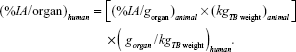Back to Journals » International Journal of Nanomedicine » Volume 10 » Issue 1
Evaluation of 188Re-labeled PEGylated nanoliposome as a radionuclide therapeutic agent in an orthotopic glioma-bearing rat model
Authors Huang F, Lee T, Chang C, Chen L, Hsu W, Chang C, Lo J
Received 17 October 2014
Accepted for publication 26 November 2014
Published 9 January 2015 Volume 2015:10(1) Pages 463—473
DOI https://doi.org/10.2147/IJN.S75955
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Thomas Webster
Feng-Yun J Huang,1 Te-Wei Lee,2 Chih-Hsien Chang,2 Liang-Cheng Chen,2 Wei-Hsin Hsu,2 Chien-Wen Chang,1 Jem-Mau Lo1
1Department of Biomedical Engineering and Environmental Sciences, National Tsing Hua University, Hsinchu, Taiwan; 2Institute of Nuclear Energy Research, Longtan, Taiwan
Purpose: In this study, the 188Re-labeled PEGylated nanoliposome (188Re-liposome) was prepared and evaluated as a therapeutic agent for glioma.
Materials and methods: The reporter cell line, F98luc was prepared via Lentivector expression kit system and used to set up the orthotopic glioma-bearing rat model for non-invasive bioluminescent imaging. The maximum tolerated dose applicable in Fischer344 rats was explored via body weight monitoring of the rats after single intravenous injection of 188Re-liposome with varying dosages before the treatment study. The OLINDA/EXM 1.1 software was utilized for estimating the radiation dosimetry. To assess the therapeutic efficacy, tumor-bearing rats were intravenously administered 188Re-liposome or normal saline followed by monitoring of the tumor growth and animal survival time. In addition, the histopathological examinations of tumors were conducted on the 188Re-liposome-treated rats.
Results: By using bioluminescent imaging, the well-established reporter cell line (F98luc) showed a high relationship between cell number and its bioluminescent intensity (R2=0.99) in vitro; furthermore, it could also provide clear tumor imaging for monitoring tumor growth in vivo. The maximum tolerated dose of 188Re-liposome in Fischer344 rats was estimated to be 333 MBq. According to the dosimetry results, higher equivalent doses were observed in spleen and kidneys while very less were in normal brain, red marrow, and thyroid. For therapeutic efficacy study, the progression of tumor growth in terms of tumor volume and/or tumor weight was significantly slower for the 188Re-liposome-treated group than the control group (P<0.05). As a result, the lifespan of glioma-bearing rats treated with 188Re-liposome was prolonged 10.67% compared to the control group.
Conclusion: The radiotherapeutic evaluation by dosimetry and survival studies have demonstrated that passive targeting 188Re-liposome via systemic administration can significantly prolong the lifespan of orthotopic glioma-bearing rats while maintaining reasonable systemic radiation safety. Therefore, 188Re-liposome could be a potential therapeutic agent for glioblastoma multiforme treatment.
Keywords: 188Re, liposome, radionuclide therapy, bioluminescent imaging, glioma
Introduction
Glioma is the most common primary tumor in the central nervous system. Glioblastoma multiforme (GBM), a World Health Organization (WHO) grade IV malignant glioma, accounts for more than half of total primary gliomas, with an average survival time ranging from 12 to 15 months.1 High morbidity and mortality make glioma the fourth most serious malignant cancer.2 The current standard treatment for GBM is essential surgical resection followed by a combination of radiation therapy plus adjuvant chemotherapy with temozolomide.3,4 Despite tremendous efforts on the treatments of GBM, prognoses remain poor, with an increase of only a few months in average survival time and the 5-year survival rate less than 10%.5
Aside from surgery and chemotherapy, radiation therapy remains an important means of cancer treatment. Approximately 50% of all cancer patients are treated with radiation therapy with varying modalities including external beam, brachytherapy, and radionuclide therapy (RNT).6 In the past decade, RNT has been increasingly adopted as a therapeutic radiopharmaceutical for treatment of cancers. In particular, radiation via RNT can be deposited to the lesion at the cellular level whereas conventional external beam therapy is limited to a gross anatomical level.7
A wide variety of nanoparticles have been developed as drug carriers for treatment of cancers. Early clinical trials of some drug-incorporated nanoparticles have shown their efficacious cancer treatments owing to the passive accumulation of nanoparticles in the tumors by enhanced permeability and retention (EPR) effect and reducing the drug side effects to the system.8 In recent years, radiolabeled nanoparticles such as 188Re-lipid nanoparticles and 186Re-liposome nanoparticles have been introduced for treatment of GBM via convection-enhanced delivery technique.9–12 These studies demonstrated that the survival of tumor-bearing rats was significantly prolonged with therapeutic radiopharmaceutical treatment. Although the convection-enhanced delivery technique affords apparent benefits for GBM treatment, it might still cause certain drawbacks in clinical practice.13,14 Moreover, the complicated drug delivery technique may not be ready and convenient for routine clinical uses. Therefore, development of a convenient and effective strategy for drug delivery is demanding for conducting RNT on GBM.
