Back to Journals » Cancer Management and Research » Volume 12
Effects of Anesthesia on Postoperative Recurrence and Metastasis of Malignant Tumors
Authors Fan X, Wang D, Chen X , Wang R
Received 1 June 2020
Accepted for publication 4 August 2020
Published 24 August 2020 Volume 2020:12 Pages 7619—7633
DOI https://doi.org/10.2147/CMAR.S265529
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Antonella D'Anneo
Xiaoqing Fan,1,2 Delong Wang,1,2 Xueran Chen,3,4 Ruiting Wang1,2
1Department of Anesthesiology, The First Affiliated Hospital of USTC, Division of Life Sciences and Medicine, University of Science and Technology of China (USTC), Hefei, Anhui 230001, People’s Republic of China; 2Department of Anesthesiology, Anhui Provincial Hospital, Hefei 230001, Anhui, People’s Republic of China; 3Anhui Province Key Laboratory of Medical Physics and Technology, Institute of Health and Medical Technology, Hefei Institutes of Physical Science, Chinese Academy of Sciences, Hefei, Anhui 230031, People’s Republic of China; 4Department of Molecular Pathology, Hefei Cancer Hospital, Chinese Academy of Sciences, Hefei, Anhui 230031, People’s Republic of China
Correspondence: Xueran Chen; Ruiting Wang Email [email protected]; [email protected]
Abstract: It is difficult to control the recurrence and metastasis of malignant tumors; furthermore, anesthesia is considered one of the main influencing factors. There has been increasing clinical attention on the effects of anesthetic drugs and methods on postoperative tumor growth and metastasis. We reviewed the effects of anesthesia on tumor recurrence and metastasis; specifically, the effects of anesthetic agents, anesthesia methods, and related factors during the perioperative period on the tumor growth and metastasis were analyzed. This study can provide reference standards for rational anesthesia formulations and cancer-related pain analgesia protocols for surgical procedures in patients with malignant tumors. Moreover, it contributes toward an experimental basis for the improvement and development of novel anesthetic agents and methods.
Keywords: anesthesia, cancer, recurrence, metastasis, prognosis, immunosuppression
Introduction
It remains difficult to control the recurrence and metastasis of malignant tumors, which could be strongly associated with multiple factors that affect prognosis, including anesthesia.1,2 There have been several worldwide studies on the relationships between anesthetic agents and methods with cancer growth and immune function in patients with cancer.3,4 These studies have provided valuable references for selecting anesthesia and perioperative management for patients with cancer. Different anesthetic agents have been shown to have different effects on immunity, recurrence, and metastasis in patients with cancer.5,6 Further, different anesthesia methods, including epidural, intravenous, inhalation, and combined intravenous and inhalation anesthesia, as well as intercostal nerve block, could have different effects on cancer recurrence or metastasis.7–9 In surgery-naïve healthy individuals, epidural anesthesia and general anesthesia were found to induce mild transient immune suppression; however, surgical stress significantly increased the risk of peri-/postoperative cancer recurrence and metastasis.1,10 Contrastingly, epidural anesthesia reduces the risk of cancer recurrence through surgical stress reduction. Although there is no report of paravertebral block reducing cancer recurrence, it is associated with a higher overall survival rate after lung cancer surgery.11 Therefore, anesthesia management of patients with cancer could significantly affect their long-term prognosis. Clinical studies have proposed several beneficial measures, including appropriate induction agent selection, minimal volatile anesthetic agent usage, and minimal combined use of opioids and cyclooxygenase inhibitors.12 Moreover, other intraoperative factors, such as blood transfusion and temperature regulation, affect the long-term prognosis of patients with cancer.13,14 This study aimed to review the effects of anesthetic agents, anesthesia methods, and intraoperative factors on cancer recurrence and metastasis.
Effect of Anesthetic Drugs on Cancer Recurrence and Metastasis
Intravenous Anesthetic Agents
Intravenous general anesthetic agents act on the central nervous system to achieve anesthesia.14–16 They are currently the main agents for anesthesia induction and maintenance. Studies have shown that most intravenous general anesthetic agents can suppress immune system function and affect cancer progression (Table 1).
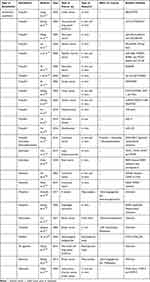 |
Table 1 The Recent Studies on the Effect of Intravenous Anesthetics on Cancers |
Propofol
Propofol is a short-acting intravenous anesthetic that is widely used as an intraoperative and postoperative sedative and hypnotic agent.17,18 Its anesthetic effects involve direct GABAA receptor activation, which slows their channel-closing time and blocks sodium channels.19,20 Li et al suggested that propofol-based total intravenous anesthesia (TIVA) for breast cancer surgery could reduce the risk of recurrence within the first 5 years after modified radical mastectomy.17 Propofol anesthesia was associated with better survival in hepatectomy for hepatocellular carcinoma patients,21 in radical prostatectomy for prostate cancer patients,22 in patients who underwent surgery for infiltrating bladder cancer,23 in pancreatic cancer surgery,24 and in open intrahepatic cholangiocarcinoma surgery.25 However, a recent retrospective cohort study showed that propofol-based TIVA was not significantly associated with a decrease in the 1-year overall or cancer-related mortality after gastric cancer surgery, as compared with inhalation anesthesia.26,27 In addition, a randomized control trial involving more than 976 women who underwent breast cancer surgery demonstrated that no significant difference in the locoregional recurrence or overall 5-year survival rates occurred after breast surgery using desflurane or propofol anesthesia.28 A retrospective study with 6305 patients demonstrated that propofol may have a survival advantage compared with sevoflurane among breast cancer patients.29 However, another study showed that paravertebral block with propofol anesthesia does not improve survival compared with sevoflurane anesthesia for breast cancer surgery.30 The inherent weaknesses of retrospective analyses were made apparent. With regard to its mechanism, propofol is considered to protect against immunosuppression during the perioperative period and has a lower inflammatory response than volatile agents.15 Propofol can induce apoptosis by activating different signaling pathways and inhibiting cancer cell growth.31,32 Deng et al reported that propofol could inhibit in vitro and in vivo colorectal cancer cell (CRC) migration through PI3K/AKT signaling activation and induction of epithelial-to-mesenchymal transition (EMT).33 By downregulating transforming growth factor β1 (TGF-β1) expression, propofol effectively inhibits osteosarcoma cell proliferation and invasion, and induces their apoptosis.34 Liu et al suggested that propofol inhibits pancreatic cancer cell (PANC-1) invasion and induces their apoptosis through microRNA (miR)-21/Slug signaling modulation.35 Only one study found a different conclusion, whereby propofol induces the proliferation and invasion of gallbladder cancer cells.36 Based on this, we found that an increasing number of studies have discovered that propofol plays an important role in cancer by regulating the expression of multiple signaling pathways, downstream molecules, microRNAs, and long non-coding RNAs. Emerging evidence has indicated that propofol can improve the anti-tumor effect of some small molecular compounds or chemotherapeutic drugs. Moreover, most clinical trials imply that propofol is related with better survival outcomes in cancer patients after surgery.
