Back to Journals » Cancer Management and Research » Volume 11
EGFR mutations are significantly associated with visceral pleural invasion development in non-small-cell lung cancer patients
Authors Shi J, Yang Y, Zhao Y, Zhu J, Song X, Jiang G
Received 24 November 2018
Accepted for publication 31 January 2019
Published 1 March 2019 Volume 2019:11 Pages 1945—1957
DOI https://doi.org/10.2147/CMAR.S195747
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Antonella D'Anneo
Jinghan Shi, Yang Yang, Yanfeng Zhao, Junjie Zhu, Xiao Song, Gening Jiang
Department of Thoracic Surgery, Shanghai Pulmonary Hospital affiliated to Tongji University, Shanghai, China
Objectives: A retrospective study was performed to investigate the association between EGFR mutations and visceral pleural invasion (VPI), and evaluate the prognostic value of EGFR in resected non-small-cell lung cancer (NSCLC) patients with VPI.
Materials and methods: Clinicopathological characteristics and follow-up information were collected from 508 consecutive patients with surgically resected stage I–III NSCLC, and EGFR mutations were detected based on real-time PCR technology. Significant results (P<0.05) from univariate logistic regression analysis were involved as covariates to adjust confounding factors in the analysis of independent factors.
Results: VPI and EGFR mutations were detected in 229 (45.1%) and 243 (47.8%) cases in NSCLC, respectively. There was a significant association between EGFR mutations and VPI development. Both 19-del (adjusted OR =2.13, 95%CI =1.13–3.99, P=0.019) and L858R (adjusted OR =2.89, 95%CI =1.59–5.29, P=0.001) could significantly increase the risk of VPI development compared with EGFR wild-type. Higher frequency of L858R (adjusted OR =2.63, 95%CI =1.42–4.88, P=0.002) was detected in VPI patients compared with non-VPI patients. 19-del (adjusted HR =0.31, 95%CI =0.12–0.80, P=0.015) was an independent prognostic factor for a better disease-free survival (DFS) in non-VPI patients. No significant association was shown between EGFR mutations and DFS in VPI patients.
Conclusion: EGFR mutations were significantly associated with VPI development in NSCLC, but no significant association was observed between EGFR mutations and DFS in the patients with VPI. 19-del was a favorable prognostic factor for DFS in non-VPI patients.
Keywords: EGFR mutations, visceral pleural invasion, non-small-cell lung cancer, association study
Introduction
Lung cancer remains the most common cause of cancer-related mortality worldwide.1 Non-small-cell lung cancer (NSCLC) accounts for ~85% of lung cancer.2 Adenocarcinoma (ADC) and squamous cell carcinoma (SCC) are two major histological subtypes of NSCLC. Although conventional treatment strategies including surgery, chemotherapy, and radiotherapy have improved the prognosis of NSCLC patients, the side effects on life quality should not be ignored. In the past few decades, studies on signaling pathways involved in the onset and progression of NSCLC have acquired great achievements, especially the ectopic activation of EGFR which plays a crucial role in the tumor growth and invasiveness. About 40% of NSCLC patients presented the dysregulation of EGFR.3 The somatic mutations of EGFR prominently locate in the exons 19–23, which encode the tyrosine kinase domain.4 Approximately 70% of Asian female nonsmoker patients with ADC harbored EGFR mutations.5 Exon 19 deletions (19-del) and exon 21 missense mutation (L858R) are the two predominant mutant subtypes of EGFR in NSCLC. Tyrosine kinase inhibitors (TKIs), which specifically target EGFR mutations (19-del and L858R), could improve the prognosis of NSCLC patients harboring EGFR mutations, and have been recommended as the first-line therapy in lung cancer patients with EGFR mutations.6 Some recent studies presented that patients harboring 19-del had a better overall survival compared with those with L858R following TKIs treatment especially in advanced-stage NSCLC, while others failed to reach this conclusion.7–10 As a result, the difference of 19-del and L858R in the prognosis of patients harboring EGFR mutations remains controversial.
