Back to Journals » Cancer Management and Research » Volume 11
Evaluation of therapeutic efficacy with CytoSorter® circulating tumor cell–capture system in patients with locally advanced head and neck squamous cell carcinoma
Authors Zheng W, Zhang Y, Guo L, Wang S, Fang M, Mao W, Lou J
Received 12 March 2019
Accepted for publication 21 May 2019
Published 27 June 2019 Volume 2019:11 Pages 5857—5869
DOI https://doi.org/10.2147/CMAR.S208409
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Harikrishna Nakshatri
Weihui Zheng,1,2 Yibiao Zhang,3 Liang Guo,2 Shengye Wang,4 Meiyu Fang,5 Weimin Mao,1 Jianlin Lou2
1Center of Oncology, the First Affiliated Hospital of Soochow University, Suzhou 215000, People’s Republic of China; 2Department of Head and Neck Surgery, Zhejiang Cancer Hospital & Creative Laboratory of Head and Neck Oncology in Zhejiang Province, Hangzhou 310022, People’s Republic of China; 3Department of Clinical Laboratory, Jinhua Guangfu Hospital, Jinhua 321000, People’s Republic of China; 4Department of Radiotherapy, Zhejiang Cancer Hospital, Hangzhou 310022, People’s Republic of China; 5Department of Medical Oncology, Zhejiang Cancer Hospital, Hangzhou 310022, People’s Republic of China
Objective: This study aimed to investigate the feasibility of utilizing CytoSorter® system to detect circulating tumor cells (CTCs) and clinical value of CTCs in patients with locally advanced head and neck squamous cell carcinoma (LAHNSCC).
Methods: 31 patients with LAHNSCC, 12 healthy volunteers, and 6 patients with benign tumor serving as controls were enrolled in this study. CTCs were enriched with the CytoSorter®, a microfluidic based immune capture system. CTC detection was performed before and after induction chemotherapy, as well as after surgery and/or radiotherapy. Correlations between CTC enumerations at different time points and survival outcome and recurrence risk were evaluated. The correlation between CTCs and clinicopathological characteristics was appraised. Follow-up of patients continued until March 2019.
Results: While CTCs were not found in the controls, they were detected in 24 of 31 LAHNSCC patients. CTCs could be used to distinguish diseased people from the healthy (P<0.0001). CTCs were statistically associated with patient age (P=0.037, >60 years old vs<60 years old) and lymph node metastasis (P= 0.034, N0N1 VS N2N3). Most patients had significantly reduced CTCs at the end of treatment. Patients with partial remission of tumor after induction therapy had more CTCs than those with complete remission of tumor. Patients with higher CTCs counts prior to treatment had higher chance of developing local recurrence of tumor after treatment (P=0.0187).
Conclusion: CTCs were successfully isolated in LAHNSCC patients using CytoSorter® system with better sensibility. CTCs can be used to differentiate LAHNSCC patients from those with benign HNSCC tumor or healthy volunteers, and as markers to monitor patient’s response to treatment and predict the local tumor recurrence after treatment. CTC detection at baseline has the greatest prognostic potency in LAHNSCC patients.
Keywords: circulating tumor cells, head and neck carcinoma, squamous cell carcinoma, induction chemotherapy
Introduction
With more than 1.45 million newly diagnosed cases globally, head and neck squamous cell carcinoma (HNSCC) comprises about 3% of all cancers and caused over 494,000 deaths in 2018.1,2 Although advances in comprehensive therapy (such as surgery, chemotherapy, and radiotherapy) have led to improvements in therapeutic efficacy and life quality of HNSCC patients, most patients still eventually develop locoregional recurrences of tumors or distal metastases, and the 5-year survival rate is <50%.3,4 Currently, a competent marker to identify effectively patients with refractory HNSCC is lacking.
