Back to Journals » Clinical Interventions in Aging » Volume 10
Effects of dexamethasone, celecoxib, and methotrexate on the histology and metabolism of bone tissue in healthy Sprague Dawley rats
Authors Liu Y , Cui Y, Chen Y, Gao X, Su Y, Cui L
Received 21 March 2015
Accepted for publication 12 May 2015
Published 5 August 2015 Volume 2015:10 Pages 1245—1253
DOI https://doi.org/10.2147/CIA.S85225
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 4
Editor who approved publication: Dr Zhi-Ying Wu
Yanzhi Liu,1,2,* Yang Cui,3,* Yan Chen,1 Xiang Gao,4 Yanjie Su,1 Liao Cui1,2
1Department of Pharmacology, Guangdong Key Laboratory for Research and Development of Natural Drugs, Guangdong Medical University, Zhanjiang, Guangdong, 2School of Traditional Chinese Medicine, Southern Medical University, Guangzhou, 3Department of Rheumatism Medicine, Guangdong General Hospital, Guangzhou, 4Stem Cell Research and Cellular Therapy Center, Affiliated Hospital of Guangdong Medical University, Zhanjiang, People’s Republic of China
*These authors contributed equally to this work
Objective: To investigate the long-term effects of three antiarthritics, namely dexamethasone, celecoxib, and methotrexate on the histology and metabolism of intact bone tissue in rats.
Methods: Thirty-two 12-week-old healthy female Sprague Dawley rats were randomly allocated into four groups: 1) control (saline, daily); 2) dexamethasone (2 mg/kg, twice weekly); 3) celecoxib (50 mg/kg, daily); and 4) methotrexate (0.5 mg/kg, twice weekly). The drugs were administered to the rats for 12 weeks and the animals were weighed on a weekly basis. The femurs and lumbar vertebrae were harvested for bone mineral density and bone mechanical properties analyses. The proximal tibiae were processed for bone histomorphometry and micro-computed tomography analyses.
Results: The following results were obtained: 1) dexamethasone strongly inhibited bone formation rate accompanied with a decrease in bone mineral density and bone biomechanical properties; 2) celecoxib stimulated bone resorption, leading to a decrease of bone mass and femur biomechanic properties; and 3) methotrexate caused bone loss and bone quality deterioration to a lesser extent due to the increase of the bone turnover rate on the proximal tibial metaphysis of the rats.
Conclusion: This study provides a comparative profile of the long-term effects of clinical doses of celecoxib, methotrexate, and dexamethasone on intact skeletons of the rats. The results indicate that the three antiarthritics have varying degrees of side effects on bone metabolism, and these findings will help physicians to learn more about the potential effects of antiarthritics on bone metabolism.
Keywords: rheumatoid arthritis, celecoxib, methotrexate, dexamethasone, bone
Introduction
Rheumatoid arthritis (RA) is an autoimmune disease that causes chronic inflammation of the joints, and mainly leads to synovitis, bone loss, and bone destruction around the affected joints.1 High prevalence of RA worldwide2 has become an important issue that is associated with significant clinical burden.3 Bone remodeling equilibrium is interfered in RA, and is accompanied with increased risk of osteoporosis and fractures.4,5 Many factors that can affect bone metabolism during the RA treatment include age, underlying disease, disease severity, comedication, and duration of treatment. However, the skeleton impairment occurring during the treatment of RA is mainly due to two factors: the effects of drugs and the special nature of RA per se.6,7 It is not clear whether the skeleton impairments are caused by the synergistic effect or antagonistic effect of the two factors. Moreover, if the combination of the two factors occurs in one individual, it becomes difficult to differentiate the effects of antiarthritics from the outcomes of the underlying disease they are used to treat, as both the drugs and the disease have significant effects on bone metabolism in inflammatory individuals. The impairments caused by the disease itself may conceal the real effects induced by the drugs or even reverse their original effects on bone, and vice versa. Thus, the two closely connected factors can lead to confounding the truth, and may thus preclude definite conclusions, which lead to the physicians’ inability to fully understand the long-term potential effects of antiarthritics on bone metabolism. Although many studies have reported limited short-term effects of antiarthritics on bone metabolism in arthritis patients or inflammatory animal models, these studies could only provide limited data for the short-term effects, but not for the long-term effects.8–15 Therefore, it is interesting to investigate the long-term effects of antiarthritics on bone alone in healthy individuals. It will help to understand the actual long-term effects of antiarthritics on bone metabolism and how the inflammation would change the original effects of antiarthritics on bone in vivo. There are four aspects of medication for RA till date, namely nonsteroidal anti-inflammatory drugs (NSAIDs), disease-modifying antirheumatic drugs (DMARDs), corticosteroids (eg, glucocorticoids [GCs]), and biologic agents involving the immune system. Dexamethasone (Dex), celecoxib (Cel), and methotrexate (MTX) are all used in the clinical treatment of RA, and they were selected to represent the GCs, NSAIDs, and DMARDs, respectively. In this study, we designed to give therapeutic doses of Dex, Cel, and MTX for a long–term administration in healthy female rats, in order to investigate whether the original effects of drugs would affect the bone metabolism in healthy condition.