The condition that tumors adjacent to the pleural might be an unfavorable prognostic factor was first observed by Brewer et al in 1958.11 Compared with tumors growing in the mid-lung zone, tumors under pleural surface had an adverse influence on survival. Since that, visceral pleural invasion (VPI) has been identified as an adverse factor for the survival of patients who underwent NSCLC resection.12 In the eighth edition of AJCC TNM classification for lung cancer, VPI was an essential factor for the T descriptors – tumors ≤3cm will be upstaged to T2 stage if they invade the visceral pleural.13
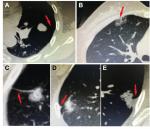
According to the previous researches, tumors with VPI presented more aggressive invasiveness, which may lead to the dissemination of tumor cells in the pleural cavity and mediastinal lymph node metastasis.14 A great number of studies have focused on the prognostic value of VPI stratified by the tumor size, especially the necessity of chemotherapy for postoperative patients with VPI in early stage NSCLC.12,15,16 Computer tomography (CT) is an important imaging method in diagnosis of NSCLC. Three types of tumor–pleural relationship could be observed on CT images including no contacting, abutting pleural, and pleural tag. Some studies suggested that tumors abutting the pleural surface can predict 77% of VPI in accuracy.17 Pleural tag, which represents the stripes stretching from the tumor margin to the pleural surface, is formed from thickened interlobular septa. This important CT feature could be classified into three types (pleural tag type I, type II, and type III) according to Hsu et al’s study. Tumors with pleural tags may be an important clue to prejudge VPI, and the positive predictive value is up to 76.2% according to different types observed on CT images.18 Although it is commonly recognized that tumors that contact the pleural presented on CT scans have large potential of VPI, it is still difficult to diagnose VPI from CT images accurately.
Numerous researches focused on the prognostic value of VPI stratified by tumor size, while limited studies shed light on the correlation of EGFR mutations and VPI. In order to clarify their relationship in NSCLC, a retrospective study was conducted to investigate the association between EGFR mutations and VPI, and evaluate their prognostic value in NSCLC patients who underwent primary tumor resection.
Materials and methods
Patient selection and follow-up
Seven hundred eleven consecutive patients who underwent primary tumor lobectomy and EGFR mutation detection were included in our current study to analyze the association between EGFR mutations and VPI as well as their roles in the prognosis of NSCLC patients in Shanghai Pulmonary Hospital from November 2013 to May 2014. The exclusion criteria were 1) pathological stage IV diagnosed after surgery; 2) administration of preoperative chemotherapy, radiation therapy, or EGFR-TKIs; 3) patients who died of surgical complications in perioperative period. Written informed consent was obtained from all patients before surgery, and the study was approved by the Review Board of Shanghai Pulmonary Hospital. This study was conducted in accordance with the Declaration of Helsinki. Finally, 508 patients with resected NSCLC were included in this study; clinicopathologic characteristics including gender, age, smoking status, tumor size, histological type, preoperative carcinoembryonic antigen (CEA) level, pathological TNM (pTNM) stage, and postoperative therapy were collected. Pathologic staging was performed according to the eighth edition of TNM classification.
Each patient received regular follow-up mainly in outpatient department or by telephone, and latest examination reports were recorded for rechecking. Consultant doctors checked radiological images of all patients who visited our outpatient department for postoperative follow-up and compared with the former ones. The examination reports for these patients were recorded in our hospital health system. For a minority of patients living far away, they would choose to be followed in the local hospital, and we would keep in touch with them by telephone every 3 months and enquired about their examination reports then recorded. For these patients, we checked their radiological reports instead. The disease-free survival (DFS) was defined as the time from the day of operation to the day of confirmation of recurrence according to clinical and radiological findings.