Ketamine
Ketamine has an immunomodulatory effect on macrophages, lymphocytes, and mast cells.37–39 A breast cancer rat model study reported that ketamine, sodium thiopental, and inhaled anesthetic agents could promote tumor metastasis and were inversely associated with NK cell activity.40 This effect was significantly reduced by pre-administering the β-blocker nadolol or through long-term low-dose immunostimulant administration. He et al reported that ketamine could induce anti-apoptotic protein Bcl-2 upregulation and promote breast cancer cell invasion and proliferation.41 Contrastingly, a recent study suggested that ketamine was an N-methyl-D-aspartate (NMDA) antagonist that inhibits pancreatic cancer cell proliferation and apoptosis.42 These indicate that ketamine has cancer-promoting effects mainly involving immune function suppression; however, its direct effect on cancer cells remains unclear.
Opioids
Given their strong analgesic effects, opioids are widely used in perioperative analgesia and treatment of postoperative pain and chronic cancer pain.43,44 Opioid receptors are located in both neurons and immune cells; further, they can be present in several tumor cell types.45 Opioid promotion of tumor metastasis is intricately associated with the role of μ receptors.46,47 Opioid receptors, especially μ receptors, are expressed on vascular endothelial cells. Binding between opioids and μ receptors promotes nitric oxide production, which is involved in angiogenesis in endothelial cells, and VEGF-mediated angiogenesis.48,49 These processes are involved in tumor proliferation, metastasis, and recurrence. In addition, opioids can activate cyclooxygenase receptors and promote prostaglandin E2 (PGE2) production.50,51 PGE2 has been shown to promote the production of endothelin 1, VEGF, and platelet-derived growth factors in breast cancer cells, which promotes cancer invasion and metastasis.52 There has been inconsistency across recent reports with regard to opioid effects on patients with cancer. For example, the antitumor morphine effects mainly occur as anti-proliferative and pro-apoptotic effects on different cancer cell types.53,54 Koodie et al found that morphine could reduce leukocyte migration across the endothelium and tumor angiogenesis in mice.55 They suggested that morphine may be beneficial in pain management of patients with cancer through its effects on angiogenesis. It has also been reported that tramadol has an antitumor effect, revealing that tramadol use is related to enhanced postoperative outcomes in breast cancer patients.56 Furthermore, continuous administration of morphine with high doses is more likely to inhibit tumor metastasis and growth in rodent models. In contrast, intermittent injection induces withdrawal-like conditions and activates the hypothalamic-pituitary-adrenal (HPA) axis known to facilitate cancer metastasis and progression. Therefore, not only is the type of opioid receptor potentially significant, but the method of dosing may influence whether opiate analgesia has a pro- or anti-tumor effect.
Non-Steroidal Anti-Inflammatory Drugs (NSAIDs)
NSAIDs are a class of analgesics that lack steroid structures.57 They can inhibit cyclooxygenase, reduce the production of PG inflammatory mediators, and exert anti-inflammatory and analgesic effects.58,59 NSAIDs are considered capable of reducing the development, recurrence, and proliferation of various cancers, including colon, breast, lung, and pancreatic cancer.60,61 NSAIDs are able to interfere with the tumor microenvironment by increasing chemo-sensitivity and apoptosis, and reducing cell migration. Furthermore, they can protect immune system function and reduce the risk for perioperative micrometastasis, and therefore are used as preoperative analgesics widely in patients with cancer. A retrospective study reported that ketorolac improved the overall survival of patients with lung cancer (p=0.05).62,63 Forget et al reported an independent association of the NSAID used at the beginning of surgery with a lower risk of metastasis after lung cancer surgery.64 Furthermore, the use of ketorolac was independently associated with longer survival. Preoperative ketorolac administration resulted in a lower cancer recurrence rate.62,64
Inhaled Anesthetic Agents
Inhaled anesthetic agents are widely clinically used given their strong anesthetic effects and ease in adjusting the anesthesia depth.16,65,66 They are currently widely considered to have adverse effects on patients with cancer by suppressing immunity and promoting tumor cell migration (Table 2).
 |
Table 2 The Recent Studies on the Effect of Volatile Anesthetics on Cancers |
Sevoflurane
Sevoflurane is an inhaled anesthetic agent widely used in cancer surgery for maintaining intraoperative anesthesia.66,67 Several retrospective studies have shown that compared with propofol anesthesia, sevoflurane anesthesia is associated with worse clinical outcomes in patients with breast, colon, rectal, and gastric cancer.68–70 Sevoflurane can suppress the immune response by regulating cytokine expression and reducing NK cell toxicity. Compared with patients undergoing radical laparoscopic hysterectomy for cervical cancer who received propofol, those who received sevoflurane had significantly lower CD3+, CD4 +, and natural killer (NK) cell counts, as well as a lower CD4+/CD8+ ratio. Regarding the mechanism of action, Shi et al demonstrated that sevoflurane promoted the self-renewal and proliferation of glioma stem cells in vitro by regulating the hypoxia-inducible factor (HIF) pathway.71 This suggests that sevoflurane may enhance tumor growth, and thus affect patient outcomes by inducing tumor stem cell proliferation. Sevoflurane has been shown to increase the expression of oncogenic protein markers, including HIF-2α and nuclear p-p38, in neck squamous cell carcinoma cells.72 This suggests that sevoflurane may play a key role in the adverse outcomes of cancer treatments, but does not eliminate the possibility that sevoflurane has different cell biological effects in various cancer types.
Isoflurane
Isoflurane is a volatile general anesthetic agent that induces and maintains general anesthesia to eliminate behavioral responses in patients undergoing tumor resection.66 Benzonana et al demonstrated that isoflurane upregulates the expression of hypoxia-inducible factors HIF1a and HIF2a, as well as vascular endothelial growth factor.73 Consequently, it promotes the growth and proliferation of RCC4 renal carcinoma cells, which may contribute to increased postoperative recurrence. Notably, propofol partially reduces the malignant capacity of cancer cells by inhibiting isoflurane-triggered HIF-1α activation. Isoflurane exposure can promote cancer cell proliferation and inhibit apoptosis in glioblastoma cells.74 Isoflurane increases non-small cell lung cancer cell proliferation via Akt-mTOR signaling pathway activation.75 Similarly, isoflurane inhibits apoptosis through caveolin-1 expression upregulation in human colon cancer cells.76 In summary, these phenomena suggest that treatment with isoflurane might be a factor promoting the progression of most types of cancers.
Desflurane
Desflurane can cause adverse outcomes in most patients undergoing cancer surgery; however, there is a need for further studies on the specific biological mechanisms.65,77 Exposure of human ovarian cancer cells to 10.3% desflurane for 2 hours promotes their migration by increasing metastasis-related gene expression, including VEGF-a, MMP-11, CXC chemokine receptor 2, and TGF-β.78 Contrastingly, Muller-Edenborn et al reported that desflurane inhibits MMP-9 release from neutrophils and inhibits the metastasis of colon cancer cells.79 Perioperative use of low-flow desflurane reduces the inhibitory effects on neutrophils and T cells; additionally, it protects immune function.80,81 As for desflurane, it played a pivotal role in adverse outcomes in most patients undergoing cancer surgery; further exploration of its specific biological mechanism is still warranted.