Materials and methods
Animal ethics and breeding conditions
This study was carried out in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of Guangdong Laboratory Animal Monitoring Institute, under the National Laboratory Animal Monitoring Institute of People’s Republic of China. The experiments have been conducted according to protocols approved for Specific Pathogen Free animal care of the Animal Center of Guangdong Medical College, and approved by the Academic Committee on the Ethics of Animal Experiments of the Guangdong Medical College, Zhanjiang, People’s Republic of China (Permit Number: SYXK (GUANGDONG) 2008-0007).
The Sprague Dawley (SD) female rats were acclimated to local vivarium conditions (temperature: 24°C–26°C, humidity: 67%) and under specific pathogen-free conditions. Rats were allowed free access to water and diet containing 1.33% calcium, 0.95% phosphorus, and 60 IU% vitamin D3.
Experimental protocols
Thirty-two 12-week-old female SD rats weighing 263.5±12 g were purchased from the Medical Laboratory Animal Center of Guangdong Province (Permit Number: SCXK [GUANG DONG] 2008-0002, Guangdong, People’s Republic of China). Rats were randomly allocated into four groups: 1) intact control group (Con, saline, daily, oral gavage); 2) Dex group (Dex [Tianjin Tianyao Pharmaceuticals Co. Ltd, Tianjin, People’s Republic of China], 2 mg/kg,16 twice weekly, tail vein injection); 3) Cel group (Cel [Pfizer, Inc., New York, NY, USA], 50 mg/kg,11 daily, oral gavage); and 4) MTX group (MTX [Shanghai Pharmaceutical Group Co. Ltd., Xinyi Pharmaceutical Co., Ltd., Shanghai, People’s Republic of China], 0.5 mg/kg,17 twice weekly, oral gavage). Eight rats in each group were administered for 12 weeks and weighed on weekly basis. Rats were given subcutaneous injections of tetracycline (50 mg/kg; Sigma-Aldrich Co, St Louis, MO, USA) on the 14th and 13th day and calcein (10 mg/kg; Sigma-Aldrich Co) on the fourth and third day before being sacrificed for the purpose of double labeling in vivo.
At the end point of the experiment, the rats were sacrificed by cardiac puncture under sodium pentobarbital anesthesia. Care of the rats was in accordance with institutional guidelines.
Serum markers assay
Serum samples were collected for measurements of serum calcium, phosphorus, glucose, total cholesterol, triglyceride (TG), and the biomarkers of bone such as alkaline phosphatase and tartrate-resistant acid phosphatase (TRACP) according to manufacturer’s instructions (Nanjing Jiancheng Bioengineering Institute, Nanjing, People’s Republic of China).
Bone mineral density determination
The left femurs and the fifth lumbar vertebrae of the rats were wrapped with saline-saturated gauze to maintain moisture and stored at −20°C. After thawing at room temperature, the femurs and the fifth lumbar vertebrae were moisturized by soaking in saline solution, and the residual muscle was removed. The whole femur and lumbar bone mineral density (BMD) was measured with a DXA system, the Lunar Prodigy Advance Bone Densitometer (GE Healthcare Bio-Sciences Corp., Piscataway, NJ, USA).
