Back to Journals » Infection and Drug Resistance » Volume 14
Evaluation of Wet Mount and Concentration Techniques of Stool Examination for Intestinal Parasites Identification at Debre Markos Comprehensive Specialized Hospital, Ethiopia
Authors Demeke G , Fenta A , Dilnessa T
Received 25 February 2021
Accepted for publication 30 March 2021
Published 9 April 2021 Volume 2021:14 Pages 1357—1362
DOI https://doi.org/10.2147/IDR.S307683
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Suresh Antony
Gebreselassie Demeke, Abebe Fenta, Tebelay Dilnessa
Department of Medical Laboratory Science, Health Science College, Debre Markos University, Debre Markos, Ethiopia
Correspondence: Gebreselassie Demeke Tel +251924338828
Email [email protected]
Background: Intestinal parasitic infection has serious medical and public health impacts in developing countries. Intestinal parasites were under reported via direct wet mount method due to its poor sensitivity. So, the selection of a more sensitive and reliable diagnostic method is essential.
Objective: The aim of the study was to evaluate the diagnostic performance of wet mount and concentration techniques of stool examination for intestinal parasites identification.
Methods: A cross-sectional study among 797 pregnant women was conducted from October 2018 to February 2019 at Debre Markos Comprehensive Specialized Hospital. The study participants were selected by using a systematic random sampling technique. Stool sample was processed by using Wet Mount (WM) and Formol-Ether Concentration (FEC) techniques. Data was entered into Epi-data version 3.1 and analysis using SPSS version 20.0. The sensitivity, specificity, positive predictive value, and negative predictive value were determined against the combined result as “Gold” standard. Kappa value was determined to estimate the agreement of the diagnostic methods.
Results: The overall prevalence of intestinal parasites (IP) was 35.2% with a combined method. By using WM and FEC the prevalence of IP was 13.1% and 25.7%, respectively. The sensitivity and negative predictive value of WM method were 37.1% and 74.6%, and for FEC method 73.5% and 87.5%, respectively. Test efficiency of direct WM and FEC were 77.9% and 90.7%, respectively. The agreement of WM and FEC techniques with combined technique were moderate (κ=0.434) and perfect (κ=0.783), respectively.
Conclusion: The prevalence of intestinal parasites was under-reported by wet mount technique. The diagnostic performance of FEC technique for the diagnosis of intestinal parasites in pregnant women was notably high as compared to WM technique in the present study. Therefore, the WM and FEC combined method should be used as a routine diagnostic technique for the diagnosis of intestinal parasites identification.
Keywords: diagnostic method comparison, intestinal parasites, pregnant women, Ethiopia
Introduction
Intestinal parasitic infection is common and has serious medical and public health impacts in developing countries. More than 3.5 billion individuals are infected by one of the intestinal parasites, and 450 million develop illness because of parasitic infection.1
Intestinal parasite infection, especially soil transmitted helminths (STH), induce deficiencies of energy, protein, folate, zinc, and iron, and anemia in pregnant women. These result in low pregnancy weight gain and intrauterine growth retardation (IUGR), and low birthweight (LBW).2
Fecal specimen examination for intestinal parasitic identification is increased by the use of concentration methods. The concentration of parasites in ova or cysts from fecal specimens can be consummate in different ways.3
Even though several diagnostic methods such as Kato-Katz and Formol-Ether Concentration (FEC) techniques are available, direct wet mount is commonly used as a reliable diagnosis method for the diagnosis of intestinal parasitic infections generally in Africa and particularly in Ethiopia.4
The direct wet mount examination of soft-to-watery fecal specimens has a unique advantage that can detect the motile trophozoite stage of the protozoan species. However, timely processing, within 1 hour of passage of a fresh specimen, is not always possible in a busy clinical laboratory, so another alternative method is recommended, like formal-ether sedimentation techniques.5,6
Sensitivity, specificity, and NPVs of wet mount method for the diagnosis of intestinal parasitic infections were 49.6%, 100%, and 56.8%, respectively. Agreements of this method the combined methods for the diagnosis of intestinal parasitic infections were moderate (k=0.439).7
The reliable diagnosis of intestinal parasitic infections requires a more rapid, easy, and sensitive method. The Wet mount method has been chosen as a routine diagnosis because it is easy to perform, had low cost and was time saving as compared to the other two techniques. The detection rate of parasites in a single stool examination using Wet mount method is very limited due to poor sensitivity.8 The FEC diagnostic method is very sensitive and reliable to apply intestinal parasite detection. Even though WM has low sensitivity it is still important to detect motile trophozoite and FEC essential for helminths ova detection. Therefore conducting this research was important to select the appropriate diagnostic method to detect different parasite species.