Systemic administration of therapeutic radiopharmaceuticals is an alternative but more feasible option of drug delivery for clinical RNT. Most recently, a new drug under investigation, 188Re-labeled PEGylated nanoliposome (188Re-liposome) has been successfully validated with a series of translational studies for treatment of colon cancer.15–20 Currently, it has also been approved by Department of Health, Taiwan for further human clinical trial for treatment of metastatic colon cancer (Phase I). According to our previous work, 188Re-liposome had been explored in orthotopic glioma-bearing rat model for diagnostic evaluation where systemic administration was employed for delivering the radiolabeled liposome nanoparticles.21
In this study, we aimed to evaluate the therapeutic efficacy of 188Re-liposome for GBM in orthotopic glioma-bearing rat model where single intravenous injection of 188Re-liposome was employed. 188Re, a VII B family element in the periodic table, has a similar physicochemical property to 99mTc. It exhibits a physical half-life of 16.9 hours. Specifically, it emits not only a diagnostic gamma ray (Eγ=155 KeV) but also a therapeutic beta radiation (Eβmax=2.1 MeV).22 In order to prove our concept for treatment of GBM tumor by 188Re-liposome via single intravenous injection, the related serial experiments including establishment of luciferase expressing reporter cell (F98luc), maximum tolerated dose (MTD), bioluminescent imaging (BLI), radiation dosimetry (OLINDA/EXM), therapeutic efficacy, and so forth, were performed in this study.
Materials and methods
Materials
Lentivector expression kit system was obtained from System Biosciences (SBI) (Mountain View, CA, USA) and XenoLight D-Luciferin potassium salt was from PerkinElmer Inc.® (Waltham, MA, USA). Distearoylphosphatidylcholine (DSPC), cholesterol and polyethylene glycol (MW ~2 kDa) – derived distearoylphosphatidylethanolamine (DSPE-PEG2000) were purchased from Genzyme (Cambridge, MA, USA). Cell culture materials were purchased from GIBCO™ BRL (Grand Island, NY, USA). PD-10 column was purchased from GE Healthcare Bio-Sciences AB (Uppsala, Sweden). N,N-bis(2-mercaptoethyl)-N′,N′-diethylethylenediamine (BMEDA) was from ABX (Radeberg, Germany). Hematoxylin and eosin (H and E) staining agents were from Sigma-Aldrich Co. (St Louis, MO, USA). Zoletil® 50 anesthetic was from Virbac (Virbac Taiwan Co., Ltd., Taiwan). All other chemicals were from Merck (Darmstadt, Germany).
Cell culture and cell growth curve
The F98 glioma cell line was kindly provided by Dr Jeng-Jong Hwang of National Yang Ming University, Taipei, Taiwan, which was originally produced by Dr Rolf F Barth of Ohio State University, Cincinnati, Ohio, USA.23 This cell line was transfected with the luciferase gene to express luciferase as reporter (F98luc) through Lentivector expression kit and the F98luc cells were sorted via green fluorescent protein (GFP) by a fluorescence-activated cell sorter (BD FACSAria™ III, San Jose, CA, USA). The F98luc cells were cultured in Dulbecco’s Modified Eagle’s Medium (DMEM) supplemented with 10% fetal bovine serum, 100 units/mL penicillin, and 100 μM/mL streptomycin. Cells were incubated at 37°C in a humidified environment with 5% CO2.
For cell growth curve study, the F98luc cells were seeded in a 12-well plate with 1×106 cells in each well. The cells were harvested after incubation for 16, 24, 48, 72, 96, and 120 hours, respectively and the numbers of cells at each well were counted by a hemacytometer. The cell doubling time (Td) was evaluated by the following formula:24
Td = (T–To)×ln2/(lnN–lnN0). | (1) |
Fischer344/F98luc glioma model
All animal studies were approved by the Institutional Animal Care and Use Committee at the Institute of Nuclear Energy Research, Taoyuan, Taiwan. The Fischer344/F98luc orthotopic glioma bearing rat model was established according to the procedure as previous reported by Mathieu et al25 but with modification. The normal Fischer344 rats were supplied by the National Laboratory Animal Center, Taipei, Taiwan and were housed in a controlled environment with food and water provided ad libitum.
