Back to Journals » Clinical Ophthalmology » Volume 13
Evaluating the efficacy of short duration Mitomycin C in safe surgery system trabeculectomy combined with cataract surgery
Authors Khandelwal R, Bijlani M, Raje D, Rathi A
Received 10 November 2018
Accepted for publication 6 March 2019
Published 22 May 2019 Volume 2019:13 Pages 849—857
DOI https://doi.org/10.2147/OPTH.S192044
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Scott Fraser
Rekha Khandelwal,1 Madhavi Bijlani,1 Dhananjay Raje,2 Anand Rathi1
1Department of Ophthalmology, NKP Salve Institute of Medical Sciences & Lata Mangeshkar Hospital, Nagpur 440019, India; 2MDS Bio Analytics, Nagpur 440010, India
Purpose: To compare the efficacy of 0.2 mg/ml Mitomycin C (MMC) applied for 1-minute versus 2-minutes in patients undergoing combined surgery for primary glaucoma coexistent with cataract.
Materials and methods: This was a randomized controlled clinical trial of 63 patients operated on for primary glaucoma (POAG or PACG) with visually significant cataract. All patients underwent safe surgery system trabeculectomy with manual small incision cataract surgery (MSIC) and implantation of PC IOL. Patients were randomized into intra-operative MMC 0.2 mg/ml for 1-minute (study group) and MMC 0.2 mg/ml for 2-minutes (control group). Success was measured on the basis of two different intraocular pressure (IOP) goals (IOP ≤21 mmHg, IOP ≤18 mmHg) and mean IOP reduction from baseline at the end of 12 months.
Results: At 12 months, significant decreases in mean IOP were observed in both groups (P<0.001).The mean IOP reduction was 31.33±9.06% in the study group, as compared to 43.32±9.38% in the control group (P<0.001). The overall success for IOP ≤21 mm Hg was 80.5% in the study group and 90.9% in the control group (P<0.05). Kaplan-Meier analysis showed an insignificant difference in overall success rates of the two groups.
Conclusion: The IOP reduction with 2-minute MMC (0.2 mg/ml) is more effective than 1-minute MMC after 12 months. It offers a decrease in anti-glaucoma medications and substantial visual recovery in combined surgery done for primary glaucoma coexistent with cataract.
Keywords: glaucoma, combined surgery, Mitomycin C, trabeculectomy, safe surgery system trabeculectomy
Introduction
Glaucoma is a multifactorial optic neuropathy, leading to irreversible visual loss. Currently, intraocular pressure (IOP) is the only modifiable risk factor in glaucoma patients. Many studies have demonstrated that visual field progression is slow when IOP is lowered.1–3 As there is trabecular dysfunction in glaucomatous eyes, provocative tests have induced a significant increase in IOP as compared to normal eyes.4,5 Lowering IOP in glaucomatous eyes by surgery is indicated when maximum tolerated medications and laser therapy fail to control progression of glaucomatous optic neuropathy. There are a variety of surgical procedures which can reduce IOP, but the long-term success depends on the wound healing response of the subconjunctival space.
The surgical outcome of filtration surgery is also associated with the long-term use of pre-operative IOP-lowering medications. Patients on antiglaucoma medications develop ocular surface disorder and dry eye due to modification of the conjunctiva associated lymphoid tissue (CALT). The tear film presents high concentrations of pro-inflammatory cytokines, which depends on the number of medications, benzalkonium chloride (BAK) exposure in the form of preservative, and duration of treatment.6 These play a crucial role in the outcome of filtration surgery.