Methods and Materials
Study Design, Period, and Area
A cross-sectional study was conducted from October 2018 to February 2019 at Debre Markos Comprehensive Specialized Hospital (DMCSH), in Debre Markos town, in the Amhara region, Ethiopia. The hospital provided all necessary services including ANC. The hospital was expected to provide services to more than 3.5 million people. Debre Markos town is located 300 km from Addis Ababa, the capital of Ethiopia, and 265 from Bahir Dart, the main city of Amhara regional state.
The Source and Study Population
The source population were all pregnant women who visited Debre Markos Comprehensive specialized hospital. Those systematically selected pregnant women who fulfilled the inclusion criteria, and gave consent were considered as the study population.
Inclusion and Exclusion Criteria
All pregnant women who visited an antenatal care (ANC) clinic at DMCSH and gave consent were included in the study, whereas pregnant women who had a history of anti-helminthic drug administration in the 2 weeks prior to screening was excluded.
Sampling Techniques and Sample Size
A systematic random sampling technique was used to select study participants. On average, 20 pregnant women per day visit the ANC clinic in DMCSH according to the ANC clinic registration book. There are 80 working days in the study period. Therefore, a total of 1,600 pregnant women attended an ANC clinic in DMCSH during the study period. First, proportional allocation of the sample size (n=797) was done based on the size of the source population (N) or the total pregnant women who visited during the study period. Next, k was obtained (k=population size/sample size=2), and finally study participants were selected by random start between the first and kth element and then proceeded with the selection of every kth element from then onwards. Therefore, a total of 797 pregnant were included in this study.
Data Collection and Processing
The pregnant women who participated in this study were informed about the purpose of the study. Approximately 2 grams of fresh stool samples was collected with a clean stool cup from each study participant and transported to the nearby health institution within an hour. The fresh stool samples were processed with WM and FEC techniques to detect the presence of intestinal parasites.
Wet Mount Method
In the wet mount, fresh stool samples (2 mg of stool) were put on a slide witha wooden applicator stick, the stool was emulsified with a drop of physiological saline (0.85%) for diarrheic and semi-solid samples. For formed stools, iodine was used. Then, they were covered with a cover slide and examined under a microscope using first 10x objectives and then 40x objectives.9 The motile stage of protozoan parasites were identified using direct wet mount, and cysts were identified using FEC.
Formol-Ether Concentration Method
Stool specimens were processed following a formol-ether standard operation procedure. One gram of stool was added to a clean conical centrifuge tube containing 7 mL, of 10% formol water. The suspension was filtered through a sieve into a 15 mL conical centrifuge tube. Then 4 mL of diethyl ether was added to the formalin solution, the content was centrifuged at 300 rpm for 1 minute. The supernatant was discarded and smear was prepared using a slide from the sediment. Finally the slide was examined under a microscope with magnification power of 10x objective first and then 40x objective.9
Performance Evaluation
The performance comparison of WM and FEC diagnostic methods in the diagnosis of intestinal parasitic infections were done by taking the combined result as a “Gold” standard.10 The test agreements of diagnostic methods were evaluated by Kappa value. Kappa values were estimated to determine the strength of agreement on the diagnostic methods and it was interpreted as slight (0.01–0.20), fair (0.21–0.40), moderate (0.41–0.60), substantial (0.61–0.80), and perfect (0.81–0.99) agreement.11
Data Quality Control
Prior to data collection, training was given for data collectors about how to collect the specimen and other sociodemographic data. The quality of reagents and instruments were checked by the principal investigator. The specimens were also checked for serial number, quantity, and procedures of collection. To eliminate observer bias, each stool sample was examined immediately by two experienced laboratory technicians. For those cases with discordant results, the principal investigator repeated the test. The results of the principal investigator were considered as the final result.