The rats (male, 12–13 weeks old) were anesthetized with isoflurane® and then administered atropine sulfate (0.1 mg/kg) via subcutaneous injection; subsequently, the rats were deeply anesthetized by intraperitoneal injection of Zoletil® 50 and Balanzine 2% mixture at a 5:2 volume ratio (0.1 mL/100 g rat body weight). After anesthesia, the hair on the rat’s head was removed from the operative field. Then, the rats were immobilized by a stereotactic frame (Stoelting®, Wood Dale, IL, USA). A 2 cm linear incision was carefully made and the immobilized for the following surgery. After removing the periosteum, a 1 mm diameter hole was created with a high-speed drill in the skull of the right brain (located at 3 mm lateral to midline and 5 mm anterior to lambda) and the dura carefully pricked with sharp tweezers. For implantation, the F98luc cells were harvested and re-suspended in Hank’s balanced salt solution plating on the ice before use. The 1×105 cells in 10 μL medium were inoculated into the brain (a depth of 5 mm from the skull bone) using a 100 μL Hamilton® syringe and 27–1/2 gauge needle through nanoliter syringe pump (KDS 310 plus; Holliston, MA, USA) with the injection rate of 3 μL/min. After seeding, the needle was retained for 2 minutes and then drawn out carefully and slowly. Finally, paraffin was used to fill the surgical hole and the incision was sutured. The rats were observed closely until completely awake.
BLI
The BLI was performed by IVIS™ Imaging System 100 Series (Xenogen®, USA). The imaging protocol was in accordance with that previously described, but with some modifications.26,27 For in vitro imaging, F98luc cells were diluted from 100,000 to 195 cells by culture medium and each group of cells (in 100 μL) was carefully loaded into a black 96-well plate. Then, the D-Luciferin substrate (in medium) was added to each well at a concentration of 150 μg/mL for 5 minutes incubation and the cells-loaded plate was imaged continuously for 1 minute with the IVIS™ system. For in vivo imaging, the tumor bearing rats were administered D-Luciferin substrate in PBS (75 mg/kg) via intraperitoneal injection. At 15 minutes post-injection, the rats were anesthetized with 3% isoflurane and then imaged continuously for 10 minutes with the IVIS™ system. To monitor Fischer344/F98luc tumor growth curve in a relationship between bioluminescent intensity and time, the tumor bearing rats were imaged from Day 3 to Day 13 post-inoculation, respectively. After Day 13 imaging, the rats were sacrificed by CO2 euthanasia and the brain tumor was dissected for histopathological examination (H and E staining). Tumoral luminescence intensity (photon/s) was quantified by conducting a region of interest assay using the Living Image software (Xenogen®).
Preparation of neutral PEGylated nanoliposomes
The lipid film hydration–extrusion combining repeated freeze–thawing method was used to prepare neutral PEGylated nanoliposomes as previously reported.28 Briefly, the mixture of DSPC: cholesterol: DSPE-PEG2000 at the molar ratio of 3:2:0.3 was dissolved in chloroform followed by removing the solvent with rotary evaporation. Then the 250 mM ammonium sulfate at pH 5.5 was used to rehydrate the dry lipid film at 60°C. After rehydration, the liposomes were extruded three times through polycarbonate membrane filters with varying pore sizes at 0.4, 0.2, 0.1, 0.05, and 0.03 μm, respectively (Sterlitech Corp., Kent, WA, USA) by a high-pressure extruder (LIPEX™, Northern Lipids Inc., Burnaby, BC, Canada). Finally, normal saline was introduced to replace extraliposomal buffer via elution through a Sephadex G-50 column (Pharmacia, Uppsala, Sweden). The size and the zeta potential of the nanoliposomes were analyzed by a dynamic light scattering analyzer (N4 plus; Beckman Coulter Inc., CA, USA). Phospholipid concentration was measured via phosphorus assay with ultraviolet-visible spectrophotometry at λ=830 nm (JascoV-530, Tokyo, Japan).29
Preparation of 188Re-liposome
188Re-liposome was prepared as described previously.15,21,30 The carrier free 188Re-perrhenate solution (Na188ReO4; 3.7–7.4 GBq) was produced from an in-house alumina-based 188W/188Re generator with the original 188W radionuclide purchased from Oak Ridge National Laboratory (Oak Ridge, TN, USA).
For 188Re-liposome preparation, the preformed PEGylated nanoliposomes and 188Re-BMEDA complex were added together into a vial at a volume ratio of 1:1 (1 mL:1 mL) and capped with rubber septum and aluminum foil. Then the solution mixture was incubated in a 60°C water bath shaking at 100 rpm for 30 minutes. After reaction, the preparation vial was taken out and cooled down at room temperature for 10 minutes. Subsequently, a PD-10 size exclusion column was utilized to purify 188Re-liposome from 188Re-BMEDA complex and free rhenium-188 (Na188ReO4) via elution with normal saline. Each 0.5 mL eluate was collected in an Eppendorf tube and the liposome opacity was visualized to monitor the position of 188Re-liposome. The radiochemical yield was calculated by the radioactivity of the collected product divided by the total initial loaded radioactivity for the separation.
MTD of 188Re-liposome in Fischer344 rats
The MTD of medication in animal trial was defined as the radioactivity dose below the dose resulting in either the death of any animal in groups of five animals or body weight loss of more than 20%.17,31 To obtain the MTD for referring therapeutic evaluation, the toxicity of 188Re-liposome in Fischer344 rats was estimated. First, fifteen normal male Fischer344 rats (12–13 weeks old) were randomly divided into three groups (five rats per group) and administered 188Re-Liposome (2.5 μmol phospholipids/ 0.5 mL) at dosage of 296, 333, and 370 MBq via single intravenous injection for respective groups. After medication, the body weight loss of rats of each group was recorded twice per week for 1 month and the lethality of radiation toxicity in rats was monitored every day for 1 month.
