Back to Journals » International Journal of Nanomedicine » Volume 9 » Supplement 1
Emerging nanotechnology approaches in tissue engineering and regenerative medicine
Authors Kim E, Ahn EH, Dvir T, Kim D
Received 23 January 2014
Accepted for publication 23 January 2014
Published 5 May 2014 Volume 2014:9(Supplement 1) Pages 1—5
DOI https://doi.org/10.2147/IJN.S61212
Checked for plagiarism Yes
Eung-Sam Kim, 1,2 Eun Hyun Ahn, 3,4 Tal Dvir, 5,6 Deok-Ho Kim 1,4,7
1Department of Bioengineering, University of Washington, Seattle, WA, USA; 2Department of Biological Sciences, Chonnam National University, Gwangju, Korea; 3Department of Pathology, 4Institute of Stem Cell and Regenerative Medicine, School of Medicine, University of Washington, Seattle, WA, USA; 5Department of Molecular Microbiology and Biotechnology, 6Center for Nanoscience and Nanotechnology, Tel Aviv University, Tel Aviv, Israel; 7Center for Cardiovascular Biology, University of Washington, Seattle, WA, USA
The history of human kind suggests that there has been a correlation between global population growth and major events in science and technology over the last three centuries. Sharp increases in the world’s population have been triggered by the industrial revolution and scientific and technological breakthroughs including: the advent of the railways, discovery of penicillin and deoxyribonucleic acid (DNA), and the invention of the computer.1 Since the 20th century, interdisciplinary areas in the physical and biological sciences have accelerated the progress of biomedical applications. The recent integration of emerging nanotechnology into biology and biomedicine has resulted in a range of innovative nanoengineering efforts for the repair and regeneration of tissues and organs.2 Thus, it is expected that nanoengineering approaches to biomedical applications can contribute to addressing the present issue of personal and global health care and its economic burden for more than 7 billion people.
Why are we paying attention to nanoengineering for biomedical applications? The size of most biomolecules ranges from 0.2 nm to 200 nm (Figure 1). Research has focused on control of the interaction and localization of biomolecules even at the single-molecule level using ever-evolving nanotechnology.3 The evidence indicates that cells can respond to nanoscale changes in the dynamic extracellular matrix and vice versa. Biomimetic nanopatterns alone can direct the differentiation of stem cells without involvement of exogenous soluble biochemical factors.4,5 This regulation of cellular behavior by nanotechnology is one of many examples demonstrating the significant applications of nanoengineering in biomedicine. This special issue includes four review papers and seven research articles that provide an insight into current nanoengineering approaches to the repair or regeneration of tissues and organs.
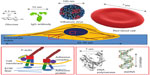  | Figure 1 Schematic size scale of biological objects.Abbreviations: dsDNA, double-stranded deoxyribonucleic acid; IgG, immunoglobulin G; ECM, extracellular matrix. |
Applications of multifunctional nanoparticles in biomedicine
Nanoparticles with a high surface to volume ratio are gaining attention because their physicochemical properties can be tailored to specific applications by changes in their size, shape, and surface chemistry.6 Moreover, synthesis of nanoparticles is fairly straightforward. Recent advances in nanotechnology have led to the development of multifunctional nanoparticles for theranostics and image-guided therapies, including drug delivery, molecular imaging, and cell labeling.