CT imaging, pathologic diagnosis, and EGFR mutation analysis
All patients underwent chest CT in our hospital within 1 month before surgery. CT images were performed using CT scanner systems (SIEMENS Somatom Definition AS; Siemens Medical Systems, Erlangen, Germany) according to the parameters as follows: section width, 2.0 mm; reconstruction interval, 1.0 mm; slice acquisition, 128×0.6 mm; rotation time, 0.5 seconds; tube voltage, 120 kVp; tube current, 300 mA. CT images were assessed by two experienced radiologists using standard lung (window width, 1,600 HU; window level, −600 HU) and mediastinal (window width, 350 HU; window level, 50 HU) window settings. The relationship of tumor and pleural on CT scans was recorded for each patient. For tumors showing pleural tags on lung window, we classified them into three types as previously reported by Hsu et al.18 Surgical specimens were reviewed by two experienced pathologists. For EGFR mutation analysis, genomic DNA was extracted from fresh tissues using QIAamp DNA Tissue Kit (Qiagen, Hilden, Germany). Mutations of EGFR were detected using commercially available kits from ACCB Diagnostics (Beijing, China). The procedure was based on amplification refractory mutation system real-time PCR technology. All experiments were performed according to the manufacturer’s instructions.
Statistical analysis
The differences in distribution of categorical variables were assessed by Pearson’s chi-squared test or the Fisher’s exact test. Independent t-test was carried out to evaluate the difference of mean value for continuous variables. Univariate logistic regression was performed to investigate the association between clinicopathologic characteristics and VPI, and multivariate logistic regression was carried out to analyze the independent risk factors for VPI.
Kaplan–Meier method was adopted to generate survival curve, and log-rank test was performed to compare the differences of survival curves between patients’ group. Cox regression model was used to assess the independent prognostic factors of VPI. Two-sided P-value <0.05 was considered statistically significant. All analyses were performed with SPSS version 17.0 (SPSS Inc., Chicago, IL, USA).
Results
Patients’ characteristics
To investigate the association between clinicopathologic characteristics and EGFR mutations as well as VPI, 508 NSCLC patients who underwent primary tumor resection in one tertiary care hospital were included. The patients’ characteristics are listed in Table 1. The median age was 61 years old (range 25–91), and 293 patients (57.7%) were >60 years when diagnosed. Male patients accounted for 52% of all cases. Sixty-one percent of patients were never smokers, and 39% of patients had a history of smoking. The mean tumor size was 27.3 mm, and most tumors were no more than 30 mm (70.1%), and only 7.1% of tumors were >50 mm. 77.6% of tumors did not contain ground-glass opacity (GGO). According to eighth edition of pathological TNM stage, 67.5% of patients were in stage I, 10.6% of patients were in stage II, and 21.9% of patients were in stage III. Twenty-two percent of patients (intrapulmonary lymph node-N1 4.7%, and mediastinal lymph node-N2 17.9%) were detected lymphatic metastasis. 80.9% of patients were diagnosed as ADC, 16.1% of patients were SCC. Based on the CT image, five different types describing tumor–pleural relationship were presented: no contacting (28.9%), abutting pleural (32.1%), pleural tag type I (13.6%), pleural tag type II (19.5%), and pleural tag type III (5.9%; Figure S1). According to pathological diagnosis, 45.1% of patients showed VPI. The results of EGFR mutation status showed that 19-del and L858R accounted for 19.5% and 24% of all the subtypes, respectively. 55.3% of patients received postoperative adjuvant therapy. The median follow-up time was 50 (range 3–57) months. The median DFS time was 49 (range 2–57) months.
Association between VPI and clinicopathologic characteristics
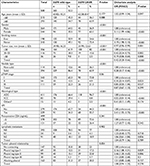
In order to investigate the potential correlation between VPI and clinicopathologic characteristics, the following data including age, gender, smoking status, tumor size, pTNM stage, histological type, tumor location, tumor–pleural relationship, lymphatic metastasis and preoperative CEA were collected to uncover the association between VPI and clinical factors. The results are listed in Table 2.