Local Anesthetic Agents
Local anesthetic agents exert their effects by blocking voltage-gated sodium channels (VGSC) on nerve cell membranes.82 Tumor cell membranes have VGSCs, which are associated with tumor cell invasion and metastasis.83–85 There have been recent studies carried out on the antitumor properties of local anesthetics (Table 3). Sakaguchi et al reported that clinical lidocaine concentration inhibited the proliferation of the human tongue cancer cell line CAL27 induced by serum and epidermal growth factor.86 Moreover, lidocaine concentrations higher than those clinically applied caused direct cytotoxicity and anti-proliferative effects. Siekmann et al reported that clinical ropivacaine concentrations inhibited colon cancer cell proliferation in vitro in a dose-dependent manner.87 Lirk et al reported that a similar lidocaine concentration caused DNA demethylation and activated tumor suppressor genes, especially in estrogen receptor-positive breast cancer cells.88 Additionally, local anesthetic agents can promote tumor cell apoptosis. Lidocaine and bupivacaine can inhibit the MAPK signaling pathway, reduce ERK1/2 activity, upregulate p38 MAPK, and promote apoptosis in human thyroid cancer cells.89 Intravenous lidocaine has been shown to have anti-inflammatory effects. Continuous intraoperative lidocaine infusion in patients undergoing radical hysterectomy reduces early lymphocyte apoptosis and maintains the interferon-γ/IL-4 ratio.90 This indicates the protective role of lidocaine in cell-mediated immunity, which contributes to tumor recurrence suppression. Lidocaine is an ideal adjuvant drug for cancer treatment given its major therapeutic advantages, strong anti-inflammatory effects, and protective effects on innate immune system surveillance.91,92 In addition, it improves the prognosis of patients with cancer. However, its use in patients with cancer warrants further clarifications and clinical validation.
 |  |  |
Table 3 The Recent Studies on the Effect of Local Anesthetics on Cancers |
Effect of Anesthesia Methods on Growth and Metastasis of Malignant Tumors
The anesthesia effect on tumor migration and invasion remains unclear. Several retrospective studies have reported that regional anesthesia reduces the risk of tumor metastasis and recurrence. This is primarily because regional anesthesia attenuates surgery-induced neuroendocrine response and reaches the central nervous system to inhibit harmful nerve impulses.93 Therefore, it maximally suppresses the perioperative immune response. Regional anesthesia can increase NK cell activity, maintain the Th1/Th2 ratio balance, and reduce intraoperative plasma cortisol and catecholamine levels.1,94 Compared with only general anesthesia, general anesthesia combined with epidural anesthesia improves the clinical survival rate and reduces tumor malignancy.95 However, other previous studies have reported contrasting findings. A multi-center randomized controlled clinical study by Short et al reported that compared with general anesthesia combined with postoperative intravenous analgesia, general anesthesia combined with postoperative epidural analgesia neither reduced the risk of tumor recurrence nor prolonged the tumor-free survival period in patients who underwent abdominal cancer surgery.96 The reduction of cancer recurrence by epidural anesthesia could be associated with systemic conditions in the body and the biological tumor characteristics. To confirm this hypothesis, there is a need for a large-sample, multi-center, randomized controlled trial with a follow-up period that allows for validation.
Furthermore, a clinical study assessed patients with primary breast cancer who underwent general anesthesia and propofol combined with intraoperative paravertebral block anesthesia.97 The study showed that local anesthesia reduces stress response, protects immune function in patients with tumors, and reduces opioid use. In addition, vascular endothelial growth factor C, TGF-β1, acidic fibroblast growth factor, basic fibroblast growth factor, and placental growth factor levels are reduced in the veins of patients who receive propofol combined with paravertebral block anesthesia.97 These growth factors promote angiogenesis and metastatic tumor formation, which indicates that the employed anesthesia method affects plasma levels of angiogenesis-related factors in patients with primary breast cancer, which affects tumor recurrence and metastasis.
Anesthesia-Related Factors and Cancer Recurrence and Metastasis
During the perioperative period, many factors affect cancer recurrence, including the immune system, blood transfusions, hypothermia, hyperglycemia, and postoperative pain.98–100
Intact Cell-Mediated Immunity
Intact cell-mediated immunity is crucial for developing resistance to tumor metastasis.101,102 The immune response is regulated by the hypothalamic-pituitary-adrenal axis and the sympathetic nervous system. Therefore, anesthesia promotes tumor metastasis through activation of the aforementioned systems and certain tumor-derived factors.2,66,103 Activation of these systems inhibits cell-mediated immunity (CMI) and the release of catecholamines and prostaglandin E2 (PGE2). Moreover, these factors increase the levels of immunosuppressive cytokines, soluble factors (eg IL-4, IL-10, TGF-β, and VEGF), and pro-inflammatory cytokines (eg IL-6 and IL- 8). In addition, inhaled anesthetic agents and opioids inhibit CMI and promote tumor cell proliferation and angiogenesis. On the other hand, propofol inhibits tumor angiogenesis but not CMI. Moreover, regional anesthesia does not affect CMI and reduces surgery-induced neuroendocrine response by weakening afferent nerve conduction in the HPA axis and the sympathetic nervous system.85,91,104 Therefore, reducing opioid usage and inhaled anesthetic agents may reduce the risk of tumor recurrence.
Intraoperative Blood Transfusion
Compared to patients who did not receive intraoperative blood transfusion, those who did had a significantly lower disease-free and overall survival rate.105–107 Therefore, blood transfusion is an independent risk factor for determining the prognosis of patients with cancer. Contrastingly, some studies have reported that blood transfusion or immunosuppression caused by blood transfusion is not responsible for the poor postoperative prognosis.108 Rather, they indicated that the prognosis is closely associated with the biological tumor characteristics and the systemic condition of the patient. This inconsistency could be attributed to the complexity of the response of inherent growth and metastasis tumor properties to blood transfusion, as well as differences in the population and experimental groups across the studies.
Hypothermia
Hypothermia is a body temperature dysregulation that commonly occurs during anesthesia and surgery.109,110 Hypothermia can have several adverse effects on the body, including affecting the prognosis of patients with cancer.111,112 Compared with normal body temperature, Benzonana et al reported that hypothermia in Wistar rats could significantly inhibit NK cell activity and increase the susceptibility to lung metastasis.73 A study on humans by Du et al demonstrated that hypothermia could reduce Th1-type cytokine levels, increase Th2-type cytokine levels, inhibit immune cell function, and accelerate tumor progression.113 This demonstrates that hypothermia can promote tumor recurrence.
Conclusion
To date, anesthesia is considered among the major factors affecting the recurrence and metastasis of malignant tumors. Certain anesthetic agents and methods have adverse effects on the immunity of patients with cancer, which further increases the risk of tumor recurrence and metastasis. There have been increasing studies carried out on the different effects of various anesthetic agents on malignant tumors.
Traditionally, anesthesiologists would perform sedation, anesthesia, and postoperative analgesia unsure of whether the anesthetic agents affect tumor recurrence and metastasis. Subsequent studies have confirmed the specific effects of anesthetic agents on malignant tumor metastasis and recurrence. Future studies should determine the biological relationship between anesthetic agents and malignant tumors, their interaction during anesthesia, and means of assessing anesthesia effects and mechanisms on tumor recurrence and metastasis at the cellular and molecular levels. This could contribute toward significantly improving the survival rate of patients with cancer. In addition, they could provide new standards regarding the proper use of anesthetic agents and experimental evidence for developing novel anesthetic agents and methods.
