Back to Journals » Therapeutics and Clinical Risk Management » Volume 15
Efficacy of denosumab therapy for a 21-year-old woman with Prader-Willi syndrome, osteoporosis and history of fractures: a case report
Authors Uehara M, Nakamura Y, Takahashi J , Suzuki T, Iijima M, Arakawa Y , Ida K, Kosho T , Kato H
Received 7 September 2018
Accepted for publication 22 January 2019
Published 25 February 2019 Volume 2019:15 Pages 303—307
DOI https://doi.org/10.2147/TCRM.S186855
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Garry Walsh
Masashi Uehara,1 Yukio Nakamura,1 Jun Takahashi,1 Takako Suzuki,1 Mari Iijima,2 Yuko Arakawa,3 Koichi Ida,4 Tomoki Kosho,5,6 Hiroyuki Kato1
1Department of Orthopaedic Surgery, Shinshu University School of Medicine; 2Department of Clinical Nutrition, Shinshu University School of Medicine; 3Department of Dentistry and Oral Surgery, Shinshu University School of Medicine; 4Department of Obstetrics and Gynecology, Shinshu University School of Medicine; 5Department of Medical Genetics, Shinshu University School of Medicine; 6Center for Medical Genetics, Shinshu University Hospital, Matsumoto, Japan
Abstract: Appropriate management for osteoporosis in adult patients with Prader-Willi syndrome (PWS) has not been established. We report on a 21-year-old woman with PWS, who underwent denosumab treatment for osteoporosis. She presented with fractures and was shown to have very low bone mineral density (BMD), while she had been treated with supplementation of growth hormone for 7–14 years of age and estrogen from 15 years of age. BMD was monitored in the total hip region by dual-energy X-ray absorptiometry. Laboratory tests included bone-specific alkaline phosphatase, urinary type I collagen amino-terminal telopeptide, tartrate-resistant acid phosphatase 5b, 1-alpha, 25-dihydroxyvitamin D3, and parathyroid hormone. BMD and laboratory data were evaluated before and at 4, 8, and 13 months of treatment. After 13 months of denosumab therapy, BMD increased by 4.5%, and bone turnover markers notably improved. No fractures occurred. To the best of our knowledge, this is the first report to describe the clinical outcomes of denosumab treatment for osteoporosis in patients with PWS. Based on our findings, denosumab could represent an effective treatment option for osteoporosis in PWS patients.
Keywords: bone mineral density, denosumab, fracture, case report, osteoporosis
Introduction
Prader-Willi syndrome (PWS) is characterized by poor feeding, hypotonia, and developmental delay in infancy, and intellectual disability, autistic behavior, hyperphagia, obesity, and hypogonadism thereafter. A decrease in bone density is common in adolescent to adult patients with PWS, which is associated with a high risk of fracture.1–3 One of the causes of this bone mineral density (BMD) reduction may be related to growth hormone (GH) and decreased gonadal function.4
Prolonged growth hormone treatment has a beneficial effect on BMD with a trend for a higher BMD in individuals with uniparental disomy.5 In a recent long-term GH study, Bakker et al6 reported that BMD remained stable in prepubertal children with PWS but decreased during adolescence due to incomplete pubertal development. Based on their findings, they suggest treating females with estrogen replacement from 11 years of age and males with testosterone from 14 years of age.6 As far as we know, there have been no reports on the efficacy of GH or anti-resorption drug for PWS with osteoporosis.
Denosumab is an anti-receptor activator of nuclear factor-kappaβ (RANK) ligand monoclonal antibody approved for the treatment of osteoporosis and prevention of skeletal metastatic complications.7 To date, there are no published accounts of denosumab treatment for PWS complicated with osteoporosis, and it remains controversial as to whether osteoporotic treatment is suitable for osteoporotic PWS patients. Although bisphosphonates are the first-line drugs for osteoporosis, the use of these medications in adolescents and young adults remains controversial.8 Studies have demonstrated the effectiveness of denosumab for bisphosphonate (BP) refractory cases in children and young adults.9–12 Therefore, we evaluated denosumab in this study.
In this report, we describe the clinical results of a 21-year-old woman with PWS accompanied by osteoporosis and a history of fractures. Improvements in BMD and bone metabolic markers were observed over 1 year of denosumab treatment.