The distribution of gender, smoking status, nodule type, histological type, preoperative CEA level, EGFR mutations, pTNM stage, lymphatic metastasis, and tumor–pleural relationship showed significant differences between VPI and non-VPI groups. Significant difference in tumor size was also observed in the two groups (P=0.022). The results of univariate logistic regression analysis showed that all positive characteristics presented in chi-squared test were significantly associated with the increased risk of VPI development (OR >1.00, P<0.05), except histological type; SCC could significantly reduce the risk of VPI occurrence (OR =0.18, 95%CI =0.10–0.33, P<0.001) compared with ADC. After adjusting the covariates in multivariate logistic regression analysis, tumor size (OR =1.02, 95%CI =1.01–1.04, P=0.041), EGFR mutations (adjusted OR =2.13, 95%CI =1.13–3.99, P=0.019 for 19-del, and adjusted OR =2.89, 95%CI =1.58–5.29, P=0.001 for L858R compared to EGFR wild-type), and tumor–pleural relationship (adjusted OR =2.54, 95%CI =2.14–3.02, Ptrend<0.001) showed independent risk factors for VPI. SCC was an independent protective factor for the development of VPI (adjusted OR =0.17, 95%CI =0.06–0.43, P<0.001 compared with ADC).
Association between EGFR mutations and clinicopathologic characteristics
To further investigate the distribution of EGFR mutations in VPI and non-VPI patients, chi-squared test and logistic regression analysis were carried out to analyze the association between clinicopathologic characteristics and EGFR mutations. The covariate factors including age, gender, smoking status, tumor size, nodule type, pTNM stage, histological type, tumor–pleural surface relationship, lymphatic metastasis, and preoperative CEA were collected in this study. The results showed that gender, smoking status, tumor size, nodule type, histological type, tumor–pleural relationship, and VPI presented significant association with EGFR mutations (Table S1), including 19-del (Table S2) and L858R (Table S3).
The prevalence of EGFR mutations was 47.8% in NSCLC in this study. More female patients (61.7% vs 38.3%, P<0.001) and never smokers (74.1% vs 25.9%, P<0.001) harbored EGFR mutations. EGFR-mutant nodules tended to be much smaller (24.47±12.92 vs 29.90±16.23 mm, P<0.001) and contained GGO component (P<0.001; Table S1). Compared with nodules containing GGO component, the mutation rate of EGFR was significantly decreased in solid tumors (OR =0.39, 95%CI =0.24–0.65, P<0.001), and only L858R presented significant association with nodule type after adjusting covariates (adjusted OR =0.33, 95%CI =0.18–0.59, P<0.001) in subgroup analysis (Table 3). Analysis for histological type showed that 96.7% of EGFR mutations presented in ADC and the mutation rate of EGFR was 57.2% in ADC (Table S1). Compared with ADC, the mutation rates of both 19-del (adjusted OR =0.05, 95%CI =0.01–0.41, P<0.001) and L858R (adjusted OR =0.05, 95%CI =0.01–0.34, P=0.003) were significantly decreased in SCC (Table 3). VPI also showed significant association with EGFR mutations (adjusted OR =2.21, 95%CI =1.36–3.59, P=0.001), and higher frequency of EGFR mutations occurred in tumors with VPI (adjusted OR =2.21, 95%CI =1.36–3.59, P=0.001). Subgroup analysis showed that only L858R showed significant association with VPI (adjusted OR =2.63, 95%CI =1.42–4.88, P=0.002), 19-del presented potential significance level of P<0.05 with VPI (P=0.053). Lymphatic metastasis analysis showed that only 19-del was an independent risk factor for intrapulmonary lymph node (N1) metastasis (adjusted OR =5.02, 95%CI =1.59–15.81, P=0.006; Table 3).