Abbreviations
TIVA, Total intravenous anesthesia; CRC, Colorectal cancer cells; PANC-1, Pancreatic cancer cells; NMDA, N-methyl-D-aspartate; PGE2, Prostaglandin E2; NSAIDS, Non-steroidal anti-inflammatory drugs; NK, Natural killer; HIF, Hypoxia-inducible factor; VGSC, Voltage-gated sodium channels; VEGF-C, Vascular endothelial growth factor C; TGF-β1, Transforming growth factor β1; HPA, Hypothalamic-pituitary-adrenal; CMI, Cell-mediated immunity; PGE2, Prostaglandin E2.
Acknowledgments
This research was supported by the National Natural Science Foundation of China (grant numbers 81872066), the Innovative Program of Development Foundation of Hefei Centre for Physical Science and Technology (grant number 2018CXFX004) and the Youth Innovation Promotion Association of the Chinese Academy of Sciences (grant number 2018487).
Disclosure
The authors declare no competing interests for this work.
References
1. Kim R. Effects of surgery and anesthetic choice on immunosuppression and cancer recurrence. J Transl Med. 2018;16(1):8. doi:10.1186/s12967-018-1389-7
2. Wang W, Xiao J, Shen S, Wang S, Chen M, Hu Y. Emerging effect of anesthesia on post-operative tumor recurrence and metastasis. J Int Med Res. 2019;47(8):3550–3558. doi:10.1177/0300060519861455
3. Sessler DI, Pei L, Huang Y, et al. Recurrence of breast cancer after regional or general anaesthesia: a randomised controlled trial. Lancet. 2019;394(10211):1807–1815. doi:10.1016/S0140-6736(19)32313-X
4. Dong J, Zeng M, Ji N, et al. Impact of anesthesia on long-term outcomes in patients with supratentorial high-grade glioma undergoing tumor resection: a retrospective cohort study. J Neurosurg Anesthesiol. 2020;32(3):227–233. doi:10.1097/ANA.0000000000000588
5. Anderson SL, Duke-Novakovski T, Singh B. The immune response to anesthesia: part 2 sedatives, opioids, and injectable anesthetic agents. Vet Anaesth Analg. 2014;41(6):553–566. doi:10.1111/vaa.12191
6. Moradkhani MR, Karimi A. Role of drug anesthesia and cancer. Drug Res (Stuttg). 2018;68(3):125–131. doi:10.1055/s-0043-119071
7. Byrne K, Levins KJ, Buggy DJ. Can anesthetic-analgesic technique during primary cancer surgery affect recurrence or metastasis? Can J Anaesth. 2016;63(2):184–192. doi:10.1007/s12630-015-0523-8
8. Dubowitz JA, Sloan EK, Riedel BJ. Implicating anaesthesia and the perioperative period in cancer recurrence and metastasis. Clin Exp Metastasis. 2018;35(4):347–358. doi:10.1007/s10585-017-9862-x
9. Vahabi S, Eatemadi A. Effects of anesthetic and analgesic techniques on cancer metastasis. Biomed Pharmacother. 2017;87:1–7. doi:10.1016/j.biopha.2016.12.073
10. Longhini F, Bruni A, Garofalo E, et al. Anesthetic strategies in oncological surgery: not only a simple sleep, but also impact on immunosuppression and cancer recurrence. Cancer Manag Res. 2020;12:931–940. doi:10.2147/CMAR.S237224
11. Lee EK, Ahn HJ, Zo JI, Kim K, Jung DM, Park JH. Paravertebral block does not reduce cancer recurrence, but is related to higher overall survival in lung cancer surgery: a retrospective cohort study. Anesth Analg. 2017;125(4):1322–1328. doi:10.1213/ANE.0000000000002342
12. Fodale V, D’Arrigo MG, Triolo S, Mondello S, La Torre D. Anesthetic techniques and cancer recurrence after surgery. ScientificWorldJournal. 2014;2014:328513. doi:10.1155/2014/328513
13. Liu X, Ma M, Huang H, Wang Y. Effect of perioperative blood transfusion on prognosis of patients with gastric cancer: a retrospective analysis of a single center database. BMC Cancer. 2018;18(1):649. doi:10.1186/s12885-018-4574-4
14. Kaye AD, Patel N, Bueno FR, et al. Effect of opiates, anesthetic techniques, and other perioperative factors on surgical cancer patients. Ochsner J. 2014;14(2):216–228.
15. Yap A, Lopez-Olivo MA, Dubowitz J, Hiller J, Riedel B. Anesthetic technique and cancer outcomes: a meta-analysis of total intravenous versus volatile anesthesia. Can J Anaesth. 2019;66(5):546–561. doi:10.1007/s12630-019-01330-x
16. Jin Z, Li R, Liu J, Lin J. Long-term prognosis after cancer surgery with inhalational anesthesia and total intravenous anesthesia: a systematic review and meta-analysis. Int J Physiol Pathophysiol Pharmacol. 2019;11(3):83–94.
17. Li R, Liu H, Dilger JP, Lin J. Effect of Propofol on breast Cancer cell, the immune system, and patient outcome. BMC Anesthesiol. 2018;18(1):77. doi:10.1186/s12871-018-0543-3
18. Jiang S, Liu Y, Huang L, Zhang F, Kang R. Effects of propofol on cancer development and chemotherapy: potential mechanisms. Eur J Pharmacol. 2018;831:46–51. doi:10.1016/j.ejphar.2018.04.009
19. Sahinovic MM, Struys M, Absalom AR. Clinical pharmacokinetics and pharmacodynamics of propofol. Clin Pharmacokinet. 2018;57(12):1539–1558. doi:10.1007/s40262-018-0672-3
20. Weir CJ, Mitchell SJ, Lambert JJ. Role of GABAA receptor subtypes in the behavioural effects of intravenous general anaesthetics. Br J Anaesth. 2017;119(suppl_1):i167–i175. doi:10.1093/bja/aex369
21. Lai HC, Lee MS, Lin C, et al. Propofol-based total intravenous anaesthesia is associated with better survival than desflurane anaesthesia in hepatectomy for hepatocellular carcinoma: a retrospective cohort study. Br J Anaesth. 2019;123(2):151–160. doi:10.1016/j.bja.2019.04.057
22. Lai HC, Lee MS, Lin KT, et al. Propofol-based total intravenous anesthesia is associated with better survival than desflurane anesthesia in robot-assisted radical prostatectomy. PLoS One. 2020;15(3):e0230290. doi:10.1371/journal.pone.0230290
23. Guerrero Orriach JL, Raigon Ponferrada A, Malo Manso A, et al. Anesthesia in combination with propofol increases disease-free survival in bladder cancer patients who undergo radical tumor cystectomy as compared to inhalational anesthetics and opiate-based analgesia. Oncology. 2020;98(3):161–167. doi:10.1159/000504807
24. Lai HC, Lee MS, Liu YT, et al. Propofol-based intravenous anesthesia is associated with better survival than desflurane anesthesia in pancreatic cancer surgery. PLoS One. 2020;15(5):e0233598. doi:10.1371/journal.pone.0233598
25. Lai HC, Lee MS, Lin KT, et al. Propofol-based total intravenous anesthesia is associated with better survival than desflurane anesthesia in intrahepatic cholangiocarcinoma surgery. Medicine (Baltimore). 2019;98(51):e18472. doi:10.1097/MD.0000000000018472
26. Oh TK, Kim HH, Jeon YT. Retrospective analysis of 1-year mortality after gastric cancer surgery: total intravenous anesthesia versus volatile anesthesia. Acta Anaesthesiol Scand. 2019;63(9):1169–1177. doi:10.1111/aas.13414
27. Schaefer MS, Raub D, Xu X, et al. Association between propofol dose and 1-year mortality in patients with or without a diagnosis of solid cancer. Br J Anaesth. 2020;124(3):271–280. doi:10.1016/j.bja.2019.11.028
28. Huang YH, Lee MS, Lou YS, et al. Propofol-based total intravenous anesthesia did not improve survival compared to desflurane anesthesia in breast cancer surgery. PLoS One. 2019;14(11):e0224728. doi:10.1371/journal.pone.0224728
29. Enlund M, Berglund A, Ahlstrand R, et al. Survival after primary breast cancer surgery following propofol or sevoflurane general anesthesia-a retrospective, multicenter, database analysis of 6305 Swedish patients. Acta Anaesthesiol Scand. 2020;64(8):1048–1054. doi:10.1111/aas.13644
30. Cata JP, Forget P. Paravertebral block with propofol anaesthesia does not improve survival compared with sevoflurane anaesthesia for breast cancer surgery: independent discussion of a randomised controlled trial. Br J Anaesth. 2020;124(1):19–24. doi:10.1016/j.bja.2019.09.039
31. Wang J, Cheng CS, Lu Y, et al. Novel findings of anti-cancer property of propofol. Anticancer Agents Med Chem. 2018;18(2):156–165. doi:10.2174/1871520617666170912120327
32. Farooqi AA, Adylova A, Sabitaliyevich UY, Attar R, Sohail MI, Yilmaz S. Recent updates on true potential of an anesthetic agent as a regulator of cell signaling pathways and non-coding RNAs in different cancers: focusing on the brighter side of propofol. Gene. 2020;737:144452. doi:10.1016/j.gene.2020.144452
33. Deng F, Ouyang M, Wang X, et al. Differential role of intravenous anesthetics in colorectal cancer progression: implications for clinical application. Oncotarget. 2016;7(47):77087–77095. doi:10.18632/oncotarget.12800
34. Xu YB, Jiang W, Zhao FR, et al. Propofol suppresses invasion and induces apoptosis of osteosarcoma cell in vitro via downregulation of TGF-β1 expression. Eur Rev Med Pharmacol Sci. 2016;20(7):1430–1435.