Testing of bone mechanical properties
After measuring BMD, the femurs were used to determine the bone mechanical properties by three-point bending test using a Bose Electro Force Testing system (Bose Electroforce 3510, Bose Corp., Eden Prairie, MN, USA). Bone samples were tested with a 1 mm indenter at a speed of 0.01 mm/s with 15 mm span (L). Force and deflection were automatically recorded. The output parameters included elastic force (the force required to cause bone specimens to deform, in units of N), maximum force (the maximum force that the bone can resist, N), fracture force (the force required to cause bone fracture, N), and the maximum deflection (maximum degree of the bone displacement, mm). The stiffness (load–displacement curve slope, N/mm), the energy to maximum load, and the energy to fracture load were also calculated based on the parameters mentioned earlier.
Micro-CT analysis
The left proximal tibias were harvested for micro-computed tomography (micro-CT) analysis. The proximal tibial metaphysis (PTM) of the rats were scanned using a Viva CT40 (Scanco Medical, Zurich, Switzerland) under high-resolution conditions (X-ray energy 70 kVp, 114 μA, 8 W; integration time 200 ms). Briefly, the region of interest was the proximal tibial growth plate and the PTM located between 1.5 and 3.5 mm distal to the growth plate epiphyseal junction. Cortical bone was excluded from the measurement. Three-dimensional (3D) images and longitudinal sections of mineralized tibia metaphyses were generated using a Gaussian filter (sigma 0.8, support 1) and a threshold of 350. The 3D image analysis was performed to determine bone volume/tissue volume (BV/TV), structure model index (SMI), bone mineral density of tissue volume (density of TV), and degree of anisotropy (DA).
Bone histomorphometry
The right proximal tibial metaphyses were processed for bone histomorphometric analyses. The samples were opened to expose the bone marrow cavity using an ISOMET Low Speed Saw (Buehler, Lake Bluff, IL, USA) and fixed in 10% phosphate buffered formalin for 24 hours. They were then dehydrated in graded ethanol, defatted in xylene, and embedded undecalcified in methyl methacrylate. Frontal sections were cut at thicknesses of 5 and 9 μm with the RM2155 hard tissue microtome (Leica, Wetzlar, Germany). The 5 μm sections were stained by Goldner’s trichrome stain for static histomorphometric measurements. The 9 μm unstained sections were used for dynamic histomorphometric analyses. A semiautomatic digitizing image analysis system (Osteometrics Inc., Decatur, GA, USA) was used for quantitative bone histomorphometric measurements. Briefly, the regions of interest were the proximal tibial growth plate and the PTM located between 1 and 4 mm distal to the growth plate–epiphyseal junction. The quantitative analysis was performed on the region of interest mentioned, in each sample. The abbreviations used for the bone histomorphometric parameters were those recommended by the ASBMR Histomorphometric Nomenclature Committee.18 Static measurements included total tissue volume (TV), trabecular bone volume (BV), trabecular bone surface (BS), and osteoclast surface (OcS). Dynamic measurements included trabecular single-labeled surface (sLS), double labeled surface (dLS), and inter-label width (IntWi). The parameters of microarchitecture included trabecular thickness (TbTh), number (TbN), and separation (TbSp), and were derived from area and perimeter measurements. Inter-label width in the growth plate (G-IntWi), sLS, and dLS were measured on unstained sections under ultraviolet light and were used to calculate the mineral apposition rate (MAR) and the ratio of mineralizing surface to bone surface (MS/BS, calculated as double plus half of single-labeled surfaces), bone formation rate per unit of bone surface (BFR/BS), and bone formation rate per unit of bone volume (BFR/BV). These dynamic parameters were used to assess bone formation. Bone resorption was assessed by the measurement of osteoclast surface per bone surface (OcS/BS). All histomorphometric parameters and procedures were in accordance with the published studies.19–22
Statistical analysis
Data were presented as mean ± standard deviation. The statistical differences among groups were evaluated using SPSS 16.0 software by analysis of variance (ANOVA) with Fisher’s protected least significant difference (PLSD) test. Probabilities (P) less than 0.05 were considered significant.
Results
Body weight
Body weight in the Dex group decreased significantly after the first administration (the second week, Figure 1) and lasted till the end. No significant differences in body weight and behavior were noted among the Con, Cel, and MTX groups throughout the experimental period.