When targeting ligands are conjugated to the surface of nanoparticles into which small-molecule drugs have been loaded or encapsulated, these nanoparticles can be taken up by target cells inside which they unload their drug cargo. If the nanoparticle is magnetic, it can be used as a contrast agent for magnetic resonance imaging to monitor the distribution of drug-loaded nanoparticles. Superparamagnetic iron oxide nanoparticles coated with small interfering ribonucleic acid (siRNA) have been used in magnetic resonance imaging for visualization of accumulation of siRNA in tumor tissue in vivo.7 Furthermore, release of anticancer drugs loaded into magnetic core silica nanoparticles can be controlled by an external magnetic field.8 Hydrogel nanoparticles (or nanogels) were developed to protect and transport siRNA into diseased cells via the intravenous route.9 For image-guided cancer surgery, a near-infrared emitting polymer nanogel was efficient enough to map sentinel lymph nodes, which cancer cells are most likely to migrate to from a primary site.10 Biodegradable nanoparticles can serve as a protective delivery vehicle for therapeutic proteins that need to face a harsh environment prior to uptake in the gastrointestinal tract after oral administration.11 In addition, nanoparticles containing self-assembled chimeric proteins can stimulate dramatic tissue growth in the setting of chronic wounds. For example, elevation of temperature causes fusion of keratinocyte growth factors and elastin-like peptides to form nanoparticles.12
When cancer-specific or cell-specific ligands are conjugated to the surface of nanoparticles, they can label and track target cells in vivo. In a murine model, due to the long-term photostability of quantum dots, polyethylene glycol-encapsulated and Tat peptide-conjugated quantum dots were injected into the tail vein to visualize the distribution of transplanted mesenchymal stem cells. Quantum dot-labeled mesenchymal stem cells were located by fluorescence microscopy in the liver and spleen, but not in the brain and kidney.13 Iron oxide nanoparticles can track stem cells by noninvasive magnetic resonance imaging, which has high spatial resolution in comparison with other clinical imaging modalities.14
In this special issue, Galanzha et al report on iron oxide nanoparticles functionalized with a urokinase plasminogen activator to capture tumor cells circulating in the bloodstream of mice.15 Circulating tumor cells can be magnetically enriched under an external magnet and detected by photoacoustic imaging. Conventional ex vivo detection of circulating tumor cells is done using a small blood sample.15,16 Formation of DNA-functionalized gold nanoparticles causes a rapid color transition in solution, which enables visual detection of a single base mismatch.17
Applications of nanoengineered scaffolds in tissue growth and regenerative medicine
It is becoming increasingly evident that interaction between cells and their microenvironment at the nanoscale level can reorganize cytoskeleton and induce specific cell signaling that regulates the fate of the cell. Thus, nanostructured scaffolds that mimic the tissue-specific microenvironment have been of great interest in nanotechnology for tissue engineering and regenerative medicine. Scaffolds with biochemical, mechanical, and electrical properties similar to those of native tissues have been nanoengineered to enhance cell adhesion, proliferation, differentiation, and even maturation, thereby fostering cell function and tissue growth.18
An extracellular matrix-like architecture can be fabricated by nanopatterning, electrospinning, self-assembly, conjugation of adhesion motifs to the matrix backbone, or sulfating the matrix backbone.19 The properties of this extracellular matrix-like architecture can be adjusted by incorporation of nanomaterials such as carbon nanotubes, nanowires, and nanoparticles.20 For instance, You et al21 developed an electrically conductive hybrid hydrogel scaffold based on gold nanoparticles homogeneously synthesized throughout a polymer template gel. The expression of connexin-43 increased in neonatal cardiomyocytes grown on the scaffold, suggesting that an electrically active scaffold impregnated with gold can enhance cardiomyocyte function.21
Nanoscale topographical features (100 nm to 1 μm in size) defined on cell culture substrates can direct cell behavior, including polarity, migration, proliferation, and differentiation. For example, nanotopographical variations in the cell adhesion substrate can regulate differentiation of human mesenchymal stem cells towards adipocytes or osteocytes.22 Contact guidance cues from preferential parallel nanoridge-induced elongation and alignment of cells along the nanopattern can reorganize the actin cytoskeleton in response to the topographical pattern density.23,24 Engraftment of a nanoridged polyethylene glycol-based hydrogel scaffold was found to promote retention and growth of transplanted heart cells and their integration into host tissue in a rat model of myocardial infarction.5 Furthermore, a graphene oxide film coating on a glass slide was shown to enhance the adhesion and osteogenic differentiation of human adipose-derived stem cells.25 Systematic understanding of the mechanisms of spatiotemporal regulation of the mechanotransduction pathways involved in cell-matrix interactions will be useful for designing and fabricating further improved biomimetic nanoscaffolds that can even release bioactive reagents in a controlled manner in vivo. Engineering of cell sheets could also be a potential tool for constructing scaffold-free, three-dimensional tissues using the more responsive polymers.26,27
Papers in this special issue
Nanoscale topography can enhance tissue growth and control cell behavior. In this special issue, Alpaslan et al review the biomimetic advance represented by nanofeatured scaffold-based tissue engineering to improve the growth of hard and soft tissues, such as the bone and bladder. As an alternative nanotopographical cue, Alon et al coated a glass surface with silver nanoparticles. Growth of human neuroblastoma cells on this silver nanoparticle-coated substrate resulted in enhanced neurite outgrowth, suggesting that silver nanoparticles can be used as biocompatible nanomaterials for neuronal tissue engineering. In addition, Ebara et al demonstrated that adherent cells can sense and gradually adapt to dynamic changes in the topographical nanopatterns of a cell culture substrate fabricated from temperature-responsive poly(µ-caprolactone). The polymer film showed surface shape-memory transition at the melting temperature from a memorized temporal pattern to the original permanent pattern, while maintaining its wettability and surface charge.