Case presentation
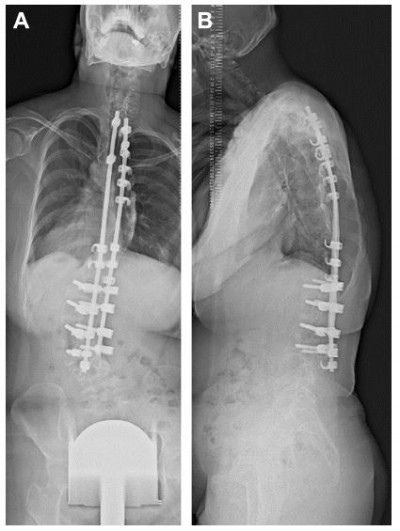
A 21-year-old woman with PWS was referred to our department for osteoporosis treatment. Her BMD and laboratory data are presented in Tables 1 and 2, respectively. She had exhibited hypotension at birth, and she was diagnosed after the confirmation of the deletion of the region located at the proximal part of chromosome 15 which is responsible for PWS. She underwent posterior spinal correction surgery for scoliosis at 12 years of age (Figure 1). She received growth hormone treatment from the age of 7–14 years due to the hypogonadism. Estrogen monotherapy was commenced at 15 years of age, and progestin therapy has been combined with this from 17 years of age. The patient’s history revealed two previous fragility fractures to the knee and one to the toe around the age of 18 years after falling.
  | Table 1 Patient characteristics and the change of total hip bone mineral density |
At age 21 years, she was referred to our hospital for transition from a pediatric clinic-based management to a multidisciplinary team approach. Careful nutritional support based on detailed dietary assessment as well as comprehensive dental care was initiated. Osteoporotic treatment was initiated for the patient’s diminished lumbar and proximal femur BMD values due to past fragility fractures. Denosumab was injected subcutaneously every 6 months (ie, at 0, 6, and 12 months), while the bone metabolism marker and BMD values were examined before and at 4, 8, and 13 months of treatment. Serum tartrate-resistant acid phosphatase 5b (TRACP-5b) and urinary type I collagen amino-terminal telopeptide (NTX) greatly decreased after the first administration (Figure 2). Serum bone-specific alkaline phosphatase (BAP) and type I procollagen N-terminal propeptide (PINP) decreased as well, while the whole- parathyroid hormone (PTH) and 1-alpha, 25-dihydroxyvitamin D3 (1,25[OH]2D) increased (Figure 2). Hypocalcemia was not observed during the observational period.
At 13 months of denosumab treatment, her total hip BMD T-score (the number of standard deviations above (+) or below (−) mean peak young adult BMD) improved from −2.3 to −2.0, and the BMD increased by 4.5% for total hips at the end of our study (Figure 3). The change of femoral neck BMD was also similar (Figure 3). There were no fractures and falls during the treatment period. Since she previously underwent surgery for scoliosis, the lumbar BMD was excluded for diagnosis and evaluation of osteoporosis.
This study was approved by the institutional ethical review board of Shinshu University School of Medicine prior to its start and was conducted in accordance with the ethical standards set forth in the 2013 Declaration of Helsinki for research involving human subjects. Written informed consent was obtained from the patient for publication prior to this study.
Discussion
This report evaluates the clinical outcomes of a patient receiving denosumab treatment for PWS complicated by osteoporosis. At 13 months of treatment, total hip BMD values had increased by 4.5%. No fractures or adverse effects occurred during the therapy, and her bone metabolism markers noticeably improved. To our knowledge, this study is the first of its kind describing the successful use of osteoporotic treatment denosumab for a patient with PWS and a history of fractures.
In adolescence and adulthood, decreased bone mineral density is common in PWS patients.1–3 Butler et al found low bone mass or osteopenia in four of the nine subjects with PWS over the age of 20 years.4 Past reports have described that growth hormone and gonadal hormone treatment is effective for elevating BMD in young PWS patients.6,13 However, a recent report suggested that GH treatment is ineffective in adult PWS patients.14 Our patient exhibited low BMD and revealed frequent fractures despite having already undergone GH and gonadal hormone treatments.
As denosumab also inhibits bone resorption, the humanized monoclonal antibody is a strong candidate for BMD improvement in growth hormone-unresponsive cases, although there have been no reports on denosumab for PWS and osteoporosis. We have demonstrated the effectiveness and safety of denosumab for BP refractory cases in children and young adults.9–12 In our patient, BMD values were very low, and the patient fractured her leg and foot after growth hormone treatment. As a result, we started denosumab treatment to prevent ensuing fractures and improve BMD after a careful discussion with her and her family. At 4 months of therapy, total hip BMD decreased since her activities were very low at that time due to the deterioration of her intellectual disabilities. However, at 13 months of denosumab treatment, total hip BMD values increased, with no additional fractures, although her low activities and the deterioration of her intellectual disabilities were not changed.
