Back to Journals » International Journal of Women's Health » Volume 12
Evaluation of the Cesarean Scar Niche In Women With Secondary Infertility Undergoing ICSI Using 2D Sonohysterography Versus 3D Sonohysterography and Setting a Standard Criteria; Alalfy Simple Rules for Scar Assessment by Ultrasound To Prevent Health Problems for Women
Authors Alalfy M , Osman OM , Salama S , Lasheen Y , Soliman M , Fikry M, Ramadan M, Alaa D, Elshemy S , Abdella R
Received 3 July 2020
Accepted for publication 7 September 2020
Published 3 November 2020 Volume 2020:12 Pages 965—974
DOI https://doi.org/10.2147/IJWH.S267691
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Everett Magann
Mahmoud Alalfy,1 Omneya M Osman,2 Sameh Salama,1 Yossra Lasheen,2 Mahmoud Soliman,2 Mohamed Fikry,2 Mohamed Ramadan,2 Doaaa Alaa,2 Shaimaa Elshemy,2 Rana Abdella2
1Reproductive Health and Family Planning Department, National Research Centre, Dokki, Cairo, Egypt; 2Obstetrics and Gynecology Department, Kasr Alainy, Faculty of Medicine, Cairo University, Egypt, Algezeera Hospital, Egypt
Correspondence: Mahmoud Alalfy
Reproductive Health and Family Planning Department, National Research Centre, P.O: 12622, Affiliation ID: 60014618, Dokki, Cairo, Egypt
Tel +20 1002611058
Email [email protected]
Background: Many expressions were used to define the defect that is seen by ultrasound after cesarean section (CS) namely scar defect, niche, isthmocele, uterine pouch or diverticula.
Objective: To compare the accuracy of 2 dimensional sonohysterography (2D SHG) to 3 dimensional sonohysterography (3D SHG) in evaluating cesarean section uterine scar depth (D), base width (BW), width (W) and residual myometrial thickness (RMT) in women with secondary infertility and establishment of a standard criteria; Alalfy simple rules for scar assessment.
Patients and Methods: This was an observational cross-sectional comparative study that was conducted on women who presented with secondary infertility and were candidates for intracytoplasmic sperm injection (ICSI) and giving a history of a previous cesarean section. Assessment of uterine scar in each woman was performed using 2D transvaginal ultrasound with sonohysterography (SHG) followed by 3D transvaginal with SHG with evaluation of niche depth, width, RMT, niche BW and RMT/depth ratio. The study was conducted at Algezeera hospital, Egypt.
Results: The present study revealed that 3D ultrasound with SHG is superior in evaluation of the RMT and niche width prior to ICSI providing better characterization of the scar niche.
Conclusion: Scar niche should be assessed by a combined integrated 2D SHG and 3D SHG scan with the specific geometrical and anatomical considerations, Alalfy simple rules for scar niche assessment that involvemeasurement of niche depth, (Base width) BW, width, RMT and RMT/depth ratio in sagittal plane, RMT in coronal plane / niche width in coronal plane ratio (ratio less than 1 denotes scar weakness with more liability for dehiscence).
Trial Registration: Clinical Trials.gov Id NCT04076904.
Keywords: cesarean, niche, two dimensional, 2D, three-dimensional, 3D, sonohysterography, SHG
Introduction
Cesarean section rate is increasing in recent practice. Women delivered by cesarean section are prone to some complications, one of which is the presence of a uterine niche which is defined as any uterine dimpling 2 mm or more at the cesarean scar site that could be visualized by ultrasound.1,2
Women with uterine niches might complain of pelvic pain, postmenstrual spotting or dysmenorrhea. It could be related to cesarean scar pregnancy or it could be implicated in decreased fertility.3,4
Many expressions were used to define the defect that is seen by ultrasound after cesarean section namely a scar defect, niche, isthmocele, uterine pouch or diverticula.2
The shape of the cesarean scar by ultrasound may be significant, but there is limited proof to correlate the appearance of the scar with the function. There is also concern about the possibility of implantation within the scar and the association with abnormal uterine bleeding and subfertility.5
The aim of this study was to compare the accuracy of 2 dimensional sonohysterography (2D SHG) to 3 dimensional sonohysterography (3D SHG) in evaluating cesarean section uterine scar depth (D), base width (BW), width and residual myometrial thickness (RMT) in women with secondary infertility and establishment of a standard criteria; Alalfy simple rules for scar assessment.