Radiation dosimetry estimation
Internal radiation-dose assessment of 188Re-liposome in nuclear medicine was simulated using OLINDA/EXM 1.1 computer software according to previous reports.26,32,33 In order to evaluate the equivalent dose (Sv) in humans, the relative organ-mass scaling method was used. Before starting computer calculation, the bio-distribution of 188Re-liposome in human (73 kg adult) organs was extrapolated from our previous results of percentage injected activity per gram for the organ (%IA/gorgan) in rats21 and then calculated according to the following formula:
|
|
The extrapolated bio-distribution data (%IA/organ) of 188Re-liposome in human organs at 1, 4, 24, 48, and 72 hours were entered into OLINDA/EXM 1.1 software. The integral (MBq · s) information from selected 14 organs including heart contents (blood), muscle, testes, pancreas, stomach, small intestine, large intestine, kidney, spleen, liver, lung, heart, brain, and remainder of the body, was used to evaluate radiation dosimetry. The activity of remainder of the body was defined as the total activity in the body minus all of the activities from selected organs. In addition, the distribution of activity of remainder of the body was presumed to be uniform. To estimate tumoral absorbed dose (Gy) at different tumor sizes ranging from 0.01 to 6,000 g, the sphere model in OLINDA/EXM 1.1 software was used. The uptake value of tumor (%IA/g) in bio-distribution study in rats21 was directly entered into OLINDA/EXM 1.1 software prgram for dosimetry estimation.
Therapeutic efficacy evaluation
To evaluate therapeutic efficacy of 188Re-Liposome in Fischer344/F98luc orthotopic glioma bearing rat model, a total of 35 rats was used herein. For survival evaluation study, 14 tumor bearing rats were randomly divided into two groups (seven rats per group). Subsequently, the rats of two groups were administered normal saline (control group) and 188Re-liposome (333 MBq/0.5 mL; 2.5 μmol phospholipid/0.5 mL) via single intravenous injection on Day 7 post-inoculation, respectively. Then the rats were monitored for survival and body weight every day until death. In addition, a decrease in body weight of 20% or more compared with weight on the day of inoculation was considered as a humane endpoint for survival evaluation. For tumor-growth inhibition study, 21 tumor bearing rats were used. Nine rats per group were administered saline (control group) and 188Re-liposome (333 MBq/0.5 mL; 2.5 μmol phospholipids/0.5 mL) via single intravenous injection on Day 7 post-inoculation, respectively. Then, three rats from each group were randomly sacrificed with CO2 euthanasia on Day 10, 14, and 17 post-inoculation, respectively (another 3 rats were sacrificed on Day 6 post-inoculation). The brain tumors were carefully dissected, measured, and weighed. The tumor volume (mm3) was calculated according to the formula, volume = 0.5 × length × width2. Meanwhile, the histopathological examination of the tumors was performed on Day 14 post-inoculation by staining with H and E according to routine staining protocol.
Statistical analysis
All data were expressed as mean ± standard error of the mean (SEM). The unpaired Student’s t-test was used for group comparisons. For therapeutic efficacy evaluation studies, survival curves were compared by use of the logrank test (SPSS software; SPSS Inc., Chicago, IL, USA). Values of P<0.05 were considered significant.
Results
Bioluminescence of F98luc cell line and in vitro growth curve
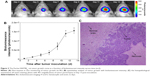
After sorting via GFP by FACS, the purity of luciferase-expressed F98luc cells was greater than 95%. The luminescence emission of F98luc cells was detected using BLI technique as shown in Figure 1A. The minimal amount of cells for effective detection using BLI device was approximately 195 cells per well. In addition, the intensity of bioluminescence was highly correlated with the total amount of F98luc cells with the linear correlation coefficient between bioluminescent intensity and cell numbers fitted R2=0.99 (Figure 1C). The in vitro growth curve of F98luc cells is shown in Figure 1B. The Td of F98luc cells was calculated to be 26.23 hours.
In vivo monitoring of Fischer344/F98luc tumor growth
For monitoring brain tumor growth in vivo, the noninvasive images of tumor growth were acquired from Fischer344/F98luc glioma-bearing rat model via BLI device at different time intervals (Figure 2A). The intensity of bioluminescence from tumor region was apparently enhanced as the time of post-inoculation increased. To understand the accurate trend of tumor growth curve, the quantitative region of interest analysis from bioluminescent images was carried out (Figure 2B). The tumor growth curve indicates that intensity of bioluminescence increased slowly and reached a plateau on Day 13 after inoculation. Furthermore, the histopathological analysis of tumor parenchyma on Day 13 after inoculation shows that the phenomenon of central necrosis was observed in the tumor (Figure 2C).