35. Liu Z, Zhang J, Hong G, Quan J, Zhang L, Yu M. Propofol inhibits growth and invasion of pancreatic cancer cells through regulation of the miR-21/Slug signaling pathway. Am J Transl Res. 2016;8(10):4120–4133.
36. Zhang L, Wang N, Zhou S, Ye W, Jing G, Zhang M. Propofol induces proliferation and invasion of gallbladder cancer cells through activation of Nrf2. J Exp Clin Cancer Res. 2012;31(1):66. doi:10.1186/1756-9966-31-66
37. Singh V, Gillespie TW, Harvey RD. Intranasal ketamine and its potential role in cancer-related pain. Pharmacotherapy. 2018;38(3):390–401. doi:10.1002/phar.2090
38. Visser E, Schug SA. The role of ketamine in pain management. Biomed Pharmacother. 2006;60(7):341–348. doi:10.1016/j.biopha.2006.06.021
39. Quibell R, Fallon M, Mihalyo M, Twycross R, Wilcock A. Ketamine. J Pain Symptom Manage. 2015;50(2):268–278. doi:10.1016/j.jpainsymman.2015.06.002
40. Melamed R, Bar-Yosef S, Shakhar G, Shakhar K, Ben-Eliyahu S. Suppression of natural killer cell activity and promotion of tumor metastasis by ketamine, thiopental, and halothane, but not by propofol: mediating mechanisms and prophylactic measures. Anesth Analg. 2003;97(5):1331–1339. doi:10.1213/01.ANE.0000082995.44040.07
41. He H, Chen J, Xie WP, et al. Ketamine used as an acesodyne in human breast cancer therapy causes an undesirable side effect, upregulating anti-apoptosis protein Bcl-2 expression. Genet Mol Res. 2013;12(2):1907–1915. doi:10.4238/2013.January.4.7
42. Malsy M, Gebhardt K, Gruber M, Wiese C, Graf B, Bundscherer A. Effects of ketamine, s-ketamine, and MK 801 on proliferation, apoptosis, and necrosis in pancreatic cancer cells. BMC Anesthesiol. 2015;15:111. doi:10.1186/s12871-015-0076-y
43. Amaram-Davila J, Davis M, Reddy A. Opioids and cancer mortality. Curr Treat Options Oncol. 2020;21(3):22. doi:10.1007/s11864-020-0713-7
44. Wong SS, Cheung CW. Optimization of opioid utility in cancer pain populations. Ann Palliat Med. 2020;9(2):558–570. doi:10.21037/apm.2020.03.04
45. Cruickshank M, Henderson L, MacLennan G, et al. Alpha-2 agonists for sedation of mechanically ventilated adults in intensive care units: a systematic review. Health Technol Assess. 2016;20(25):v–117. doi:10.3310/hta20250
46. Levins KJ, Prendeville S, Conlon S, Buggy DJ. The effect of anesthetic technique on µ-opioid receptor expression and immune cell infiltration in breast cancer. J Anesth. 2018;32(6):792–796. doi:10.1007/s00540-018-2554-0
47. Connolly C, Buggy DJ. Opioids and tumour metastasis: does the choice of the anesthetic-analgesic technique influence outcome after cancer surgery? Curr Opin Anaesthesiol. 2016;29(4):468–474. doi:10.1097/ACO.0000000000000360
48. Singleton PA, Lingen MW, Fekete MJ, Garcia JG, Moss J. Methylnaltrexone inhibits opiate and VEGF-induced angiogenesis: role of receptor transactivation. Microvasc Res. 2006;72(1–2):3–11. doi:10.1016/j.mvr.2006.04.004
49. Yamamizu K, Furuta S, Hamada Y, et al. к Opioids inhibit tumor angiogenesis by suppressing VEGF signaling. Sci Rep. 2013;3:3213. doi:10.1038/srep03213
50. Chong D, Shao L, Yang Y, Wang R, Yang C, Zhang B. Correlations of cancer pain degree with levels of β-EP, CGRP and PGE2 and the effects of oxycontin on them. J BUON. 2018;23(5):1552–1557.