To avoid reactive scarring in the subconjunctival space in the post-operative period, cytotoxic antimetabolites like Mitomycin C (MMC) and 5-fluorouracil (5-FU) have been used in filtering surgery.7–9
The role of these antifibrotic agents has been well documented by non-invasive modern techniques using in vivo confocal microscopy (IVCM) of the conjunctiva and three-dimensional laser scanning confocal analysis of the conjunctival microcysts before and after surgery.10,11
The adjunctive use of antifibrotic agents has been a major advance in reducing subconjunctival fibrosis, thereby lowering IOP. A review done to assess the effects of intraoperative application of MMC in the eyes of patients undergoing trabeculectomy reported that, although the IOP lowering effect is better with a higher concentration and longer duration of MMC application, the complication rates are also higher.7
In developing countries, glaucoma is generally diagnosed at a very advanced stage, only when the patient seeks advice for cataract surgery. Trabeculectomy surgery has been associated with progressive cataract in the majority of cases.12 There has been strong evidence in favor of combined surgery for long-term IOP control.13 Trabeculectomy is, thus, performed as a primary procedure with cataract extraction in many developing countries.14,15 It reduces the risk of early severe post-operative pressure elevation, provides long-term intraocular pressure control, and decreases the need for glaucoma medication.16 Combined surgery is effective, but also carries the risk of failure and complications.17 Using MMC in glaucoma surgery is a double-edged sword as it prevents bleb fibrosis on the one hand but can cause severe blinding side-effects on the other. It has been shown to cause bleb avascularity, transconjunctival oozing (TCO), delayed bleb leaks, etc.18 The safe surgery system trabeculectomy using wide and posterior application of MMC, tight closure of the scleral flap, adjustable/releasable sutures, and watertight conjunctival closure has decreased surgical failures significantly.19
Routinely, Mitomycin C (0.2 mg/ml) is used in trabeculectomy for 2−3 minutes. A recent study on the efficacy of Mitomycin C in glaucoma filtration surgery showed that lower concentrations of MMC and shorter exposure times are as effective as higher concentrations/prolonged exposure time in achieving optimum IOP levels. Also, higher MMC concentrations and prolonged exposure times may be associated with delayed risk of complications.20 An in vitro study suggested that fibroblast inhibition was mainly dependent on the concentration rather than the duration of MMC exposure.21 Many recent studies have shown that reducing the concentration and duration of Mitomycin C was safe and effective in lowering IOP with a lower rate of post-operative complications.22,23 Hence, this study was designed to evaluate the efficacy of a short exposure time of Mitomycin C (0.2 mg/ml for 1-minute) as compared to the standard exposure time (0.2 mg/ml for 2-minutes), keeping the same concentration, in primary trabeculectomy combined with cataract surgery.
Materials and methods
This was a comparative randomized interventional study on patients with primary glaucoma who underwent safe surgery system trabeculectomy with cataract extraction (MSICS) from January 2015 to December 2016 at a rural-based tertiary care hospital of a single academic institute in Central India. The study was approved by the Ethics Committee of NKP Salve Institute of Medical Sciences and Research Center and Lata Mangeshkar Hospital (ECR/27/Inst/MH/2015), and followed the tenets of the Declaration of Helsinki. All participants provided written informed consent to be part of the study. Patients more than 40 years of age, having primary open angle glaucoma or primary angle closure glaucoma (as per ISGEO definitions) uncontrolled on medication or/with laser associated with visually significant cataract were included. Patients who were non-compliant for medical treatment or who had poor healthcare accessibility were also included for primary trabeculectomy. By definition, all POAG and PACG cases with intraocular pressure (IOP) more than 21 mm Hg with a cup:disc ratio ≥0.7 or asymmetry ≥0.2 between two eyes and visual field loss were included in the study. Patients who could not perform reliable field testing but had severe glaucomatous disc damage (cup: disc ratio ≥0.8) with IOP >21 mmHg were also included. Patients with secondary glaucoma (lens induced, pseudoexfoliation, neovascular, steroid induced, pigmentary, uveitic, angle recession glaucoma), previous failed trabeculectomy, major intra-operative complications like vitreous loss, posterior capsular rent, zonular dialysis, dropped nucleus, etc., were excluded from the study. Patients with less than 12 months of follow-up were excluded.