Circulating tumor cells (CTCs) are TCs that are released from a primary tumor or metastasis sites into circulation, either spontaneously or during medical procedures. CTCs surviving in circulation can colonize a distal site in the body. The 7th edition of the American Joint Committee on Cancer (AJCC) staging manual introduced a new premetastasis stage, cM0(i+), where CTCs are found in the blood or bone marrow without any symptom or sign of metastasis was observed. Furthermore, the 8th edition of the AJCC cancer guidelines said CTCs can be used as a prognostic marker in breast cancer to predict patients’ survival outcome, i.e progression free survival and overall survival. Studies have shown that CTCs have prognostic values not only in breast cancer, but also in colorectal cancer, prostate cancer, and lung cancer.5–8 CTCs can be used as a monitoring tool to evaluate patient response to treatment and to see whether tumor recurrence occurs.9–11 Dynamic monitoring of CTCs and subtyping of CTCs allow doctors to adjust treatment regimen accordingly.12,13
CTCs are rare in the blood and thus, many methods have been developed to enrich CTCs from the blood based on their physical or biological properties.14 Currently, CellSearch is still the only US Food and Drug Administration (FDA)–approved CTC-detection system. However, studies have shown CTC-detection rate in locally advanced HNSCC (LAHNSCC) patients with CellSearch system was <40%.15–18 Preliminary data with CytoSorter® (Hangzhou Watson Biotech, Hangzhou, China) CTC-capture system in LAHNSCC patients showed a CTC-detection rate of >70%. Therefore, we decided to use CytoSorter® system in this study. CytoSorter® is a noninvasive system for the capture, enumeration, isolation, and retrieval of CTCs. CytoSorter® technology employs the positive selection utilizing a streptavidin nanoarray, CytoChipNano, which can be coated with biotin-labelled CTCs capture antibody, and immunofluorescence staining, in order to capture and identify CTCs for cell enumeration. Capture and identification antibodies used here are antiepithelial cell adhesion molecules (EpCAM) and pan-cytokeratin (PanCK), respectively. CytoSorter® system has been used to capture CTCs in patients with breast, liver, lung, colorectal, and pancreatic cancers (unpublished data).10 However, utilization of CytoSorter® system to identify CTCs in patients with HNSCC has not yet been investigated.
31 patients with LAHNSCC, 6 patients with benign HNSCC, and 12 healthy volunteers as controls were enrolled in this study. For the patients with LAHNSCC, CTC enumerations were performed before and after induction chemotherapy and once more after whole procedure was completed (surgery or/and radiotherapy). For those with benign HNSCC and healthy volunteers, CTC enumeration was performed only once. The aims of this study were to assess the feasibility of CTCs detection in LAHNSCC patients using CytoSorter® system, to correlate CTCs to LAHNSCC patients’ clinicopathological findings, to evaluate CTCs as a marker to monitor LAHNSCC patients’ response to the treatment, and to evaluate CTCs as a prognostic marker for survival outcome, local recurrence of tumors, and distal metastasis.
Materials and methods
Ethics
The study followed the principles established in the Declaration of Helsinki and approved by both ethics committees of Zhejiang Cancer Hospital and Zhejiang Jinhua Guangfu Hospital. All patients and healthy volunteers involved provided written informed consent to participate in this research and to publication of their case details.
Cell lines
The human-tongue squamous cell carcinoma cell line, UT-SCC-24A (RRID:CVCL_7826), was obtained from the ATCC (Manassas, VA, USA). UT-SCC-24A cells were maintained in DMEM (Thermo Fisher Scientific, Waltham, MA, USA), supplemented with 10% fetal bovine serum (Thermo Fisher Scientific, Waltham, MA, USA) in the presence of penicillin and streptomycin. UT-SCC-24A cells were housed in 37°C incubators with 5% CO2 saturation.
Patients
Between March 2016 and December 2017, 31 patients with LAHNSCC — 27 males and 4 females — median age of 57.64 years (range: 45–71 years) were enrolled in this study. Inclusion criteria were pathologically or cytologically diagnosed with LAHNSCC at the hypopharynx, oropharynx, larynx or mouth (including LAHNSCC with concomitant esophageal cancer), tumor staged at III–IV (III, IVA and IVB) according to the TNM-staging system of the AJCC (seventh edition), age -OBrk-75 years, no prior chemotherapy, radiotherapy, or surgery for LAHNSCC, no other malignant tumors (except cervical carcinoma in situ, cutaneous basal carcinoma, SCC, and other cancers that had been cured by surgery and no recurrence within 5 years), and compliance with treatments and examinations. Exclusion criteria were pregnant or breast-feeding, prior or concomitant disease, unstable heart disease after treatment, myocardial infarction within prior 6 months, history of mental illness (such as dementia or seizures), active or uncontrollable infection, active gastrointestinal ulcer, and chronic obstructive pulmonary disease requiring hospitalization within prior 1 year. Hypopharyngeal carcinoma was found in 17 patients, oropharyngeal carcinoma in 13, laryngeal cancer in 2 and concomitant esophageal cancer in 5. In addition, peripheral blood was also collected from 12 healthy volunteers and 6 patients with benign HNSCC for the detection of CTCs.
Blood collection and preparation
A 23G needle was used to collect vein-blood sample. The first 2 mL of collected blood was discarded to avoid potential skin cell contamination from venipuncture. Collected blood was stored in a heparin tube (BD) at room temperature before use. Blood has a maximum preservation time of 6 hrs at room temperature. CTCs were enriched by CytoSorter® system. Blood preprocessing procedure was as per the manufacture protocol described. In brief, 4 mL peripheral blood sample was diluted at 1:1 ratio with 1⨰ PBS to final volume of 8 mL, then transferred equally into 2 separate Leucosep® tubes containing 3 mL of Histopaque®-1077 (Sigma-Aldrich) density-gradient media. After density-gradient centrifugation, peripheral blood mononuclear cell (PBMCs) layer was isolated and washed twice with washing medium (WM, 5% FBS DMEM). Final cell pellets were resuspended in 190 μL of WM and ready to use.
