Back to Journals » Journal of Experimental Pharmacology » Volume 10
Effects of blackberry (Morus nigra) fruit juice on levodopa-induced dyskinesia in a mice model of Parkinson's disease
Authors Fahimi Z , Jahromy MH
Received 6 January 2018
Accepted for publication 16 April 2018
Published 4 July 2018 Volume 2018:10 Pages 29—35
DOI https://doi.org/10.2147/JEP.S161782
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Bal Lokeshwar
Zahra Fahimi, Mahsa Hadipour Jahromy
Herbal Pharmacology Research Center, Department of Pharmacology, Faculty of Medicine, Tehran Medical Sciences Branch, Islamic Azad University, Tehran, Iran
Background and objective: Levodopa-induced dyskinesia (LID) is a movement disorder that occurs due to levodopa consumption for a long period to attenuate Parkinsonism. Plants have been the basis for medical treatments in human history and still widely practiced. Blackberry (Morus nigra) is one of the fruits rich in anthocyanin. The present study examined the effect of blackberry fruit juice on LID in 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-induced Parkinson’s disease in mice.
Materials and methods: In this study, 42 male mice were used, which were divided into six groups equally: one control group and five groups receiving MPTP injection. After confirmation of Parkinsonism in MPTP groups, one group was preserved without treatment and four other groups were treated with levodopa (50 mg/kg ip). After the onset of LID (2 weeks), one group was kept without additional treatment and three other groups were treated with three different doses of blackberry fruit juice (5, 10, and 15 mL/kg) with levodopa orally for 7 days. Abnormal involuntary movement scale (AIMS) and cylinder behavioral test were carried out according to the schedule. The collected data were analyzed using the SPSS software with the significant level of P<0.05.
Results: Parkinson’s disease was confirmed with AIMS test on the fourth day after MPTP injection. The onset of LID was observed after 2 weeks of levodopa treatment using both behavioral tests. The result of administration of M. nigra fruit juice for 1 week showed that this addition is useful in hindering LID. These effects were more pronounced at doses 10 and 15 mL/kg with nearly the same results on attenuating AIMS. Low dose of the fruit juice does not seem to affect LID significantly.
Conclusion: M. nigra fruit juice is effective to attenuate LID in an MPTP-induced Parkinson mice model.
Keywords: Morus nigra, Parkinsonism, levodopa, LID, mice
Introduction
Parkinson’s disease is one of the most common degenerative brain disorders with motor dysfunction symptoms, and it is the second neurodegenerative disease after Alzheimer’s disease.1 Today, this disease is among the most important causes of disability worldwide, especially in old societies.
The prevalence of Parkinson’s disease is ~18–418/100,000 worldwide.2 Investigations indicated that the prevalence ratio of Parkinsonism among men and women is 3:2.1
Typically, the prevalence of this disease increases after the age of 50 years. The most common clinical sign of the disease is tremor at rest in one hand accompanied with decreased hand movements (arm swing) while walking and pain in the shoulder. Other major symptoms of the disease are muscle stiffness (rigidity) and motor slowness (bradykinesia).3–5 As symptoms develop, patient is unable to get up from the seat or turn over in the bed. An increasing motor dysfunction may occur in walking and the balance of the patient, which leads him/her to fall repeatedly. Moreover, behavioral and cognitive changes are very common.5 Patients suffering from Parkinsonism experience a reduced quality of life due to the chronic course of the disease as well as the adverse effects due to receiving different drugs.7
Several animal models have been introduced to help in better understanding the mechanisms involved in Parkinsonism. The fact that 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) causes Parkinsonism in humans is a major reason for using this toxin chemical substance to induce it in animals. The administration of MPTP to mice is one of the most common regimen that causes apoptosis and depletes striatal dopamine (DA) terminal nerves, and the dopaminergic lesion stabilizes after its administration for a month.6,7 Therefore, in this study, we first induced Parkinson’s disease in mice, using MPTP.