Baseline demographic data, history, and clinical characteristics were recorded. Age, sex, type of glaucoma, pre-operative number of antiglaucoma drops, and best-corrected visual acuity by Standard Snellen’s chart were noted (converted to LogMAR acuity for analysis). Slit-lamp biomicroscopy (Topcon, Oakland, NJ, USA) was done in all cases to rule out secondary glaucoma and visual impairment due to other eye diseases. Peripheral anterior chamber depth (ACD) was estimated by Van Herrick technique. Gonioscopy was performed using four-mirror Sussman gonioscope (Ocular Instruments, Inc; Bellevue, WA). Fundus evaluation was done by indirect ophthalmoscopy to rule out retinal and macular pathology. The stereoscopic optic disc evaluation was done using a +78D lens (x16 magnification). The vertical cup-disc ratio (VCDR) was measured and morphological features of glaucomatous optic neuropathy were noted. Intraocular pressure (IOP) was measured using Goldmann applanation tonometer (Inami and Co, Tokyo, Japan). Visual field examination using a Humphrey Field Analyzer (SITA 24–2; Humphrey Field Analyzer II; Carl Zeiss Meditec, Inc; Oberkochen, Germany) was attempted whenever possible.
The glaucomatous visual field defect was considered to be present if 1) the glaucomatous hemifield test was outside normal limit, and 2) a cluster of three or more non-edge, contiguous points, not crossing the horizontal meridian with a probability of <5%, was found on a pattern deviation plot on two separate tests. The patients were categorized as POAG and PACG on the basis of ISGEO classification. Only one eye of the selected patient was included in the study. Patients using anti-glaucoma drugs like beta-blockers in the other eye (not selected eye for surgery) were shifted to other antiglaucoma drugs which had no crossover effects of lowering IOP.
Eligible patients were divided into two groups as follows. The study group, comprising of a short exposure time of MMC 0.2 mg/ml for 1-minute and a control group of MMC 0.2 mg/ml for 2-minutes (standard exposure time). For randomization, two consecutive cases were selected for the study group (1 minute), and the third case was selected for the control group (2 minutes), thus maintaining the study-to-control ratio of 2:1.
Statement of ethics
The authors certify that all applicable institutional and governmental regulations concerning the ethical use of human volunteers were followed during this research.
Surgical technique
In the procedure of combined surgery, both trabeculectomy and cataract extraction were done in the same sitting. Under peribulbar anesthesia (2% Xylocaine, 2% Bupivacaine, Hyaluronidase 37.5 IU/ml), the periocular area was cleaned with 5% Povidone iodine for 3 minutes. A fornix based conjunctival flap was dissected superiorly, and a superior rectus traction suture was taken with 6–0 silk. The tenon capsule was separated and conjunctiva dissected backwards for 8–10 mm for the wide application of antimetabolite sponges. Three MMC (0.2 mg/ml) soaked sponges were applied below the conjunctiva, keeping the edges at a distance. After either 1 minute (study group) or 2 minutes (control group) as per randomization, the sponges were removed. The surgical area was thoroughly irrigated.
For MSICS, a partial thickness scleral tunnel (flap) of 5.5–7 mm was made 3 mm posterior to the limbus with a crescent blade. The dissection was extended 1–2 mm into the clear cornea, and scleral pockets were made in the same plane, keeping the sides of the scleral tunnel intact. A capsulorhexis (5–6 mm) was performed and 3.2 mm keratome was used to enlarge the inner lip of the tunnel. The nucleus was delivered using the sandwich technique, and the cortex was aspirated using a simcoe two way-irrigation-aspiration cannula. A polymethyl methacrylate (PMMA) IOL (single piece, optic diameter 5.5 or 6 mm, modified C loop with 10° angulation, S3602; Aurolab, Madurai, India) was placed in the bag and rotated to a horizontal position. After the cataract extraction (MSICS), intracameral acetylcholine was injected to constrict the pupil.
The trabeculectomy was performed using a Kelley’s punch followed by iridectomy. Two adjustable 10–0 monofilament nylon sutures (with four throws) were taken on either side of the punch area to close the scleral tunnel, as described in safe surgery system trabeculectomy. Additional interrupted sutures were taken when a section was enlarged for a large brown and hard nuclear cataract. Conjunctival closure was achieved by 10–0 nylon using anchoring corneal suture technique in a watertight manner under tension. The patency of trabeculectomy was tested at the end of the surgery, by injecting balanced salt solution (BSS) through the side port and observing the formation of diffuse conjunctival bleb without any leak. At the end of surgery, a subconjunctival injection of steroid (Dexamethasone) and antibiotic (Gentamicin sulphate) was given 180° away from the trabeculectomy site and eye patched for 24 hours.