51. Farooqui M, Li Y, Rogers T, et al. COX-2 inhibitor celecoxib prevents chronic morphine-induced promotion of angiogenesis, tumour growth, metastasis and mortality, without compromising analgesia. Br J Cancer. 2007;97(11):1523–1531. doi:10.1038/sj.bjc.6604057
52. Schoos A, Gabriel C, Knab VM, Fux DA. Activation of HIF-1α by δ-opioid receptors induces COX-2 expression in breast cancer cells and leads to paracrine activation of vascular endothelial cells. J Pharmacol Exp Ther. 2019;370(3):480–489. doi:10.1124/jpet.119.257501
53. Tuerxun H, Cui J. The dual effect of morphine on tumor development. Clin Transl Oncol. 2019;21(6):695–701. doi:10.1007/s12094-018-1974-5
54. Dupoiron D. Intrathecal therapy for pain in cancer patients. Curr Opin Support Palliat Care. 2019;13(2):75–80. doi:10.1097/SPC.0000000000000427
55. Koodie L, Yuan H, Pumper JA, et al. Morphine inhibits migration of tumor-infiltrating leukocytes and suppresses angiogenesis associated with tumor growth in mice. Am J Pathol. 2014;184(4):1073–1084. doi:10.1016/j.ajpath.2013.12.019
56. Kim MH, Oh JE, Park S, et al. Tramadol use is associated with enhanced postoperative outcomes in breast cancer patients: a retrospective clinical study with in vitro confirmation. Br J Anaesth. 2019;123(6):865–876. doi:10.1016/j.bja.2019.09.004
57. Feng Z, Lu X, Gan L, Zhang Q, Lin L. Xanthones, a promising anti-inflammatory scaffold: structure, activity, and drug likeness analysis. Molecules. 2020;25(3):598. doi:10.3390/molecules25030598
58. Zappavigna S, Cossu AM, Grimaldi A, et al. Anti-inflammatory drugs as anticancer agents. Int J Mol Sci. 2020;21(7):2605. doi:10.3390/ijms21072605
59. Ye Y, Wang X, Jeschke U, von Schönfeldt V. COX-2-PGE(2)-EPs in gynecological cancers. Arch Gynecol Obstet. 2020;301(6):1365–1375. doi:10.1007/s00404-020-05559-6
60. Majidi A, Na R, Dixon-Suen S, Jordan SJ, Webb PM. Common medications and survival in women with ovarian cancer: a systematic review and meta-analysis. Gynecol Oncol. 2020;157(3):678–685. doi:10.1016/j.ygyno.2020.03.028
61. Woolbright BL, Pilbeam CC, Taylor JA. Prostaglandin E2 as a therapeutic target in bladder cancer: from basic science to clinical trials. Prostaglandins Other Lipid Mediat. 2020;148:106409. doi:10.1016/j.prostaglandins.2020.106409
62. Choi JE, Villarreal J, Lasala J, et al. Perioperative neutrophil: lymphocyte ratio and postoperative NSAID use as predictors of survival after lung cancer surgery: a retrospective study. Cancer Med. 2015;4(6):825–833. doi:10.1002/cam4.428
63. Wen QP, Miao Z, Wu P, et al. Whole-course application of dexmedetomidine combined with ketorolac in nonnarcotic postoperative analgesia for patients with lung cancer undergoing thoracoscopic surgery: a randomized control trial. Pain Physician. 2020;23(2):E185–E193.
64. Forget P, De Kock M. Perspectives in anaesthesia for cancer surgery. J Cancer Res Clin Oncol. 2014;140(3):353–359. doi:10.1007/s00432-013-1522-1
65. Jiao B, Yang C, Huang NN, Yang N, Wei J, Xu H. Relationship between volatile anesthetics and tumor progression: unveiling the mystery. Curr Med Sci. 2018;38(6):962–967. doi:10.1007/s11596-018-1970-6
66. Xu Y, Jiang W, Xie S, Xue F, Zhu X. The role of inhaled anesthetics in tumorigenesis and tumor immunity. Cancer Manag Res. 2020;12:1601–1609. doi:10.2147/CMAR.S244280
67. Evans DR, Fowler-Williams C, Ma D. Is volatile anesthesia during cancer surgery likely to increase the metastatic risk? Int Anesthesiol Clin. 2016;54(4):92–107. doi:10.1097/AIA.0000000000000115
68. Mitsui Y, Hou L, Huang X, Odegard KC, Pereira LM, Yuki K. Volatile anesthetic sevoflurane attenuates toll-like receptor 1/2 activation. Anesth Analg. 2020;131(2):631–639. doi:10.1213/ANE.0000000000004741
69. de Sousa GC, Cruz FF, Heil LB, et al. Intraoperative immunomodulatory effects of sevoflurane versus total intravenous anesthesia with propofol in bariatric surgery (the OBESITA trial): study protocol for a randomized controlled pilot trial. Trials. 2019;20(1):300. doi:10.1186/s13063-019-3399-z
70. Liu S, Gu X, Zhu L, et al. Effects of propofol and sevoflurane on perioperative immune response in patients undergoing laparoscopic radical hysterectomy for cervical cancer. Medicine (Baltimore). 2016;95(49):e5479. doi:10.1097/MD.0000000000005479
71. Shi QY, Zhang SJ, Liu L, et al. Sevoflurane promotes the expansion of glioma stem cells through activation of hypoxia-inducible factors in vitro. Br J Anaesth. 2015;114(5):825–830. doi:10.1093/bja/aeu402
72. Hirai T, Konishi Y, Mizuno S, Rui Z, Sun Y, Nishiwaki K. Differential effects of sevoflurane on the growth and apoptosis of human cancer cell lines. J Anesth. 2020;34(1):47–57. doi:10.1007/s00540-019-02701-w
73. Benzonana LL, Perry NJ, Watts HR, et al. Isoflurane, a commonly used volatile anesthetic, enhances renal cancer growth and malignant potential via the hypoxia-inducible factor cellular signaling pathway in vitro. Anesthesiology. 2013;119(3):593–605. doi:10.1097/ALN.0b013e31829e47fd
74. Zhu M, Li M, Zhou Y, et al. Isoflurane enhances the malignant potential of glioblastoma stem cells by promoting their viability, mobility in vitro and migratory capacity in vivo. Br J Anaesth. 2016;116(6):870–877. doi:10.1093/bja/aew124
75. Zhang W, Shao X. Isoflurane promotes non-small cell lung cancer malignancy by activating the Akt-mammalian target of rapamycin (mTOR) signaling pathway. Med Sci Monit. 2016;22:4644–4650. doi:10.12659/MSM.898434
76. Kawaraguchi Y, Horikawa YT, Murphy AN, et al. Volatile anesthetics protect cancer cells against tumor necrosis factor-related apoptosis-inducing ligand-induced apoptosis via caveolins. Anesthesiology. 2011;115(3):499–508. doi:10.1097/ALN.0b013e3182276d42
77. Sneyd JR. Thiopental to desflurane - an anaesthetic journey. Where are we going next? Br J Anaesth. 2017;119(suppl_1):i44–i52. doi:10.1093/bja/aex328
78. Iwasaki M, Zhao H, Jaffer T, et al. Volatile anaesthetics enhance the metastasis related cellular signalling including CXCR2 of ovarian cancer cells. Oncotarget. 2016;7(18):26042–26056. doi:10.18632/oncotarget.8304
79. Müller-Edenborn B, Roth-Z’graggen B, Bartnicka K, et al. Volatile anesthetics reduce invasion of colorectal cancer cells through down-regulation of matrix metalloproteinase-9. Anesthesiology. 2012;117(2):293–301. doi:10.1097/ALN.0b013e3182605df1
80. Reichle FM, Conzen PF. Halogenated inhalational anaesthetics. Best Pract Res Clin Anaesthesiol. 2003;17(1):29–46. doi:10.1053/bean.2002.0265
81. Woo JH, Baik HJ, Kim CH, et al. Effect of propofol and desflurane on immune cell populations in breast cancer patients: a randomized trial. J Korean Med Sci. 2015;30(10):1503–1508. doi:10.3346/jkms.2015.30.10.1503
82. Heavner JE. Local anesthetics. Curr Opin Anaesthesiol. 2007;20(4):336–342. doi:10.1097/ACO.0b013e3281c10a08
83. Liu H, Dilger JP, Lin J. Effects of local anesthetics on cancer cells. Pharmacol Ther. 2020;212:107558. doi:10.1016/j.pharmthera.2020.107558
84. Pérez-González O, Cuéllar-Guzmán LF, Soliz J, Cata JP. Impact of regional anesthesia on recurrence, metastasis, and immune response in breast cancer surgery: a systematic review of the literature. Reg Anesth Pain Med. 2017;42(6):751–756. doi:10.1097/AAP.0000000000000662
85. Sen S, Koyyalamudi V, Smith DD, et al. The role of regional anesthesia in the propagation of cancer: a comprehensive review. Best Pract Res Clin Anaesthesiol. 2019;33(4):507–522. doi:10.1016/j.bpa.2019.07.004
86. Sakaguchi M, Kuroda Y, Hirose M. The antiproliferative effect of lidocaine on human tongue cancer cells with inhibition of the activity of epidermal growth factor receptor. Anesth Analg. 2006;102(4):1103–1107. doi:10.1213/01.ane.0000198330.84341.35
87. Siekmann W, Tina E, Von Sydow AK, Gupta A. Effect of lidocaine and ropivacaine on primary (SW480) and metastatic (SW620) colon cancer cell lines. Oncol Lett. 2019;18(1):395–401. doi:10.3892/ol.2019.10332
88. Lirk P, Hollmann MW, Fleischer M, Weber NC, Fiegl H. Lidocaine and ropivacaine, but not bupivacaine, demethylate deoxyribonucleic acid in breast cancer cells in vitro. Br J Anaesth. 2014;113(Suppl 1):i32–38. doi:10.1093/bja/aeu201
89. Chang YC, Hsu YC, Liu CL, Huang SY, Hu MC, Cheng SP. Local anesthetics induce apoptosis in human thyroid cancer cells through the mitogen-activated protein kinase pathway. PLoS One. 2014;9(2):e89563. doi:10.1371/journal.pone.0089563
90. Zanni MP, Mauri-Hellweg D, Brander C, et al. Characterization of lidocaine-specific T cells. J Immunol. 1997;158(3):1139–1148.