Nanocarriers can control the release of bioactive reagents ranging from small chemicals to proteins. Julani et al decoupled and controlled these release profiles in response to temperature changes using dual drug-loaded bicompartmental nanofibers, which were fabricated using an electrohydrodynamic coinjecting system. Lim et al developed a peptide-based amphiphile nanomatrix that releases nitric oxide and promotes viability and functionality of pancreatic islet cells. The amphiphile peptide was self-assembled into a three-dimensional nanomatrix to provide cells with biomimetic and bioactive cues, such as sustained release of nitric oxide. La et al found that bone formation in a mouse with a calvarial defect was enhanced by local release of bone morphogenetic protein-2 and substance P using graphene oxide-coated titanium implants.
The molecular mechanisms of cellular uptake and excretion of nanosized particles are reviewed by Oh et al. The effects of nanoparticle size, shape, and surface chemistry on endocytosis and exocytosis in various cell types are summarized, providing guidelines for developing clinically safe nanoparticles for targeted drug delivery, bioimaging, and elimination from the body. Katagiri et al discuss present and potential strategies that could be used to develop stealth carbon nanotubes capable of evading opsonization and sequestration in the hepatobiliary system, with improved blood circulation time and biocompatibility.
Other studies have focused on the development of cell-loading peptide hydrogels, microwell arrays for monitoring cell–cell interactions, and optical stimulation of neurons. Kim et al encapsulated bone marrow-derived mesenchymal stem cells in self-assembled peptide hydrogels and showed the clinical potential of this nanostructured peptide-cell complex to prevent osteoarthritis of the knee in a rat model. Choi et al used polydimethylsiloxane-based microwell arrays to investigate antiproliferative effects of mesenchymal stem cells on CD4+ T-cells. These microwell arrays can generate a microenvironment to control and monitor real-time cell–cell communications, whereas most bulk arrays have limits with regards to reflecting the heterogeneous nature of mesenchymal stem cells. Bareket-Keren et al review recent advances in light-directed approaches for neuronal stimulation to improve retinal implants, which currently use electrical stimulation with extracellular electrodes.
Conclusion and perspectives
We have seen an exponential growth in science and technology since the 18th century. The industrial revolution was based on the principle of classical mechanics and allowed human kind to perform macroscale engineering feats, such as development of the Watt steam engine. Subsequent developments in microscale engineering led to the microelectronics revolution in the 20th century. The integration of biology and nanotechnology will significantly impact tissue engineering and regenerative medicine. If issues such as toxicity and biodistribution of organic or inorganic nanomaterials can be overcome, nanomaterial-based particles, nanostructured scaffolds, and drug delivery systems will revolutionize the diagnosis and treatment of human disease and allow regeneration of failing organs (Figure 2). Nanoengineering for well-defined and precisely controlled synthesis and fabrication of nanotechnological platforms will realize Feynman’s vision in the 1950s, ie, “there’s plenty of room at the bottom”.28
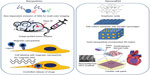  | Figure 2 Applications of nanomaterials in biomedicine.Abbreviations: QDs, quantum dots; IR, infrared; 3D, three-dimensional; PEG, polyethylene glycol; AC, alternating current. |
Acknowledgments
This work was supported by the new faculty startup fund at the University of Washington (to DHK), an American Heart Association Scientist Development grant (to DHK), and a Muscular Dystrophy Association research grant (to DHK).
Disclosure
The authors report no conflicts of interest in this work.