Data Analysis
Data was entered by using Epi-data version 3.1 and data analysis was done using SPSS version 20.0 statistical software. Since there was no reference diagnostic method for intestinal parasites, the sensitivity (Sn), specificity (Sp), negative predictive value (NPV), and positive predictive value (PPV) were determined for each diagnostic method by taking the combined results as the diagnostic “Gold“ standard. Kappa value was calculated to determine the strength of agreement on the diagnostic method. Finally, a P-value<0.05 was considered as statistically significant.
Ethical Consideration
The ethical clearance was obtained from the College of Health Science ethical review committee (RF: DMU/CHSRERC/31/11/2018) of Debre Markos University. Permission was obtained from DMCSH administrative bodies. Written informed consent was obtained from every study participant. Intestinal parasite positive individuals were treated with anti-parasitic drugs depending on the types of species identification. This study was complied with the Declaration of Helsinki.
Results
Socio-Demographic Characteristics of the Study Participants
In the present study, a total of 797 pregnant women were enrolled with a 0.1% non-response rate. The mean age was 26.45 years and it ranged from 19–37 years with a standard deviation of 3.98 years. Seven hundred and ten (89.1%) of the study participants were urban dwellers.
Prevalence of Intestinal Parasitic Infections
The overall prevalence of intestinal parasites was 35.2% (95% CI=31.9–38.5) with the combined method. The prevalence of intestinal parasites by using wet mount and FEC were 13.1% (95% CI=10.7–15.6) and 25.7% (95% CI=22.8–28.7), respectively.
The prevalence of A. lumbricoides, Hookworm, T. trichiura, Taenia species, S. stercoralis, H. nana, E. histolytica, and G. lamblia infections by the combined method was 3.3%, 14.3%, 1.4%, 4.3%, 1.0%, 1.4%, 9.7%, and 3.5%, respectively. Hookworm was the most common intestinal parasite from our findings (Table 1).
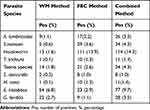 |
Table 1 Prevalence of Intestinal Parasite Species Identified by the WT, FEC, and Combination of WM and FEC Techniques among Pregnant Women at DMCSH, Ethiopia |
Performance Evaluation of Diagnostic Methods in the Diagnosis of Intestinal Parasites
The FEC technique had more sensitivity (73.5%) and NPV (87.5%) than WM sensitivity (37.1%) and NPV (74.6%) in the detection of intestinal parasites (Table 2).
 |
Table 2 The Performance of Wet Mount and Formol-Ether Concentration Techniques to Diagnose Intestinal Parasite against the Gold Standard among Pregnant Women in DMCSH, Ethiopia |
Test Efficiency (TE) of Diagnostic Methods
The overall ability of direct WM and FEC to correctly diagnose intestinal parasites (TE) were 77.9% and 90.7%, respectively (Table 2).
Test Agreement (TA) of Diagnostic Methods
The agreement of FEC technique with the combined result was perfect in detecting intestinal parasites (κ=0.783). On the other hand, the WM technique agreed moderately in intestinal parasites (κ=0.434), detection with combined technique (Table 2).
Discussion
Direct wet mount is commonly used as a diagnostic method for the diagnosis of both protozoal and helminthic infections generally in Africa and particularly in Ethiopia.12 The sensitivity of the FEC was higher than that of the Kato-Katz method and wet mount for the diagnosis of intestinal parasites except Schistosoma mansoni.13 Therefore, the use FEC technique for routine laboratory test is critical for the correct detection and management of intestinal parasite infection.
In the present study a number of intestinal parasites were detected using FEC and Wet mount methods. We used the two combined results to compare the sensitivity and negative predictive value because the combined results have better detection ability than the single methods. In this study, formal-ether concentration techniques have better outperformance than the wet mount.