Post-operatively, the patients were treated with moxifloxacin 0.5% 4-times daily for 2 weeks and prednisolone acetate 1% drops 6-times per day for 1 week; and then tapered over the next 2 months. The supplemental use of antiglaucoma medications and cycloplegics was based on the IOP and the level of intraocular inflammation. Patients were evaluated on post-operative day 1, day 7, at 1 month, 3 months, 6 months, and 12 months, with additional visits as and when required. Follow-up visit day ±2 days was also considered for analysis purposes. At each visit, visual acuity, intraocular pressure (IOP), bleb morphology, and complications including the need for anti-glaucoma medications were recorded.
Surgical outcome
Success was measured on the basis of two different IOP goals (IOP ≤21 mmHg, IOP ≤18 mmHg) and mean IOP reduction from baseline. A complete success (CS) was defined as IOP control without medication, while IOP control with medication was treated as a qualified success (QS). Overall success was defined as IOP control with or without medications. Sustained IOP <5 mmHg on two visits with anatomic changes or No PL vision developed post-operatively was considered a failure.
An ideal bleb was defined as mildly elevated, diffused with normal vascularity. Seidel positive blebs seen within the first month of follow-up were reported as wound leaks, and those after 1 month were considered as bleb leaks. Bleb-related infection included bleb-related endophthalmitis and blebitis (discharging eye with opacification of bleb). Visual outcome was defined as the presence or absence of improvement in visual acuity as compared to the pre-operative acuity of vision.
Statistical analysis
The demographic and clinical profile of patients treated with MMC at 1 minute and 2 minutes was compared using t-test for independent samples for continuous variables, and chi-square test of homogeneity for categorical variables. The mean IOP and visual acuity (logMAR) were compared using t-test of independent samples at both pre- and post-treatment; while within-group comparisons were made using paired t-test between pre- and post-treatment. Antiglaucoma medication used between two groups was compared using Mann-Whitney U-test, while within group comparison was performed using Wilcoxon signed rank test. The mean percent reduction of IOP was compared using t-test of independent samples. Further, the success achieved in two groups was tested using chi-square test of homogeneity. Kaplan-Meier analysis was performed, and a log-rank test was used to evaluate the statistical significance of difference in the survival plots in two groups. All the analyses were performed in SPSS ver 20.0 (IBM Corp., Armonk, NY, USA), and the significance was tested at 5% level.
Results
In our study, there were 41 cases in the study group (0.2 mg/ml MMC, 1-minute) and 22 cases in the control group (0.2 mg/ml MMC, 2-minutes). The mean follow-up was 18.5 months (range=12–24 months). The two study groups were similar in age, sex, type of glaucoma, and minimum follow-up period. The demographic characteristics of patients in the two treatment groups are shown in Table 1.
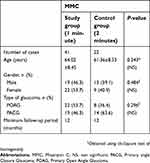 | Table 1 Baseline characteristics of patients undergoing combined surgery augmented with intraoperative 0.2 mg/ml MMC for 1-minute vs 2-minutes |
The pre-operative IOP, visual acuity (logMAR scale), and number of anti-glaucoma medications (AGM) did not differ significantly between groups, as shown in Table 2. After 12 months of follow-up, the mean IOP was 19.80±2.41 mmHg in the study group (1-minute) and 17.27±3.24 mmHg in the control group (2-minutes), which showed a statistically significant reduction from the pre-operative IOP levels in both groups (P<0.001). The post-operative mean IOP was significantly lower in the control group as compared to the study group (P<0.003). The difference in logMAR visual acuity between pre- and post-operative visit was significant in both treatment groups (P<0.001). The logMAR acuity after follow-up was comparable and did not differ significantly between the two groups (P=0.949). The number of anti-glaucoma medications (AGM) was significantly reduced in both treatment groups, but the number of AGM requirements in the study group was comparatively higher than the control group, with a P-value of 0.004. The mean IOP reduction was 31.3% (±9.06%) in the study group, while it was 43.3% (±9.38%) in the control group, and the difference was statistically significant (P<0.001). The pre- and post-operative comparison between two study groups is shown in Table 2.