Patients and Methods
This was a prospective observational cross-sectional comparative study that was conducted on women who presented with secondary infertility and were candidates for ICSI and had a history of previous cesarean section. Assessment of uterine scar in each woman was performed using 2D transvaginal ultrasound with sono hysterography (SHG) followed by 3D transvaginal with SHG with evaluation of niche depth, width, RMT, niche BW and RMT/depth ratio. The study was conducted at Algezeera hospital, Egypt from October 2019 till end of February 2020.
Inclusion criteria were women complaining of secondary infertility and history previous one cesarean section. Exclusion criteria included the following: menstruating women or any current abnormal vaginal bleeding, any uterine pathology (like myoma) or previous placenta previa, previous myomectomy, postpartum hemorrhage. This research included 110 women who were recruited from the infertility clinic complaining of secondary infertility who were candidates for ICSI and fulfilling the inclusion and exclusion criteria. They were counseled to participate in the study after explaination of the procedures to be performed was provided, then informed written consent was obtained. All participants were subjected to 2D vaginal examination to exclude uterine pathology followed by 2D and 3D transvaginal ultrasound with SHG for assessment of the uterine scar saline infusion sonography was performed using 20mL of sterile Na Cl 0.9% solution injected through the cervix using a 2 mm sterile disposable catheter. The procedure was done postmenstrually. All cases were performed using a 3D/4D endocavitary probe RIC5-9W-RS GE voluson P 8 and E8 ultrasound system equipped with a 6–12 MHz 3D (General electric voluson, 2014, USA). In every case, where the 2D SHG image and 3D SHG rendered image were taken, measurement of niche depth, width and RMT were made with great precision by the same expert sonographer to avoid inter observer variability. No adverse effects occurred from this maneuver. The presence of uterine niche was recognized as a triangular anechoic filling defect behind the bladder recess, in the lower segment at the classic site at which low-transverse cesarean section was performed. By transvaginal sonography, the scar was recognized in the sagittal plane of the uterus and the following features were evaluated: RMT described as the remaining anterior uterine wall thickness in front of the scar. Width of the base of triangular hypoechoic niche (‘W of B’); described as a distance from the outer and inner parts of the myometrium of the anterior uterine wall. Depth of triangular hypoechoic niche (‘D’); described as a space from the surface of the endometrial- endocervical layer of the anterior wall of uterus to the tip of the hypoechoic triangle.6
Statistical Analysis
Collected data were analyzed and processed using SPSS version 19. Quantitative data was expressed as mean ± SD while Qualitative data were expressed as numbers and percentages. Chi Square, Student t-test and Mann Whitney U-tests were used to test Significance of difference for qualitative variables. A probability value (p-value) <0.05 was considered statistically significant. Data were analyzed and appropriately presented in tables and graphs.
Consent
Informed consent was obtained from the study participants before participation in the research and the study conforms to the provisions of the Declaration of Helsinki.
The study was conducted after taking the ethical committee approval of Algezeera Hospital With Approval number 0062.
Results
The present study was conducted at Algezeera hospital, Egypt from October 2019 till the end of February 2020.
The present study was conducted on women who presented with secondary infertility and a history of previous cesarean delivery who were candidates for ICSI. Before starting the treatment protocol, evaluation of the integrity of the cesarean scar as demonstrated in Figures 1 and 2 was carried out, with comparison of the ability of 2D and 3D SHG in its evaluation and assessment of possible scar dehiscence and if it needs intervention before ICSI aiming at a better reproductive outcome.