Drug treatments such as levodopa and DA agonists improve the early symptoms of the disease. As the disease progresses and levodopa treatment continues, the drug eventually becomes ineffective that characterized by involuntary movements called dyskinesia. Therefore, levodopa-induced dyskinesia (LID) is a form of dyskinesia that often involves hyperkinetic movements. Development of LID is reported to be related to pulsatory stimulation of DA.8,9
Extensive researches have been conducted on the effectiveness of different herbs and supplements to treat Parkinsonism and improve LID.8–11 Blackberry (Morus nigra) is one of the fruits that have been reported for its effectiveness in Parkinsonism. It belongs to Moraceae family, native to southwest Asia.12 Planting this shrub in its native habitants has a very long history. Therefore, natural and wild species cannot be precisely specified. This fruit is rich in glucose, vitamins A and C, tannins, and minerals. Concerning antioxidant activity, it is the best one, which has strong phytochemicals such as anthocyanin. The major component of anthocyanin in blackberry is cyanidin-3-glucoside.13,14 Despite many studies that have been carried out on the various effects of M. nigra, no research has been reported about its effect on reducing the symptoms of LID. Thus, this study aims to investigate the effect of blackberry fruit juice on LID in an animal model of MPTP-induced Parkinson’s disease.
Materials and methods
Animals
Forty-two male BALB/c mice, weighing 25–30 g, were housed under the following laboratory conditions: temperature 23±1°C, humidity 40%–60%, 12 h:12 h light/dark cycle (lights on at 07:00 h). Food and water were available ad libitum. All the experiments were carried out between 10 am and 3 pm in testing rooms adjacent to the animal rooms. Mice were treated in accordance with the current law and the National Institutes of Health Guide for the Care and Use of Laboratory Animals. The ethical and legal approval for animal experiments of the present study has been obtained from Research and Technology Department of Azad University, Tehran Medical Sciences Branch, Tehran, Iran.
Mice were divided into six groups equally: a control group and five other groups that received MPTP injection. After confirmation of Parkinsonism in MPTP groups, one group was preserved without treatment and four other groups treated with levodopa (50 mg/kg ip). After the onset of LID (2 weeks), one group was kept without additional treatment and three other groups were treated with three different doses of blackberry fruit juice (5, 10, and 15 mL/kg) with levodopa orally for 7 days. Abnormal involuntary movement scale (AIMS) and cylinder behavioral tests were carried out according to the schedule.
AIMS test
On test days, animals were placed in a cage (30 cm×20 cm×20 cm) on a mirror to observe any abnormal involuntary movements in a total time of 3 h with 30 min intervals for 1 min each time (totally, seven times in 3 h). Abnormal involuntary movements including axial, orolingual, limbs, and locomotor movements were considered. Severities of 0–4 were assigned for each movement. A score of 0 was assigned for the absence of AIMS; 1 was assigned for occasional AIMS (<50% of observation time); 2 was assigned for frequent AIMS (>50% of observation time); 3 was assigned for AIMS that was continuous but interrupted by strong sensory stimuli; and 4 was assigned for continuous, uninterrupted AIMS. For all AIMS category, the scores were summed at each time point and then the average of multiple observations is calculated and reported.15
Cylinder test
The cylinder test is designed to evaluate locomotor function or spontaneous activity in rodent models of central nervous system disease. As the animal moves in an open-top, clear cylinder, its forelimb activity while rearing against the wall of the cylinder is recorded. The use of forelimbs is defined by the placement of the whole forepaws on the wall of the cylinder, which indicates their use for body support. The contacts of forelimbs while rearing are scored with a total of 20 contacts recorded for each animal in the maximum time of 3 min. The numbers of impaired and nonimpaired forelimbs’ contacts are calculated as a percentage of total contacts.15 The test performed on days 11, 15, 18, and 26 after levodopa treatment.
Drugs and fruit
MPTP (98%) was purchased from SERVA (FeinBiochemica, Heidelburg, Germany), and levodopa was purchased from Ramopharmin Company (Tehran, Iran). Blackberry fruit were harvested from a garden in North Khorasan province, then washed, chilled to 4°C, and stored. The whole juice was extracted with the aid of electric juicer and then filtered and stored in clean jars in fridge (33%, w/v). The major anthocyanin in blackberry is cyanidin-3-glucoside. The total anthocyanin content ranged from 70 to 200 mg/100 g with a mean of 137 mg/100 g.16
Developing Parkinsonism
To create Parkinsonism model, 15 mL/kg of MPTP was injected intraperitoneally to all mice in MPTP groups. AIMS behavioral test was performed on the second and fourth days post-MPTP injection, which finally confirmed the disorder.