Biochemical markers in serum
Compared to the Con group, TRACP and glucose increased in the Dex group, while serum calcium, phosphorus, and TG decreased significantly. Elevated serum TRACP levels were found in the Cel group, while there was no difference in the levels of glucose and TG; however, glucose levels decreased and TG levels increased in the Cel and the Dex groups, respectively. Serum calcium and glucose levels increased significantly in the MTX group (Table 1).
Bone histomorphometry in proximal tibial metaphysis
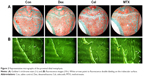
Compared to the Con group, Dex significantly decreased BV/TV and TbN by 19.8% and 15.2%, respectively, and increased TbSp. Dex also significantly decreased MS/BS by 55%, MAR by 45.2%, BFR/BS by 75.2%, and BFR/BV by 74.56%. Besides, Dex treatment significantly increased OcS by 42.1% and OcS/BS by 47.8%. Treatment by Cel markedly decreased BV/TV by 30.3% and TbN by 20.7% with TbSp increased by 49%, whereas no significant change was found in bone formation parameters when compared to the control group; however, Cel treatment significantly increased OcS by 42.1% and OcS/BS by 104.3%. MTX significantly decreased BV/TV by 18.1% and TbN by 11.1% as well as increased TbSp by 25.3% when compared to the Con treatment. Yet, MTX significantly increased select PTM cancellous bone formation parameters MS/BS, MAR, BFR/BS, and BFR/BV by 32.7%, 26.4%, 69.2%, and 77%, respectively, and simultaneously increased OcS by 47.3% and OcS/BS by 91.3% (Tables 2 and 3, Figure 2A).
Compared to the control group, fluorescence micrographs of tibial metaphyses revealed that fluorescence was markedly decreased in the Dex group because of normal bone formation was strongly inhibited. Compared to Dex group, stronger fluorescence and double labeling was seen in the Cel and MTX groups (Figure 2B, Table 3).
Micro-CT analysis of tibial metaphyses trabecular
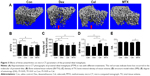
Micro-CT data demonstrated that Dex significantly decreased BV/TV by 14% and mean mineral density of TV by 16.2%, while SMI and DA in the Dex group demonstrated no significant differences compared to the Con group. Compared to the Con group, Cel decreased BV/TV by 25% and mean mineral density of TV by 19.1% significantly. Furthermore, the SMI was significantly higher than in the other groups, suggesting that Cel reduced bone mass and changed trabecular bone structure from the plate to the rod shape. The MTX-treated rats showed no significant difference in BV/TV, SMI, and DA, whereas mineral density of TV decreased by 16.4%. On the other hand, the DA and SMI in the MTX group significantly differed from the Cel group (Figure 3).
Bone mineral density in femur and lumbar
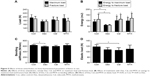
BMD of femur in Dex, Cel, and MTX groups significantly decreased by 7.3%, 9.3%, and 6.9%, respectively, compared to the Con group. No significant difference was found in BMD of lumbar vertebrae (Figure 4).
Bone biomechanical property parameters of femur
The three-point bending test of the femur demonstrated that Dex treatment significantly reduced energy to maximum load by 34.4% and energy to fracture load by 46.3% compared to the Con treatment (Figure 5). Significant reductions were also found in these parameters when compared to Cel and MTX treatments. Cel treatment decreased elastic load by 10.9%.
Discussion
Long-term high-dose administration of GCs is linked to serious adverse effects, including osteoporosis, fractures, peptic ulcer disease, infections, and cardiovascular disease among others. Thus, in the past, GC treatment in RA therapy was merely being used as a temporary “bridge” until the disease-modifying therapy became effective. Randomized controlled trials have demonstrated the ability of GC to retard radiographic progression in RA and prevent development of new erosions, confirming disease-modifying effects when used in low doses.23,24 The disease-modifying effects of low-dose GC encouraged its use for longer durations in RA therapy, with an acceptable adverse effect profile, at least, in the short to medium term. However, the adverse effects of GCs should not be ignored.10 A retrospective cohort study using the General Practice Research Database of the UK has shown that the rate of clinical vertebral fractures increased by 55% for a dose of prednisolone of less than 2.5 mg/day and by over 400% if the dose exceeded 7.5 mg/day.25 Although the occurrence of fractures in RA patients may be associated with an increased incidence of osteoporosis induced by disease per se or falls, the chronic use of GC further amplifies this increased risk by a factor of 2.26 Our data demonstrated that even in low dose, with the absence of inflammatory disease, GC still induced significant osteogenic inhibition with bone resorption increasing significantly. The biomechanical results revealed that the energy to maximum load, energy to fracture load, and bending stiffness were decreased significantly (Figure 5). These results suggested that the effects of bone loss and bone formation inhibition induced by low doses of GC treatment would also deteriorate bone quality and increase the risk of fracture. The 2010 American Society for Bone and Mineral Research task force report on fractures identified glucocorticoid treatment as one potential cofactor.27 Possibly, GCs in RA, leading to decreased disease activity, may cause less bone loss than they would have in the absence of inflammatory disease. However, evidences mentioned earlier indicate that low dose of chronic GC administration remains the cause of risk for osteoporosis and fracture irrespective of the presence or absence of the inflammatory disease. Therefore, people treated with chronic GC therapy may still need prescribed co-therapy to prevent bone loss no matter in high dose or low dose.