Characterizations of PEGylated liposome and 188Re-liposome
The concentration of phospholipid in the prepared PEGylated liposome was 21.2±1.95 μmol/mL (n=3). The hydrodynamic size of PEGylated liposome was 80.12±0.83 nm (n=3) and the charge on the liposomal surface was −2.02±0.96 mV (n=3). High purity 188Re-BMEDA complex was prepared at high radiochemical yield (>98%) and used to react with PEGylated liposome to form 188Re-liposome. No significant change of particle size was observed after the PEGylated liposome was further radio-labeled with 188Re. The radiochemical yield of 188Re-liposome was 88.75%±1.46% (n=3) and the radiochemical purity was greater than 95%.
MTD
Figure 3 shows 1 month body weight monitoring for MTD estimation with 296, 333, and 370 MBq of 188Re-liposome via single intravenous injection in normal Fischer344 rats, respectively. All treatment groups had body weight decrease (less than 20% changes) at first and then the body weight gradually recovered to normal after Day 7 post-injection. However, one rat died after receiving 370 MBq treatment. Given the above experimental data, the MTD value for therapeutic evaluation with 188Re-liposome in the Fischer344 rats was estimated to be 333 MBq.
Radiation dosimetry
Table 1 shows the organ equivalent dose estimation of 188Re-liposome for humans via orthotopic glioma-bearing rat model. The high equivalent doses were found in the spleen (6.96 mSv/MBq) and the kidneys (1.2 mSv/MBq). Moderate equivalent doses were observed in the heart wall (0.89 mSv/MBq), liver (0.81 mSv/MBq), lung (0.43 mSv/MBq), small intestine (0.2 mSv/MBq), lower large intestine (0.14 mSv/MBq), pancreas (0.13 mSv/MBq), and stomach wall (0.12 mSv/MBq) etc. Slight equivalent doses were found in the normal brain (0.02 mSv/MBq), red marrow (0.05 mSv/MBq) and thyroid (0.07 mSv/MBq) and so forth. In addition, the tumoral absorbed doses were estimated to be 122 to 13.6 mGy/MBq corresponding to the tumor sizes of 1 to 10 g, respectively.
Therapeutic efficacy
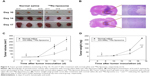
Figure 4 shows the survival curves for 188Re-liposome and normal saline treatment on a Fischer344/F98luc glioma-bearing rat model. The result shows that the lifespan for the 188Re-liposome-treated group was significantly increased by 10.67% compared to control group (P=0.007). The statistical analysis for therapeutic efficacy evaluation is summarized in Table 2. The maximum survival time for the 188Re-liposome-treated group and control group was 24 and 19 days, respectively. In addition, the median survival time was 20.75 and 18.75 days, respectively.
  | Table 2 Therapeutic efficacy evaluation of 333 MBq 188Re-liposome in orthotopic glioma-bearing rat model |
To monitor tumor growth inhibition, the treated rats were euthanized at predetermined times and the tumor masses were removed for further analysis. Figure 5 shows tumor growth inhibition after treating with 188Re-liposome and normal saline. All of the tumor masses removed from rats treated with 188Re-liposome were found to be significantly smaller than the tumor masses removed from control group (Figure 5A), consistent with the images observed in the pathomorphological examination (Figure 5B). In addition, the quantitative analysis from the removed tumors also showed that the tumor volume from the 188Re-liposome treated group declined significantly at Day 14 (P=0.0291) and Day 17 (P=0.0177) compared to control group (Figure 5C), as well as the tumoral mass weight at Day 10 (P=0.0294), Day 14 (P=0.0489), and Day 17 (P=0.0007) (Figure 5D).
Discussion
The bio-distribution, pharmacokinetics, and imaging of 188Re-liposome in the orthotopic glioma-bearing rat model have been studied in our recent report.21 Previous work revealed that 188Re-liposome is retained in the GBM tumor area with approximately 2 %ID/g with T/N (tumor-to-normal brain) uptake ratio as high as 32.5 at 24 hours post-injection. To elucidate the mechanism underlying the significant accumulation of injected 188Re-liposome in a tumor, it is presumed that 188Re-liposome (~80 nm) can cross leaky blood–brain barrier (BBB) near the glioma followed by retention in the tumor parenchyma by EPR effect. Leaky BBB structure in the Fischer344/F98 rat has also been confirmed via immunohistochemistry assay of the brain.34 Moreover, as previously reported, the neutral, negatively-charged, and/or PEGylated liposome may display larger retention volume in the brain than positive charged/unshielded liposome.35 The critical exclusion size of the endothelial gap in human primary GBM (U87 MG cell line) was estimated to be 7–100 nm, which may allow the passage of the 80 nm sized 188Re-liposome nanoparticles.36 Based on the aforementioned substantial evaluation, 188Re-liposome is worthy of further development as a therapeutic agent in orthotopic glioma-bearing rat model via single intravenous injection as discussed in this study.