Developing LID
On the fifth day, one group remained without treatment, whereas other four groups were treated with ip injection of 50 mg/kg levodopa. Administration of high dose of levodopa showed rapid induction of dyskinesia in short period. Dyskinesia tests including AIMS and cylinder tests were done on days 8, 11, 15, and 18 till LID was confirmed (after 2 weeks of levodopa treatment).
Administration of blackberry fruit juice
After confirming LID on day 18, four groups receiving levodopa were divided randomly as follows: one group kept without fruit juice treatment and three other groups treated with three different doses of blackberry fruit juice (5, 10, and 15 mL/kg). Blackberry juice was administered orally using gavage needle for 7 consecutive days, started from day 19. Moreover, AIMS behavioral tests were carried out after 1 week of treatment, ie, on day 26.
Figure 1 represents the classification of experimental groups.
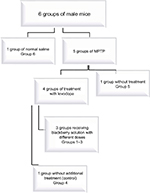  | Figure 1 Classification of experimental groups. Abbreviation: MPTP, 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine. |
Statistical analysis
To analyze the results, SPSS software (24th edition; IBM Corporation, Armonk, NY, USA) was used. The one-way analysis of variance, followed by Tukey’s tests when applicable, was performed, and P<0.05 was considered statistically significant.
Results
Parkinson’s disease induction
The first evaluation was carried out on the second day of the study. Average score of AIMS test in all groups was nearly the same indicating that no changes in motor function behaviors happened (control group 0.13±0.028 and one MPTP group 0.14±0.535). On the fourth day, changes were observed in scores, and the analysis of scores showed that Parkinson’s disease was confirmed in five other groups (P=0.033, df=1.176, F(8,54)=62). Results of AIMS scores in one MPTP-induced Parkinsonism group are shown in Table 1, from day 2 to the end of the study when compared with the control group (P<0.05).
LID induction
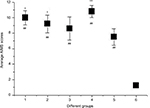
On day 5, levodopa injection was started for groups 1–4 for 14 continuous days, and AIMS and cylinder tests were performed on days 8, 11, 15, and 18. There was no significant difference between the first four groups and group 5 on days 8 and 11, while on days 15 and 18 movement disorders were different. Average scores of AIMS test after levodopa treatment on day 15 increased significantly in groups 1–4, which were 6.17±0.708, 5.67±0.823, 6.57±1.327, and 7.80±1.453, respectively. Average scores of AIMS in six groups on day 18 are presented in Figure 2. Tukey’s test was used for multiple comparisons among groups. The analysis of AIMS scores indicated that the differences between all four groups (1–4), group 5 (without levodopa treatment), and control (group 6) are significant (P<0.05) (Figure 2).
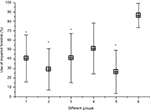
In cylinder test, when we analyzed the statistical results, the differences were only significant between groups 1, 2, 3, and 5 when compared with control, but no differences were reported between groups receiving levodopa treatment (1–4) and group 5 that was without treatment (Figure 3).
Effect of blackberry fruit juice on LID
Blackberry fruit juice at doses 5, 10, and 15 mL/kg was administered to groups 1–3, respectively, from day 19 for 7 consecutive days. Group 4 received only water. The observation showed that as doses of blackberry fruit juice increased, average AIMS scores decreased. The average scores in groups 1–4 were 7.27±1.82, 4.35±1.94, 5.18±1.49, and 8.85±1.74, respectively. One-way analysis of variance showed a significant difference among groups. Tukey’s test indicated that there is a significant difference (P<0.0.5) between groups 2 and 3 when compared with group 4. It was observed that mice in group 4 (without blackberry) and group 1 (low dose of blackberry) had undesirable movement condition compared to other groups. Statistical difference was not significant between groups 1 and 4 (P>0.05). The difference suggested that blackberry fruit juice at doses 10 and 15 mL/kg had similar effects on attenuating the LID. Lack of difference between groups 1 and 4 showed that blackberry fruit juice at dose 5 mL/kg had no effect on LID (Figure 4).
