Back to Journals » Clinical, Cosmetic and Investigational Dermatology » Volume 17
Eruptive Cherry Angiomas After Nitrogen Mustard Treatment for Vitiligo
Received 10 November 2023
Accepted for publication 8 February 2024
Published 28 February 2024 Volume 2024:17 Pages 477—481
DOI https://doi.org/10.2147/CCID.S446781
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Jeffrey Weinberg
Huiying Wang,1 Ruzhi Zhang2
1Department of Dermatology, The Third Affiliated Hospital of Soochow University, Changzhou, People’s Republic of China; 2Department of Dermatology, The Second Affiliated Hospital of Wannan Medical College, Wuhu, People’s Republic of China
Correspondence: Ruzhi Zhang, Department of Dermatology, The Second Affiliated Hospital of Wannan Medical College, Wuhu, 241000, People’s Republic of China, Tel +8618761161826, Email [email protected]
Abstract: A 43-year-old male was diagnosed with vitiligo and had been treated with topical nitrogen mustard at the age of 13. Following two years of treatment, eruptive cherry angiomas developed and presented as widely distributed red papules throughout his trunk and proximal limbs. Ceasing the use of nitrogen mustard slowed the emergence of lesions. This case highlights the potential adverse effects associated with nitrogen mustard treatment in individuals with susceptibility, as it may lead to the onset of eruptive cherry angiomas.
Keywords: eruptive cherry angiomas, nitrogen mustard, vitiligo
Introduction
Cherry angiomas (CAs), also known as senile angiomas due to their association with aging, are the most common vascular skin tumors. They typically emerge in individuals aged 40 and older and are characterized by a gradual increase in both quantity and size over time. The etiology and pathogenesis of CAs are not yet fully elucidated. Recent studies suggested a potential involvement of HHV-8 in the development of CAs, particularly in individuals with systemic or local immunosuppression.1 Decreased expression of mir-424 and elevated levels of MEK1 or cyclin E1 in CAs may contribute to abnormal cell proliferation within the tumor.2 Additionally, GNAQ- and GNA11-mediated signaling pathways are implicated in the pathogenesis of CAs.3 Eruptive cherry angioma (ECA) refers to the sudden and widespread emergence of cherry angiomas. This condition is relatively uncommon, with only limited reported cases. The presentation of ECA has been found to be associated with advanced age, skin malignancies such as cutaneous melanoma, and chronic iatrogenic immunosuppression. The disruption in skin immune competence, arising from both iatrogenic influences and age-related changes, could potentially serve as a significant predisposing factor in the development of CAs.4
In this report, we present a case involving a vitiligo patient, who developed ECA subsequent to the administration of nitrogen mustard at a young age. This case highlights the potential adverse effects associated with NM treatment in individuals with susceptibility, as it may lead to the onset of ECA.
Case Presentation
At the age of 13, a 43-year-old male was diagnosed with vitiligo and received treatment with a nitrogen mustard solution at a local hospital. After using this solution, the patient experienced mild itching and redness in the vitiliginous areas, subsequently leading to the emergence of partial repigmented spots. However, one year later, the patient also observed the development of several red papules near the treated areas, and these papules gradually increased in number over time. As a result of this unforeseen progression, the patient discontinued the two-year vitiligo treatment. At that time, hundreds of papules had already appeared. After discontinuation of nitrogen mustard treatment, a noticeable deceleration in the growth rate of papules was observed. Subsequently, the patient did not pursue specialized treatment for vitiligo or ECA. Notably, the patient has a family history of lupus erythematosus in his father, with no other noteworthy medical history, such as malignancy. Imaging studies of internal organs and detection of HHV-8 revealed no evident abnormalities.
Physical examination revealed over 400 dome-shaped, smooth-surfaced, and ruby-colored papules on his chest, abdomen, back, and proximal extremities. The diameters of these papules ranged from 1 to 5 mm (Figure 1). Dermatoscopy revealed the homogeneous red background containing clustered red lacunae (Figure 2). A biopsy of a chest lesion was performed, revealing dilated vascular spaces in the papillary dermis and superficial dermis. These vessels exhibited erythrocyte congestion, in accordance with the diagnosis of cherry angiomas (Figure 3).