CTC enrichment
CTCs were enriched by CytoSorter® epithelial cell detection kit. In brief, The CytoChipNano was first rinsed with 95% ethanol, followed by 2 times 1⨰ PBS wash, and then incubated with EpCAM capture antibody working solution for 1 hr at room temperature. The CytoChipNano was washed 3 times with 1⨰ PBS to remove the residual capture antibody working solution, and then incubated with blocker working solution for 1 hr at room temperature. The CytoChipNano was washed 3 times with 1⨰ PBS to remove the residual blocker working solution before placing onto CytoSorter® system. PBMC sample solution was transferred into SCx Spiral sample tube. Instruction of CTC capture was as described in CytoSorter® user manual.
CTC identification
The CytoChipNano was incubated with staining blocking solution for 20 mins, followed by 3 times washes with 1⨰ PBS, and then incubated with CTC detection antibody cocktail (PanCK-FITC & CD45-PE) for 1 hr at room temperature washed 3 times with 1⨰ PBS to remove the residual detection-antibody cocktail, then incubated with DAPI solution for 30 mins, then washed another 3 times with 1⨰ PBS to remove the residual DAPI solution. Olympus scanning microscope (Olympus BX61, Tokyo, Japan) and CytoView™ software were used to scan CytoChipNano for potential CTCs, and then Nikon microscope (Nikon ECLIPSE Ti) was used to confirm CTC staining and localization.
Assessment of therapeutic efficacy and statistical analysis
Therapeutic efficacy was evaluated according to the Response Evaluation Criteria in Solid Tumor (RECIST 1.1 edition). Measurement of the tumor response by imaging was qualitatively classified into four categories: complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD). Imaging methods used here were computed tomography, positron-emission tomography–computed tomography, magnetic resonance imaging, and laryngoscopy. Responsive was defined as CR and PR, while nonresponsive was defined as stable disease and PD. Dominant response was defined as a very small residual lesion after induction chemotherapy, but absence of measurable lesion on imaging examination. Statistical analysis was performed with SPSS version 19.0.
Results and analysis
Identification of CTCs in LAHNSCC patients
UT-SCC-24A cell line was used as a quality control to evaluate the efficiency of CytoSorter® system. Using the CytoSorter® epithelial cell-detection kit, a capture rate of 88% was achieved for UT-SCC-24A cells (data not shown). CTC detection was performed on 31 LAHNSCC patients, 6 patients with benign tumor, and 12 healthy volunteers. A summary of patients’ clinicopathological characteristics, including age, gender, tumor site, TNM classification, tumor stage, treatment received, and CTC count before treatment, is provided in Table 1. Upon capture, CTCs were stained for PanCK, CD45, and DAPI to identify epithelial type of cells, white blood cells (WBCs), and nuclei, respectively, and defined as PanCK-positive, CD45-negative, and DAPI-positive cells (Figure 1). CTCs were detected in 24 of 31 patients with prior LAHNSCC treatment and the average CTCs count per 7.5 mL blood was 20.71. As no CTCs were found in patients with benign tumor or healthy volunteers, this result indicated that CTC enumeration is able to differentiate LAHNSCC patients from healthy volunteers and patients with benign tumors (P<0.0001), as shown in Figure 1.
 |
Table 1 LAHNSCC patient clinicopathological characteristics and CTC enumerations before treatment |
Correlation of CTCs with patients’ clinicopathological characteristics
In order to assess whether CTCs were associated with any clinicopathological characteristic, patients were divided into groups according to their age, gender, TNM stages, tumor location, or tumor differentiation state. Analysis results showed CTCs were related with patients’ age and lymph node metastasis as shown in Table 2. More CTCs were found in aged patients (P=0.037, >60 years old) and in patients at advanced nodal stage (P=0.0287, N2N3). No significant correlation of CTCs with gender, or tumor stage, or tumor differentiation state was found. Interestingly, CTCs were slightly inversely proportional to the tumor T stage, although there was no statistical significance. More CTCs were detected in LAHNSCC patients at T1T2 stage compared to those at T3T4 stage as shown in Figure 2.