Previous report indicated that Cel treatment inhibited osteoclastic bone resorption in pathological conditions (eg, RA, estrogen deficiency, or their joint condition), but not in normal conditions. A clinical study suggested that Cel may inhibit osteoclastic bone resorption in postmenopausal women with RA, but not in premenopausal women or in men with RA.28 Another study also reported that orally administered Cel could decrease serum levels of C-telopeptide, a marker of bone resorption, in ovariectomy mice but not sham-operated mice.29 These reports indicated that RA and estrogen deficiency are important factors for Cel to exhibit its ability to inhibit bone resorption. However, in the absence of these pathological conditions, Cel may not demonstrate such an ability. To date the inhibition of Cel on osteoclast still has different voice. Kawashima et al30 demonstrated that Cel acts directly on circulating human osteoclast precursors to inhibit osteoclast formation without cytotoxic effect in vitro. They suggested that the direct effect of Cel on osteoclast precursors may be related to the COX-2–PGE-2 pathway.30 However, Igarashi et al reported that TRACP-positive cell formation in the single culture with sRANKL and M-CSF did not decline with Cel treatment.31 In this study, we investigated Cel treatment in female rats without RA and estrogen deficiency background, and found significantly elevated osteoclast surface and a decline of bone mass as well as BMD. The elastic load of femur decreased significantly in bone biomechanical test due to bone loss occurred in Cel treatment group. However, no further serious deterioration in bone biomechanical property occurred by Cel treatment, which suggested the ratio of matrix and mineral composition might not deteriorate significantly in the skeleton. On the other hand, we also noticed that bone formation was not significantly affected by Cel treatment, which was consistent with the previous studies.11,29 NSAIDs would induce different effects on osteogenic differentiation of Mesenchymal stem cells (MSCs) under inflammatory and noninflammatory conditions.32 Some studies reported that COX-2 selective inhibitors suppress bone remodeling and repair in vivo, resulting from the suppression of angiogenesis as well as the potential interference with osteoblast and osteoclast functions.33,34 Yet, Muller et al35 reported that only a partial inhibitory effect of NSAIDs can be seen on the osteogenic differentiation. It is speculated that NSAID effects on bone cells or precursors might be mediated by the inhibition of prostaglandin synthesis, since PGE-2 stimulated bone formation.35 In this study, our data suggested that Cel treatment may induce bone loss in female rats without RA and estrogen deficiency background. To understand the mechanism by which Cel switches its role in different conditions during bone metabolism, more in-depth studies need to be conducted.