 | Table 2 Comparison of clinical parameters in patients undergoing combined surgery augmented with intraoperative 0.2 mg/ml MMC for 1-minute vs 2-minutes |
The complete, qualified, and overall success were obtained for two IOP goals in both the treatment groups. The overall success for IOP ≤21 mmHg was achieved in 33 eyes out of 44 eyes in the study group (80.5%) and 20 eyes out of 22 eyes in the control group (90.9%). For IOP ≤18 mmHg, overall success was achieved in 15 eyes in the study group (36.6%) and 15 eyes in the control group (68.2%). The surgical outcomes for IOP ≤21 mmHg and IOP ≤18 mmHg are shown in Table 3.
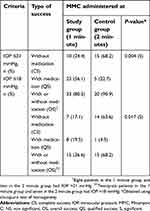 | Table 3 Success achieved in patients undergoing combined surgery augmented with intraoperative 0.2 mg/ml MMC for 1-minute vs 2-minutes |
The post-operative complications and number of patients requiring IOP-lowering procedures (bleb massage, bleb needling, and bleb revision) were comparable in the two groups (P>0.05), as shown in Table 4.
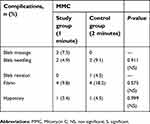 | Table 4 Post-operative complications in patients undergoing combined surgery augmented with intraoperative 0.2 mg/ml MMC for 1-minute vs 2-minutes |
The comparison of mean IOP at different time periods between two treatment groups is shown in Figure 1A. Up until 1 month, the difference was insignificant (P>0.05); however, subsequently the mean IOP in the study group was higher than the control group, until the last follow-up (P<0.05).
 | Figure 1 Kaplan-Meier plots for overall success in two treatment groups. |
Kaplan-Meier survival analysis was performed for IOP ≤21 mmHg and ≤18 mmHg, as shown in Figure 1B. For IOP ≤21 mmHg, the overall success rate in the study group decreased rapidly with mean time of 290.48 (±19.75) days as compared to the control group with a mean time of 353.86 (±11.15) days, although the difference between the success rates was statistically insignificant with a P-value of 0.573. Also, for IOP ≤18 mmHg, the overall success rate in the study group decreased faster with a mean time of 274.32 (±21.58) days, as compared to the control group with a mean of 315 (±23.48) days; however, the difference between the two success rates was statistically insignificant, with a P-value of 0.372.
Discussion
Trabeculectomy is the standard primary surgical treatment for intraocular pressure (IOP) reduction in medically uncontrolled glaucoma. In developing countries, glaucoma is diagnosed when the patient seeks advice for cataract surgery. Trabeculectomy is, thus, performed as a combined procedure using antifibrotic agents to have better long-term IOP control.14,15
With the higher concentration and long duration of MMC application, the IOP lowering effect was better, but the risk of long-term complications increased. To minimize MMC-related complications, safe surgery system trabeculectomy was developed by Khaw et al,19 which could be combined with cataract extraction (phacoemulsification/MSICS) with good surgical outcome.24
The effect of brief exposure to MMC on viability and proliferation of cultured human Tenon’s capsule fibroblast showed that even a 1 minute exposure of a 0.04% (0.4 mg/ml) MMC might be as effective as a 5 minute exposure.21 Earlier studies also confirmed that a reduced concentration and duration of MMC was safe and effective in lowering IOP with less post-operative complications.22,23
In this study, we evaluated the IOP lowering efficacy of 1 minute MMC exposure time with standard exposure time, keeping the concentration the same for both groups in combined surgery.
We found that direct head-to-head comparison between studies was difficult due to different definitions of success, study design, varying time of antimetabolite exposure, and different follow-up period.