References
Fogel RW. Catching up with the economy. Am Econ Rev. 1999;89(1):1–21. | |
Kim HN, Jiao A, Hwang NS, et al. Nanotopography-guided tissue engineering and regenerative medicine. Adv Drug Deliv Rev. 2013;65(4):536–558. | |
Kim ES, Ahn EH, Chung E, Kim DH. Recent advances in nanobiotechnology and high-throughput molecular techniques for systems biomedicine. Mol Cells. 2013;36:477–484. | |
Kim DH, Provenzano PP, Smith CL, Levchenko A. Matrix nanotopography as a regulator of cell function. J Cell Biol. 2012;197(3):351–360. | |
Kim DH, Kshitiz, Smith RR, et al. Nanopatterned cardiac cell patches promote stem cell niche formation and myocardial regeneration. Integr Biol (Camb). 2012;4(9):1019–1033. | |
Brannon-Peppas L, Blanchette JO. Nanoparticle and targeted systems for cancer therapy. Adv Drug Deliv Rev. 2004;56(11):1649–1659. | |
Medarova Z, Pham W, Farrar C, Petkova V, Moore A. In vivo imaging of siRNA delivery and silencing in tumors. Nat Med. 2007;13(3):372–377. | |
Thomas CR, Ferris DP, Lee JH, et al. Noninvasive remote-controlled release of drug molecules in vitro using magnetic actuation of mechanized nanoparticles. J Am Chem Soc. 2010;132(31):10623–10625. | |
Smith MH, Lyon LA. Multifunctional nanogels for siRNA delivery. Acc Chem Res. 2012;45(7):985–993. | |
Noh YW, Kong SH, Choi DY, et al. Near-infrared emitting polymer nanogels for efficient sentinel lymph node mapping. ACS Nano. 2012;6(9):7820–7831. | |
Bakhru SH, Furtado S, Morello AP, Mathiowitz E. Oral delivery of proteins by biodegradable nanoparticles. Adv Drug Deliv Rev. 2013;65(6):811–821. | |
Koria P, Yagi H, Kitagawa Y, et al. Self-assembling elastin-like peptides growth factor chimeric nanoparticles for the treatment of chronic wounds. Proc Natl Acad Sci U S A. 2011;108(3):1034–1039. | |
Lei Y, Tang H, Yao L, Yu R, Feng M, Zou B. Applications of mesenchymal stem cells labeled with Tat peptide conjugated quantum dots to cell tracking in mouse body. Bioconjug Chem. 2008;19(2):421–427. | |
Hachani R, Lowdell M, Birchall M, Thanh NT. Tracking stem cells in tissue-engineered organs using magnetic nanoparticles. Nanoscale. 2013;5(23):11362–11373. | |
Galanzha EI, Shashkov EV, Kelly T, Kim JW, Yang L, Zharov VP. In vivo magnetic enrichment and multiplex photoacoustic detection of circulating tumour cells. Nat Nanotechnol. 2009;4(12):855–860. | |
Kim E-S, Kim S, Choi K, Han K-H. Micro-/nanotechnology-based isolation and clinical significance of circulating tumor cells. Biomed Eng Lett. 2012;2(2):78–87. | |
Han MS, Lytton-Jean AK, Oh BK, Heo J, Mirkin CA. Colorimetric screening of DNA-binding molecules with gold nanoparticle probes. Angew Chem Int Ed Engl. 2006;45(11):1807–1810. | |
Yang HS, Ieronimakis N, Tsui JH, et al. Nanopatterned muscle cell patches for enhanced myogenesis and dystrophin expression in a mouse model of muscular dystrophy. Biomaterials. 2014;35(5):1478–1486. | |
Kim DH, Lee H, Lee YK, Nam JM, Levchenko A. Biomimetic nanopatterns as enabling tools for analysis and control of live cells. Adv Mater. 2010;22(41):4551–4566. | |
Dvir T, Timko BP, Kohane DS, Langer R. Nanotechnological strategies for engineering complex tissues. Nat Nanotechnol. 2011;6(1):13–22. | |
You JO, Rafat M, Ye GJ, Auguste DT. Nanoengineering the heart: conductive scaffolds enhance connexin 43 expression. Nano Lett. 2011;11(9):3643–3648. | |
Ahn EH, Kim Y, Kshitiz, et al. Spatial control of adult stem cell fate using nanotopographic cues. Biomaterials. 2014;35(8):2401–2410. | |
Kim DH, Han K, Gupta K, Kwon KW, Suh KY, Levchenko A. Mechanosensitivity of fibroblast cell shape and movement to anisotropic substratum topography gradients. Biomaterials. 2009;30(29):5433–5444. | |
Kim DH, Seo CH, Han K, Kwon KW, Levchenko A, Suh KY. Guided cell migration on microtextured substrates with variable local density and anisotropy. Adv Funct Mater. 2009;19(10):1579–1586. | |
Kim J, Choi KS, Kim Y, et al. Bioactive effects of graphene oxide cell culture substratum on structure and function of human adipose-derived stem cells. J Biomed Mater Res A. 2013;101(12):3520–3530. | |
Takahashi H, Shimizu T, Nakayama M, Yamato M, Okano T. The use of anisotropic cell sheets to control orientation during the self-organization of 3D muscle tissue. Biomaterials. 2013;34(30):7372–7380. | |
Lee B, Jiao A, Yu S, You JB, Kim DH, Im SG. Initiated chemical vapor deposition of thermoresponsive poly(N-vinylcaprolactam) thin films for cell sheet engineering. Acta Biomater. 2013;9(8):7691–7698. | |
Feynman RP. There’s plenty of room at the bottom. J Microelectromech Syst. 1992;1:60–66. |
 © 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2014 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