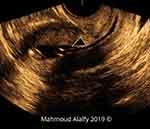 |
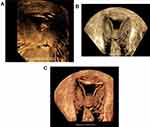
Figure 1 2D SHG image in a sagittal section of the uterus showing the triangular cesarean scar niche. |
 |
Figure 2 2D SHG image in a sagittal section of the uterus showing the triangular cesarean scar niche. |
One hundred ten women were recruited to the study, with the mean age was 28.2 years and mean BMI was 23.94. Age, BMI of studied women are shown in Table 1.
 |
Table 1 Demographic Data of the Women Included in the Study |
The difference in age, BMI between the two groups were statistically insignificant.
In the present study, the comparison between 2D SHG and 3D SHG in evaluation of CS niche BW shows a statistically significant P value (<0.001) for intraclass coefficient (0.826) and Cronbach’s Alpha (0.905) as shown in Table 2. The niche BW is demonstrated in figure 4 and in Figure 7.
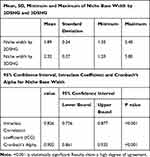 |
Table 2 Degree of Agreement Between 2D SHG and 3D SHG in Detection of Niche BW |
In the present study, the comparison between 2DSHG and 3D SHG in evaluation of the RMT in front of the scar revealed a statistically significant P value (<0.001) with intraclass correlation coefficient (0.423) and Cronbach’s Alpha (0.594) (as shown in Table 4), the RMT is demonstrated in Figures 8 and 9.
 |
Table 3 Degree of Agreement Between 2DSHG and 3D SHG in Detection of Niche Depth |
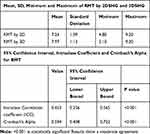 |
Table 4 Degree of Agreement Between 2D SHG and 3D SHG in Detection of RMT |
Also in the current study, the comparison between 2D SHG and 3D SHG in evaluation of CS niche depth shows a statistically significant P value (<0.001) with intraclass correlation coefficient (0.971) and Cronbach’s Alpha (0.985) with high values as shown in Table 3, the niche depth is demonstrated in Figures 3 and 6.
 |
Figure 3 2DSHG image in a sagittal section of the uterus showing the cesarean scar niche depth. |
In the present study, the comparison between 2D SHG and 3D SHG in evaluation of CS niche width shows a statistically significant P value for intraclass coefficient and cronbach s Alpha but with low values 0.251 and 0.402 respectively as shown in Table 5, the niche base width is demonstrated in Figures 4 while niche width is shown in figures 5 A and 5 B and 5C . figure 5C demonstrates 3D HD live renderin in the
 |
Table 5 Degree of Agreement Between 2D SHG and 3D SHG in Measurement of Niche Width |
 |
Figure 4 2D SHG image in a sagittal section of the uterus showing the cesarean scar niche BW. |
 |
Figure 6 3D SHG image of the uterus showing the cesarean scar niche depth. |
 |
Figure 7 3D SHG image of the uterus showing the cesarean scar niche BW. |
 |
Figure 8 2D SHG image of a sagittal section of the uterus showing the RMT. |
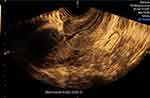 |
Figure 9 3D SHG image in the sagittal cut that demonstrates the RMT with adhesions in front of the niche. |
2D SHG in the sagittal plane has a high degree of agreement with 3D SHG in the sagittal cut of the 3D volume in evaluation of niche depth and BW.
2D SHG and 3D SHG in the sagittal plane have moderate agreement in evaluation of RMT.
