow-wave sleep (SWS) correlated negatively with the amount of Internalizing behaviors (r = −0.41, p = 0.046), and the reciprocal social interaction score ADOS (r = −0.807, p = 0.003). Sleep latency, which was defined as the first occurrence of 10 consecutive minutes of stage 1 sleep or the first epoch of any other sleep stage, and SWS were associated with low levels of internalizing behavior and the social functioning of children as determined through ADOS measurements. Poor sleep quality is also associated with lower mood.27 Kirkpatrick et al48 examined the impact of insomnia, using focus groups (n = 2) that involved family members (n = 15). Their findings revealed that 26% percent of parents said that tiredness negatively impacted the mood of their children. Some evidence suggests that lower sleep quality may lead to poor prosocial behavior and social withdrawal.28,69,71 One study examined the association between sleep problems and the quality of peer relationships.62 This involved a small sample of verbally fluent autistic adolescents (n = 19) that measured seven nights’ sleep using actigraphy. The questionnaires, but not the actigraph data indicated significant associations between sleep problems and discordant peer relationships (0.59 p < 0.01). Participants with more sleep problems reported discord with peers but the closeness of peer relationships was not significantly associated with sleep quality. They concluded that autistic adolescents can be vulnerable to negativity in peer relationships when they experience sleep problems and that they may have difficulty regulating social interaction.
In one study, social withdrawal, and stereotypic behaviors accounted for 17% and 36% of the variance, respectively, in sleep disturbances.71 It has also been suggested that the need for social interaction can decrease with increased sleep problems.67
Aspects of Sleep Quality
Particular aspects of sleep quality including sleep onset delay/sleep latency, short sleep duration, and shorter continuous sleep have been associated with social functioning.22,68,71 For example, Tudor et al68 found a positive correlation between stereotyped behavior with sleep onset delay (0.31 p < 0.01), and social interaction (0.40 p < 0.01). Sleep duration also correlated with stereotyped behavior (0.32 p < 0.01) and social Interaction (0.33 p < 0.01). Also, when controlling for the variance contributed by other sleep problems, sleep onset delay (pr = 0.27, p < 0.01) and night wakings (pr = 0.22, p < 0.05) were significantly strong predictors of stereotypical behaviors.68
There is some evidence that shorter sleep latency relates to poorer social functioning. Naito et al58 examined the relationship between body movements at night and social ability, in a small sample of relatively young autistic children (n = 17) arguing that movement would correspond to periods of light rather than deep sleep. Dissimilar to previous studies that used PSG, they measured the body movement of participants sleeping in their own homes for 3 nights using an accelerometer attached to the waist. They found that a higher rate of body movement at 0.5 to 1 hour after onset body stillness was associated with lower social ability. They suggested that higher social symptoms severity was associated with shorter latency in the first nocturnal peak of movement and in the latter half of the night. This study did not evaluate motor function or measure the amount of daytime activity that may impact movements at night and they did not measure body movements for 5 nights, which is optimal to estimate body movement patterns.72
Other evidence indicates that the duration of uninterrupted continuous sleep is important. Delahaye (2014) found the largest correlations were between sleep duration and quality of life total (−0.36, p < 0.001), and psychosocial summary (−0.36, p < 0.001) scores, when they investigated the relationship between sleep problems and quality of life, child age, gender, autism diagnosis, internalizing and externalizing behavior, and autism severity. Furthermore, extremely short sleep duration increased core autism symptom severity22 and a short duration of uninterrupted sleep may be more impactful than sleep latency.65,71 These results concur with those of Yavuz-Kodat et al,71 who examined the relationship between sleep, circadian rest-activity rhythm to problem behaviors. This study found children with shorter continuous sleep had high irritability in comparison with those with lower irritability (−60 min, p = 0.04), and high stereotypic behaviors compared to children with fewer stereotypies (−75 min, p = 0.006). Shorter continuous sleep was 60 minutes shorter in the higher irritability group and 75 minutes shorter in the higher stereotypy group. Yavuz-Kodat et al determined that children with shorter continuous sleep completed approximately one less complete sleep cycle of REM and non-REM sleep. Further evidence of the importance of uninterrupted sleep can be found in a study that evaluated the effects of slow-release melatonin treatment on sleep and behavior.65 Schroder et al65 found a modest correlation between the number of difficulties experienced by participants and increase in total sleep time from baseline (Spearman’s rank correlation R − 0.229 p = 0.024). Schroder et al65 attributed this to the increased duration of uninterrupted sleep (Spearman’s rank correlation R − 0.21 p = 0.047) but not with the shortening of sleep latency (Spearman’s rank correlation R 0.09 p = 0.379). Taking the slow-release melatonin drug resulted in the Ped- PRM-treated participants sleeping for an average of 57.5 minutes longer. The placebo group had an increase of 9.14 minutes longer. The sleep latency of the Ped- PRM-treated participants decreased by 39.6 minutes on average compared to 12.5 minutes with placebo.