Disease-free survival analysis in non-VPI and VPI patients
To investigate the association between survival and VPI as well as EGFR mutations, DFS analysis were carried out in this study. Survival curve analysis showed that VPI was significantly associated with DFS (log-rank P=0.047; Figure 1A). EGFR mutations presented better DFS compared with EGFR wild-type, but did not reach the significance level of 0.05 (Figure 1B). Subgroup analysis in VPI and non-VPI showed that whether 19-del or L858R was significantly associated with DFS (log-rank P=0.004 for 19-del, and log-rank P=0.024 for L858R, respectively; Figure 1C) in non-VPI group; however, no significant association was found between EGFR mutations and DFS (log-rank P>0.05) in VPI group (Figure 1D). There was no significant difference between VPI and non-VPI in EGFR wild-type group (Figure 1E). Survival curve analysis in EGFR mutation groups showed that the significant difference between VPI and non-VPI was present only in 19-del group (log-rank P=0.002), but not in L858R (log-rank P=0.090; Figure 1F).
The Cox regression analysis showed that after adjusting the significant factors in univariate analysis, EGFR 19-del (adjusted HR =0.31, 95%CI =0.12–0.80, P=0.015) could significantly decrease the risk of DFS, and was the independent prognosis factor for DFS in non-VPI group; preoperative CEA level (adjusted HR =2.32, 95%CI =1.40–3.85, P=0.001) and lymphatic metastasis (adjusted HR =1.78, 95%CI =1.36–2.32, Ptrend<0.001) were independent risk factors for DFS in non-VPI group. In VPI group, only lymphatic metastasis (adjusted HR =2.36, 95%CI =1.78–3.14, Ptrend<0.001) was the independent risk factor for DFS (Table 4).
Discussion
EGFR is one of the most well-studied mutant genes in NSCLC. According to a recent meta-analysis, the average mutation rate of EGFR ranged from 9.6% to 82.2% worldwide, which was higher in Asian population than in other races on average (43.5% vs 37.9%).19 VPI was considered as an adverse prognostic factor in NSCLC; for this reason, tumors <3 cm will be upstaged to T2 stage if they invade the visceral pleural according to the eighth edition of AJCC TNM classification for lung cancer.13 However, limited researches were focused on the association between EGFR mutations and VPI. In this retrospective study, 508 patients who underwent primary tumor lobectomy were enrolled to analyze the association between EGFR mutations and VPI as well as their roles in the prognosis of NSCLC patients. Our results showed that 47.8% NSCLC patients harbored EGFR mutations, among which 19-del and L858R are the two main subtypes. 45.1% of patients presented VPI. EGFR mutations were significantly associated with VPI, and higher frequency of EGFR mutations was found in VPI patients. Tumor–pleural relationship indicated to be an important CT feature as it showed great association with VPI. In 19-del group, VPI showed significant association with DFS and was identified to be an independent risk factor. At the same time, 19-del was found to have an important prognostic value for a better DFS in non-VPI patients. Lymphatic metastasis was an independent risk factor for DFS in both VPI and non-VPI patients.
Tumor that abuts the pleural surface does not necessarily mean pleural invasion. The possibility of VPI can be further predicted by 1) the obtuse angle between pleural surface and lesion; 2) contact length >3 cm, and 3) thickening of adjacent pleural. The combination of all these image markers can reach a sensitivity of 87% and a specificity of 68%.20 Hsu et al18 first reported that for tumors that do not abut the pleural surface, pleural tags might be a sensitive predictor of VPI on CT image. The authors classified pleural tags into three types in their study, and showed that the pleural tag increased its accuracy of VPI invasion from the order of type I, type III, and type II, which was consistent with ours. Our study also showed that no VPI was diagnosed in the tumors without pleural contact on CT scans, and tumors abutted the pleural surface was an independent risk factor for the development of VPI.
The influence of VPI on the prognosis of resected NSCLC patients has not reached an agreement. Some previous studies revealed that VPI was recognized as an adverse independent factor for the prognosis in NSCLC patients who underwent tumor resection.15,21 David et al excluded the association between VPI and OS or DFS in tumors <5 cm.22 Yanagawa et al found that VPI failed to be an independent predictor for decreased survival in resected stage I patient.23 In our study, we observed that VPI had an adverse effect on the DFS in NSCLC patients. Further stratified analysis in VPI and non-VPI subgroups identified that only regional lymph node metastasis actually decreased survival independently in VPI group. In non-VPI group, EGFR mutation was an independent factor for a better DFS.