Discussion
Assessment of the cesarean scar integrity and prediction of subsequent scar pregnancy complications as uterine rupture or implantation on a scar is feasible using ultrasound. The evaluation of various ultrasonographic parameters in demonstrating scar weakness is available nowadays.6–10
Isthmocele can cause postmenstrual bleeding, dysmenorrhea or pelvic pain, also in isthmocele, the presence of retained menstrual blood may result in a site for chronic inflammation impairing fertility.11
Ultrasonographic evaluation of the lower uterine segment (LUS) thickness has been the method employed in prediction of cesarean scar rupture at various gestational weeks according to many recent studies.6–8 Ultrasonography provides a non-invasive way to scan the uterus and scar.12
A recent meta-analysis was conducted and recommended the implementation of the LUS thickness for prediction of uterine rupture during VBAC.13,14
Naji et al (2012) revealed that rupture uterine scar is associated with lesser RMT and emphasized the necessity to describe the scar exactly with great precision and to discover their clinical importance in subsequent pregnancies. They suggested that the dimensions shown in their research which were the RMT, niche depth and width would be a suitable method, which were also the parameters we adopted for investigation in our current study but we added the BW and D/RMT ratio.5 They concluded that employing a uniform technique to describe the shape of scars, could make future research on the complications related to them reliable.4
Previous studies reported that transvaginal US is better than transabdominal US in scar assessment.15,16
In the present study, the comparison between 2D and 3D ultrasound in evaluation of CS niche BW showed a high degree of agreement as demonstrated in Table 2.
Another previously studied parameter of clinical importance in prediction of CS scar rupture in the upcoming gestation was the RMT/D ratio where the larger the depth of the niche (D) and the lesser the thickness of the myometrium, the more likely risk of dehiscence of cesarean section scars as seen in Figure 10 (shows the defective niche) and in Figure 11 (RMT/D ratio less than 1).17,18
 |
Figure 10 2D SHG image showing a sagittal section in 3rd degree RVF uterus with a markedly defective cesarean scar niche. |
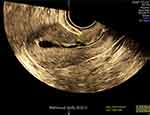 |
Figure 11 3D SHG image in the sagittal cut that demonstrates RMT/Depth ratio (less than 1) with a thin RMT with adhesions in front of the niche. |
The acceptable analysis of the role of the D/RMT ratio in expecting dehiscence of scar is that the D plus RMT form the whole thickness of the anterior wall of uterus at the scar site.10
Figure 12: Shows 3 D HD live Rendering image illustrating the niche in coronal view.
Also in the current study, the comparison between 2D SHG and 3D SHG in evaluation of CS niche depth shows a high degree of agreement with a statistically significant P values for intraclass coefficient and cronbach s Alpha with high values as shown in Table 3.
A previous study made by Ejub Basic concluded that ultrasound assessment of the quality of the scar has practical use in the decision on the mode of delivery in women who delivered by cesarean section.19
Moreover, in the present study, the comparison between 2DSHG and 3D SHG in evaluation of RMT in front of the scar shows a moderate degree of agreement reflecting the higher accuracy of 3D SHG in evaluation of the scar niche (as shown in Table 4).
So, evaluation of the RMT/depth ratio is better to be evaluated by 3DSHG.
A previous study that reviewed the capability of imaging methods to expect incomplete healing of uterine scars on ladies who underwent previous cesarean section, assessed by hysterography, SHG, or transvaginal ultrasound.20
In the present study, the comparison between 2D SHG and 3D SHG in evaluation of CS niche width showed a statistically significant P value for intraclass coefficient and cronbach s Alpha but with low values denoting a mild degree of agreement as shown in Table 5.
In a meta analysis that involved 21 research studies, the magnitudes of supposed scar dehiscence diagnosed by hysterography, SHG, and TVU were 58% (33 to 70), 59% (58 to 85), and 37% (20 to 65), respectively.20
In the current study, we found that there is a high agreement between 2D and 3D SHG in describing the niche BW and depth while 3D SHG has the upper hand in giving a precise data about the RMT which is so important in management and in predicting outcome for future pregnancies.