Evidence Suggesting Causal Relationships
Studies that have examined the relationship between sleep quality and social functioning longitudinally or with social functioning and behavior problems as outcomes during the evaluation of sleep interventions provide evidence that suggests improving sleep quality may improve these outcomes.56 For example, Malow et al53 conducted an RCT that used parental reporting and no placebo to assess the response, tolerability, safety of a slow-release Melatonin. After 14 weeks, sleep latency significantly decreased and there were significant improvements in sleep duration, social withdrawal, affective, and ADHD subscales. Another double-blind RCT evaluated the efficacy of prolonged-release melatonin mini-tablets (PedPRM; 2–5 mg) in improving sleep onset, duration, and child behavior.65 Participants had either autism or Smith-Magenis syndrome (n = 125). After 13-weeks, melatonin effectively improved sleep quality and behavior in comparison to a placebo. There were statistically significant improvements in externalizing behavior, which decreased on average − 0.70 units in the Ped- PRM-treated (N = 54), compared to worsening with an increase of 0.13 units in the placebo-treated group (N = 49). In addition, 53.7% of patients in the PedPRM group improved by 1 or more units on externalizing behavior score compared to 27.7% in the placebo group (Odds ratio 3.0; p = 0.008). However, there was no significant difference in the mean scores of attributes that related to Internalizing behaviors (peer relationships and emotional behavior). Overall, 47% of subjects had a reduction of 1 or more in total strengths and difficulties score in the PedPRM group, compared to 27.1% in the placebo group (p = 0.035). This suggests that hyperactivity/inattention may be relatively more responsive to the sleep improvements that result from treatment with melatonin than Internalizing behaviors. However, it is noteworthy that this study was powered to detect differences in effects on sleep but not behavior. Papadopoulos et al60 conducted an RCT pilot study that evaluated the efficacy of a brief behavioral sleep intervention on sleep and child functioning. Participants who were children with an ADHD-Autism co-diagnosis were randomized to the sleep intervention group (n = 28), or the group receiving usual care (n = 33). This study assessed the mean difference in change scores between baseline to 3 and 6 months between the groups. They found that the intervention group improved emotional functioning to a greater degree according to parent reports at 3 months: (mean difference = −1.0, 95% CI = [−2.0, −0.1]; ES = −0.6, p = 0.04), and teacher reports at 3 months (mean difference =−1.2, 95% CI = [−2.3, −0.1]; ES = −0.5, p = 0.04). However, by 6 months, there was no significant difference in emotional functioning, but teacher reports suggested there was an improvement in conduct problems. Small improvements in peer relationships at 6 months were detected by both parents and teachers (mean difference = −0.6, 95% CI = [−1.6, 0.4]; ES = −0.3, p = 0.22) and teacher report (mean difference = −1.0, 95% CI = [−2.3, 0.3]; ES = −0.4, p = 0.11). Papadopoulos et al60 concluded that the intervention resulted in large improvements in sleep and moderate improvements in behavioral function at 3 and 6 months.
Related Factors
The Severity of Autism Core Symptoms
Some studies have examined social functioning focusing on the severity of autism symptoms. These studies provide mixed evidence concerning the relationship between sleep quality and the autism severity. The majority of studies have found that a positive association exists between them.20,22,37,64,67,68 For example, Sare & Smith (2020),63 in a study involving secondary data analysis of a large parent-reported data set (n = 19,104), found a strong association between sleep problems and the severity of autism-related behaviors. However, in two studies, sleep disturbance scores did not correlate with autism severity33,54 and in another, a small amount of variance in daytime symptoms was explained by sleep problems.64
The evidence suggests that hyper-arousal and increased sensitivity to stimuli in the sleep environment are associated with sleep disturbance.54 Manelis-Baram et al54 found that the sensory sensitivity score exhibited the strongest correlation with sleep duration, and sleep disturbances correlated with sensory problems correlated (r = 0.57, p < 0.001), sensation avoiding (r = 0.49, p < 0.001), low registration (r = 0.37, p < 0.001), and sensation seeking (r = 0.24, p = 0.014) scores. Further evidence of the relationship between sensitivity and sleep problems was obtained through a study that found significant associations between sleep problems and increased seeking, touch, movement processing patterns, conduct, and attentional responses to sensory processing.20 Mazurek et al55 also found sleep problems correlated with sensory over-responsivity, and the study results suggested that sensory over-responsivity was a longitudinal predictor of sleep problems and inattention/hyperactivity in very young, but not older children.
Cognitive Ability
Several studies found that the relationship between sleep quality and social functioning exists regardless of cognitive ability and the presence/absence of ID.28,41,47,52,54,63 In contrast, a small number of studies found that the quantity and quality of sleep are associated with cognitive performance50 and that the presence of ID was an important predictor in the relationship between sleep quality and emotional and behavioral disturbance.47 Krakowiak et al49 also found, in a large population-based study, that lower cognitive and adaptive function is associated with increased delayed sleep onset, night awakening, and shorter sleep duration. In addition, two other studies found that individuals with an unstable sleep phenotype had a relatively lower IQ and adaptive functioning level than those with a stable phenotype.37,38
Anxiety
There is some evidence that anxiety contributes to the association between sleep and social functioning.21,46,47,57,68 However, Mazurek et al’s55 results did not indicate a significant predictive relationship between anxiety and sleep problems after accounting for covariates, even though anxiety correlated with sleep in both older and younger children. However, a causal relationship between anxiety and sleep quality was described by eighty-six percent of parents (n = 13) in one study.48 Parents said that their children experienced sleep-related anxiety that resulted in delayed bedtimes, unwanted co-sleeping, night awakening disruptive night-time behaviors.48 They reported that children were afraid to sleep alone, anxious about darkness, worried about forthcoming events or school and that this anxiety contributed to reduced and poor quality sleep. Therefore, anxiety may impact sleep quality by delaying sleep onset and pre-bedtimes routines.44 Indeed, the consistency of pre-bedtime routines can be negatively associated with externalizing behaviors (b = 0.55, t(110) = 6.76, p < 0.001) and sleep quality. The consistency of routines is related to problematic pre-bedtime behavior and bedtime resistance40,48 and children with higher irritability have more difficulties regarding bedtime routine.41,44,71 Goldman et al42 also found that this was a major concern by parents of children who were categorized as poor sleepers. Bedtime resistance was also identified as a risk factor for repetitive behavior in preschool autistic children and it explained a large proportion of variance in repetitive behavior scores (adjusted R2 = 0.206).46 In addition, linear regression showed that bedtime resistance and sleep onset delay explained a large and considerable proportion of the variance in the total repetitive behavior score (20.6% and 17.3%), respectively.46