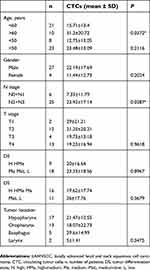 |
Table 2 Correlation of CTC enumerations with LAHNSCC clinicopathological characteristics |
CTCs can be used as a marker to monitor patients’ response to the treatment
LAHNSCC patients are usually treated with surgery, chemotherapy, and radiotherapy. In this study, most patients were treated first with induction chemotherapy, followed by surgery and/or radiotherapy, as shown in Table 3. To evaluate whether CTCs could be used as an indicator of treatment efficacy, CTC enumerations were performed before and after induction chemotherapy and after the whole treatment, and results are listed in Table 3. Follow-up was up until March 2019, by which time 8 patients had died, 5 had developed distal metastasis, and 12 had local recurrences. For the 5 patients without induction chemotherapy, CTC detection was performed before surgery and after surgery or radiotherapy. 7 patients had no CTCs detected before treatment, and none developed local recurrence or metastasis, or died. These 7 patients were excluded from the following analysis. As shown in Figure 3, all patients had CTCs decreased after induction chemotherapy/surgery and decreased even more after the treatment had been completed. The highest number of CTCs detected in the end of treatment was 5 CTCs per 7.5mL blood.
 |
Table 3 CTC enumerations in LAHNSCC patients before, during, and after the treatment |
Image analysis showed that most patients were responsive to the treatment, ie, CR or PR of tumor in the end of treatment, as shown in Figure 3. Only 2 patients had PD when the treatment was finished. Between these 2 patients, 1 had CTCs decreased from 44 to 25, while the other patient had CTCs decreased from 24 to 4 after induction chemotherapy. Although patients proceed further to surgery or radiotherapy, both patients died within 4 months after treatment. Most patients without any evidence of disease in the end of treatment had only 0–2 CTCs detected. Even for the patient with 5 CTCs detected in the end of treatment, imaging examination showed CR of disease. For patients who underwent induction chemotherapy, the average CTC count dropped from 28.42 to 8. 16 patients showed PR, with average CTC count of 9, while 5 patients showed CR, with average CTC count of 4.6. However, there was no statistical significance (P=0.281). One patient had CTC count drop from 4 to 0 after induction chemotherapy and did not take any further treatment. Image examination showed no sign of disease in this patient in the end of treatment. As such, CTCs can be served as a marker to monitor response to the treatment.
CTCs can be used as a prognosis marker for local recurrence of tumor after treatment
Based on results in pancreatic cancer and the unpublished data of breast cancer and physical examinations for cancer screening, a CTC-cutoff value of 2 can be used to distinguish diseased people from the healthy.10 When the CTC cutoff was set at 1, the ROC curve of CytoSorter® CTC-detection system in LAHNSCC patients gave an AUC value of 0.887 as shown in Figure 4. Therefore, both CTC cutoffs of 1 and 2 were used to see whether CTCs were correlated with survical outcome, the local recurrence risk, and distant metastasis after treatment. One patient's death was not tumor related and was thus excluded from the survival analysis. As shown in Figure 4 and Table 4, LAHNSCC patients with higher CTC counts tend to have higher chances of developing local recurrence of tumor or distal metastasis, or die, and CTC enumeration at baseline (prior treatment) has the highest prognostic potency with the lowest P-values compared to CTC enumerations after induction chemotherapy or treatment. Statistical analysis showed CTC counts at baseline can be used to predict the local recurrence risk of tumor after treatment in LAHNSCC patients (P=0.0187). There was no statistical significance regarding CTC enumeration at baseline with distal metastasis or survival outcome of LAHNSCC patients after treatment.
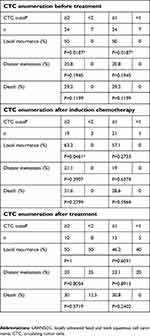 |
Table 4 Local recurrence rate, distant metastasis rate, and death rate of LAHNSCC patients based on different CTC cutoff value |
Discussion
Although advances in diagnosis and treatment procedures have resulted in improved local control for HNSCC patients, overall survival of patients has not changed much in the past few decades.2 Most patients pass away due to distant metastases. LAHNSCC patients who tend to have symptoms, such as local invasion, accompanied by cervical lymph node metastasis, are often at higher risk for distant metastases. LAHNSCC patients usually have poor prognostic outcomes and are more difficult to treat for it is hard to identify micrometastases at early stage using traditional imaging methods. A reliable alternative that allows identification of patients with higher risk of disseminated diseases prior to the development of distinguishable metastasis would have clear clinical relevance.
