Back to Journals » Clinical Interventions in Aging » Volume 14
Effects of oral environment on frailty: particular relevance of tongue pressure
Authors Satake A, Kobayashi W , Tamura Y, Oyama T, Fukuta H, Inui A , Sawada K, Ihara K, Noguchi T, Murashita K, Nakaji S
Received 23 April 2019
Accepted for publication 2 September 2019
Published 12 September 2019 Volume 2019:14 Pages 1643—1648
DOI https://doi.org/10.2147/CIA.S212980
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Richard Walker
Anna Satake,1 Wataru Kobayashi,1 Yoshihiro Tamura,1 Toshiaki Oyama,1 Haruka Fukuta,1 Akinari Inui,2 Kahori Sawada,2 Kazunari Ihara,2 Takao Noguchi,1 Koichi Murashita,3 Shigeyuki Nakaji2
1Department of Oral and Maxillofacial Surgery, Hirosaki University Graduate School of Medicine, Hirosaki, Aomori, Japan; 2Department of Social Medicine, Hirosaki University Graduate School of Medicine, Hirosaki, Aomori, Japan; 3COI Research Initiatives Organization, Hirosaki University Graduate School of Medicine, Hirosaki, Aomori, Japan
Correspondence: Anna Satake
Department of Oral and Maxillofacial Surgery, Hirosaki University Graduate School of Medicine, 53 Honcho, Hirosaki-shi, Aomori, Japan
Tel +81 172 39 5127
Fax +81 17 239 5128
Email [email protected]
Purpose: Oral frailty or the loss of oral functionality can be a symptomatic precursor of overall frailty. Previous studies have suggested that decreased tongue pressure causes a decline in ingesting and swallowing function and poor nutrition. This study investigated what factor(s) contribute to tongue pressure, thereby leading to frailty.
Patients and methods: For the purposes of the present study, 467 residents of Hirosaki city in northern Japan aged≥60 years who completed a questionnaire about frailty and underwent an intraoral assessment, which included number of teeth, presence or absence of periodontitis, tongue pressure, and oral diadochokinesis (ODK) were recruited.
Results: Of the 467 participants with complete data sets, frailty was identified in 13 (7.5%) of 173 males and in 34 (11.6%) of 294 females. Significantly fewer teeth, lower tongue pressure, and a reduced diadochokinetic syllable rate were more prevalent among frail than among healthy residents. Multivariable logistic regression analysis revealed that age, body mass index, number of teeth, and tongue pressure significantly contributed to frailty, whereas ODK did not. Multiple regression analysis showed that tongue pressure was positively associated with muscle index and number of teeth.
Conclusion: The results of the present study suggest that fewer teeth and lower tongue pressure, but not ODK function, are risk factors for developing overall frailty among older residents.
Keywords: aging, low nutrition, number of teeth, oral diadochokinesis
Introduction
O’Brien1 described “frailty” in 1986 as a weakness that was becoming more prevalent among older persons. Fried2 described frailty as a clinical syndrome that meets at least three of the following criteria: unintentional weight loss (10 pounds within 1 year), self-reported exhaustion, muscle weakness (determined as grip strength), slow gait, and low levels of physical activity.
The prevalence of frailty increases with advancing age, conferring a high risk for adverse health outcomes such as falls, hospitalization, institutionalization, and mortality.2 Frailty should be addressed as soon as it is discovered to prevent progression to serious health issues and to maintain and improve quality of life. Early detection is important because oral frailty, namely the loss of oral function, is believed to be a precursor or accelerator of general frailty.3,4 We previously reported that the loss of teeth affects general muscle strength, which can lead to disrupted swallowing function.5 The tongue plays an important role in swallowing. Aging-related changes in tongue motion during swallowing have been reported, as has a relationship between tongue pressure and swallowing.6 From these findings, a loss in tongue pressure appears to lead to frailty because of the associated declines in ingestion, swallowing function, and nutrition. Therefore, the aim of the present cross-sectional study was to identify the relationship between tongue pressure and frailty.