Age
As suggested above, the relationship between sleep quality and social functioning may differ according to age and behavior problems and a later sleep midpoint of older children was associated with challenging behavior.34 Veatch et al20 evaluated the effects of age on associations of sleep duration and autism traits in children who were 4–5 years old (17.7%); primary school age (63.6%), and secondary school age (18.5%). This study found that age is significantly related to short sleep duration, anxiety, depression, Internalizing problems, and the severity scores for affective problems. Goldman et al43 also found an association between parasomnias and behavior across the age span with an inverse relationship between age and behavior problems. However, in contrast, Johnson et al28 found no age difference between good sleepers and poor sleepers in terms of disruptive behaviors, and parasomnias have been found to persist across an age range of 3–18 years.43 Furthermore, even adjusted for age, poor sleepers were found to be at the highest risk of self-injurious behaviors.34
Temporal Aspects
A few studies have examined the relationship between sleep and functioning over time. May et al21 investigated the bi-directional relationship between sleep disturbance and behavioral, and emotional problems, longitudinally, at baseline (T1) and 1 year later (T2). At both time points, sleep problems correlated with social difficulties, aggression, and hyperactivity. These results concurred with Mazurek et al,55 who examined a large well-characterized sample of autistic children at baseline (T1) and M = 3.8 years later (T2). Mazurek et al found that sleep problems were significantly associated with most co-occurring symptoms at T1 and T2.
For individuals over time, the evidence varies as to whether the relationship between the trajectories of sleep problems and social functioning can persist over time. They can be persistent, but children with greater sleep problems at the second time point (aged 4) had higher trajectories for repetitive behaviors.47 In addition, several studies identified a high degree of individual variability.34,37,40
It is likely that the chronicity of disturbed sleep may have an accumulative effect on social functioning. Abel et al34 used objective measures of sleep and behavior to explore night-to-night fluctuations in sleep patterns and challenging behavior, ie, whether challenging behavior would follow the day after a poor quality night’s sleep. This study found, using multilevel models for predictive analysis, that across eight nights, rather than a single night, sleep quality predicted the subsequent challenging behavior. This finding concurred with Cohen et al,38 who examined data from five days/nights measurement of sleep and challenging behaviors. They collected sleep data using structured observations and clinicians worked one on one with participants to observe and record behavior hourly each day for up to 21 hours. They found that the previous night’s sleep did not strongly predict next-day problem behaviors. Instead, behavior was affected after several nights of poor sleep, and the accuracy of predicting behavior increased with the number of nights measured. This finding concurs with qualitative research in which a participant parent said planned family trips to the cinema were canceled after: “a few late nights and early mornings. Well! He [the autistic child] can be awkward and it’s just not worth it” (Kirkpatrick et al, 2019, Parent 13 p. 64).48
Discussion
Summary of Key Findings
The evidence presented in this review reveals that the social functioning and problem behaviors of autistic children, with or without ID, are associated with their sleep quality. A relatively small number of studies have evaluated social function and behavior problems as outcomes of sleep interventions, but these reveal evidence that suggests causal relationships and that social functioning may improve as sleep improves but that the improvements may be temporary.53,60,65
Social functioning is associated with the tendency for autistic children to have shorter latency in the first nocturnal peak, less deep, and more fragmented sleep than those with NT.58 Therefore, the relationship between sleep quality and social functioning is probably unique to autistic individuals.68
More specifically, chronic and accumulative poor sleep quality can increase externalizing behavior problems including hyperactivity and inattentiveness. However, a few of the study samples examined contained autistic participants who were also diagnosed with ADHD, and the majority of studies did not out rule the presence of ADHD. Therefore, definitive conclusions cannot be reliably drawn due to the high comorbidity rate between autism and ADHD.73 The evidence also reveals that poor sleep is negatively associated with problematic Internalizing behavior, increased negative mood, and social withdrawal. However, further research needs to investigate the impact of sleep on Internalizing behavior as discussed below.
Overall, the evidence suggests that anxiety negatively impacts sleep onset, duration, and it increases sleep disturbance. Therefore, the relationship between sleep quality and social functioning is bi-directional. This is an important finding as anxiety disorders are common in autistic individuals with prevalence rates from 42% to 79%.74 Related to anxiety, hyper-arousal and sensitivity are associated with sleep disturbance.20,54,55 Hyper-arousal has also been associated with insomnia in autistic adults.75 This finding also supports the presence of a bi-directional relationship between sleep quality and social functioning in autistic children and it supports the hypothesis that particularly in young children, sleep disturbance and sensory sensitivities are likely to be co-dependent, and generated by interacting physiological mechanisms.54
There is mixed evidence regarding the relationship between sleep quality and social functioning and its association with autism severity and age. On balance, the evidence reveals an association with the severity of autism symptoms, and that the relationship changes according to age. Previous related research has identified that autistic individuals have age-related variations in core symptom severity76 and sleep behavior.73
The majority of studies reviewed used a cross-sectional research design and a small proportion were longitudinal.21,34,40,54–56,60 Therefore, there is little evidence with which to determine causality and its direction with any degree of certainty. It is also notable that only one study employed qualitative research methodology.48 Most studies had relatively small sample sizes. However, there were a few large population-based studies.22,43,49
Examining the relationship between sleep quality and social functioning is complex as it is impacted by comorbidities that are common and experienced by up to 46% of autistic individuals.38,77 The studies reviewed varied hugely regarding which confounding factors researchers controlled for or excluded through participant selection. As discussed above, studies did not all exclude children with co-diagnosis of ADHD, and only some controlled for medical comorbidities,46,53 and pharmacological agents.54,57 There was a similar variety regarding other confounding factors. For example, few studies assessed the impact of daytime sleep or daytime activities and none recorded whether participants slept in the same room as other children and if the latter had sleep problems.57 Kelmanson et al47 were an exception in recruiting only singletons without severe comorbidities.