91. Chamaraux-Tran TN, Piegeler T. The amide local anesthetic lidocaine in cancer surgery-potential antimetastatic effects and preservation of immune cell function? A narrative review. Front Med (Lausanne). 2017;4:235. doi:10.3389/fmed.2017.00235
92. Azuma Y, Ohura K. Immunological modulation by lidocaine-epinephrine and prilocaine-felypressin on the functions related to natural immunity in neutrophils and macrophages. Curr Drug Targets Immune Endocr Metabol Disord. 2004;4(1):29–36. doi:10.2174/1568008043339974
93. Kim R. Anesthetic technique for cancer surgery: harm or benefit for cancer recurrence? Eur J Surg Oncol. 2018;44(5):557–558. doi:10.1016/j.ejso.2018.02.207
94. Iwasaki M, Edmondson M, Sakamoto A, Ma D. Anesthesia, surgical stress, and “long-term” outcomes. Acta Anaesthesiol Taiwan. 2015;53(3):99–104. doi:10.1016/j.aat.2015.07.002
95. Tseng KS, Kulkarni S, Humphreys EB, et al. Spinal anesthesia does not impact prostate cancer recurrence in a cohort of men undergoing radical prostatectomy: an observational study. Reg Anesth Pain Med. 2014;39(4):284–288. doi:10.1097/AAP.0000000000000108
96. Short TG, Leslie K, Chan MT, Campbell D, Frampton C, Myles P. Rationale and design of the balanced anesthesia study: a prospective randomized clinical trial of two levels of anesthetic depth on patient outcome after major surgery. Anesth Analg. 2015;121(2):357–365. doi:10.1213/ANE.0000000000000797
97. Looney M, Doran P, Buggy DJ. Effect of anesthetic technique on serum vascular endothelial growth factor C and transforming growth factor β in women undergoing anesthesia and surgery for breast cancer. Anesthesiology. 2010;113(5):1118–1125. doi:10.1097/ALN.0b013e3181f79a69
98. Poeran J, Mazumdar M, Memtsoudis SG. Anesthesia, outcomes, and public health: changing health care while “asleep”. Reg Anesth Pain Med. 2014;39(3):192–194. doi:10.1097/AAP.0000000000000085
99. Tai YH, Wu HL, Mandell MS, Tsou MY, Chang KY. The association of allogeneic blood transfusion and the recurrence of hepatic cancer after surgical resection. Anaesthesia. 2020;75(4):464–471. doi:10.1111/anae.14862
100. Le-Wendling L, Nin O, Capdevila X. Cancer recurrence and regional anesthesia: the theories, the data, and the future in outcomes. Pain Med. 2016;17(4):756–775. doi:10.1111/pme.12893
101. Neri D, Sondel PM. Immunocytokines for cancer treatment: past, present and future. Curr Opin Immunol. 2016;40:96–102. doi:10.1016/j.coi.2016.03.006
102. Budhwani M, Mazzieri R, Dolcetti R. Plasticity of type I interferon-mediated responses in cancer therapy: from anti-tumor immunity to resistance. Front Oncol. 2018;8:322. doi:10.3389/fonc.2018.00322
103. Lewis AL, Chaft J, Girotra M, Fischer GW. Immune checkpoint inhibitors: a narrative review of considerations for the anaesthesiologist. Br J Anaesth. 2020;124(3):251–260. doi:10.1016/j.bja.2019.11.034
104. Lusty AJ, Hosier GW, Koti M, et al. Anesthetic technique and oncological outcomes in urology: a clinical practice review. Urol Oncol. 2019;37(12):845–852. doi:10.1016/j.urolonc.2019.08.004
105. Chowdappa R, Tiwari AR, Arjunan R, Althaf S, Kumar RV, Chunduri S. Perioperative mortality in cancer esophagus-a case control study at a high-volume regional cancer center in South India. Indian J Surg Oncol. 2019;10(1):83–90. doi:10.1007/s13193-018-0825-8
106. Cata JP, Gutierrez C, Mehran RJ, et al. Preoperative anemia, blood transfusion, and neutrophil-to-lymphocyte ratio in patients with stage I non-small cell lung cancer. Cancer Cell Microenviron. 2016;3(1):e1116.
107. Brown KGM, Solomon MJ, Koh CE. Pelvic exenteration surgery: the evolution of radical surgical techniques for advanced and recurrent pelvic malignancy. Dis Colon Rectum. 2017;60(7):745–754. doi:10.1097/DCR.0000000000000839
108. Ma QM, Han GS, Li BW, Li XJ, Jiang T. Effectiveness and safety of the use of antifibrinolytic agents in total-knee arthroplasty: a meta-analysis. Medicine (Baltimore). 2020;99(20):e20214. doi:10.1097/MD.0000000000020214
109. Soehle M, Dehne H, Hoeft A, Zenker S. Accuracy of the non-invasive Tcore™ temperature monitoring system to measure body core temperature in abdominal surgery. J Clin Monit Comput. 2019. doi:10.1007/s10877-019-00430-9
110. Laitman BM, Ma Y, Hill B, et al. Mild hypothermia is associated with improved outcomes in patients undergoing microvascular head and neck reconstruction. Am J Otolaryngol. 2019;40(3):418–422. doi:10.1016/j.amjoto.2019.03.010
111. Cajigas I, Mahavadi AK, Shah AH, et al. Analysis of intra-operative variables as predictors of 30-day readmission in patients undergoing glioma surgery at a single center. J Neurooncol. 2019;145(3):509–518. doi:10.1007/s11060-019-03317-6
112. Aoki Y, Atsumi K, Kora M, Koh N, Yokoyama J. Sugammadex and amino acid infusion can contribute to safe anesthetic management of variegate porphyria. JA Clin Rep. 2018;4(1):49. doi:10.1186/s40981-018-0187-9
113. Du G, Liu Y, Li J, Liu W, Wang Y, Li H. Hypothermic microenvironment plays a key role in tumor immune subversion. Int Immunopharmacol. 2013;17(2):245–253. doi:10.1016/j.intimp.2013.06.018
114. Liang B, Dong T. Effects of propofol on invasion and migration of colon cancer cells and JAK2/STAT3 signaling pathway. Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2020;45(3):290–296. doi:10.11817/j.issn.1672-7347.2020.180704
115.. Zheng X, Dong L, Zhao S, et al. Propofol affects non-small-cell lung cancer cell biology by regulating the miR-21/PTEN/AKT pathway in vitro and in vivo. Anesth Analg. 2020;Publish Ahead of Print. doi:10.1213/ANE.0000000000004778.