Early intensive treatment strategies of RA with MTX as an anchor antiarthritic are recommended in the Western world, and its effectiveness is well established.36,37 The 2012 RA treatment guidelines published by the American College of Rheumatology (ACR) recommend MTX as monotherapy or in combination with other agents for initial management of established RA with low disease activity and poor prognosis or with moderate or high disease activity, with the treatment goal of reaching low disease activity or remission.38 MTX, used originally as a chemotherapeutic drug for cancer, has been reported to suppress skeletal growth and increase bone resorption in high doses.39–41 But when low dose of MTX was used for RA treatment, skeleton impairment was more likely due to local disease activity, adjacent joint damage, and therapy drug (eg, glucocorticoid), rather than a toxic effect of MTX in an RA patient. A previous study reported that a low dose of MTX caused little or no deterioration in bone growth and osteoblast differentiation.42 A clinical trial of over 5-years, demonstrated no adverse effects of low-dose MTX (10 mg/week) on bone formation and bone density in RA patients. Minaur et al43 also suggested that local inflammatory joint disease was a more important determinant of bone loss than MTX therapy for RA, and they observed an interesting result that MTX therapy increased the period of bone formation. However, they did not believe the result was clinically relevant.43 In this study, our results demonstrated that MTX treatment in a low dose in healthy rats increased bone formation rate while stimulating bone resorption, which induced a decline in mineral density. However, bone biomechanical parameters of the femur revealed that bone quality in the MTX treatment group did not change significantly compared to the control group. We noticed that histomorphometry results demonstrated bone loss in MTX rats but micro-CT data did not reveal such changes. The reason may be that the regions of interest in micro-CT analysis were smaller than those in histomorphometry. The DA data in micro-CT analysis suggested that the spatial distribution of trabecular bone may contribute to maintain bone biomechanical property in MTX-treated rats.
In summary, we compared long-term effects of three antiarthritics (GCs, NSAIDs, and DMARDs) on the skeleton of healthy female rats. Dex inhibited bone formation and increased bone resorption, leading to osteopenia; Cel increased bone resorption and induced bone loss; and MTX caused slight bone loss as both bone formation and bone resorption increased simultaneously. This comparative study not only demonstrated the effects of the three antiarthritics in healthy models but also compared the findings with previous reports using inflammation model.
Conclusion
This study provides a comparative profile of the long-term effects of therapeutic doses of Cel, MTX, and Dex on intact skeletons of rats. The results indicate that the three antiarthritics have varying degrees of side effects on bone metabolism, and these findings may help physicians figure out whether appropriate measure will be needed to better prevent the occurrence of osteopenia in RA treatment.
Acknowledgments
This study was supported by the National Natural Science Foundation of China (No: 81273518), the Guangdong Province Science and Technology Project (No: 2012B060300027), and Guangdong Medical College Youth Fund (No: Q2012010).
Author contributions
Yanzhi Liu and Yang Cui performed the animal experiments, analyzed data, participated in study design, and wrote the manuscript. Yan Chen contributed to histomorphometry analyses. Xiang Gao and Yanjie Su made contributions to micro CT analysis. Liao Cui conceived the study, and participated in its design and coordination and helped to revise the manuscript. All authors contributed toward data analysis, drafting and revising the paper and agree to be accountable for all aspects of the work. All authors read and approved the final manuscript for publication.
Disclosure
The authors report no conflicts of interest in this work.
References
Gravallese EM. Bone destruction in arthritis. Ann Rheum Dis. 2002;61 Suppl 2:ii84-6. | ||