Most of the studies relied on parental reporting for measuring sleep and/or behavior although three studies assessed daytime social functioning with teacher/caregivers reports that were blinded to the sleep quality.37,38,60 The use of parental reporting means most assessments were unblinded and were therefore, subject to reporter bias. Findings using parent-reported sleep measures can concur with those from objective measures.20,23,27,42 But, they do not always do so,35 and sleep duration is the most reliable sleep trait reported by parents.22 Furthermore, the use of parental measures for behavior, and not including reports from child/adolescent participants, is likely to underestimate Internalizing behavior, emotion, anxiety, and feelings of sleepiness.65,67 Notably, only two studies in this review collected data directly from autistic adolescents.62,67 Underestimation of Internalizing behavior is concerning particularly as self-injurious behavior is a high risk for poor sleepers of all ages43 and worry/rumination can predict the later development of Internalizing and externalizing problems in autistic children.78 But, it may be more problematic for adolescents because the reported somatic complaints of individuals increase over time with age.78 There are methodological challenges in involving people with autism who have an ID but these can be minimized to facilitate their participation in future research.79
Only one study measured both sleep and social functioning objectively,34 and two related studies used structured sleep and behavior observations.37,38 The use of objective methods potentially reduces bias and several studies measured sleep objectively using actigraphy,20,36–38,40,53,58,62,80 and PSG.33,42,50 Moore et al81 reviewed the literature and reported that actigraphy can underestimate SOL in autistic children, but that there is good compliance with this method and minimal data loss. The validity of actigraphy against PSG has been assessed in autistic children (n = 26) during one night in a hospital setting.82 This study found good agreement between measures for SOL, TST, and moderate agreement regarding WASO and SE. However, in this review, there was a large variation regarding whether sleep diaries were used to provide contextual data, how children and parents were prepared for PSG and actigraphy, and regarding the duration of the measurement. These are important considerations for the validity of measurements because usual sleep behavior may be distorted if it is measured in ways that are challenging for autistic children who have sensory issues and a preference for sameness.3 PSG may be challenging because it involves sensors being attached to the face and scalp, and it requires children to sleep in an unfamiliar environment laboratory environment. Malow et al83 found that autistic children who were poor sleepers had a significantly poorer SOL and SE in comparison to autistic good sleepers and NT control groups measured on one night by PSG. However, after subsequent nights of PSG measurement, there was less difference between the groups. Malow et al83 recommended that the validity of PSG can increase if autistic children are acclimatized to PSG for two nights and then have their sleep measured during a third night in the laboratory.
Future Research
Future research needs to explore further the impact of confounding variables on the relationship between sleep quality and social functioning. Doing so could be helpful through explaining the variability between individual children.34,37,40 For example, the onset of puberty may be a potential confounder22 and this has not been investigated to date. Other factors, including gastrointestinal symptoms,70 dietary and eating habits,48 and breathing problems,59 have been afforded minimal attention. Future work should also clarify how gender impacts sleep quality and social functioning. Except for May et al,21 whose sample had a more equal gender ratio, the reviewed studies contained samples of predominantly males, as is typical within the autism population.2
Sleep quality and social functioning are both impacted by a mixture of internal and external factors, including interpersonal interactions.38 They are also associated with wider societal policies and cultural practices.47 To date, research has focused on issues that are personal to and within individual autistic children. Future research should explore the impact of wider socio-cultural factors in general, and specifically parental stress, the parent–child relationship, and co-sleeping practices. It can be comparatively more stressful to parent an autistic child than an NT child84 and this may be aggravated through a child’s poor sleep quality.48 The use of mixed methods/qualitative research methodology may facilitate in-depth exploration of potential environmental and societal issues.
Sleep quality and social functioning have predominantly been studied using cross-sectional cohorts with data collected at one point in time. More longitudinal and prospective research are required to confirm causal relationships. These designs are also necessary to increase understanding of the trajectory over time and age. Knowledge from this research will facilitate clinicians to offer individualized interventions.
Future research using Delphi survey techniques85 would also be helpful to increase consensus regarding the management of confounding factors in future research and to facilitate the development of best practice guidelines regarding the use of actigraphy and PSG with autistic individuals.
Implications of Findings
Because autistic children may have poor quality sleep and not complain about this,50 all parents of autistic children may benefit from receiving information that aims to increase sleep quality, because this is likely to maximize their child’s social functioning potential. Also, sleep quality should be assessed for autistic children who present to services with behavioral problems and sleep interventions should be integrated into treatment plans.27,52,57,59
Limitations and Strengths
This review used systematic and robust methods and the methodological strengths and weaknesses of studies were critically appraised using a recognized tool. However, the review only included studies published in English that focused on sleep quality, social functioning, and behavior problems. Therefore, some studies could have been excluded that might have been informative, particularly those that focused on related concepts including communication, and quality of life.