Discussion and conclusion
Blackberry is among the best food regarding antioxidant activity. It has strong phytochemicals such as anthocyanin. Although many studies have been carried out on various effects of blackberry (M. nigra),17–24 including its effect on reducing the symptoms of Parkinsonism,20 no research has been conducted on its effect to control LID. The results of this study indicated that the blackberry fruit juice has positive effect on controlling LID in Parkinson’s disease. Some similar studies are presented here.
Strathearn et al19 in the USA suggested that the blackberry plant has anthocyanin, a strong antioxidant, which can be effective in the treatment of neurodegenerative disease such as Parkinson’s disease. In Gao et al’s analytic study,20 consumption of food by 130,000 people was examined, and it was declared that blackberry is effective in reducing the risk of Parkinson’s disease. Rahman et al21 in an experimental research in Japan reported that the use of anthocyanin available in blackberry with the dose of 100 mg/kg can adjust the level of neurotransmitters such as DA, which is one of the affecting factors in Parkinson’s disease. Vauzour et al22 stated that flavonoids available in various plants including blackberry are effective to prevent Parkinson’s disease.
Ziaei et al23 suggested that blackberry can reduce the symptoms of Parkinsonism and decrease oxidative stress markers and death of dopaminergic cells. Turguta et al investigated the effect of blackberry fruit juice in the treatment of cognitive disorder and oxidative stress in healthy Mus musculus. The results showed that blackberry can potentially lead to cognitive improvement and aging prevention due to decreasing the activity of free radicals and high antioxidant capacity.24
Albarracin et al25 investigated the effect of natural antioxidants (fruits, vegetables, plants, and various drinks) in the treatment of Alzheimer’s and Parkinson’s diseases and confirmed the efficiency of such food diet.
Some other therapeutic effects of blackberry have also been reported. Del Rio et al26 and Neto27 investigated the diet rich in polyphenols including blackberry and its positive protective effect on the prevention of cardiovascular disease and cancers.
Nikkhah et al28 examined the effect of some chemical factors on the stability of anthocyanin extracted from blackberry. In this study, anthocyanin pigments extracted from blackberry were exposed to different sugar concentration, H2O2, SO2, and ascorbic acid. The effect of these materials on the stability of anthocyanin was examined, and it was concluded that each of these substances has different effects on the stability of anthocyanin.
Many studies confirmed the antioxidant activities and healthy benefits of anthocyanin in different fruits and vegetables.29–31 Anthocyanin has been accepted as a drug in many countries. Studies suggested that natural anthocyanin available in fruits and vegetables is effective on many vascular degenerative diseases. Other beneficial effects of anthocyanin are anti-angiogenesis, anti-hyperlipemia and anti-inflammatory effects.31–39
It is also believed that LID has consistently been related to excessive DA,9 although not strictly paralleled by a raise in striatum DA tone.40 Although DA provision is the main and most important therapy for Parkinson’s disease, its excessive amounts after many years of therapy could impose bradykinesia or dyskinesia via unknown mechanism probably due to its oxidation. This might be the reason why antioxidants postpone LID when coadministered with levodopa in clinic.40–42
The results obtained from this study showed that blackberry fruit juice at doses ≥10 mL/kg can be effective in controlling the LID. Investigation of the effect of blackberry fruit juice on human samples is recommended, and the efficiency of flavonoids available in other plants can be considered as future studies.
Disclosure
The authors report no conflicts of interest in this work.