National Collaborating Centre for Chronic Conditions (UK). Rheumatoid Arthritis: National Clinical Guideline for Management and Treatment in Adults. London; 2009. | ||
Huelin R, Pokora T, Foster TS, Mould JF. Economic outcomes for celecoxib: a systematic review of pharmacoeconomic studies. Expert Rev Pharmacoecon Outcomes Res. 2012;12:505–523. | ||
Guler-Yuksel M, Klarenbeek NB, Goekoop-Ruiterman YP, et al. Accelerated hand bone mineral density loss is associated with progressive joint damage in hands and feet in recent-onset rheumatoid arthritis. Arthritis Res Ther. 2010;12:R96. | ||
Kim SY, Schneeweiss S, Liu J, et al. Risk of osteoporotic fracture in a large population-based cohort of patients with rheumatoid arthritis. Arthritis Res Ther. 2010;12:R154. | ||
Vis M, Guler-Yuksel M, Lems WF. Can bone loss in rheumatoid arthritis be prevented? Osteoporos Int. 2013;24:2541–2553. | ||
Schett G, Teitelbaum SL. Osteoclasts and arthritis. J Bone Miner Res. 2009;24:1142–1146. | ||
Noguchi M, Kimoto A, Sasamata M, Miyata K. Micro-CT imaging analysis for the effect of celecoxib, a cyclooxygenase-2 inhibitor, on inflammatory bone destruction in adjuvant arthritis rats. J Bone Miner Metab. 2008;26:461–468. | ||
Durez P, Malghem J, Nzeusseu Toukap A, et al. Treatment of early rheumatoid arthritis: a randomized magnetic resonance imaging study comparing the effects of methotrexate alone, methotrexate in combination with infliximab, and methotrexate in combination with intravenous pulse methylprednisolone. Arthritis Rheum. 2007;56:3919–3927. | ||
Da Silva JA, Jacobs JW, Kirwan JR, et al. Safety of low dose glucocorticoid treatment in rheumatoid arthritis: published evidence and prospective trial data. Ann Rheum Dis. 2006;65:285–293. | ||
Tsuboi H, Nampei A, Matsui Y, et al. Celecoxib prevents juxta-articular osteopenia and growth plate destruction adjacent to inflamed joints in rats with collagen-induced arthritis. Mod Rheumatol. 2007;17:115–122. | ||
Cranney AB, McKendry RJ, Wells GA, et al. The effect of low dose methotrexate on bone density. J Rheumatol. 2001;28:2395–2399. | ||
di Munno O, Mazzantini M, Sinigaglia L, et al. Effect of low dose methotrexate on bone density in women with rheumatoid arthritis: results from a multicenter cross-sectional study. J Rheumatol. 2004;31:1305–1309. | ||
Zweers MC, de Boer TN, van Roon J, Bijlsma JW, Lafeber FP, Mastbergen SC. Celecoxib: considerations regarding its potential disease-modifying properties in osteoarthritis. Arthritis Res Ther. 2011;13:239. | ||
Odio CM, Ramirez T, Arias G, et al. Double blind, randomized, placebo-controlled study of dexamethasone therapy for hematogenous septic arthritis in children. Pediatr Infect Dis J. 2003;22:883–888. | ||
Wang D, Miller SC, Liu XM, Anderson B, Wang XS, Goldring SR. Novel dexamethasone-HPMA copolymer conjugate and its potential application in treatment of rheumatoid arthritis. Arthritis Res Ther. 2007;9:R2. | ||
Jurcovicova J, Svik K, Scsukova S, Bauerova K, Rovensky J, Stancikova M. Methotrexate treatment ameliorated testicular suppression and anorexia related leptin reduction in rats with adjuvant arthritis. Rheumatol Int. 2009;29:1187–1191. | ||
Dempster DW, Compston JE, Drezner MK, et al. Standardized nomenclature, symbols, and units for bone histomorphometry: a 2012 update of the report of the ASBMR Histomorphometry Nomenclature Committee. J Bone Miner Res. 2013;28:2–17. | ||
Cui L, Li T, Liu Y, et al. Salvianolic acid B prevents bone loss in prednisone-treated rats through stimulation of osteogenesis and bone marrow angiogenesis. PloS one. 2012;7:e34647. | ||
Lin SE, Huang JP, Wu LZ, Wu T, Cui L. Prevention of osteopenia and dyslipidemia in rats after ovariectomy with combined aspirin and low-dose diethylstilbestrol. Biomed Environ Sci. 2013;26:249–257. | ||
Cui L, Wu T, Li QN, Lin LS, Liang NC. Preventive effects of ginsenosides on osteopenia of rats induced by ovariectomy. Acta pharmacol Sin. 2001;22:428–434. | ||
Lin S, Huang J, Zheng L, et al. Glucocorticoid-induced osteoporosis in growing rats. Calcif Tissue Int. 2014;95:362–373. | ||
Bakker MF, Jacobs JW, Welsing PM, et al. Low-dose prednisone inclusion in a methotrexate-based, tight control strategy for early rheumatoid arthritis: a randomized trial. Ann Intern Med. 2012;156:329–339. | ||