Materials and methods
Participants
The overall objective of a community-based project entitled “The Iwaki Health Promotion Project” was to promote health among rural residents of the Hirosaki city of northern Japan in 2016. The present study enrolled community-dwelling volunteers aged ≥60 years who were part of that project. Participants with missing data, a medical history of cancer, stroke, ischemic heart disease, or Parkinson’s disease were excluded from the analysis. This study was approved by the Hirosaki University School of Medicine Ethics Committee (approval no. 2016–028), and all residents voluntarily provided written, informed consent to participate. This study was conducted in accordance with the Declaration of Helsinki.
Questionnaire
We used a structured questionnaire to survey the residents. Standardized interviews ascertained age, sex, smoking history (pack-years), alcohol consumption history, and exercise habits. Body mass index (BMI) was calculated as weight (kg)/height (m2), and muscle index as muscle mass (kg)/height (m2).
Oral examinations
Dentists counted the number of teeth in each resident. Periodontal pockets were assessed using the World Health Organization Community Periodontal Index probe. A periodontal probing depth of ≤3 mm was defined as normal and ≥4 mm as periodontitis. In addition, all dentists were given training beforehand to minimize errors among them.
Tongue pressure measurements
Tongue pressure was measured using a disposable, handheld balloon probe (JF-TPP; JMS, Tokyo, Japan). The residents placed a balloon on the anterior part of the palate, closed their mouths around a hard ring, and bit it using the upper and lower incisors. A probe was placed in the mouths of the residents, who then pushed the tips of their tongues against the hard palate using maximal pressure. The amount of applied pressure was measured in kPa using a digital voltmeter (TPM-01; JMS) attached to a tongue pressure manometer.
Oral diadochokinesis (ODK)
Oral function was evaluated using a ODK measuring tool (Kenko-kun; Takei Scientific Instruments Co., Niigata, Japan). The participants repeatedly pronounced the monosyllables/pa/,/ta/, and/ka/for 5 s as fast as possible. Hypofunctional ODK was diagnosed when at least one of the three syllables was below 6.
Evaluation of frailty
The simple “FRAIL” questionnaire screening tool consists of five components proposed by Morley to indicate frailty status.7 The letter “F” stands for Fatigue (Are you fatigued?), “R” for Resistance (Are you unable to walk up one flight of stairs?), “A” for Aerobic (Are you unable to walk one block?), “I” for Illnesses (Do you have more than five illnesses?), and “L” for Loss of weight (Have you lost more than 5% of your weight in the past 6 months?). Scores on the FRAIL questionnaire scale range from 0 to 5 and represent frail (3–5), pre-frail (1–2), and robust (0) health status.
Statistical analysis
The characteristics of the residents were statistically assessed using the Mann–Whitney U, chi-squared, and Fisher’s exact tests. Univariate analysis was first used to verify the association between frailty and the following variables; age, sex, BMI, muscle index, smoking history (pack-years), alcohol consumption history, exercise habit, number of teeth, periodontitis, tongue pressure, ODK hypofunction, and monosyllables /pa/,/ta/, and /ka/.
Next, factors associated with frailty were investigated using multivariate logistic regression. Based on a scatter diagram, we confirmed that no variables showed an apparent collinearity with each other (|r|<0.9). Factors associated with frailty were investigated using age, BMI, muscle index, smoking history, alcohol consumption history, number of teeth, tongue pressure, and diadochokinetic syllable rate as independent variables. The Hosmer–Lemeshow test was used to calculate 95% confidence intervals (CIs). Multiple regression analysis was used to investigate associations between tongue pressure and the following independent variables: age, number of teeth, muscle index, and BMI. All data were analyzed using SPSS (version 25.0; IBM Japan, Tokyo, Japan), and P-values<0.05 were considered to indicate statistical significance.
Results
Comparison of variables between frail and healthy residents (univariate analysis)
The study sample included 467 residents (females, n=294; males, n=173) aged≥60 years. The residents’ background characteristics and results of oral examinations are summarized in Table 1. Among 47 (10.1%) of the 467 respondents who were diagnosed with frailty, 34 (72.3%) were female and 13 (27.7%) were male, with a mean age of 74.4±7.8 years. The frailty rate was 1.5-fold higher among women than among men (11.6% [34/294] vs 7.5% [13/173]). Significant correlations were observed between frailty and age, number of teeth, tongue pressure, and the diadochokinetic syllable rate; however, no correlations were seen between frailty and muscle index or periodontitis.