In order to determine the appropriate treatment dose of 188Re-liposome in glioma rat model, the MTD estimation of 188Re-liposome in normal Fischer344 rats was conducted before the therapeutic experiments. According to the related studies in the previous reports, the acute toxicity of 188Re-liposome in rats was estimated as the following.37,38 The rats administered 185 MBq 188Re-liposome only, via intravenous injection, showed slight body weight loss (without causing any death) until 1 month. In this study, three higher dosages (296, 333, and 370 MBq) of 188Re-liposome were tested separately for the MTD estimation experiment. The results showed none of the rats in each group lost body weight over 20% in the period of 1 month after injection except for one rat which died in the group with the highest dosage (370 MBq). From the above experiment, the MTD of 188Re-liposome for Fischer344 rats was estimated to be 333 MBq.
For BLI experiments, the F98 cells expressing luciferase were successfully constructed by lentiviral-mediated transfection. By suspending a serial number of F98luc cells in the 96-well plate, the in vitro BLI showed a good linear relationship (R=0.99) between the cell number and the photo intensity (Figure 1C); meanwhile, the minimum detectable bioluminescent signal was shown to locate at the cell number 195. These results are comparable to those reported in the literature.27 As shown in the BLI study, the GBM tumor images can be clearly displayed via F98luc reporter at different time points after inoculation; meanwhile, the tumor growth could be monitored by the intensity of bioluminescence. In vivo monitoring by BLI declared that the tumor growth in orthotopic Fischer344/F98luc model rose gradually until Day 13 post-inoculation and then plateaued (Figure 2B). The presentation of the plateau could reveal the phenomena as follows. First, the larger tumor size (>10 mm) could have shielded some extent of the bioluminescence. Second, the tumoral central core could have had areas of necrosis with hypoxia which would cause decreased expression of luciferase.39 The imaging information of in vivo BLI in Fischer344/F98luc model is in good compliance with a previous report in the literature.40 In short, the well-established Fischer344/F98luc model can provide convenient, rapid, and non-invasive monitoring of the orthotopic tumor growth.
The lifespan of the glioma-bearing rats treated with 188Re-liposome was found to have 10.67% prolongation compared to control group (P<0.05). In addition, the tumor volume or tumor weight monitoring after treatments displayed significant difference (P<0.05) between the 188Re-liposome-treated and control group. Although only ~2 %ID/g 188Re-liposome was retained in the tumor area,21 the dosage of 333 MBq was sufficient to damage the cancer cells, resulting in prolongation of the lifespan of the rats. Once 188Re-liposome was accumulated in the tumor area by EPR effect, the beta particle radiation emitted from 188Re could powerfully damage the surrounding tumor cells, even spreading to the cells in the vicinity (>1 cm). This wide range of killing tumor capability by emitted β ray or electrons is known as “cross fire” effect,41 by destroying a tumor without necessarily targeting every cell within the tumor. Moreover, the cross fire effect may compensate for the insufficient extent for heterogeneous tumor uptake.42
In combination with chemotherapy, 188Re-liposome could be further incorporated with doxorubicin or other drugs to multiply the therapeutic efficacy.43 Moreover, further modifications of liposome surface with lactoferrin may be beneficial for its trafficking across BBB, which may further improve the therapeutic efficacy.28 All of these novel designs would be added in our future work for developing the radio-labeled liposome system as a potential therapeutic agent for GBM for clinical use.
Conclusion
In this study, the MTD of 188Re-liposome on Fischer344 rats was estimated to be 333 MBq. Fischer344/F98luc glioma model could be efficiently used for tumor growth monitoring via non-invasive BLI. The in vivo evaluation of therapeutic efficacy by dosimetry estimation and survival studies have shown that passive targeting 188Re-liposome via systemic administration can contribute to significant prolonged lifespan of orthotopic glioma-bearing rats with reasonable systemic radiation safety. Taken together, 188Re-liposome is worthy for further development as a potential diagnostic agent of RNT against GBM.
Acknowledgments
The authors would like to thank WL Wu for directing transfected work of F98 cell expressing luciferase as reporter (F98luc) and Dr Tsai-Yueh Luo for providing the rhenium-188 from Division of Isotope Application, Institute of Nuclear Energy Research (INER). In addition, we also thank CY Yu, YJ Chang, WC Lee, CL Ho, WC Hsu, TJ Chang, CH Chuang, SJ Chen, and SP Chiu from Radiopharmacology Laboratory, Division of Isotope Application, INER, for the technical support and meaningful discussion. This study was financially supported by INER, Longtan, Taiwan and was partially supported by National Health Research Institute, Taiwan (NHRI-EX103-10221EC) and Ministry of Science and Technology, Taiwan (102-2113-M-007-006-MY2).
Disclosure
The authors declare no conflicts of interest in this work.