In our study, we included all primary glaucoma patients having moderate-to-severe/end stage disease with significant cataract. After a minimum follow-up of 12 months, we found that all patients in both groups showed a significant IOP reduction from the pre-operative level (P<0.05). Although the inter-group difference was not significant until the end of first month, later it was significant until the last follow-up (P<0.05).
The IOP reduction was 31.3% in the study group (1-minute), as compared to 43.3% in the control group (2-minutes). Mittal et al14 found a mean IOP reduction of 31% in a retrospective analysis of combined surgery cases using MMC for 2 minutes, and Khandelwal et al24found a 40% reduction with MMC for 2 minutes in combined surgery cases after 12 months follow-up.
For IOP ≤21 mmHg, the overall success was 80.5% in the study group and 90.9% for the control group, similar to earlier studies.14,22–24 However when more stringent criteria of IOP ≤18 mmHg, was used, it was found that the overall success was only 36.6% for the study group and 68.2% for the control group. A retrospective study done by Simon et al22using brief exposure of MMC (15 seconds, 0.4 mg/mL) in trabeculectomy reported an overall success of 93.7% for IOP ≤21 mmHg.
A study done by Sihota et al23showed that a 1 minute exposure of 0.1 mg/ml MMC was as effective as 0.2 mg/ml in achieving a target IOP of 15 mm Hg in primary adult glaucoma. The success rate of was 92% in the 0.1 mg/ml group and 88% in the 0.2 mg/ml group at a follow-up of 24 months.
Lee et al25 studied the benefit of titrating the concentration and exposure time of Mitomycin C (MMC) based on the patient’s risk factors for surgical failure, and found that there were no significant differences between the outcome groups and MMC exposure time.
Thus, there was inadequate evidence in the literature to claim superiority of any particular MMC protocol, with or without titration.25 A study done by Megevand et al26showed that a 2-minute intraoperative application of MMC (0.2 mg/ml) was as effective as a 5 minute exposure. The overall success for IOP ≤21 mmHg was 88% in the 2-minute group, as compared to 84% in the 5-minute group, with an average follow up of 20.2 months in this study.
Kim et al27 reported that application of 0.5 mg/ml of MMC for 0.5– to 1 minsute was optimal for the successful outcome of primary phakic trabeculectomy compared with no MMC or longer application of it. He also found that the incidence of hypotony was significantly higher with longer duration exposure of MMC during the follow-up of 3– to 12 months. A retrospective study conducted by Alwitry et al28 reported 83.1% patients had IOP ≤16 mm Hg at 12 months follow -up using 0.1 mg/ml MMC for 1 minute.Annen and Sturmer29found an IOP ≤21 mm Hg at the end of 12 months in 88% patients when MMC 0.2 mg/ml was used for 1 minute.
There were many studies titrating the concentration of MMC in trabeculectomy, but to the best of our knowledge this is the first study comparing the short exposure time of MMC in combined surgery. We did not report any statistical difference in the need of IOP lowering procedures between the two groups. There were no major sight threatening complications, like blebitis, bleb-related endophthalmitis, or prolonged hypotony, in any group during the follow-up of 12 months.
There are certain limitations of our study. The sample size was small and was from rural areas. Due to our strong peripheral activities, many rural cases admitted for cataract surgery were also diagnosed with established glaucoma at our tertiary care center. The majority of these cases had undetected moderate-to-severe glaucoma with high pre-operative IOP at the time of presentation. Such a sample of patients might have resulted in a significant inter-group difference between 1-minute and 2-minutes MMC exposure time, even when the concentration (0.2 mg/mL) was the same, unlike earlier studies.26–28 Other limitations are non-inclusion of a placebo group without MMC application for comparison with the 1-minute and 2-minutes MMC groups. The IOP criteria of ≤15 mmHg and ≤12 mmHg could have been used, as there is slower progression of advanced glaucoma with lower IOP. The filtration bleb grading could have improved the interpretation of results. We have not used a standardized preparation of Mitomycin C (FDA approved Mitosol kit) in our patients. The efficacy of MMC in glaucoma surgery depends on many factors like dose delivered to the tissues, volume, duration of exposure, site of application, type of microsurgical sponge, and MMC toxicity to the ciliary epithelium. The preparation of MMC, storage, and application procedure may induce variability in the results. The availability of a standardized preparation of MMC will help in comparing and quantifying the results in a better way. The long-term results of short MMC exposure time could not be evaluated in this study, and need further research and detailed analysis.