 |
Table 1 Univariate analysis of the residents’ characteristics |
Factors affecting frailty (multivariate analysis)
Multivariate logistic regression analysis (Table 2) revealed significant correlations between frailty and age (P=0.003; odds ratio [OR], 1.077; 95% CI, 1.025–1.132), BMI (P=0.007; OR, 1.138; 95% CI, 1.036–1.250), number of teeth (P=0.032; OR, 0.963; 95% CI, 0.930–0.997), and tongue pressure (P=0.029; OR, 0.956; 95% CI, 0.919–0.996). Thus, these factors can be considered risk factors for progression to frailty. No correlations were found between frailty and the diadochokinetic syllables/pa/,/ta/, and/ka/, muscle index, smoking history, or alcohol consumption history.
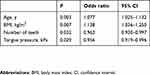 |
Table 2 Multivariate analysis of factors affecting frailty (logistic regression) |
Factors affecting tongue pressure (multivariate analysis)
Among the four risk factors for frailty presented above, only tongue pressure could be a factor improvable by seniors from a dentistry standpoint. To determine factors involved in tongue pressure, multiple regression analysis was used (Table 3). The results indicated that tongue pressure was inversely associated with age, and positively associated with muscle index and number of teeth.
 |
Table 3 Multivariate analysis of factors affecting tongue pressure (multiple regression) |
Discussion
Japanese society has progressed into the realm of ultra-population aging, with individuals aged≥65 years accounting for>20% of the total population.8
This is a concern from the perspective of public health because aging is associated with increased physical disability, organ dysfunction, and a subsequent decrease in quality of life that could shift to high-risk frailty, requiring nursing care.
Fried noted that the prevalence of frailty has been estimated to be 6.9% among older community-dwelling adults in the US, and that the prevalence of frailty within that population has increased from 3.2% among those aged 65–70 years to 23.1% among those aged≥90 years.2 The reported frequency of frailty among community-dwelling older persons in Japan is 11.3%.9 Oral frailty caused by aging is found in individuals with weakened oral status, such as a decreasing interest in teeth and oral health, dental caries, periodontitis, decreased fluency, choking, and difficulties with chewing.2 Pre-frailty often progresses to oral frailty, sarcopenia, locomotive syndrome (decreased locomotive function with aging), and finally, to overall frailty, an irreversible state requiring nursing care. The early detection of decreasing oral performance and physiological function, accompanied by timely interventions, might help prevent oral frailty. Although oral cavity function is generally assessed based on oral hygiene status, xerostomia, occlusal force, ODK, tongue pressure, mastication, and swallowing function,10 no standard criteria for oral frailty have been established.
In the present study, the results of the univariate analysis verified that frailty was correlated with number of teeth, tongue pressure, and diadochokinetic syllables. The tendency for females to have a higher frailty rate than men was also observed, which is in line with a previous report.11 The results of the univariate analysis also showed that the frequency of periodontitis in the frail group was the same or even lower than that of the healthy group; this could be because of the significantly lower number of teeth among members of the frailty compared with the healthy group (10.9 vs 17.3, respectively), which provides frail individuals with less space for periodontitis lesions.
The results of the multivariate logistic analysis conducted to determine factors affecting frailty revealed significant associations between frailty and age, BMI, number of teeth, and tongue pressure, but not diadochokinetic syllables (Table 2). The ORs for age, BMI, number of teeth, and tongue pressure were 1.077, 1.138, 0.963, and 0.956, respectively. Because tongue elevation and exercise of the tongue root play important roles in the oral and pharyngeal phases of swallowing, it is considered that the loss of muscle mass, dysphagia, and decreased tongue pressure are closely related. In fact, Yoshida et al12 and Hirota et al13 have reported an association between tongue pressure and dysphagia. Maeda et al14 reported an association between decreased tongue pressure and sarcopenia, namely the aging-related loss of muscle mass. These results suggest that tongue pressure is an essential indicator of oral frailty, which is known as a precursor to general frailty. Reduced tongue pressure is thought to cause a decline in ingestion and swallowing function, leading to malnutrition and low nutrient intake. In fact, Xue et al15 proposed that the frailty cycle should include metabolism-related items such as basal metabolism, energy consumption, and nutrient intake.