References
Dolecek TA, Propp JM, Stroup NE, Kruchko C. CBTRUS statistical report: primary brain and central nervous system tumors diagnosed in the united states in 2005–2009. Neuro Oncol. 2012;14 Suppl 5:v1–v49. | ||
Niclou SP, Fack F, Rajcevic U. Glioma proteomics: status and perspectives. J Proteomics. 2010;73(10):1823–1838. | ||
Vredenburgh JJ, Desjardins A, Reardon DA, et al. The addition of bevacizumab to standard radiation therapy and temozolomide followed by bevacizumab, temozolomide, and irinotecan for newly diagnosed glioblastoma. Clin Cancer Res. 2011;17(12):4119–4124. | ||
Perlstein B, Finniss SA, Miller C, et al. TRAIL conjugated to nanoparticles exhibits increased anti-tumor activities in glioma cells and glioma stem cells in vitro and in vivo. Neuro Oncol. 2013;15(1):29–40. | ||
Quick A, Patel D, Hadziahmetovic M, Chakravarti A, Mehta M. Current therapeutic paradigms in glioblastoma. Rev Recent Clin Trials. 2010;5(1):14–27. | ||
Baskar R, Lee KA, Yeo R, Yeoh KW. Cancer and radiation therapy: current advances and future directions. Int J Med Sci. 2012;9(3):193–199. | ||
Dash A, Knapp FF, Pillai MR. Targeted radionuclide therapy – an overview. Curr Radiopharm. 2013;6(3):152–180. | ||
Davis ME, Chen ZG, Shin DM. Nanoparticle therapeutics: an emerging treatment modality for cancer. Nat Rev Drug Discov. 2008; 7(9):771–782. | ||
Vanpouille-Box C, Lacoeuille F, Belloche C, et al. Tumor eradication in rat glioma and bypass of immunosuppressive barriers using internal radiation with Re-188-lipid nanocapsules. Biomaterials. 2011;32(28):6781–6790. | ||
Phillips WT, Goins B, Bao A, et al. Rhenium-186 liposomes as convection-enhanced nanoparticle brachytherapy for treatment of glioblastoma. Neuro Oncol. 2012;14(4):416–425. | ||
Bobo RH, Laske DW, Akbasak A, Morrison PF, Dedrick RL, Oldfield EH. Convection-enhanced delivery of macromolecules in the brain. Proc Natl Acad Sci U S A. 1994;91(6):2076–2080. | ||
Allard E, Passirani C, Benoit JP. Convection-enhanced delivery of nanocarriers for the treatment of brain tumors. Biomaterials. 2009;30(12):2302–2318. | ||
Chen MY, Lonser RR, Morrison PF, Governale LS, Oldfield EH. Variables affecting convection-enhanced delivery to the striatum: a systematic examination of rate of infusion, cannula size, infusate concentration, and tissue-cannula sealing time. J Neurosurg. 1999;90(2): 315–320. | ||
Raghavan R, Brady ML, Rodriguez-Ponce MI, Hartlep A, Pedain C, Sampson JH. Convection-enhanced delivery of therapeutics for brain disease, and its optimization. Neurosurg Focus. 2006;20(4):E12. | ||
Chen LC, Chang CH, Yu CY, et al. Biodistribution, pharmacokinetics and imaging of Re-188-BMEDA-labeled pegylated liposomes after intraperitoneal injection in a C26 colon carcinoma ascites mouse model. Nucl Med Biol. 2007;34(4):415–423. | ||
Chen LC, Chang CH, Yu CY, et al. Pharmacokinetics, micro-SPECT/CT imaging and therapeutic efficacy of (188)Re-DXR-liposome in C26 colon carcinoma ascites mice model. Nucl Med Biol. 2008;35(8):883–893. | ||
Chang YJ, Chang CH, Yu CY, et al. Therapeutic efficacy and microSPECT/CT imaging of (188)Re-DXR-liposome in a C26 murine colon carcinoma solid tumor model. Nucl Med Biol. 2010;37(1):95–104. | ||
Tsai CC, Chang CH, Chen LC, et al. Biodistribution and pharmacokinetics of Re-188-liposomes and their comparative therapeutic efficacy with 5-fluorouracil in C26 colonic peritoneal carcinomatosis mice. Int J Nanomedicine. 2011;6:2607–2619. | ||
Chang YJ, Hsu CW, Chang CH, Lan KL, Ting G, Lee TW. Therapeutic efficacy of Re-188-liposome in a C26 murine colon carcinoma solid tumor model. Invest New Drug. 2013;31(4):801–811. | ||
Chang CH, Liu SY, Lee TW. Pharmacokinetics of BMEDA after intravenous administration in beagle dogs. Molecules. 2014;19(1):538–549. | ||
Huang FY, Lee TW, Kao CH, et al. Imaging, autoradiography, and biodistribution of (188)Re-labeled PEGylated nanoliposome in orthotopic glioma bearing rat model. Cancer Biother Radiopharm. 2011;26(6):717–725. | ||
Phillips WT, Bao A, Brenner AJ, Goins BA. Image-guided interventional therapy for cancer with radiotherapeutic nanoparticles. Adv Drug Deliver Rev. 2014;76:39–59. | ||