Metastases develop when detached TCls invade and migrate into circulation, then colonize distant sites and start to proliferate. CTCs represent the process of metastasis. Studies have shown that CTCs may be used as markers to reflect the invasion and metastasis of tumors.19–21 Even more, assessment of CTCs is an earlier, more reproducible indication of disease status than the current imaging methods.22 Monitoring of CTCs provides valuable information for evaluation of disease progression, therapeutic efficacy, and prognosis. Studies of CTCs in HNSCC patients have shown that CTC enumeration is correlated with patient survival.23 A high level of CTCs indicate reduced disease-free survival, reduced progression free survival, and overall survival.23
There are more than 50 techniques available to enrich CTCs from millions of blood cells, by either physical parameters (size or density) or biological characteristics (cell surface markers) or combining both properties.14 Microfluidic based methods are the most advanced technique for CTCs enrichment with high efficiency. Microfluidic methods allow for gentle capturing of live CTCs so that further downstream analysis can be performed using cellular, microscopic, or molecular approaches, to meet the clinical or research need. Microfluidic based CTC-capture devices can be modified to exploit physical and/or biological properties between CTCs and the background cells, thus enabling isolation.24 On the basis of microfluidic technique, CytoSorter® CTC-capture system employs an antibody-immobilized chip for immune capture of CTCs, which are then verified by immunofluorescence staining for PanCK, CD45, and DAPI. The detection rate of CTCs in HNSCC patients is 15–71%, depending on the detection method used.25 As for LAHNSCC, Nichols et al used CellSearch system to detect CTCs in 15 patients, and CTCs were identified in 6 patients (40%; 1–2 CTCs/7.5 mL blood).15 Moreover, Tinhofer et al conducted a prospective study to investigate the CTCs in 144 LAHNSCC patients, and CTCs were detected in 42(29%).26 In this study, CTC detection was performed in 31 LAHNSCC patients, and the detection rate of CTCs was 77% (24/31), which is much higher than previous studies.15–26
While no CTCs were found in either patients with benign tumor or healthy volunteers, CTCs were detected in 24 of 31 LAHNSCC patients with an average CTC count of 20.71 per 7.5 mL blood. The results indicated that CTC enumeration could be used as a tool to differentiate LAHNSCC patients from healthy/patients with benign tumors. A recent study in pancreatic tumor with CytoSorter® CTC capture system also showed that CTC enumeration could be used to distinguish patients with malignant tumors from patients with benign tumors or healthy volunteers.10 It implied that CTC enumeration should be able to reflect tumor stages. Therefore, the relationship of CTCs with clinicopathological characteristics of LAHNSCC patients was evaluated. CTCs were found to be correlated with patients’ age and lymph node metastasis. Patients with lymph node metastasis at N2N3 stage had more CTCs compared to those at N0N1 stage (23.73 vs 7.73). In addition, our results indicated CTCs were associated with age: more CTCs were detected in patients over 60 years old compared to those younger than 60 years old (31.2 VS 15.71). Theoretically, higher CTC counts usually indicated more invasive tumor. Therefore, patients with advanced lymph node metastasis should have more CTCs. Currently, there is no study supporting our finding that aged patients have more CTCs. Tumors tend to occur in older people, especially for those >50 years old.1,2 The effect of aging renders the immune system less effective in killing abnormal/TCs.27 Therefore, older people might have more severe symptoms or more CTCs when they have cancers. There might be some confounding factors or statistical bias as well due to the small sample size. Lymph node metastasis often predicts a high risk for distal metastasis; therefore, more CTCs detected in patients over 60 years old might indicate they were at higher risk for distal metastasis, which needs to be further confirmed in more studies with large sample size and longer follow up.
Although there is no statistical significance, our results show that quantities of CTCs were associated negatively with tumor stages in accordance with a recent study which evaluated 109 patients with lung cancer.28 It has been reported that CTCs undergoing epithelial–mesenchymal transition (EMT) had a greater potential for metastasis and patients with late-stage tumor usually have more mesenchymal CTCs.29 In this study, EpCAM antibodies were used to capture CTCs, and recognized only epithelial type of cells. The reason that fewer CTCs were detected in LAHNSCC patients at later tumor stage is that few of the mesenchymal CTCs were captured by the EpCAM antibody. One advantage of CytoSorter® system is that any choice of biotin-labeled capture antibody can be used to isolate desired cells in the blood. Studies have shown cell-surface vemintin (CSV) antibody can be used to specifically capture mesenchymal CTCs.30,31 CytoSorter® mesenchymal cell–capture kit which uses CSV antibody should be used next time to capture EMT type of CTCs and to confirm whether CTCs are correlated with tumor stages in LAHNSCC patients.
Winter et al used immunomagnetic bead–based CTC enrichment and RT-PCR for CTC detection and reported that CTCs were detectable in 68% of LAHNSCC patients receiving surgery, and they had no relationship with overall and disease-free survival.32 Jatana et al employed immunomagnetic bead–based CTCs enrichment and immunocytochemical staining for CTCs detection.33 Their results showed CTCs were closely related to the development and metastasis of HNSCC. Hristozova et al used flow cytometry and RT-PCR to detect the EpCAM+/CK+/CD45- CTCs and found CTCs were not related to the T stage of primary cancer.34 Buglione et al found the change in CTC number before and after treatment could be used for the prediction of therapy response and disease progression.35 Meta analysis conducted by Harris et al showed CTCs could be used as a marker for the evaluation of therapeutic efficacy in patients with HNSCC.36 Grisanti et al used CellSearch technique to detect CTCs in 53 patients with recurrent/metastatic head and neck cancer, and multivariate analysis showed that one or more CTCs before treatment predicted poorer disease-free survival and poorer overall survival.18 Therefore, CTCs can be used as a potent predictor of therapeutic efficacy in HNSCC patients.