Glaucoma patients require lifelong follow-up for adequate control of IOP. In developing countries, frequent follow-ups may not always be possible and, hence, a long-term solution in the form of filtration surgery yields excellent results in control of IOP.
From our study, we cannot justify the use of a short exposure time of Mitomycin C (1-minute) in all primary glaucoma cases, irrespective of glaucoma status, but our findings point towards an optimal success rate in combined surgery. Although Kaplan-Meier analysis did not show a significant difference between the two groups, we found a lower post-surgical IOP with 2-minutes MMC as compared to 1-minute MMC in our study population of moderate-to-severe glaucoma. As 2-minute applications did not increase post-operative complications, this study showed that a 2-minute application is better than a 1-minute application in small incision cataract surgery combined with trabeculectomy.
The only disadvantage of 2-minute applications is the 1 minute longer surgical time than the 1-minute MMC application.
In summary, both groups (1-minute vs 2-minutes MMC 0.2 mg/ml) resulted in an excellent IOP control, decrease in anti-glaucoma medications, and substantial visual recovery in trabeculectomy, combined with MSIC surgery over 12 months follow-up.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Odberg T. Visual field prognosis in advanced glaucoma. Acta Ophthalmol. 1987;65:27–29. doi:10.1111/j.1755-3768.1987.tb02583.x
2. Mikelberg FS, Schulzer M, Drance SM, Lau W. The rate of progression of scotomas in glaucoma. Am J Ophthalmol. 1986;101:1–6.
3. Grant WM, Burke JF
4. Giuffrè I, Taverniti L, Di Staso S. The effects of 2% ibopamine eye drops on the intraocular pressure and pupil motility of patients with open-angle glaucoma. Eur J Ophthalmol. 2004;14:508–513.
5. Virno M, Sampaolesi R, Pecori Giraldi J, et al. Ibopamine: D1-dopaminergic agonist in the diagnosis of glaucoma. J Glaucoma. 2013;22:5–9. doi:10.1097/IJG.0b013e3182311db4
6. Mastropasqua R, Agnifili L, Fasanella V, et al. The conjunctiva-associated lymphoid tissue in chronic ocular surface diseases. Microsc Microanal. 2017;23:697–707. doi:10.1017/S1431927617000538
7. Wilkins M, Indar A, Wormald R. Intra-operative mitomycin C for glaucoma surgery. Cochrane Database Syst Rev. 2005;4:CD002897.
8. Chen CW, Huang HT, Bair JS, Lee CC. Trabeculectomy with simultaneous topical application of mitomycin C in refractory glaucoma. J Ocul Pharmacol. 1990;6:175–182.
9. Rotham RF, Liebmann JM, Ritch R. Low dose 5-fluorouracil trabeculectomy as initial surgery in uncomplicated glaucoma: long-term follow up. Ophthalmology. 2000;107:1184–1190.
10. Mastropasqua R, Fasanella V, Brescia L, et al. In vivo confocal imaging of the conjunctiva as a predictive tool for the glaucoma filtration surgery outcome. Invest Ophthalmol Vis Sci. 2017;58:BIO114–BIO120. doi:10.1167/iovs.17-21795
11. DI Staso S, Agnifili L, DI Gregorio A, et al. Three-dimensional laser scanning confocal analysis of conjunctival microcysts in glaucomatous patients before and after trabeculectomy. In Vivo. 2017;31:1081–1088. doi:10.21873/invivo.11173
12. Jampel HD, Solus JF, Tracey PA, et al. Outcomes and bleb-related complications of trabeculectomy. Ophthalmology. 2012;119:712–722. doi:10.1016/j.ophtha.2011.09.049
13. Friedman DS, Jampel HD, Lubomski LH, et al. Surgical strategies for coexisting glaucoma and cataract. Ophthalmology. 2002;109:1902–1913.