Kirwan JR, Bijlsma JW, Boers M, Shea BJ. Effects of glucocorticoids on radiological progression in rheumatoid arthritis. Cochrane Database Syst Rev. 2007:CD006356. | ||
Van Staa TP, Leufkens HG, Abenhaim L, Zhang B, Cooper C. Use of oral corticosteroids and risk of fractures. June, 2000. J Bone Miner Res. 2005;20:1487–1494; discussion 6. | ||
Saag KG, Koehnke R, Caldwell JR, et al. Low dose long-term corticosteroid therapy in rheumatoid arthritis: an analysis of serious adverse events. Am J Med. 1994;96:115–123. | ||
Shane E, Burr D, Ebeling PR, et al. Atypical subtrochanteric and diaphyseal femoral fractures: report of a task force of the American Society for Bone and Mineral Research. J Bone Miner Res. 2010;25:2267–2294. | ||
Tsuji S, Tomita T, Nakase T, Hamada M, Kawai H, Yoshikawa H. Celecoxib, a selective cyclooxygenase-2 inhibitor, reduces level of a bone resorption marker in postmenopausal women with rheumatoid arthritis. Int J Rheum Dis. 2014;17:44–49. | ||
Kasukawa Y, Miyakoshi N, Srivastava AK, et al. The selective cyclooxygenase-2 inhibitor celecoxib reduces bone resorption, but not bone formation, in ovariectomized mice in vivo. Tohoku J Exp Med. 2007;211:275–283. | ||
Kawashima M, Fujikawa Y, Itonaga I, Takita C, Tsumura H. The effect of selective cyclooxygenase-2 inhibitor on human osteoclast precursors to influence osteoclastogenesis in vitro. Mod Rheumatol. 2009;19:192–198. | ||
Igarashi K, Woo JT, Stern PH. Effects of a selective cyclooxygenase-2 inhibitor, celecoxib, on bone resorption and osteoclastogenesis in vitro. Biochem Pharmacol. 2002;63:523–532. | ||
Yoon DS, Yoo JH, Kim YH, Paik S, Han CD, Lee JW. The effects of COX-2 inhibitor during osteogenic differentiation of bone marrow-derived human mesenchymal stem cells. Stem Cells Dev. 2010;19:1523–1533. | ||
Goodman S, Ma T, Trindade M, et al. COX-2 selective NSAID decreases bone ingrowth in vivo. J Orthop Res. 2002;20:1164–1169. | ||
Murnaghan M, Li G, Marsh DR. Nonsteroidal anti-inflammatory drug-induced fracture nonunion: an inhibition of angiogenesis? J Bone Joint Surg Am. 2006;88 Suppl 3:140–147. | ||
Muller M, Raabe O, Addicks K, Wenisch S, Arnhold S. Effects of non-steroidal anti-inflammatory drugs on proliferation, differentiation and migration in equine mesenchymal stem cells. Cell Biol Int. 2011;35:235–248. | ||
Visser K, Katchamart W, Loza E, et al. Multinational evidence-based recommendations for the use of methotrexate in rheumatic disorders with a focus on rheumatoid arthritis: integrating systematic literature research and expert opinion of a broad international panel of rheumatologists in the 3E Initiative. Ann Rheum Dis. 2009;68:1086–1093. | ||
Cronstein BN. Low-dose methotrexate: a mainstay in the treatment of rheumatoid arthritis. Pharmacol Rev. 2005;57:163–172. | ||
Singh JA, Furst DE, Bharat A, et al. 2012 update of the 2008 American College of Rheumatology recommendations for the use of disease-modifying antirheumatic drugs and biologic agents in the treatment of rheumatoid arthritis. Arthritis Care Res. 2012;64:625–639. | ||
Xian CJ, Cool JC, Scherer MA, et al. Cellular mechanisms for methotrexate chemotherapy-induced bone growth defects. Bone. 2007;41:842–850. | ||
Fan CM, Foster BK, Hui SK, Xian CJ. Prevention of bone growth defects, increased bone resorption and marrow adiposity with folinic acid in rats receiving long-term methotrexate. PloS one. 2012;7:e46915. | ||
Xian CJ, Cool JC, Scherer MA, Fan C, Foster BK. Folinic acid attenuates methotrexate chemotherapy-induced damages on bone growth mechanisms and pools of bone marrow stromal cells. J Cell Physiol. 2008;214:777–785. | ||
Fan C, Cool JC, Scherer MA, et al. Damaging effects of chronic low-dose methotrexate usage on primary bone formation in young rats and potential protective effects of folinic acid supplementary treatment. Bone. 2009;44:61–70. | ||
Minaur NJ, Kounali D, Vedi S, Compston JE, Beresford JN, Bhalla AK. Methotrexate in the treatment of rheumatoid arthritis. II. In vivo effects on bone mineral density. Rheumatology. 2002;41:741749. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.