To assess whether CTCs could be used as an indicator of therapeutic efficacy as well in LAHNSCC patients, CTC detection was performed before and after induction chemotherapy, as well as after surgery or/and radiotherapy. Results showed that patients with detectable CTCs at baseline had reduced CTCs after induction chemotherapy, and CTCs decreased even more after surgery or/and radiotherapy. Patients with CR of tumors after induction had fewer CTCs than those with PR (4.6 vs 9). Imaging results showed that patients with CTCs less than 5 in the end of treatment were responsive to the treatment. Among the 9 patients who had CTCs reduced to below 5 after induction chemotherapy, 2 patients continued to surgery and radiotherapy, 3 patients took only radiotherapy, and 3 patients had only surgery afterward. The last patient had no CTCs detected after induction chemotherapy, and did not take any further treatment. Only one patient with radiotherapy following induction chemotherapy in this group showed PD, while the others showed CR in the end of treatment. For the 12 patients with CTC counts higher than 5 after induction chemotherapy, 3 patients undergoing radiotherapy had CTCs drop from 6–8 to 2, 8 patients undergoing surgery and radiotherapy had CTCs drop from 7–18 to 0–3, and all showed CR or PR of tumor. The last patient who had 25 CTCs after induction therapy died within 2 months after surgery due to tumor progression.
Wang et al used flow cytometry to isolate CTCs in LAHNSCC patients before and after chemoradiotherapy and found that CTC counts were significantly reduced within 2–4 weeks in the therapy-responsive patients.37 CTCs can be used as a marker to monitor patients’ response to treatment. To avoid an overtreatment effect after induction chemotherapy, CTCs can assist the doctors in adjusting the successive treatment plans for patients based on CTC enumeration. This idea was actually adopted by Goodman et al who proposed CTC-based management for radiotherapy after breast-conserving surgery in women with early-stage breast cancer.12 Dynamic monitoring of CTCs during treatment would allow doctors to identify refractory patients and change their treatment regimen accordingly.
Studies in HNSCC and LAHNSCC patients have shown CTCs were associated with patients’ survival outcome. Patients with higher CTC counts would have reduced disease free survival, progression-free survival, or overall survival. To assess whether CTCs could be used as a prognostic marker and CTCs detection at which time point would give the greatest prognostic potency, correlations of CTC enumerations with survival, locoreginal recurrence risk, and distal metastasis were evaluated. Results showed CTC enumeration at baseline had the greatest prognostic power, with the lowest P-values on survival, local recurrence risk, and metastasis analysis. Although there was no statistical significance regarding CTCs with paitents’ survival outcome and distal metastasis risk in our study, a tendency was still observed that patients with higher CTC counts had poor survival and higher chance for distal metastasis. The reason for the lack of statistical significane might have been the small sample size and short follow-up. In accordance with studies in lung, breast, and liver cancers, our results showed that CTC detection at baseline could predict local recurrence of tumor after treatment.11,38,39
Conclusion
The results of this study showed that CytoSorter® system can successfully isolate CTCs in LAHNSCC patients with better sensibility and that CTCs can serve as markers to monitor response to treatment and predict local recurrence of tumor after treatment. However, the relationship of CTCs to long-term therapeutic efficacy, metastasis, and survival still need to be evaluated. More studies on larger patient population with longer follow-up are needed to confirm our findings and to elucidate the clinical value of utilizing CTCs as a diagnostic, therapeutic and prognostic indicator in LAHNSCC patients.
Bullet Point Summary
- CTC enumeration could be used as a tool to differentiate LAHNSCC patients from healthy/patients with benign tumors.
- CytoSorter® system can be used to isolate CTCs in LAHNSCC patients.
- CTCs can be used as a monitoring marker for LAHNSCC patient response to the treatment.
- CTCs can be used as a predicting marker for LAHNSCC local recurrence after treatment.