Patients with EGFR mutation are recommended for TKIs target therapy; however, the prognostic value of EGFR mutations in resected NSCLC patients is still under debate. Kim et al suggested that EGFR mutation is not a prognostic factor for patients after tumor resection,24 whereas the research published by Takamochi et al8 demonstrated that EGFR mutations contributed to better DFS and OS. Lee et al25 retrospectively reviewed 117 patients who underwent curative resection and pointed out that EGFR mutations may benefit patients’ DFS. D’Angelo et al26 studied 1,118 postoperative patients with stage I–III lung cancers and showed that EGFR mutations lowered death rate significantly. The underlying mechanism for this prognostic significance of EGFR mutations in NSCLC remains unclear. It is speculated that the progression of EGFR-mutated lung cancer cells was less likely to be interacted with other oncogenes, making the survival of these patients better than those who exhibit higher nonsynonymous mutation burden.27
Exon 19 (19-del) and Exon 21 (L858R) account for >80% of EGFR gene mutations. Li et al reported that mutation frequencies in exon 19 were significantly higher in early stage lung cancer (I/II), whereas L858R were more common in tumors having lymph node metastasis and late stage (III/IV).28 Renaud et al analyzed 108 19-del and 88 L858R surgically treated NSCLC patients and concluded that exon19 confers a better OS than exon 21 in stage II and III NSCLC.7 However, Takamochi et al compared the prognosis between L858R and 19-del in resected tumors, and no differences in OS and DFS were observed.8 Our data showed that both 19-del and L858R could significantly increase the DFS in non-VPI patients. However, after adjusting for covariates, only 19-del was identified as an independent prognostic factor for better DFS in non-VPI patients. No significant results were found in VPI group. It is possible that in VPI patients, the merit of EGFR mutations in improving DFS could be eliminated by strong invasive potential to visceral pleural; whereas, the favorable prognostic significance of EGFR mutations, especially 19-del, could be displayed in patients without VPI.
EGFR mutations occur more frequently in patients with malignant pleural effusion than those without,29 which may indicate that EGFR mutation is closely related to the invasiveness of NSCLC. To the best of our knowledge, few studies have analyzed the relationship between VPI and EGFR mutations. Le et al30 suggested that EGFR signaling pathway may promote the development of VPI through its downstream effector miR-135b. Hence, the clinical meaning of EGFR mutations correlated with VPI should be studied. Lin et al observed no association between EGFR mutations and visceral pleural surface invasion in 172 patients with tumors no more than 2 cm.31 In our study, we found that both 19-del and L858R were significantly associated with VPI as well as independent risk factors for VPI. Furthermore, we identified that the frequency of EGFR mutations was increased from the order of pleural tag type I, type III, and type II, which consisted with the predictive accuracy of VPI occurrence. This result infers that EGFR mutations might play important roles in the development of VPI. We also demonstrated that only 19-del was an independent factor for the increased DFS in non-VPI patient.
Our study also has several limitations. The data were based on a retrospective study in a single center, which may cause selection bias inevitably. We did not show the depth of VPI infiltration using elastic staining. VPI-positive patients in our study were defined as PL1 and PL2.32 Besides, we did not show the overall survival in this study. A variety of methods and therapeutic strategies after disease recurrence including chemotherapy, radiotherapy, target therapy, and palliative care were decided by patients, and we considered that this discrepancy may interfere with the overall survival.
Conclusion
In this study, we identified that there was a significant association between EGFR mutations and VPI. EGFR mutations could significantly increase the risk of VPI, and higher frequency of L858R was detected in VPI patients. EGFR mutations might not benefit the survival of patients with VPI, but conversely 19-del was an independent favorable prognostic factor for DFS in patients without VPI.
Acknowledgment
This work was supported by the Shanghai Hospital Development Center (grant number: 16CR2016A, 2017).
Disclosure
The authors report no conflicts of interest in this work.