A previous study that described 2D evaluation of a scar, reported that the hypoechoic space with a triangular shape, is denoting an interruption of the myometrium at the site of the previous cesarean scar which is better to be evaluated at postmenstrual scan and they defined the niche as a myometrial defect with a depth of at least two mm. A large niche is defined as a defect with a depth of at least more than fifty percent of the anterior myometrial wall and ≤ 2.5 mm when assessed by SHG).9,21
In the current study, 3D SHG in coronal plane is the best at evaluating niche width with a mild agreement of 2D SHG with 3D SHG.
Using 3D ultrasound, many niche parameters, as depth (both perpendicular to niche base and maximal depth), maximal width, width at niche base and RMT can be measured with a high level of accuracy, in particular if measured in the longitudinal plane.22
Transvaginal 3D SHG could be a routine non invasive outpatient method for evaluation of uterine cavity lesions as polyps, intra cavitary myomas, intrauterine synechia and congenital uterine abnormalities as septum in women with infertility, also in addition to conventional transvaginal 3D US which is important in evaluation of women with gynecological problems as bleeding, ovarian cysts or tubal lesions as hydrosalpinx.23–25
SHG is recommended to be used in combination with transvaginal US as an initial investigation of choice in patients presenting with abnormal uterine bleeding.26
The error in measurements of the niche by 2D US could be eliminated by off line analysis of the 3D volume image and getting the optimal slice before measurement and in case of RVF uterus.
Hysteroscopic repair of isthmocele is safe when RMT is more than 3 mm.27
While if RMT is smaller than 3 mm, the laparoscopic repair is recommended to avoid perforation of the uterus and injury to the bladder, especially in women with infertility.28
According to data in the literature, there is no solid conclusion about the best recommended time for subsequent pregnancy and about the mode of delivery after isthmocele repair. Considering the lack of available evidence and the need for a clear indication to advise women, the Consensus Statement From the Global Congress on Hysteroscopy Scientific Committee recommends that women should postpone pregnancy for three months post isthmocele repair with a prophylactic delivery by planned CS no later than 38 weeks of pregnancy to decrease the risk of uterine rupture.29,30
Methods for repair of isthmocele consist of resection of the upper and lower borders of the defect and ablation of the isthmic endometrium, also dissecting the upper border of the isthmocele only is accepted.31,32
Laganà et al assumed that although 3D SHG has a high diagnostic accurateness and less invasiveness, but they advised that it could not replace hysteroscopy in endocavitary lesions diagnosis, and might be considered as a good way for screening to select women to do hysteroscopy for verification of the lesions.33
According to the literature, the studies published describing the uterine niche and the role of both 2D and 3D in diagnosis are not sufficient for having a solid comparison, and there is no available data in the literature comparing both 2D and 3D SHG.
Alonso Pacheco et al carried out a study to assess the T-shaped uterus through 3D US and found 3 different types of T-shaped uterus; T-shaped (thick lateral walls and normal fundus (with no septum or subseptate uterus) and interostial distance; the Y-shaped uterus (thick lateral walls, fundal septum or subseptum, and reduced interostial distance) and the I-shaped uterus (very thick lateral walls (even above the isthmus) and severe reduction of the interostial distance (tubular appearance of the whole uterus).34
Alonso Pacheco et al studied women who have unexplained infertility and T-shaped uterus diagnosed by 3D US then a hysteroscopic metroplasty was made to they found that women with primary infertility with T-shaped uterus, hysteroscopic metroplasty is thought to be useful to increase reproductive outcomes.35
To the best of our knowledge, our study design is unique regarding the evaluation of the cesarean scar comparing 2D SHG versus 3D SHG in the pre-pregnancy period in women with secondary infertility where most conducted studies evaluated the scar during pregnancy for fear of subsequent dehiscence or rupture in the ongoing pregnancy. We were concerned with the implication of the scar niche in secondary subfertility and in future pregnancy.
Conclusion
The present study documented the comparable high accuracy of 2DSHG and 3D SHG in evaluation of CS niche regarding the depth and BW of CS niche.