The loss of teeth is considered to cause difficulties in food ingestion. Loss of teeth can affect dysphagia, leading to low nutrient intake, a basic status of frailty, as proposed by Xue et al. Dysphagia can lead to aspiration, which is believed to be involved in accelerating frailty. Aspiration is directly related to mortality rates because of aspiration pneumonia.16–18 In the present study, no association was found between frailty and ODK; this is possibly because the fine and complex tongue movements required for speech do not require robust muscle strength.
The results of the multiple regression analysis revealed a significant correlation between tongue pressure and number of teeth and muscle index. Optimal occlusal stability is needed for safe swallowing, and can be achieved by preserving more teeth, greater occlusal contact, and a larger area of support. While swallowing, the mandible is fixed, the hyoid bone is lifted anteriorly by the muscle attached to the mandible, and the tongue is pushed up against the palate. We consider that this mechanism of muscle movement during swallowing serves as the basis for the association between tongue pressure and the number of teeth and frailty. Sarcopenia is thought to affect the association between tongue pressure and muscle index.
Age was shown to be an important factor relating to frailty in our study. It is possible that inflammatory cytokines have some roles in decreasing tongue pressure and muscle index in older persons. It has been reported that inflammatory cytokines such as interleukin (IL)-6 and tumor necrosis factor-alpha (TNF-α) correlate with muscle mass and strength.19 Moreover, several recent reports have established that monocytes produce more IL-1, IL-6, and TNF-α in older than in younger persons.20–22 The chronic low-grade, systemic inflammation that is closely associated with aging is called “inflammaging”. Although the mechanism underlying inflammaging is unclear, it is plausible that muscle weakness developing in older persons with high cytokine levels could contribute to a loss of muscle mass and oral function.
This study did have some limitations. First, it had a cross-sectional design; thus, no definite associations between frailty and the oral factors evaluated in this study could be determined. Longer-term cohort studies are needed to elucidate what kinds of changes in lifestyle or the intraoral environment lead to frailty. Second, because of time constraints, only tongue pressure and ODK were used for functional evaluations. The implementation of testing for variables such as oral dryness (xerostomia), bite force, and masticatory performance could enable more accurate diagnoses of oral dysfunction.
Conclusion
The results of the present study revealed that the oral factors of tongue pressure and tooth loss were associated with a higher risk of general frailty among older persons; however, ODK was not. Decreased tongue movement and deteriorating swallowing function are reversible, but not tooth loss. Therefore, to help prevent frailty, maintaining natural teeth is very important. These findings further suggest that to prevent overall frailty and avoid reaching a state where nursing care is required, the early detection of oral frailty is necessary. Moreover, standardized diagnostic criteria and treatment guidelines need to be established for oral frailty.
Abbreviations
ODK, oral diadochokinesis; BMI, body mass index; CI, confidence interval.
Acknowledgments
This study was supported by grants-in-aid for Scientific Research (JP16K11879, JP17K17576) from the Japanese Ministry of Education. The authors specially thank all participants of the study.
Author contributions
WK designed the study. AS collected and analyzed data and wrote the manuscript. All other authors have contributed to the interview and oral examination of residents.
All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. O’Brien TD, Roberts J, Brackenridge GR, Lloyd WH. Some aspects of community care of the frail and elderly: the need for assessment. Gerodontol Clin. 1968;10:215–227.
2. Fried LP, Tangen CM, Walston J, et al. Frailty in older adults: evidence for a phenotype. J Gerontol A Biol Sci Med Sci. 2001;56:M146–M156. doi:10.1093/gerona/56.3.M146
3. Tanaka T, Hirano H, Takahashi K, et al. Oral frailty as a risk factor for physical frailty and mortality in community-dwelling elderly. J Gerontol A Biol Sci Med Sci. 2018;73:1661–1667. doi:10.1093/gerona/glx225
4. Tôrres LH, Tellez M, Hilgert JB, Hugo FN, de Sousa MD, Ismail AI. Frailty, frailty components, and oral health: asystematic review. J Am Geriatr Soc. 2015;63:2555–2562. doi:10.1111/jgs.13826
5. Inui A, Takahashi I, Sawada K, et al. Teeth and physical fitness in a community-dwelling 40 to 79-year-old Japanese population. Clin Interv Aging. 2016;11:873–878. doi:10.2147/CIA.S108498
6. Robbins J
7. Morley JE, Vellas B, Van Kan GA, et al. Frailty consensus: a call to action. J Am Med Dir Assoc. 2013;14:392–397. doi:10.1016/j.jamda.2013.03.022
8. Population estimation. [homepage on the Internet]. Japan; 2019. Available from: https://www.stat.go.jp/data/jinsui/pdf/201907.pdf.