References
Wirdefeldt K, Adami HO, Cole P, Trichopoulos D, Mandel J. Epidemiology and etiology of Parkinson’s disease: a review of the evidence. Eur J Epidemiol. 2011;26(Suppl 1):S1–S58. | ||
Jankovic J. Parkinson’s disease: clinical features and diagnosis. J Neurol Neurosurg Psychiatry. 2008;79(4):368–376. | ||
Massano J, Bhatia KP. Clinical approach to Parkinson’s disease: features, diagnosis, and principles of management. Cold Spring Harb Perspect Med. 2012;2(6):a008870. | ||
Goetz CG. The history of Parkinson’s disease: early clinical descriptions and neurological therapies. Cold Spring Harb Perspect Med. 2011;1(1):a008862. | ||
Martinez-Martin P, Rodriguez-Blazquez C, Forjaz MJ, Kurtis MM. Impact of pharmacotherapy on quality of life in patients with Parkinson’s disease. CNS Drugs. 2015;29(5):397–413. | ||
Lundblad M, Usiello A, Carta M, Håkansson K, Fisone G, Cenci M. Pharmacological validation of a mouse model of l-DOPA-induced dyskinesia. Exp Neurol. 2005;194:66–75. | ||
Jackson-Lewis V, Przedborski S. Protocol for the MPTP mouse model of Parkinson’s disease. Nat Protoc. 2007;2:141–151. | ||
Schober A. Classic toxin-induced animal models of Parkinson’s disease: 6-OHDA and MPTP. Cell Tissue Res. 2004;318:215–224. | ||
Yang X, Chen Y, Hong X, et al. Levodopa/benserazide microspheres reduced levodopa-induced dyskinesia by downregulating phosphorylated GluR1 expression in 6-OHDA-lesioned rats. Drug Des Devel Ther. 2012;6:341. | ||
Chung V, Liu L, Bian Z, et al. Efficacy and safety of herbal medicines for idiopathic Parkinson’s disease: a systematic review. Mov Disord. 2006;21(10):1709–1715. | ||
Li XZ, Zhang SN, Liu SM, Lu F. Recent advances in herbal medicines treating Parkinson’s disease. Fitoterapia. 2013;84:273–285. | ||
Kim TH, Cho KH, Jung WS, Lee MS. Herbal medicines for Parkinson’s disease: a systematic review of randomized controlled trials. PLoS One. 2012;7(5):e35695. | ||
Shim YH, Park JY, Choi WW, et al. Herbal medicine treatment for drug-induced Parkinsonism. J Altern Complement Med. 2015;21(5):273–280. | ||
Naderi GA, Asgary S, Sarraf-Zadegan N, Oroojy H, Afshin-Nia F. Antioxidant activity of three extracts of Morus nigra. Phytother Res. 2004;18(5):365–369. | ||
Cui G, Yang X, Wang X, et al. Ranitidine reduced levodopa-induced dyskinesia in a rat model of Parkinson’s disease. Neuropsychiatr Dis Treat. 2014;10:39–46. | ||
Lee J. Rosaceae products: anthocyanin quality and comparisons between dietary supplements and foods. NFS J. 2016;4:1–8. | ||
Mesquita Padilha M, Vilela FC, da Silva MJ, dos Santos MH, Alves-da-Silva G, Giusti-Paiva A. Antinociceptive effect of the extract of Morus nigra leaves in mice. J Med Food. 2009;12(6):1381–1385. | ||
Volpato GT, Calderon IM, Sinzato S, Campos KE, Rudge MV, Damasceno DC. Effect of Morus nigra aqueous extract treatment on the maternal-fetal outcome, oxidative stress status and lipid profile of streptozotocin-induced diabetic rats. J Ethnopharmacol. 2011;138(3):691–696. | ||
Strathearn KE, Yousef GG, Grace MH, et al. Neuroprotective effects of anthocyanin- and proanthocyanidin-rich extracts in cellular models of Parkinson’s disease. Brain Res. 2014;1555:60–77. | ||
Gao X, Cassidy A, Schwarzschild MA, Rimm EB, Ascherio A. Habitual intake of dietary flavonoids and risk of Parkinson disease. Neurology. 2012;78(15):1138–1145. | ||
Rahman MM, Ichiyanagi T, Komiyama T, Sato S, Konishi T. Effects of anthocyanins on psychological stress-induced oxidative stress and neurotransmitter status. J Agric Food Chem. 2008;56(16):7545–7550. | ||