Conclusions
This review examined 46 studies to elucidate understanding of the relationship between sleep quality, social functioning, and behavior problems. Evidence revealed that poor sleep quality is associated with social functioning and behavior problems. A small amount of evidence suggests the existence of a bi-directional causal relationship between sleep quality and social functioning. Delayed sleep onset, reduced sleep duration, and fragmented sleep without continuous periods of uninterrupted sleep, over several days, appear to increase problematic externalizing and Internalizing behavior. However, anxiety, hyper-arousal, and sensitivity may negatively impact sleep quality. RCTs and prospective studies are required to establish causality. Future research also needs to further examine confounding factors, increase consensus for the objective measurement of sleep with autistic individuals, explore the impact of socio-cultural practices, and increase understanding about the Internalizing behavioral consequences of poor sleep quality.
Funding
This research did not receive any grant from any funding agency.
Disclosure
All the authors declare that they have no conflicts of interest in this research.
References
1. Goldman S, Alder M, Burgess H, et al. Characterizing sleep in adolescents and adults with autism spectrum disorders. J Autism Dev Disord. 2017;47(6):1682–1695. doi:10.1007/s10803-017-3089-1
2. Christensen DL, Braun KVN, Baio J, et al. Prevalence and characteristics of autism spectrum disorder among children aged 8 years—autism and developmental disabilities monitoring network, 11 sites, United States, 2012. MMWR Surveill Summaries. 2018;65(13):1. doi:10.15585/mmwr.ss6513a1
3. American Psychiatric Association D, Association AP. Diagnostic and Statistical Manual of Mental Disorders: DSM-5. Washington, DC: American Psychiatric Association; 2013.
4. Pallathra AA, Calkins ME, Parish‐Morris J, et al. Defining behavioral components of social functioning in adults with autism spectrum disorder as targets for treatment. Autism Res. 2018;11(3):488–502. doi:10.1002/aur.1910
5. Dawson G, Bernier R. Development of social brain circuitry in autism. In: Coch D, Dawson G, Fischer KW, editors. Human Behavior, Learning, and the Developing Brain: Atypical Development. New York: Guildford Publications; 2007:28–55.
6. Dawson G, Meltzoff AN, Osterling J, Rinaldi J. Neuropsychological correlates of early symptoms of autism. Child Dev. 1998;69(5):1276–1285. doi:10.2307/1132265
7. Mundy P, Sigman M, Ungerer J, Sherman T. Defining the social deficits of autism: the contribution of non‐verbal communication measures. J Child Psychol Psychiatr. 1986;27(5):657–669. doi:10.1111/j.1469-7610.1986.tb00190.x
8. Golan O, Baron-Cohen S, Hill JJ, Golan Y. The “reading the mind in films” task: complex emotion recognition in adults with and without autism spectrum conditions. Soc Neurosci. 2006;1(2):111–123. doi:10.1080/17470910600980986
9. Nielsen M, Carpenter M. Reflecting on imitation in autism: introduction to the special issue. J Exp Child Psychol. 2008;101(3):165–169. doi:10.1016/j.jecp.2008.06.004
10. Rosset DB, Rondan C, Da Fonseca D, Santos A, Assouline B, Deruelle C. Typical emotion processing for cartoon but not for real faces in children with autistic spectrum disorders. J Autism Dev Disord. 2008;38(5):919–925. doi:10.1007/s10803-007-0465-2
11. Deruelle C, Rondan C, Gepner B, Tardif C. Spatial frequency and face processing in children with autism and Asperger syndrome. J Autism Dev Disord. 2004;34(2):199–210. doi:10.1023/B:JADD.0000022610.09668.4c
12. Bishop-Fitzpatrick L, Mazefsky CA, Eack SM, Minshew NJ. Correlates of social functioning in autism spectrum disorder: the role of social cognition. Res Autism Spectr Disord. 2017;35:25–34. doi:10.1016/j.rasd.2016.11.013
13. El Shakankiry HM. Sleep physiology and sleep disorders in childhood. Nat Sci Sleep. 2011;3:101. doi:10.2147/NSS.S22839
14. Mindell JA, Owens JA, Carskadon MA. Developmental features of sleep. Child Adolesc Psychiatr Clin N Am. 1999;8(4):695–725. doi:10.1016/S1056-4993(18)30149-4
15. Owens JA, Spirito A, McGuinn M, Nobile C. Sleep habits and sleep disturbance in elementary school-aged children. J Develop Behav Pediatr. 2000;21(1):27–36. doi:10.1097/00004703-200002000-00005
16. Foley JE, Weinraub M. Sleep, affect, and social competence from preschool to preadolescence: distinct pathways to emotional and social adjustment for boys and for girls. Front Psychol. 2017;8:711. doi:10.3389/fpsyg.2017.00711