In comparison to parasite recovery from stool specimen, our results confirmed that the prevalence of intestinal parasite among pregnant women by using combined method was 35.2%, FEC was 25.7%, and by wet mount preparation was 13.1%.
This result is similar with other studies conducted in other parts of Ethiopia among pregnant women previously.14 This confirmed that using FEC technique for routine stool examination is essential for better detection of intestinal parasite and good management intestinal parasite infections. Intestinal parasite infected pregnant women may develop anemia; this may lead to infant underweight.
In our study the sensitivity for Wet mount was 37.1 (95% CI=31.7–42.9). This is lower than another similar study conducted in Ethiopia where the sensitivity of wet mount was 48.9% (95% CI=48.8–49.1). But NPV was 74.6% (95% CI=71.2–77.7) and higher than in another similar study, in which the sensitivity was 33.7 (95% CI=33.5–33.9).15
In the present study the sensitivity and negative predictive value of FEC was 73.5% (95% CI=68–78.3) and 87.5% (95% CI=84.6–89.9), respectively.16 Our result was smaller than the other finding which was conducted in other parts of Ethiopia that showed FEC sensitivity and negative predictive value was 85% (95% CI=70.2–94.3) and 97.6% (95% CI=95.98.8),16 respectively. The NPV of FEC was higher than another study conducted in Gondar, Ethiopia in which the NPV was 64.4%.17 The variation may be due to parasite prevalence, reagent preparation, and level of parasite identification experience difference.
In our finding the overall ability of WM and FEC test efficiency or correctly diagnosing intestinal parasite was 77.9% and 87.5%, respectively, but in another study, the test efficiency of WM and FEC was 50.3% and 79.1%.17 Our finding was higher than other research conducted by other scholars. This may due to the sample size and reagents difference.
Moderate test agreement (k=0.434) was found for wet mount and FEC (k=0.783), and perfect agreement was recorded against the combined result. Similar test agreement was recorded by other scholars. Moderate test agreement was recorded for wet mount (k=0.48) and FEC (k=0.89) was excellent test agreement against gold standard.16,18
Our study finding showed that direct wet mount exhibited very low sensitivity for the detection of intestinal parasites as compared to the FEC. This was similar with another study.19 This suggested that the use of direct wet mount alone as an intestinal parasitic infections identification is insufficient and may lead to false negative results.
Conclusion
The prevalence of intestinal parasites was under-reported by direct wet mount microscopy. The diagnostic performance of FEC technique for the diagnosis of intestinal parasites in pregnant women was significantly high as compared to WM technique in the present study. Therefore, the FEC method should be used in combination with WM as a routine diagnostic technique in health facility laboratories for the diagnosis of intestinal parasites, especially in pregnant women.
Abbreviations
FEC, formal-ether concentration; NPV, negative predictive value; PPV, positive predictive value; WM, wet mount.
Data Sharing Statement
The data can be accessed upon reasonable request.
Acknowledgments
We would like to highly acknowledge Debre Markos University for covering the budget of the project. We also acknowledge all study participants that gave stool samples and Debre Markos Comprehensive specialized hospital administrative bodies who gave permission, and staff members for unreserved support.
Author Contributions
All authors contributed to the data analysis, drafting or revising of the article, have agreed on the journal to which the article will be submitted, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Funding
This work was supported by Debre Markos University.
Disclosure
The authors declare that they have no competing interests in this work.
References
1. Daumerie D, Savioli L. Working to Overcome the Global Impact of Neglected Tropical Diseases: First WHO Report on Neglected Tropical Diseases. Vol. 1. World Health Organization; 2010.
2. Stephenson LS, Latham MC, Ottesen E. Malnutrition and parasitic helminth infections. Parasitology. 2000;121(S1):S23–S38. doi:10.1017/S0031182000006491
3. Parameshwarappa K, Chandrakanth C, Sunil B. The Prevalence of Intestinal Parasitic Infestations and the Evaluation of Different Concentration Techniques of the Stool Examination. J Clin Diagnostic Res. 2012;6(7).