Bone resorption markers, such as urinary NTX and TRACP-5b, and bone formation markers, including PINP and BAP, are often used to monitor the effects of osteoporosis treatment.15 BAP and PINP decreased to approximately 50% with denosumab, while TRACP-5b decreased to approximately 22% with slight increase in NTX. These results indicated that denosumab strongly suppressed bone resorption in our PWS patient with accompanying osteoporosis and presumably produced her gains in bone density.
Limitations
The limitation of this study was the lack of literature on long-term effects of denosumab on PWS with osteoporosis and its sample size. Further assessment of this case will also be necessary to validate our findings.
Conclusion
Thirteen months of denosumab treatment improved BMD values and bone turnover markers in a PWS patient with osteoporosis. The drug, therefore, represents an effective treatment option in such cases and warrants further study.
Disclosure
The authors report no conflicts of interest in this work.
References
Höybye C, Hilding A, Jacobsson H, Thorén M. Metabolic profile and body composition in adults with Prader-Willi syndrome and severe obesity. J Clin Endocrinol Metab. 2002;87(8):3590–3597. | ||
Sinnema M, Maaskant MA, van Schrojenstein Lantman-de Valk HMJ, et al. Physical health problems in adults with Prader-Willi syndrome. Am J Med Genet A. 2011;155A(9):2112–2124. | ||
Butler JV, Whittington JE, Holland AJ, Boer H, Clarke D, Webb T. Prevalence of, and risk factors for, physical ill-health in people with Prader-Willi syndrome: a population-based study. Dev Med Child Neurol. 2002;44(4):248–255. | ||
Butler MG, Haber L, Mernaugh R, et al. Decreased bone mineral density in Prader-Willi syndrome: comparison with obese subjects. Am J Med Genet. 2001;103(3):216–222. | ||
Khare M, Gold JA, Wencel M, et al. Effect of genetic subtypes and growth hormone treatment on bone mineral density in Prader-Willi syndrome. J Pediatr Endocrinol Metabol. 2014;27:511–518. | ||
Bakker NE, Kuppens RJ, Siemensma EP, et al. Bone mineral density in children and adolescents with Prader-Willi syndrome: a longitudinal study during puberty and 9 years of growth hormone treatment. J Clin Endocrinol Metab. 2015;100(4):1609–1618. | ||
Diédhiou D, Cuny T, Sarr A, Norou Diop S, Klein M, Weryha G. Efficacy and safety of denosumab for the treatment of osteoporosis: a systematic review. Ann Endocrinol. 2015;76(6):650–657. | ||
Prader-Willi Syndrome Association (USA). Osteoporosis Evaluation and Therapy in Prader-Willi Syndrome. Consensus statement of the PWSA (USA) Clinical Advisory Board. The gathered view (ISSN 1077-9965). 2008;94:3763–3771. | ||
Kumaki D, Nakamura Y, Suzuki T, et al. Efficacy of denosumab for osteoporosis in two patients with adult-onset still’s disease-denosumab efficacy in osteoporotic still’s disease patients. J Clin Med. 2018;7(4):63. | ||
Uehara M, Nakamura Y, Takahashi J, et al. Efficacy of denosumab therapy for neurofibromatosis type I with osteoporosis and history of fractures: a case report. Mod Rheumatol Case Rep. 2018;2(1):92–96. | ||
Isobe F, Nakamura Y, Suzuki T, et al. Effects of denosumab on osteoporosis in three cases with anorexia nervosa and a review of the literature. Mod Rheumatol Case Rep. 2018;2(1):104–106. | ||
Uehara M, Nakamura Y, Takahashi J, et al. Efficacy of denosumab for osteoporosis in three female patients with osteogenesis imperfecta. Tohoku J Exp Med. 2017;242(2):115–120. | ||
Duran AT, Wilson KS, Castner DM, Tucker JM, Rubin DA. Association between physical activity and bone in children with Prader-Willi syndrome. J Pediatr Endocrinol Metab. 2016;29(7):819–826. | ||
Donze SH, Kuppens RJ, Bakker NE, van Alfen-van der Velden JAEM, Hokken-Koelega ACS. Bone mineral density in young adults with Prader-Willi syndrome: a randomized, placebo-controlled, crossover GH trial. Clin Endocrinol. 2018;88(6):806–812. | ||
Vasikaran S, Eastell R, Bruyère O, et al. Markers of bone turnover for the prediction of fracture risk and monitoring of osteoporosis treatment: a need for international reference standards. Osteoporos Int. 2011;22(2):391–420. |
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.