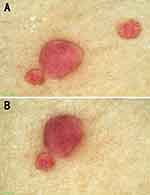 |
Figure 2 (A and B) The homogeneous red background containing clustered red lacunae (black arrows) was observed. |
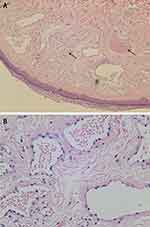 |
Figure 3 (A) A biopsy revealed dilated vascular spaces (black arrows) in the papillary dermis and superficial dermis (H&E, 100x). (B) These vessels exhibited erythrocyte congestion (H&E, 400x). |
Discussion
In this case, the patient presented with a rare and remarkable condition characterized by the rapid development of hundreds of cherry angioma (CA) lesions at a young age, occurring over just several months. Given the patient’s age, typical age-related factors are unlikely to be contributing to this condition, prompting consideration of immune-related factors as the potential underlying cause. After two years of nitrogen mustard (NM) treatment, the patient rapidly developed numerous CA lesions. Notably, upon discontinuing NM usage, the growth of these lesions appeared to slow down. This observation strongly prompted the suspicion of a potential association between the use of NM and the development of eruptive cherry angioma (ECA). Furthermore, it is plausible that the growth pattern of these lesions post-discontinuation may be influenced by age-related factors.
Ma et al reported a case of ECAs in a vitiligo patient following nitrogen mustard (NM) use, with lesion progression halting upon drug discontinuation.5 Similarly, Zhu et al described another case of ECAs and concurrent skin malignancy in a long-term NM user.6 Upon comparison, we have identified common features in these patients: a higher number of lesions burden at a younger age, often around or within vitiligo lesions and treatment sites. These reports substantiate the link between ECAs and NM utilization in vitiligo treatment.
NM is a topical chemotherapeutic agent frequently employed in vitiligo treatment. However, its use may lead to contact dermatitis, and prolonged exposure increases the risk of cutaneous malignancies, including basal cell carcinoma (BCC), squamous cell carcinoma (SCC), keratoacanthoma, actinic keratosis, and lentigo maligna.6 There are now safer and more effective alternatives for treating vitiligo, such as topical treatments (corticosteroids and calcineurin inhibitors) and phototherapy. Consequently, the utilization of NM in vitiligo treatment has gradually waned. In a study involving human bronchial epithelial cells and pulmonary endothelial cells, exposure to NM vapor led to an upregulation of vascular endothelial growth factor A (VEGF-A) and placental growth factor (PlGF) expression compared to a phosphate-buffered solution (PBS) control.7 VEGF-A is known for its pro-angiogenic properties, promoting cell migration and increasing vascular permeability, while PlGF is also capable of inducing angiogenesis. Hence, it is reasonable to propose that the emergence of ECAs in this context could be associated with the heightened expression of VEGF-A and PlGF induced by the use of NM. Furthermore, the possible contribution of external stimuli triggered by NM and the concurrent disruption of the immune system may act as potential initiators.
Conclusion
The use of topical NM has significantly declined due to its potential for cutaneous complications, notably the relatively rare occurrence of ECAs. Physicians must exercise caution when considering NM therapy for vitiligo. Moreover, this case study may offer valuable insights into the etiology and pathogenesis of ECAs.
Data Sharing Statement
The data that support the findings of this study are available from the corresponding author upon request.
Consent Statement
Written informed consent was obtained from the patient for the publication of the case details including the images. Institutional approval was not required for publishing these case details.
Funding
This work was supported by the Frontier Technology of Cell Therapy Foundation of Changzhou Xitaihu, Grant/Award Number: 2022-P-014.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Borghi A, Benedetti S, Corazza M, et al. Detection of human herpesvirus 8 sequences in cutaneous cherry angiomas. Arch Dermatol Res. 2013;305:659–664. doi:10.1007/s00403-013-1346-5
2. Nakashima T, Jinnin M, Etoh T, et al. Down-regulation of mir-424 contributes to the abnormal angiogenesis via MEK1 and cyclin E1 in senile hemangioma: its implications to therapy. PLoS One. 2010;5:e14334. doi:10.1371/journal.pone.0014334
3. Klebanov N, Lin WM, Artomov M, et al. Use of targeted next-generation sequencing to identify activating hot spot mutations in cherry angiomas. JAMA Dermatol. 2019;155:211–215. doi:10.1001/jamadermatol.2018.4231
4. Borghi A, Minghetti S, Battaglia Y, Corazza M. Predisposing factors for eruptive cherry angiomas: new insights from an observational study. Int J Dermatol. 2016;55:e598–e600. doi:10.1111/ijd.13330
5. Ma HJ, Zhao G, Shi F, Wang YX. Eruptive cherry angiomas associated with vitiligo: provoked by topical nitrogen mustard? J Dermatol. 2006;33:877–879. doi:10.1111/j.1346-8138.2006.00200.x
6. Zhu LL, Zheng S, Wei H, et al. Multiple cutaneous malignancies and cherry hemangiomas in a vitiligo patient treated with topical nitrogen mustard. Dermatol Ther. 2014;27:52–54. doi:10.1111/dth.12045
7. McGraw MD, Kim SY, White CW, Veress LA. Acute cytotoxicity and increased vascular endothelial growth factor after in vitro nitrogen mustard vapor exposure. Ann N Y Acad Sci. 2020;1479:223–233. doi:10.1111/nyas.14367
 © 2024 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2024 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.