9. Shimada H, Makizako H, Doi T, et al. Combined prevalence of frailty and mild cognitive impairment in a population of elderly Japanese people. J Am Med Dir Assoc. 2013;14:518–524. doi:10.1016/j.jamda.2013.03.010
10. Minakuchi S, Tsuga K, Ikebe K, et al. Deterioration of oral function in the elderly the position paper of Japanese society of gerodontology in 2016. Jpn Soc Gerodontology Acad Committee. 2016;31:81–99.
11. Yuki A, Otsuka R, Tange C, et al. Shimokata H. Epidemiology of frailty in elderly Japanese. J Phys Fit Sports Med. 2016;5:301–307. doi:10.7600/jpfsm.5.301
12. Yoshida M, Kikutani T, Tsuga K, Utanohara Y, Hayashi R, Akagawa Y. Decreased tongue pressure reflects symptom of dysphagia. Dysphagia. 2006;21:61–65. doi:10.1007/s00455-005-9011-6
13. Hirota N, Konaka K, Ono T, et al. Reduced tongue pressure against the hard palate on the paralyzed side during swallowing predicts dysphagia in acute stroke. Stroke. 2010;41:2982–2984. doi:10.1161/STROKEAHA.109.572750
14. Maeda K, Akagi J. Decreased tongue pressure is associated with sarcopenia and sarcopenic dysphagia in the elderly. Dysphagia. 2015;30:80–87. doi:10.1007/s00455-014-9577-y
15. Xue QL, Bandeen-Roche K, Varadhan R, Zhou J, Fried LP. Initial manifestations of frailty criteria and the development of frailty phenotype in the women’s health and aging study II. J Gerontol A Biol Sci Med Sci. 2008;63:984–990. doi:10.1093/gerona/63.9.984
16. Aida J, Kondo K, Hirai H, et al. Association between dental status and incident disability in an older Japanese population. J Am Geriatr Soc. 2012;60:338–343. doi:10.1111/j.1532-5415.2011.03791.x
17. Hayasaka K, Tomata Y, Aida J, Watanabe T, Kakizaki M, Tsuji I. Tooth loss and mortality in elderly Japanese adults: effect of oral care. J Am Geriatr Soc. 2013;61:815–820. doi:10.1111/jgs.2013.61.issue-5
18. Inui A, Takahashi I, Kurauchi S, et al. Oral conditions and dysphagia in Japanese, community-dwelling middle- and older- aged adults, independent in daily living. Clin Interv Aging. 2017;12:515–521. doi:10.2147/CIA.S132637
19. Roubenoff R, Freeman LM, Smith DE, Abad LW, Dinarello CA, Kehayias JJ. djuvant arthritis as a model of inflammatory cachexia. Arthritis Rheum. 1997;40:534–539. doi:10.1002/1529-0131(199707)40:7<1267::AID-ART11>3.0.CO;2-L
20. Fagiolo U, Cossarizza A, Scala E, et al. Increased cytokine production in mononuclear cells of healthy elderly people. Eur J Immunol. 1993;23:2375–2378. doi:10.1002/eji.1830230950
21. Gupta S, Su H, Agrawal S, Gollapudi S. Molecular changes associated with increased TNF-α-induced apoptotis in naïve (TN) and central memory (TCM) CD8+ T cells in aged humans. Immun Ageing. 2018;15. doi:10.1186/s12979-017-0109-0
22. Cohen HJ, Pieper CF, Harris T, Rao KMK, Currie MS. The association of plasma IL-6 levels with functional disability in community-dwelling elderly. J Gerodontol A Biol Sci Med Sci. 1997;52:M201–M208. doi:10.1093/gerona/52A.4.M201
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