However, 3D SHG is superior in evaluation of the RMT and niche width prior to ICSI providing better characterization of the scar niche.
Scar niche should be assessed by a combined integrated 2D SHG and 3D SHG scan with a new specific geometrical and anatomical considerations; Alalfy simple rules for scar niche assessment that involve measurement of niche depth, width, RMT, (Base width) BW, a new parameter and RMT/depth ratio in sagittal section (ratio less than 1 denotes scar weakness with more liability for dehiscence Also a new parameter, RMT in coronal plane / niche Width in coronal plane ratio (ratio less than 1 denotes scar weakness with more liablity for dehiscence).
From the geometrical point of view, the cesarean scar niche has a specific shape that can be evaluated using a combined technique with applying both 2D SHG and adding the 3D SHG, each one of these techniques has the ability to delineate a specific parameter that assess the niche (Niche depth, width, width of the base of niche and RMT).
Assessment of the scar should be considered in women with secondary infertility and in the pre-ICSI work up with previous cesarean scar. An abnormal markedly dehiscent scar could be implicated as a cause of subfertility or subsequent cesarean scar ectopic pregnancy, morbidly adherent placenta or uterine rupture which may need surgical repair prior to ICSI or pregnancy which could be subject to future research, also the usefulness of our recommendations needs to be studied in future research.
Abbreviations
CS, cesarean section; SHG, Sonohysterography; RMT, Residual myometrial thickness; D, Depth; W, Width; BW, Base width; ICSI, Intracytoplasmic sperm injection; 2D SHG, 2 dimensional sonohysterography; 3D SHG, 3 dimensional sonohsterography.
Data Availability
The study was registered in clinicaltrials. gov with identification Clinical trials. gov ID: NCT04076904.
The data that support the results reported in the manuscript are present but will not be shared because it has no additive information to the research but we are ready to reveal it when necessary.
Data Sharing Statement
The authors do not intend to share individual identified participant data.
There are no other study-related documents will be made available
Corresponding author Mahmoud Alalfy, Orcid ID: 0000-0002-8429-6376
Acknowledgment
We would like to thank all staff members who helped us in completing this research.
Author Contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Funding
There is no source of funding.
Disclosure
All authors declare that they have no financial or personal relationships with other people or organizations that could inappropriately influence (bias) this work.
References
1. Betran AP, Torloni MR, Zhang JJ, Gülmezoglu AM. WHO working group on caesarean section WHO statement on caesarean section rates. BJOG. 2015;123(5):667–670. doi:10.1111/1471-0528.13526.
2. Naji O, Abdallah Y, et al. Cesarean birth: surgical techniques glob libr women’s med. 2010. doi:10.3843/GLOWM.10133.
3. Zhang J, Landy HJ, Branch DW, et al. Consortium on safe labor. Contemporary patterns of spontaneous labor with normal neonatal outcomes. Obstet Gynecol. 2010;116(6):1281–1287. doi:10.1097/AOG.0b013e3181fdef6e
4. ACOG/SMFM Obstetric Care Consensus. Safe prevention of the primary cesarean delivery. Obstet Gynecol. 2014;123:693–711. doi:10.1097/01.AOG.0000444441.04111.1d
5. Naji O. Standardized approach for imaging and measuring Cesarean section scars using ultrasonography. Ultrasound Obstet Gynecol. 2012;39:252–259.