14. Mittal S, Mittal A, Ramakrishnan R. Safety and efficacy of manual small-incision cataract surgery combined with trabeculectomy: comparison with phacotrabeculectomy. Asian J Ophthalmol. 2008;10:221–229.
15. Bowman RJ, Hay A, Wood ML, Murdoch IE. Combined cataract and trabeculectomy surgery for advanced glaucoma in East Africa: visual and intraocular pressure outcomes. Eye. 2010;24:573–577. doi:10.1038/eye.2010.7
16. Thomas R, Paul P, Muliyil J. Glaucoma in India. J Glaucoma. 2003;12:81–87. doi:10.1097/00061198-200302000-00016
17. Edmunds B, Thompson JR, Salmon JF, Wormald RP. The National Survey of Trabeculectomy. III. Early and late complications. Eye (Lond). 2002;16:297–303. doi:10.1038/sj/eye/6700148
18. Anand N, Arora S, Clowes M. Mitomycin C augmented glaucoma surgery: evolution of filtering bleb avascularity, transconjunctival oozing, and leaks. Br J Ophthalmol. 2006;90:175–180. doi:10.1136/bjo.2005.077800
19. Khaw PT, Chiang M, Shah P, Sii F, Lockwood A, Khalili A. Enhanced trabeculectomy: the Moorfields safer surgery system. Dev Ophthalmol. 2012;50:1–28.
20. Al Habash A, Aljasim LA, Owaidhah O, Edward DP. A review of the efficacy of mitomycin C in glaucoma filtration surgery. Clin Ophthalmol. 2015;9:1945–1951.
21. Jampel HD. Effect of brief exposure to mitomycin C on viability and proliferation of cultured human Tenon‘s capsule fibroblasts. Ophthalmology. 1992;99:1471–1476.
22. Md G, Glovinsky Y. Trabeculectomy with brief exposure to mitomycin C. Clin Exp Ophthalmol. 2006;34:765–770. doi:10.1111/j.1442-9071.2006.01305.x
23. Sihota R, Angmo D, Chandra A, Gupta V, Sharma A, Pandey RM. Evaluating the long-term efficacy of short-duration 0.1 mg/ml and 0.2 mg/ml MMC in primary trabeculectomy for primary adult glaucoma. Graefes Arch Clin Exp Ophthalmol. 2015;253:1153–1159. doi:10.1007/s00417-015-3028-9
24. Khandelwal RR, Raje D, Rathi A, Agashe A, Majumdar M, Khandelwal R. Surgical outcome of safe surgery system trabeculectomy combined with cataract extraction. Eye. 2015;29:363–370. doi:10.1038/eye.2014.294
25. Lee SJ, Paranhos A, Shields MB. Does titration of mitomycin C as an adjunct to trabeculectomy significantly influence the intraocular pressure outcome? Clin Ophthalmol. 2009;3:81–87.
26. Mégevand GS, Salmon JF, Scholtz RP, Murray AD. The effect of reducing the exposure time of mitomycin C in glaucoma filtering surgery. Ophthalmology. 1995;102:84–90.
27. Kim YY, Sexton RM, Shin DH, et al. Outcomes of primary phakic trabeculectomies without versus with 0.5- to 1 min versus 3- to 5 min mitomycin C. Am J Ophthalmol. 1998;126:755–762.
28. Alwitry A, Abedin A, Patel V, Moodie J, Rotchford A, King A. Primary low-risk trabeculectomy augmented with low-dose mitomycin-C. Eur J Ophthalmol. 2018;19:971–976. doi:10.1177/112067210901900612
29. Annen DJ, Stürmer J. Follow-up of a pilot study of trabeculectomy with low dosage mitomycin C (0.2 mg/ml for 1 min). Independent evaluation of a retrospective nonrandomized study. Klin Monbl Augenheilkd. 1995;206:300–302. doi:10.1055/s-2008-1035446
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