Acknowledgments
We would like to thank all the members of multidisciplinary team and specialist nursing team of head and neck cancer center of our department. This study was supported by funding as follows, Major Science and Technology Projects of Zhejiang Medical and Health Program (WKJ-ZJ-1712, 2015ZDA007), General research projects of Zhejiang Medical and Health Program (2018KY310, 2017KY029, 2015KYA036).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin. 2019;69(1):7–34. doi:10.3322/caac.21551
2. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424. doi:10.3322/caac.21492
3. Gillison ML, Koch WM, Capone RB, et al. Evidence for a causal association between human papillomavirus and a subset of head and neck cancers. J Natl Cancer Inst. 2000;92(9):709–720. doi:10.1093/jnci/92.9.709
4. Kumar B, Cordell KG, Lee JS, et al. EGFR, p16, HPV titer, bcl-xL and p53, sex, and smoking as indicators of response to therapy and survival in oropharyngeal cancer. J Clin Oncol. 2008;26(19):3128–3137. doi:10.1200/JCO.2007.12.7662
5. Mansouri S, Hesari PM, Naghavi-Al-Hosseini F, Majidzadeh-A K, Farahmand L. The prognostic value of circulating tumor cells in primary breast cancer prior to any systematic therapy: a systematic review. Curr Stem Cell Res Ther. 2019. doi:10.2174/1574888X14666190306103759
6. Wang L, Zhou S, Zhang W, et al. Circulating tumor cells as an independent prognostic factor in advanced colorectal cancer: a retrospective study in 121 patients. Int J Colorectal Dis. 2019;34(4):589–597. doi:10.1007/s00384-018-03223-9
7. Syrigos K, Fiste O, Charpidou A, Grapsa D. Circulating tumor cells count as a predictor of survival in lung cancer. Crit Rev Oncol Hematol. 2018;125:60–68. doi:10.1016/j.critrevonc.2018.03.004
8. Josefsson A, Linder A, Flondell Site D, et al. Circulating tumor cells as a marker for progression-free survival in metastatic castration-naïve prostate cancer. Prostate. 2017;77(8):849–858. doi:10.1002/pros.23325
9. Jiang T, Zhao J, Zhao C, et al. Dynamic monitoring and predictive value of circulating tumor cells in EGFR-mutated advanced non-small-cell lung cancer patients treated with first-line EGFR tyrosine kinase inhibitors. Clin Lung Cancer. 2019;20(2):124–133. doi:10.1016/j.cllc.2018.11.014
10. Wei T, Zhang X, Zhang Q, et al. Vimentin-positive circulating tumor cells as a biomarker for diagnosis and treatment monitoring in patients with pancreatic cancer. Cancer Lett. 2019;452:237–243. doi:10.1016/j.canlet.2019.03.009
11. Sparano J, O’Neill A, Alpaugh K, et al. Association of circulating tumor cells with late recurrence of estrogen receptor-positive breast cancer: a secondary analysis of a randomized clinical trial. JAMA Oncol. 2018;4(12):1700–1706. doi:10.1001/jamaoncol.2018.2574
12. Goodman CR, Seagle BL, Friedl TWP, et al. Association of circulating tumor cell status with benefit of radiotherapy and survival in early-stage breast cancer. JAMA Oncol. 2018;4(8):e180163. doi:10.1001/jamaoncol.2017.4473
13. Yue C, Jiang Y, Li P, et al. Dynamic change of PD-L1 expression on circulating tumor cells in advanced solid tumor patients undergoing PD-1 blockade therapy. Oncoimmunology. 2018;7(7):e1438111. doi:10.1080/2162402X.2018.1490854
14. Shen Z, Wu A, Chen X. Current detection technologies for circulating tumor cells. Chem Soc Rev. 2017;46(8):2038–2056. doi:10.1039/c6cs00803h
15. Nichols AC, Lowes LE, Szeto CC, et al. Detection of circulating tumor cells in advanced head and neck cancer using the cell search system. Head Neck. 2012;34(10):1440–1444. doi:10.1002/hed.21941
16. Bozec A, Ilie M, Dassonville O, et al. Significance of circulating tumor cell detection using the CellSearch system in patients with locally advanced head and neck squamous cell carcinoma. Eur Arch Otorhinolaryngol. 2013;270(10):2745–2749. doi:10.1007/s00405-013-2399-y
17. He S, Li P, He S, et al. Detection of circulating tumour cells with the CellSearch system in patients with advanced-stage head and neck cancer: preliminary results. J Laryngol Otol. 2013;127(8):788–793. doi:10.1017/S0022215113001412
18. Grisanti S, Almici C, Consoli F, et al. Circulating tumor cells in patients with recurrent or metastatic head and neck carcinoma: prognostic and predictive significance. PLoS One. 2014;9(8):e103918. doi:10.1371/journal.pone.0103918
19. Cabel L, Proudhon C, Gortais H, et al. Circulating tumor cells: clinical validity and utility. Int J Clin Oncol. 2017;22(3):421–430. doi:10.1007/s10147-017-1105-2