6. Bastani P, Pourabolghasem S, Abbasalizadeh F, Motvalli L. Comparison of neonatal and maternal outcomes associated with head-pushing and head-pulling methods for impacted fetal head extraction during cesarean delivery. Int J Gynaecol Obstet. 2012;118(1):1. doi:10.1016/j.ijgo.2012.03.005
7. Bennich G, Rudnicki M, Wilken-Jensen C, Lousen T, Lassen PD, Wøjdemann K. Impact of adding a second layer to a single unlocked closure of a Cesarean uterine incision: randomized controlled trial. Ultrasound Obstet Gynecol. 2016;47:417–422. doi:10.1002/uog.15792
8. Bij de Vaate AJ, Brolmann HA, van der Voet LF. Ultrasound evaluation of the Cesarean scar: relation between a niche and postmenstrual spotting. Ultrasound Obstet Gynecol. 2011;37:93–99. doi:10.1002/uog.8864
9. Bij de Vaate AJ, van Der, Voet LF, Naji O. Prevalence, potential risk factors for development and symptoms related to the presence of uterine niches following Cesarean section: systematic review. Ultrasound Obstet Gynecol. 2014;43:372–382. doi:10.1002/uog.13199
10. Dahlke JD, Mendez-Figueroa H, Rouse DJ, Berghella V, Baxter K. Evidence-based surgery for cesarean delivery: an updated systematic review. Am J Obstet Gynecol. 2013;209(4):294. doi:10.1016/j.ajog.2013.02.043
11. Tower AM, Frishman GN. Cesarean scar defects: an underrecognized cause of abnormal uterine bleeding and other gynecologic complications. J Minim Invasive Gynecol. 2013;20(05):562–572. doi:10.1016/j.jmig.2013.03.008
12. Vikhareva Osser O, Valentin L. Clinical importance of appearance of cesarean hysterotomy scar at transvaginal ultrasonography in nonpregnant women. Obstet Gynecol. 2011;117:525–532. doi:10.1097/AOG.0b013e318209abf0
13. DHS program—demographic and health surveys: USAID;
14. Gibbons L, Belizan JM, Lauer JA, Betran AP, Merialdi M, Althabe F. Inequities in the use of cesarean section deliveries in the world. Am J Obstet Gynecol. 2012;206(4):331e119. doi:10.1016/j.ajog.2012.02.026.
15. Gurol-Urganci I, Bou Antoun S, Lim CP, Cromwell DA, Mahmood TA, Templeton A. Impact of Cesarean section on subsequent fertility: a systematic review and meta-analysis. Hum Reprod. 2013;28:1943–1952. doi:10.1093/humrep/det130
16. Gyhagen M, Bullarbo M, Nielsen TF, Milsom I. The prevalence of urinary incontinence 20 years after childbirth: a national cohort study in singleton primiparae after vaginal or cesarean delivery. BJOG. 2013;120(2):144. doi:10.1111/j.1471-0528.2012.03301.x
17. Kok N, Wiersma IC, Opmeer BC, de Graaf IM, B W M, Pajkrt E. The ability of sonographic measurement of the lower uterine segment thickness to predict uterine rupture during a trial of labour in women with a previous Cesarean section: a meta-analysis. Ultrasound Obstet Gynecol. 2013;42:132–139. doi:10.1002/uog.12479
18. Miller ES, Hahn K, Grobman WA. Society for maternal-fetal medicine health policy committee. Consequences of a primary elective cesarean delivery across the reproductive life. Obstet Gynecol. 2013;121(4):789. doi:10.1097/AOG.0b013e3182878b43
19. Ejub B, Vesna B-C, Hadzo K. Ultrasound evaluation of uterine scar after cesarean section. Acta Inform Med. 2012;20(3):149–153. doi:10.5455/aim.2012.20.149-153
20. Jastrow N, Chaillet N, Roberge S, Morency AM, Lacasse Y, Bujold E. Sonographic lower segment thickness and risk of uterine scar defect: a systematic review. Journal of Obstetrics and Gynecology Canada. 2010;32(4):321–327
21. Vervoort AJ, Uittenbogaard LB, Hehenkamp WJ, Brölmann HA, Mol BW. Why do niches develop in Caesarean uterine scars? Hypotheses on the aetiology of niche development. Hum Reprod. 2015;30:2695–2702.