17. Tomisaki E, Tanaka E, Watanabe T, et al. The relationship between the development of social competence and sleep in infants: a longitudinal study. Child Adolesc Psychiatry Ment Health. 2018;12(53). doi:10.1186/s13034-018-0258-8
18. Astill RG, Van der Heijden KB, Van IJzendoorn MH, Van Someren EJ. Sleep, cognition, and behavioral problems in school-age children: a century of research meta-analyzed. Psychol Bull. 2012;138(6):1109. doi:10.1037/a0028204
19. Reynaud E, Vecchierini MF, Heude B, Charles MA, Plancoulaine S. Sleep and its relation to cognition and behaviour in preschool‐aged children of the general population: a systematic review. J Sleep Res. 2018;27(3):e12636. doi:10.1111/jsr.12636
20. Tyagi V, Juneja M, Jain R. Sleep problems and their correlates in children with autism spectrum disorder: an Indian study. J Autism Dev Disord. 2019;49(3):1169–1181. doi:10.1007/s10803-018-3820-6
21. May T, Cornish K, Conduit R, Rajaratnam SM, Rinehart NJ. Sleep in high-functioning children with autism: longitudinal developmental change and associations with behavior problems. Behav Sleep Med. 2015;13(1):2–18. doi:10.1080/15402002.2013.829064
22. Veatch OJ, Sutcliffe JS, Warren ZE, Keenan BT, Potter MH, Malow BA. Shorter sleep duration is associated with social impairment and comorbidities in ASD. Autism Res. 2017;10(7):1221–1238. doi:10.1002/aur.1765
23. Vriend JL, Corkum PV, Moon EC, Smith IM. Behavioral interventions for sleep problems in children with autism spectrum disorders: current findings and future directions. J Pediatr Psychol. 2011;36(9):1017–1029. doi:10.1093/jpepsy/jsr044
24. Elrod MG, Hood BS. Sleep differences among children with autism spectrum disorders and typically developing peers: a meta-analysis. J Develop Behav Pediatr. 2015;36(3):166–177. doi:10.1097/DBP.0000000000000140
25. Díaz-Román A, Zhang J, Delorme R, Beggiato A, Cortese S. Sleep in youth with autism spectrum disorders: systematic review and meta-analysis of subjective and objective studies. Evid Based Ment Health. 2018;21(4):146–154. doi:10.1136/ebmental-2018-300037
26. Delahaye J, Kovacs E, Sikora D, et al. The relationship between health-related quality of life and sleep problems in children with autism spectrum disorders. Res Autism Spectr Disord. 2014;8(3):292–303. doi:10.1016/j.rasd.2013.12.015
27. Allik H, Larsson J-O, Smedje H. Insomnia in school-age children with Asperger syndrome or high-functioning autism. BMC psychiatr. 2006;6(1):1–11. doi:10.1186/1471-244X-6-18
28. Johnson CR, Smith T, DeMand A, et al. Exploring sleep quality of young children with autism spectrum disorder and disruptive behaviors. Sleep Med. 2018;44:61–66. doi:10.1016/j.sleep.2018.01.008
29. Shea N, Payne E, Russo N. Brief report: social functioning predicts externalizing problem behaviors in autism spectrum disorder. J Autism Dev Disord. 2018;48(6):2237–2242. doi:10.1007/s10803-017-3459-8
30. Bornstein MH, Hahn CS, Haynes OM. Social competence, externalizing, and internalizing behavioral adjustment from early childhood through early adolescence: developmental cascades. Dev Psychopathol. 2010;22(4):717–735. doi:10.1017/S0954579410000416
31. Eriksen MB, Frandsen TF. The impact of patient, intervention, comparison, outcome (PICO) as a search strategy tool on literature search quality: a systematic review. J Med Library Assoc. 2018;106(4):420. doi:10.5195/jmla.2018.345
32. Noyes J, Booth A, Flemming K, et al. Cochrane qualitative and implementation methods group guidance series—paper 3: methods for assessing methodological limitations, data extraction and synthesis, and confidence in synthesized qualitative findings. J Clin Epidemiol. 2018;97:49–58. doi:10.1016/j.jclinepi.2017.06.020
33. Aathira R, Gulati S, Tripathi M, et al. Prevalence of sleep abnormalities in Indian children with autism spectrum disorder: a cross-sectional study. Pediatr Neurol. 2017;74:62–67. doi:10.1016/j.pediatrneurol.2017.05.019
34. Abel EA, Schwichtenberg AJ, Brodhead MT, Christ SL. Sleep and challenging behaviors in the context of intensive behavioral intervention for children with Autism. J Autism Dev Disord. 2018;48(11):3871–3884. doi:10.1007/s10803-018-3648-0
35. Anders T, Iosif AM, Schwichtenberg AJ, Tang K, Goodlin-Jones B. Sleep and daytime functioning: a short-term longitudinal study of three preschool-age comparison groups. Am J Intellect Dev Disabil. 2012;117(4):275–290. doi:10.1352/1944-7558-117.4.275
36. Chong PLH, Ebel E, Peo R, McCormick EB, Schwichtenberg AJ. Sleep dysregulation and daytime electrodermal patterns in children with autism: a Descriptive Study. J Genet Psychol. 2021;1–4.