Barth RF, Kaur B. Rat brain tumor models in experimental neuro-oncology: the C6, 9L, T9, RG2, F98, BT4C, RT-2 and CNS-1 gliomas. J Neurooncol. 2009;94(3):299–312. | ||
Chow TH, Lin YY, Hwang JJ, et al. Diagnostic and therapeutic evaluation of In-111-vinorelbine-liposomes in a human colorectal carcinoma HT-29/luc-bearing animal model. Nucl Med Biol. 2008;35(5):623–634. | ||
Mathieu D, Lecomte R, Tsanaclis AM, Larouche A, Fortin D. Standardization and detailed characterization of the syngeneic Fischer/F98 glioma model. Can J Neurol Sci. 2007;34(3):296–306. | ||
Chen LC, Wu YH, Liu IH, et al. Pharmacokinetics, dosimetry and comparative efficacy of Re-188-liposome and 5-FU in a CT26-luc lung-metastatic mice model. Nucl Med Biol. 2012;39(1):35–43. | ||
Bryant MJ, Chuah TL, Luff J, Lavin MF, Walker DG. A novel rat model for glioblastoma multiforme using a bioluminescent F98 cell line. J Clin Neurosci. 2008;15(5):545–551. | ||
Huang FY, Chen WJ, Lee WY, Lo ST, Lee TW, Lo JM. In vitro and in vivo evaluation of lactoferrin-conjugated liposomes as a novel carrier to improve the brain delivery. Int J Mol Sci. 2013;14(2):2862–2874. | ||
Bartlett GR. Phosphorus assay in column chromatography. J Biol Chem. 1959;234(3):466–468. | ||
Bao A, Goins B, Klipper R, Negrete G, Phillips WT. Re-186-liposome labeling using Re-186-SNS/S complexes: in vitro stability, imaging, and biodistribution in rats. J Nucl Med. 2003;44(12):1992–1999. | ||
Reilly RM, Chen P, Wang J, Scollard D, Cameron R, Vallis KA. Preclinical pharmacokinetic, biodistribution, toxicology, and dosimetry studies of In-111-DTPA-human epidermal growth factor: an auger electron-emitting radiotherapeutic agent for epidermal growth factor receptor-positive breast cancer. J Nucl Med. 2006;47(6):1023–1031. | ||
Stabin MG, Sparks RB, Crowe E. OLINDA/EXM: The second-generation personal computer software for internal dose assessment in nuclear medicine. J Nucl Med. 2005;46(6):1023–1027. | ||
Chang CH, Stabin MG, Chang YJ, et al. Comparative dosimetric evaluation of nanotargeted (188)Re-(DXR)-liposome for internal radiotherapy. Cancer Biother Radiopharm. 2008;23(6):749–758. | ||
Poduslo JF, Curran GL, Wengenack TM, Malester B, Duff K. Permeability of proteins at the blood–brain barrier in the normal adult mouse and double transgenic mouse model of Alzheimer’s disease. Neurobiol Dis. 2001;8(4):555–567. | ||
MacKay JA, Deen DF, Szoka FC Jr. Distribution in brain of liposomes after convection enhanced delivery; modulation by particle charge, particle diameter, and presence of steric coating. Brain Res. 2005;1035(2):139–153. | ||
Hobbs SK, Monsky WL, Yuan F, et al. Regulation of transport pathways in tumor vessels: role of tumor type and microenvironment. Proc Natl Acad Sci U S A. 1998;95(8):4607–4612. | ||
Liu CM, Chang CH, Chang YJ, et al. Preliminary evaluation of acute toxicity of Re-188-BMEDA-liposome in rats. J Appl Toxicol. 2010;30(7):680–687. | ||
Chi-Mou L, Chia-Che T, Chia-Yu Y, et al. Extended acute toxicity study of Re-188-liposome in Rats. J Appl Toxicol. 2013;33(9):886–893. | ||
Towner RA, Gillespie DL, Schwager A, et al. Regression of glioma tumor growth in F98 and U87 rat glioma models by the Nitrone OKN-007. Neuro Oncol. 2013;15(3):330–340. | ||
Xi GF, Rajaram V, Mania-Farnell B, et al. Efficacy of vincristine administered via convection-enhanced delivery in a rodent brainstem tumor model documented by bioluminescence imaging. Child Nerv Syst. 2012;28(4):565–574. | ||
Barendsen GW. Dose-survival curves of human cells in tissue culture irradiated with alpha-, beta-, 20-Kv X- and 200-Kv X-radiation. Nature. 1962;193:1153–1155. | ||
Sofou S. Radionuclide carriers for targeting of cancer. Int J Nanomedicine. 2008;3(2):181–199. | ||
Hsu WH, Liu SY, Chang YJ, Chang CH, Ting G, Lee TW. The PEGylated liposomal doxorubicin improves the delivery and therapeutic efficiency of (188)Re-Liposome by modulating phagocytosis in C26 murine colon carcinoma tumor model. Nucl Med Biol. 2014;41(9): 765–771. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.