References
Siegel R, Ma J, Zou Z, Jemal A. Cancer statistics, 2014. CA Cancer J Clin. 2014;64(1):9–29. | ||
Travis WD, Brambilla E, Riely GJ. New pathologic classification of lung cancer: relevance for clinical practice and clinical trials. J Clin Oncol. 2013;31(8):992–1001. | ||
Dacic S, Shuai Y, Yousem S, Ohori P, Nikiforova M. Clinicopathological predictors of EGFR/KRAS mutational status in primary lung adenocarcinomas. Mod Pathol. 2010;23(2):159–168. | ||
Yang SY, Yang TY, Chen KC, et al. EGFR L858R mutation and polymorphisms of genes related to estrogen biosynthesis and metabolism in never-smoking female lung adenocarcinoma patients. Clin Cancer Res. 2011;17(8):2149–2158. | ||
Ren S, Kuang P, Zheng L, et al. Analysis of driver mutations in female non-smoker Asian patients with pulmonary adenocarcinoma. Cell Biochem Biophys. 2012;64(2):155–160. | ||
Keedy VL, Temin S, Somerfield MR, et al. American Society of clinical oncology provisional clinical opinion: epidermal growth factor receptor (EGFR) mutation testing for patients with advanced non-small-cell lung cancer considering first-line EGFR tyrosine kinase inhibitor therapy. J Clin Oncol. 2011;29(15):2121–2127. | ||
Renaud S, Seitlinger J, Guerrera F, et al. Prognostic value of exon 19 versus 21 EGFR mutations varies according to disease stage in surgically resected non-small cell lung cancer adenocarcinoma. Ann Surg Oncol. 2018;25(4):1069–1078. | ||
Takamochi K, Oh S, Matsunaga T, Suzuki K. Prognostic impacts of EGFR mutation status and subtype in patients with surgically resected lung adenocarcinoma. J Thorac Cardiovasc Surg. 2017;154(5):1768–1774. | ||
Lee CK, Wu YL, Ding PN, et al. Impact of specific epidermal growth factor receptor (EGFR) mutations and clinical characteristics on outcomes after treatment with EGFR tyrosine kinase inhibitors versus chemotherapy in EGFR-mutant lung cancer: a meta-analysis. J Clin Oncol. 2015;33(17):1958–1965. | ||
Kuan FC, Kuo LT, Chen MC, et al. Overall survival benefits of first-line EGFR tyrosine kinase inhibitors in EGFR-mutated non-small-cell lung cancers: a systematic review and meta-analysis. Br J Cancer. 2015;113(10):1519–1528. | ||
Brewer LA, Bai AF, Little JN, Rabago Y, Pardo G. Carcinoma of the lung; practical classification for early diagnosis and surgical treatment. J Am Med Assoc. 1958;166(10):1149–1155. | ||
Lakha S, Gomez JE, Flores RM, Wisnivesky JP. Prognostic significance of visceral pleural involvement in early-stage lung cancer. Chest. 2014;146(6):1619–1626. | ||
Rami-Porta R, Bolejack V, Crowley J, et al. The IASLC lung cancer staging project: proposals for the revisions of the T descriptors in the forthcoming eighth edition of the TNM classification for lung cancer. J Thorac Oncol. 2015;10(7):990–1003. | ||
Manac’h D, Riquet M, Medioni J, Le Pimpec-Barthes F, Dujon A, Danel C. Visceral pleura invasion by non-small cell lung cancer: an underrated bad prognostic factor. Ann Thorac Surg. 2001;71(4):1088–1093. | ||
Chang YL, Lin MW, Shih JY, Wu CT, Lee YC. The significance of visceral pleural surface invasion in 321 cases of non-small cell lung cancers with pleural retraction. Ann Surg Oncol. 2012;19(9):3057–3064. | ||
Tian D, Pei Y, Zheng Q, et al. Effect of visceral pleural invasion on the prognosis of patients with lymph node negative non-small cell lung cancer. Thorac Cancer. 2017;8(2):97–105. | ||