37. Cohen S, Fulcher BD, Rajaratnam SM, et al. Behaviorally-determined sleep phenotypes are robustly associated with adaptive functioning in individuals with low functioning autism. Sci Rep. 2017;7(1):1–8. doi:10.1038/s41598-017-14611-6
38. Cohen S, Fulcher BD, Rajaratnam SM, et al. Sleep patterns predictive of daytime challenging behavior in individuals with low‐functioning autism. Autism Res. 2018;11(2):391–403. doi:10.1002/aur.1899
39. Fadini CC, Lamônica DA, Fett-Conte AC, et al. Influence of sleep disorders on the behavior of individuals with autism spectrum disorder.Frontiers in human neuroscience. Front Hum Neurosci. 2015;9:397. doi:10.3389/fnhum.2015.00347
40. Fletcher FE, Foster-Owens MD, Conduit R, Rinehart NJ, Riby DM, Cornish KM. The developmental trajectory of parent-report and objective sleep profiles in autism spectrum disorder: associations with anxiety and bedtime routines. Autism. 2017;21(4):493–503. doi:10.1177/1362361316653365
41. Gunes S, Ekinci O, Feyzioglu A, Ekinci N, Kalinli M. Sleep problems in children with autism spectrum disorder: clinical correlates and the impact of attention deficit hyperactivity disorder. Neuropsychiatr Dis Treat. 2019;15:763. doi:10.2147/NDT.S195738
42. Goldman SE, Surdyka K, Cuevas R, Adkins K, Wang L, Malow BA. Defining the sleep phenotype in children with autism. Dev Neuropsychol. 2009;34(5):560–573. doi:10.1080/87565640903133509
43. Goldman SE, McGrew S, Johnson KP, Richdale AL, Clemons T, Malow BA. Sleep is associated with problem behaviors in children and adolescents with autism spectrum disorders. Res Autism Spectr Disord. 2011;5(3):1223–1229. doi:10.1016/j.rasd.2011.01.010
44. Henderson JA, Barry TD, Bader SH, Jordan SS. The relation among sleep, routines, and externalizing behavior in children with an autism spectrum disorder. Res Autism Spectr Disord. 2011;5(2):758–767. doi:10.1016/j.rasd.2010.09.003
45. Hoshino Y, Watanabe H, Yashima Y, Kaneko M, Kumashiro H. An investigation on sleep disturbance of autistic children. Psychiatry Clin Neurosci. 1984;38(1):45–51. doi:10.1111/j.1440-1819.1984.tb00353.x
46. Kang Y-Q, Song X-R, Wang G-F, Su -Y-Y, Li P-Y, Zhang X. Sleep problems influence emotional/behavioral symptoms and repetitive behavior in preschool-aged children with Autism spectrum disorder in the unique social context of China. Front psychiatr. 2020;11:273. doi:10.3389/fpsyt.2020.00273
47. Kelmanson IA. Sleep disturbances and their associations with emotional/behavioral problems in 5-year-old boys with autism spectrum disorders. Early Child Dev Care. 2018;4:56–78.
48. Kirkpatrick B, Gilroy SP, Leader G. Qualitative study on parents’ perspectives of the familial impact of living with a child with autism spectrum disorder who experiences insomnia. Sleep Med. 2019;62:59–68. doi:10.1016/j.sleep.2019.01.032
49. Krakowiak P, Goodlin‐Jones BETH, Hertz‐Picciotto IRVA, Croen LA, Hansen R. Sleep problems in children with autism spectrum disorders, developmental delays, and typical development: a population‐based study. J Sleep Res. 2008;2:197–206.
50. Lambert A, Tessier S, Rochette A-C, Scherzer P, Mottron L, Godbout R. Poor sleep affects daytime functioning in typically developing and autistic children not complaining of sleep problems: a questionnaire-based and polysomnographic study. Res Autism Spectr Disord. 2016;23:94–106. doi:10.1016/j.rasd.2015.11.010
51. MacDuffie KE, Munson J, Greenson J, et al.Sleep problems and trajectories of restricted and repetitive behaviors in children with neurodevelopmental disabilities. J Autism Dev Disord. 2020;50(11):
52. Malhi P, Kaur A, Singhi P, Sankhyan N. Sleep dysfunction and behavioral daytime problems in children with autism spectrum disorders: a comparative study. Indian J Pediatr. 2019;86(1):12–17. doi:10.1007/s12098-018-2731-z
53. Malow B, Adkins KW, McGrew SG, et al. Melatonin for sleep in children with autism: a controlled trial examining dose, tolerability, and outcomes. J Autism Dev Disord. 2012;42(8):1729–1737. doi:10.1007/s10803-011-1418-3
54. Manelis-Baram L, Meiri G, Ilan M, et al. Sleep disturbances and sensory sensitivities co-vary in a longitudinal manner in pre-school children with autism spectrum disorders. J Autism Dev Disord. 2021;5:1–15.
55. Mazurek MO, Dovgan K, Neumeyer AM, Malow BA. Course and predictors of sleep and co-occurring problems in children with autism spectrum disorder. J Autism Dev Disord. 2019;49(5):2101–2115. doi:10.1007/s10803-019-03894-5
56. McLay LL, France KG, Blampied NM, et al. Collateral child and parent effects of function‑based behavioral interventions for sleep problems in children and adolescents with autism. J Autism Dev Disord. 2021;5:1–16.
57. Mutluer T, Karakoc Demirkaya S, Abali O. Assessment of sleep problems and related risk factors observed in Turkish children with Autism spectrum disorders. Autism Res. 2016;9(5):536–542. doi:10.1002/aur.1542
58. Naito N, Kikuchi M, Yoshimura Y, et al. Atypical body movements during night in young children with autism spectrum disorder: a pilot study. Sci Rep. 2019;9(1):1–10. doi:10.1038/s41598-019-43397-y
59. Ng R, Heinrich K, Hodges E. Do you really lose when you snooze? Sleep correlates of externalizing and attention problems among pediatric patients with ADHD, ASD, and comorbid diagnosis. J Ment Health Res Intellect Disabil. 2020;13(3):231–249. doi:10.1080/19315864.2020.1760973
60. Papadopoulos N, Sciberras E, Hiscock H, Mulraney M, McGillivray J, Rinehart N. The efficacy of a brief behavioral sleep intervention in school-aged children with ADHD and comorbid autism spectrum disorder. J Atten Disord. 2019;23(4):341–350. doi:10.1177/1087054714568565
61. Patzold LM, Richdale AL, Tonge BJ. An investigation into sleep characteristics of children with autism and Asperger’s disorder. J Paediatr Child Health. 1998;34(6):528–533. doi:10.1046/j.1440-1754.1998.00291.x
62. Phung JN, Goldberg WA. Poor sleep quality is associated with discordant peer relationships among adolescents with Autism Spectrum Disorder. Res Autism Spectr Disord. 2017;34:10–18. doi:10.1016/j.rasd.2016.11.008
63. Saré RM, Smith CB. Association between sleep deficiencies with behavioral problems in autism spectrum disorder: subtle sex differences. Autism Res. 2020;13(10):1802–1810. doi:10.1002/aur.2396
64. Schreck KA, Mulick JA, Smith AF. Sleep problems as possible predictors of intensified symptoms of autism. Res Dev Disabil. 2004;25(1):57–66. doi:10.1016/j.ridd.2003.04.007
65. Schroder CM, Malow BA, Maras A, et al. Pediatric prolonged-release melatonin for sleep in children with autism spectrum disorder: impact on child behavior and caregiver’s quality of life. J Autism Dev Disord. 2019;49(8):3218–3230. doi:10.1007/s10803-019-04046-5
66. Segawa M, Katoh M, Katoh J, Nomura Y. Early modulation of sleep parameters and its importance in later behavior. Brain Dys. 1992;5(3):211–223.