20. Massagué J, Obenauf AC. Metastatic colonization by circulating tumour cells. Nature. 2016;529(7586):298–306. doi:10.1038/nature17038
21. Micalizzi DS, Maheswaran S, Haber DA. A conduit to metastasis: circulating tumor cell biology. Genes Dev. 2017;31(18):1827–1840. doi:10.1101/gad.305805.117
22. Budd GT, Cristofanilli M, Ellis MJ, et al. Circulating tumor cells versus imaging–predicting overall survival in metastatic breast cancer. Clin Cancer Res. 2006;12(21):6403–6409. doi:10.1158/1078-0432.CCR-05-1769
23. Wu XL, Tu Q, Faure G, Gallet P, Kohler C, Bittencourt MDC. Diagnostic and prognostic value of circulating tumor cells in head and neck squamous cell carcinoma: a systematic review and meta-analysis. Sci Rep. 2016;6:20210. doi:10.1038/srep20210
24. Zou D, Cui D. Advances in isolation and detection of circulating tumor cells based on microfluidics. Cancer Biol Med. 2018;15(4):335–353. doi:10.20892/j.issn.2095-3941.2018.0256
25. McMullen KP, Chalmers JJ, Lang JC, Kumar P, Jatana KR. Circulating tumor cells in head and neck cancer: a review. World J Otorhinolaryngol Head Neck Surg. 2016;2(2):109–116. doi:10.1016/j.wjorl.2016.05.003
26. Tinhofer I, Konschak R, Stromberger C, et al. Detection of circulating tumor cells for prediction of recurrence after adjuvant chemoradiation in locally advanced squamous cell carcinoma of the head and neck. Ann Oncol. 2014;25(10):2042–2047. doi:10.1093/annonc/mdu271
27. Valiathan R, Ashman M, Asthana D. Effects of ageing on the immune system: infants to elderly. Scand J Immunol. 2016;83(4):255–266. doi:10.1111/sji.12413
28. Que Z, Luo B, Zhou Z, et al. Establishment and characterization of a patient-derived circulating lung tumor cell line in vitro and in vivo. Cancer Cell Int. 2019;19:21. doi:10.1186/s12935-019-0735-z
29. Lowes LE, Goodale D, Xia Y, et al. Epithelial to-mesenchymal transition leads to disease-stage differences in circulating tumor cell detection and metastasis in pre-clinical models of prostate cancer. Oncotarget. 2016;7(46):76125–76139. doi:10.18632/oncotarget.12682
30. Satelli A, Mitra A, Brownlee Z, et al. Epithelial-mesenchymal transitioned circulating tumor cells capture for detecting tumor progression. Clin Cancer Res. 2015;21(4):899–906. doi:10.1158/1078-0432.CCR-14-0894
31. Satelli A, Brownlee Z, Mitra A, Meng QH, Li S. Circulating tumor cell enumeration with a combination of epithelial cell adhesion molecule- and cell-surface vimentin-based methods for monitoring breast cancer therapeutic response. Clin Chem. 2015;61(1):259–266. doi:10.1373/clinchem.2014.228122
32. Winter SC, Stephenson SA, Subramaniam SK, et al. Long term survival following the detection of circulating tumour cells in head and neck squamous cell carcinoma. BMC Cancer. 2009;9:424. doi:10.1186/1471-2407-9-424
33. Jatana KR, Lang JC, Chalmers JJ. Identification of circulating tumor cells: a prognostic marker in squamous cell carcinoma of the head and neck? Future Oncol. 2011;7(4):481–484. doi:10.2217/fon.11.19
34. Hristozova T, Konschak R, Stromberger C, et al. The presence of circulating tumor cells (CTCs) correlates with lymph node metastasis in nonresectable squamous cell carcinoma of the head and neck region (SCCHN). Ann Oncol. 2011;22(8):1878–1885. doi:10.1093/annonc/mdr130
35. Buglione M, Grisanti S, Almici C, et al. Circulating tumour cells in locally advanced head and neck cancer: preliminary report about their possible role in predicting response to non-surgical treatment and survival. Eur J Cancer. 2012;48(16):3019–3026. doi:10.1016/j.ejca.2012.05.007
36. Harris AT, Derbyshire S, Wilson J, et al. Circulating and disseminated tumour cells in head and neck cancer. J Laryngol Otol. 2015;129(9):826–830. doi:10.1017/S0022215115001784
37. Wang HM, Wu MH, Chang PH, et al. The change in circulating tumor cells before and during concurrent chemoradiotherapy is associated with survival in patients with locally advanced head and neck cancer. Head Neck. 2019. doi:10.1002/hed.25744
38. Bayarri-Lara C, Ortega FG, de Guevara ACL, et al. Circulating tumor cells identify early recurrence in patients with non-small cell lung cancer undergoing radical resection. PLoS One. 2016;11(2):e0148659. doi:10.1371/journal.pone.0148659
39. Von Felden J, Schulze K, Krech T, et al. Circulating tumor cells as liquid biomarker for high HCC recurrence risk after curative liver resection. Oncotarget. 2017;8(52):89978–89987. doi:10.18632/oncotarget.21208
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.