22. Marjolein AJ, Bij de Vaate IH, Linskens Lucet F. Huirne Reproducibility of three - dimensional ultrasound for the measurement of a niche in a cesarean scar and assessment of its shape.Eur J Obstet Gynecol Reprod Biol. 2015;188:39–44. doi:10.1016/j.ejogrb.2015.02.018
23. Salim R, Jurkovic D. Assessing congenital uterine anomalies: the role of threedimensional ultrasonography. Best Pract Res Clin Obstet Gynaecol. 2004;18(1):29–36. doi:10.1016/j.bpobgyn.2003.09.001
24. Saravelos SH, Cocksedge KA, Li TC. Prevalence and diagnosis of congenital uterine anomalies in women with reproductive failure: a critical appraisal. Hum Reprod Update. 2008;14(5):415–429. doi:10.1093/humupd/dmn018
25. Pundir J, Toukhy TE. Uterine cavity assessment prior to IVF Women’s Health (Lond). 2010;6(6):841–848 doi:10.2217/whe.10.61.PMID:21118042.
26. Mohamed SM, Anyona AA, Onyambu CK, Muriithi IM, Mutala TM. Utility of sonohysterography in evaluation of patients with abnormal uterine bleeding. Obstet Gynecol Rep. 2018;2(2):1–7. doi:10.15761/OGR.1000127
27. Di Spiezio Sardo A, Zizolfi B, Calagna G. Hysteroscopic isthmoplasty: step-by-step technique. J Minim Invasive Gynecol. 2018;25:338–339. doi:10.1016/j.jmig.2017.09.002
28. Donnez O, Donnez J, Orellana R, Dolmans MM. Gynecological and obstetrical outcomes after laparoscopic repair of a cesarean scar defect in a series of 38 women. Fertil Steril. 2017;107:289–296. doi:10.1016/j.fertnstert.2016.09.033
29. van der Voet LF, Vervoort AJ, Veersema S, BijdeVaate AJ, Brölmann HA, Huirne JA. Minimally invasive therapy for gynaecological symptoms related to a niche in the caesarean scar: a systematic review. BJOG. 2014;121:145–156. doi:10.1111/1471-0528.12537
30. Laganà AS, Pacheco LA, Tinelli A, et al. Optimal timing and recommended route of delivery after hysteroscopic management of isthmocele? A consensus statement from the global congress on hysteroscopy scientific committee. J Minim Invasive Gynecol. 2018;25(4):558. doi:doi:10.1016/j.jmig.2018.01.018
31. Laganà AS, Alonso Pacheco L, Tinelli A, Haimovich S, Carugno J, Ghezzi F. Authors’ Reply. J Minim Invasive Gynecol. 2018;25(6):1112–1113. doi:doi:10.1016/j.jmig.2018.04.009
32. Raimondo G, Grifone G, Raimondo D, Seracchioli R, Scambia G, Masciullo V. Hysteroscopic treatment of symptomatic cesareaninduced isthmocele: a prospective study. J Minim Invasive Gynecol. 2015;22:297–301. doi:10.1016/j.jmig.2014.09.011
33. Laganà AS, Ciancimino L, Mancuso A, Chiofalo B, Rizzo P, Triolo O. 3D sonohysterography vs hysteroscopy: a cross-sectional study for the evaluation of endouterine diseases. Arch Gynecol Obstet. 2014;290(6):1173–1178. doi:doi:10.1007/s00404-014-3366-6
34. Alonso Pacheco L, Laganà AS, Ghezzi F, Haimovich S, Azumendi Gómez P, Carugno J. Subtypes of T-shaped uterus. Fertil Steril. 2019;112(2):399–400. doi:doi:10.1016/j.fertnstert.2019.04.020
35. Alonso Pacheco L, Laganà AS, Garzon S, Pérez Garrido A, Flores Gornés A, Ghezzi F. Hysteroscopic outpatient metroplasty for T-shaped uterus in women with reproductive failure: results from a large prospective cohort study. Eur J Obstet Gynecol Reprod Biol. 2019;243:173–178. doi:doi:10.1016/j.ejogrb.2019.09.023
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.