67. Thenhausen N, Kuss M, Wiater A, Schlarb AA. Sleep problems in adolescents with Asperger syndrome or high-functioning autism. Somnologie. 2017;21(1):28–36. doi:10.1007/s11818-016-0078-0
68. Tudor ME, Hoffman CD, Sweeney DP. Children with autism: sleep problems and symptom severity. Focus Autism Other Dev Disabl. 2012;27(4):254–262. doi:10.1177/1088357612457989
69. Wang G, Liu Z, Xu G, et al. Sleep disturbances and associated factors in Chinese children with autism spectrum disorder: a retrospective and cross-sectional study. Child Psychiatry Hum Dev. 2016;47(2):248–258. doi:10.1007/s10578-015-0561-z
70. Yang X-L, Liang S, Zou M-Y, et al. Are gastrointestinal and sleep problems associated with behavioral symptoms of autism spectrum disorder? Psychiatry Res. 2018;259:229–235. doi:10.1016/j.psychres.2017.10.040
71. Yavuz-Kodat E, Reynaud E, Geoffray -M-M, et al. Disturbances of continuous sleep and circadian rhythms account for behavioral difficulties in children with autism spectrum disorder. J Clin Med. 2020;9(6):1978. doi:10.3390/jcm9061978
72. Acebo C, Sadeh A, Seifer R, et al. Estimating sleep patterns with activity monitoring in children and adolescents: how many nights are necessary for reliable measures? Sleep. 1999;22(1):95–103. doi:10.1093/sleep/22.1.95
73. Mannion A, Leader G. Sleep problems in autism spectrum disorder: a literature review. Rev J Autism Develop Disorders. 2014;1(2):101–109. doi:10.1007/s40489-013-0009-y
74. Kent RSE. Prevalence of anxiety in autism spectrum disorders. In: Anxiety in Children and Adolescents with Autism Spectrum Disorder. Academic Press, Elsevier Inc; 2017:5–32.
75. Baker EK, Richdale AL. Sleep patterns in adults with a diagnosis of high-functioning autism spectrum disorder. Sleep. 2015;38(11):1765–1774. doi:10.5665/sleep.5160
76. van den Bergh SF, Scheeren AM, Begeer S, Koot HM, Geurts HM. Age related differences of executive functioning problems in everyday life of children and adolescents in the autism spectrum. J Autism Dev Disord. 2014;44(8):1959–1971. doi:10.1007/s10803-014-2071-4
77. Mannion A, Leader G. An investigation of comorbid psychological disorders, sleep problems, gastrointestinal symptoms and epilepsy in children and adolescents with autism spectrum disorder: a two year follow-up. Res Autism Spectr Disord. 2016;22:20–33. doi:10.1016/j.rasd.2015.11.002
78. Bos MG, Diamantopoulou S, Stockmann L, Begeer S, Rieffe C. Emotion control predicts internalizing and externalizing behavior problems in boys with and without an autism spectrum disorder. J Autism Dev Disord. 2018;48(8):2727–2739. doi:10.1007/s10803-018-3519-8
79. Tager-Flusberg H, Plesa Skwerer D, Joseph RM, et al. Conducting research with minimally verbal participants with autism spectrum disorder. Autism. 2017;21(7):852–861. doi:10.1177/1362361316654605
80. Frazier TW, Krishna J, Klingemier E, Beukemann M, Nawabit R, Ibrahim S. A randomized, crossover trial of a novel sound-to-sleep mattress technology in children with autism and sleep difficulties. J Clin Sleep Med. 2017;13(1):95–104. doi:10.5664/jcsm.6398
81. Moore M, Evans V, Hanvey G, Johnson C. Assessment of sleep in children with autism spectrum disorder. Children. 2017;4(8):72. doi:10.3390/children4080072
82. Yavuz-Kodat E, Reynaud E, Geoffray MM, et al. Validity of actigraphy compared to polysomnography for sleep assessment in children with autism spectrum disorder. Front Psychiatr. 2019;551. doi:10.3389/fpsyt.2019.00551
83. Malow BA, Marzec ML, McGrew SG, Wang L, Henderson LM, Stone WL. Characterizing sleep in children with autism spectrum disorders: a multidimensional approach. Sleep. 2006;29:1563–1571. doi:10.1093/sleep/29.12.1563
84. Hoffman CD, Sweeney DP, Lopez-Wagner MC, Hodge D, Nam CY, Botts BH. Children with autism: sleep problems and mothers’ stress.. Focus Autism Other Dev Disabl. 2008;23(3):155–165. doi:10.1177/1088357608316271
85. Hsu CC, Sandford BA. The Delphi technique: making sense of consensus. Pract Assess, Res, Eval. 2007;12(1):10.
86. Liberati A, Altman D, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol. 2009;62(10):e1–e34. doi:10.1016/j.jclinepi.2009.06.006
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