Ebara K, Takashima S, Jiang B, et al. Pleural invasion by peripheral lung cancer: prediction with three-dimensional CT. Acad Radiol. 2015;22(3):310–319. | ||
Hsu JS, Han IT, Tsai TH, et al. Pleural tags on CT scans to predict visceral pleural invasion of non-small cell lung cancer that does not abut the pleura. Radiology. 2016;279(2):590–596. | ||
Zhang SM, Zhu QG, Ding XX, et al. Prognostic value of EGFR and KRAS in resected non-small cell lung cancer: a systematic review and meta-analysis. Cancer Manag Res. 2018;10:3393–3404. | ||
Glazer HS, Duncan-Meyer J, Aronberg DJ, Moran JF, Levitt RG, Sagel SS. Pleural and chest wall invasion in bronchogenic carcinoma: CT evaluation. Radiology. 1985;157(1):191–194. | ||
Neri S, Yoshida J, Ishii G, et al. Prognostic impact of microscopic vessel invasion and visceral pleural invasion in non-small cell lung cancer: a retrospective analysis of 2657 patients. Ann Surg. 2014;260(2):383–388. | ||
David E, Thall PF, Kalhor N, et al. Visceral pleural invasion is not predictive of survival in patients with lung cancer and smaller tumor size. Ann Thorac Surg. 2013;95(6):1872–1877. | ||
Yanagawa N, Shiono S, Abiko M, Ogata SY, Sato T, Tamura G. Prognostic impact and initial recurrence site of lymphovascular and visceral pleural invasion in surgically resected stage I non-small-cell lung carcinoma. Eur J Cardiothorac Surg. 2013;44(3):e200–e206. | ||
Kim YT, Seong YW, Jung YJ, et al. The presence of mutations in epidermal growth factor receptor gene is not a prognostic factor for long-term outcome after surgical resection of non-small-cell lung cancer. J Thorac Oncol. 2013;8(2):171–178. | ||
Lee YJ, Park IK, Park MS, et al. Activating mutations within the EGFR kinase domain: a molecular predictor of disease-free survival in resected pulmonary adenocarcinoma. J Cancer Res Clin Oncol. 2009;135(12):1647–1654. | ||
D’Angelo SP, Janjigian YY, Ahye N, et al. Distinct clinical course of EGFR-mutant resected lung cancers: results of testing of 1118 surgical specimens and effects of adjuvant gefitinib and erlotinib. J Thorac Oncol. 2012;7(12):1815–1822. | ||
Govindan R, Ding L, Griffith M, et al. Genomic landscape of non-small cell lung cancer in smokers and never-smokers. Cell. 2012;150(6):1121–1134. | ||
Li M, Zhang Q, Liu L, et al. The different clinical significance of EGFR mutations in exon 19 and 21 in non-small cell lung cancer patients of China. Neoplasma. 2011;58(1):74–81. | ||
Zou J, Bella AE, Chen Z, et al. Frequency of EGFR mutations in lung adenocarcinoma with malignant pleural effusion: implication of cancer biological behaviour regulated by EGFR mutation. J Int Med Res. 2014;42(5):1110–1117. | ||
Le H, Wang X, Zha Y, et al. Peripheral lung adenocarcinomas harboring epithelial growth factor receptor mutations with microRNA-135b overexpression are more likely to invade visceral pleura. Oncol Lett. 2017;14(6):7931–7940. | ||
Lin MW, Wu CT, Shih JY, Chang YL, Yang PC. Clinicopathologic characteristics and prognostic significance of EGFR and p53 mutations in surgically resected lung adenocarcinomas ≤2 cm in maximal dimension. J Surg Oncol. 2014;110(2):99–106. | ||
Travis WD, Brambilla E, Rami-Porta R, et al. Visceral pleural invasion: pathologic criteria and use of elastic stains: proposal for the 7th edition of the TNM classification for lung cancer. J Thorac Oncol. 2008;3(12):1384–1390. |
Supplementary materials
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.