Vauzour D, Vafeiadou K, Rodriguez-Mateos A, Rendeiro C, Spencer JP. The neuroprotective potential of flavonoids: a multiplicity of effects. Genes Nutr. 2008;3(3–4):115–126. | ||
Ziaei A, Nasri S, Roughani M, Niknami Z. The Effect of Blackberry Extract on Parkinson [MS thesis]. Payame Noor University of Tehran, Tehran; 2012. | ||
Turguta NH, Mert DG, Kara H, et al. Effect of black mulberry (Morus nigra) extract treatment on cognitive impairment and oxidative stress status of D-galactose-induced aging mice. Pharm Biol. 2016;54(6):1052–1064. | ||
Albarracin SL, Stab B, Casas Z, et al. Effects of natural antioxidants in neurodegenerative disease. Nutr Neurosci. 2012;15(1):1–9. | ||
Del Rio D, Rodriguez-Mateos A, Spencer JP, Tognolini M, Borges G, Crozier A. Dietary (poly) phenolics in human health: structures, bioavailability, and evidence of protective effects against chronic diseases. Antioxid Redox Signal. 2013;18(14):1818–1192. | ||
Neto CC. Cranberry and blueberry: evidence for protective effects against cancer and vascular diseases. Mol Nutr Food Res. 2007;51(6):652–664. | ||
Nikkhah E, Heidari R, Khayami M. Investing the effect of some chemical factors on stability of anthocyanis extracted from blackberry (Morus nigra). Iranian J Biol. 2012;25(1):32–43. | ||
Brenes CH, Del Pozo-Insfran D, Talcott S. Stability of copigmented anthocyanins and ascorbic acid in a grape juice model system. J Agric Food Chem. 2005;53(1):49–56. | ||
Huang DJ, Lin CD, Chen HG, Lin YH. Antioxidant and antiproliferative activities of sweet potato. Bot Bull Acad Sin. 2004;45:179–186. | ||
Netzel M, Strass G, Herb M, et al. The excretion and biological antioxidant activity of elderberry antioxidants in healthy humans. Food Res Intern. 2005;38:905–910. | ||
Panovsaka TK, Kulevanova S, Stefova M. In vitro antioxidant activity of some Teucrium species (Lamiaceae). Acta Pharm. 2005;55:207–214. | ||
Buchert J, Koponen JM, Suutarinen M, et al. Effect of enzyme-aided pressing on anthocyanin yield and profiles in bilberry and blackcurrant juices. J Sci Food Agric. 2005;85:2548–2556. | ||
Kamei H, Kojima T, Hasegawa M, et al. Suppression of tumor cell growth by anthocyanins in vitro. Cancer Invest. 1995;13(6):590–594. | ||
Kähkönen MP, Heinämäki J, Ollilainen V, Heinonen M. Berry anthocyanins: isolation, identification and antioxidant activities. J Sci Food Agric. 2003;83:1403–1411. | ||
Knekt P, Kumpulainen J, Järvinen R, et al. Flavonoid intake and risk of chronic diseases. Am J Clin Nutr. 2002;76(3):560–568. | ||
Kong J, Chia L, Goh N, Chia T, Brouillard R. Analysis and biological activities of anthocyanins. Phytochemistry. 2003;64:923–933. | ||
Rossi A, Serraino I, Dugo P, et al. Protective effects of anthocyanins from blackberry in a rat model of acute lung inflammation. Free Radic Res. 2003;37(8):891–900. | ||
Viljanen K, Kivikari R, Heinonen M. Protein-lipid interactions during liposome oxidation with added anthocyanin and other phenolic compounds. J Agric Food Chem. 2004;52(5):1104–1111. | ||
Carocho M, Ferreira IC. A review on antioxidants, prooxidants and related controversy: natural and synthetic compounds, screening and analysis methodologies and future perspectives. Food Chem Toxicol. 2013;51:15–25. | ||
Lambert JD, Elias RJ. The antioxidant and pro-oxidant activities of green tea polyphenols: a role in cancer prevention. Arch Biochem Biophys. 2010;501(1):65–72. | ||
Park H-Y, Ryu Y-K, Go J, Son E, Kim K-S, Kim MR. Palmitol serotonin inhibits levodopa induced abnormal involuntary movements in mouse Parkinson model. Exp Nerobiol. 2016;25(4):174–184. |
 © 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2018 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.