4. Utzinger J, Rinaldi L, Lohourignon LK, et al. FLOTAC: a new sensitive technique for the diagnosis of hookworm infections in humans. Trans R Soc Trop Med Hyg. 2008;102(1):84–90. doi:10.1016/j.trstmh.2007.09.009
5. Melvin DM. Laboratory Procedures for the Diagnosis of Intestinal Parasites. US Department of Health and Human Services, Public Health Service, Centers; 1982.
6. Moges EM. Evaluation of Sediment Transport Equations and Parameter Sensitivity Analysis Using the SRH-2D Model. MS Thesis: Universität Stuttgart; 2010.
7. Melaku M, Bayayibign B, Gelaye W. The Performance evaluation of three laboratory diagnostic methods for intestinal parasitic infections at rural Bahir Dar, Northwest Ethiopia: a cross-sectional study. J Health Sci. 2018.
8. Ritchie LS. An ether sedimentation technique for routine stool examinations. Bull U S Army Med Dep. 1948;8(4):326.
9. Williams J. District Laboratory Practice in Tropical Countries. Part 1. Monica Cheesbrough. Doddington. Cambridgeshire: tropical Health Technology, 1998: viii+ 456pp. Price£ 10.90 (surface post) or£ 19.00 (airmail). ISBN 9507434-4-5 [Also available from Cambridge University Press (£ 35.00; ISBN 0-521-66547-7).]: Royal Society of Tropical Medicine and Hygiene; 2000.
10. Dendukuri N, Rahme E, Belisle P, Joseph L. Bayesian sample size determination for prevalence and diagnostic test studies in the absence of a gold standard test. Biometrics. 2004;60(2):
11. Landis JR, Koch GG. An application of hierarchical kappa-type statistics in the assessment of majority agreement among multiple observers. Biometrics. 1977;33(2):363–374. doi:10.2307/2529786
12. Santos FL, Cerqueira EJ, Soares NM. Comparison of the thick smear and Kato-Katz techniques for diagnosis of intestinal helminth infections. Rev Soc Bras Med Trop. 2005;38(2):196–198. doi:10.1590/S0037-86822005000200016
13. Knopp S, Rinaldi L, Khamis IS, et al. A single FLOTAC is more sensitive than triplicate Kato-Katz for the diagnosis of low-intensity soil-transmitted helminth infections. Trans R Soc Trop Med Hyg. 2009;103(4):347–354. doi:10.1016/j.trstmh.2008.11.013
14. Mengist HM, Demeke G, Zewdie O, et al. Diagnostic performance of direct wet mount microscopy in detecting intestinal helminths among pregnant women attending ante-natal care (ANC) in East Wollega, Oromia, Ethiopia. BMC Res Notes. 2018;11(1):276. doi:10.1186/s13104-018-3380-z
15. Yimer M, Hailu T, Mulu W, et al. Evaluation performance of diagnostic methods of intestinal parasitosis in school age children in Ethiopia. BMC Res Notes. 2015;8(1):820. doi:10.1186/s13104-015-1822-4
16. Alemu M, Zigta E, Derbie A. Under diagnosis of intestinal schistosomiasis in a referral hospital, North Ethiopia. BMC Res Notes. 2018;11(1):1–5. doi:10.1186/s13104-018-3355-0
17. Moges F, Belyhun Y, Tiruneh M, et al. Comparison of formol-acetone concentration method with that of the direct iodine preparation and formol-ether concentration methods for examination of stool parasites. Ethiopian J Health Dev. 2010;24(2). doi:10.4314/ejhd.v24i2.62965
18. Sheyin Z, Bigwan E, Galadima M. Comparison of formol-ether, direct smear and nigrosine methylene blue for the diagnosis of human intestinal parasites; 2013.
19. Hailu T, Abera B. Performance evaluation of direct saline stool microscopy, Formol ether concentration and Kato Katz diagnostic methods for intestinal parasitosis in the absence of gold standard methods. Trop Doct. 2015;45(3):178–182. doi:10.1177/0049475515581127
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