116. Wang H, Jiao H, Jiang Z, Chen R. Propofol inhibits migration and induces apoptosis of pancreatic cancer PANC-1 cells through miR-34a-mediated E-cadherin and LOC285194 signals. Bioengineered. 2020;11(1):510–521. doi:10.1080/21655979.2020.1754038
117. Liu F, Qiu F, Fu M, Chen H, Wang H. Propofol reduces epithelial to mesenchymal transition, invasion and migration of gastric cancer cells through the microRNA-195-5p/snail axis. Med Sci Monit. 2020;26:e920981. doi:10.12659/MSM.923909
118. Li M, Qu L, Chen F, Zhu X. Propofol upregulates miR-320a and reduces HMGB1 by downregulating ANRIL to inhibit PTC cell malignant behaviors. Pathol Res Pract. 2020;216(4):152856. doi:10.1016/j.prp.2020.152856
119. Yu X, Shi J, Wang X, Zhang F. Propofol affects the growth and metastasis of pancreatic cancer via ADAM8. Pharmacol Rep. 2020;72(2):418–426. doi:10.1007/s43440-019-00015-y
120. Li F, Chen W. Propofol inhibits cell proliferation, migration, and invasion via mir-410-3p/transforming growth factor-β receptor type 2 (TGFBR2) axis in glioma. Med Sci Monit. 2020;26:e919523.
121. Su Z, Liu HL, Qi B, Liu Y. Effects of propofol on proliferation and apoptosis of cardia cancer cells via MAPK/ERK signaling pathway. Eur Rev Med Pharmacol Sci. 2020;24(1):428–433. doi:10.26355/eurrev_202001_19942
122. Zhang YF, Li CS, Zhou Y, Lu XH. Effects of propofol on colon cancer metastasis through STAT3/HOTAIR axis by activating WIF-1 and suppressing Wnt pathway. Cancer Med. 2020;9(5):1842–1854. doi:10.1002/cam4.2840
123. Zhang YF, Li CS, Zhou Y, Lu XH. Propofol facilitates cisplatin sensitivity via lncRNA MALAT1/miR-30e/ATG5 axis through suppressing autophagy in gastric cancer. Life Sci. 2020;244:117280. doi:10.1016/j.lfs.2020.117280
124. Chu CN, Wu KC, Chung WS, et al. Etomidate suppresses invasion and migration of human A549 lung adenocarcinoma cells. Anticancer Res. 2019;39(1):215–223. doi:10.21873/anticanres.13100
125. Chen HT, Zhou J, Fan YL, et al. Anesthetic agent etiomidate induces apoptosis in N2a brain tumor cell line. Mol Med Rep. 2018;18(3):3137–3142. doi:10.3892/mmr.2018.9298
126. Hu J, Duan W, Liu Y. Ketamine inhibits aerobic glycolysis in colorectal cancer cells by blocking the NMDA receptor-CaMK II-c-Myc pathway. Clin Exp Pharmacol Physiol. 2020;47(5):848–856. doi:10.1111/1440-1681.13248
127. Duan W, Hu J, Liu Y. Ketamine inhibits colorectal cancer cells malignant potential via blockage of NMDA receptor. Exp Mol Pathol. 2019;107:171–178. doi:10.1016/j.yexmp.2019.02.004
128. Grandhi RK, Lee S, Abd-Elsayed A. Does opioid use cause angiogenesis and metastasis? Pain Med. 2017;18(1):140–151. doi:10.1093/pm/pnw132
129. Zhang J, Yao N, Tian S. Morphine stimulates migration and growth and alleviates the effects of chemo drugs via AMPK-dependent induction of epithelial-mesenchymal transition in esophageal carcinoma cells. Biol Pharm Bull. 2020;43(5):774–781. doi:10.1248/bpb.b19-00779
130. Cui JH, Jiang WW, Liao YJ, Wang QH, Xu M, Li Y. Effects of oxycodone on immune function in patients undergoing radical resection of rectal cancer under general anesthesia. Medicine (Baltimore). 2017;96(31):e7519. doi:10.1097/MD.0000000000007519
131. Gaspani L, Bianchi M, Limiroli E, Panerai AE, Sacerdote P. The analgesic drug tramadol prevents the effect of surgery on natural killer cell activity and metastatic colonization in rats. J Neuroimmunol. 2002;129(1–2):18–24. doi:10.1016/S0165-5728(02)00165-0
132. Wang HW, Wang LY, Jiang L, Tian SM, Zhong TD, Fang XM. Amide-linked local anesthetics induce apoptosis in human non-small cell lung cancer. J Thorac Dis. 2016;8(10):2748–2757. doi:10.21037/jtd.2016.09.66
133. Retsky M, Rogers R, Demicheli R, et al. NSAID analgesic ketorolac used perioperatively may suppress early breast cancer relapse: particular relevance to triple negative subgroup. Breast Cancer Res Treat. 2012;134(2):881–888. doi:10.1007/s10549-012-2094-5
134. Chen Y, Wang Q, Li Z, et al. Naproxen platinum(iv) hybrids inhibiting cycloxygenases and matrix metalloproteinases and causing DNA damage: synthesis and biological evaluation as antitumor agents in vitro and in vivo. Dalton Trans. 2020;49(16):5192–5204. doi:10.1039/D0DT00424C
135. Zhang W, Sheng B, Chen S, et al. Sevoflurane enhances proliferation, metastatic potential of cervical cancer cells via the histone deacetylase 6 modulation in vitro. Anesthesiology. 2020;132(6):1469–1481. doi:10.1097/ALN.0000000000003129
136. Han XC, Zhang YJ, Dong X, Xing QZ, Li KH, Zhang L. Sevoflurane modulates the cancer stem cell-like properties and mitochondrial membrane potential of glioma via Ca(2+)-dependent CaMKII/JNK cascade. Life Sci. 2020;253:117675. doi:10.1016/j.lfs.2020.117675
137. Kang K, Wang Y. Sevoflurane inhibits proliferation and invasion of human ovarian cancer cells by regulating JNK and p38 MAPK signaling pathway. Drug Des Devel Ther. 2019;13:4451–4460. doi:10.2147/DDDT.S223581
138. Li R, Huang Y, Lin J. Distinct effects of general anesthetics on lung metastasis mediated by IL-6/JAK/STAT3 pathway in mouse models. Nat Commun. 2020;11(1):642. doi:10.1038/s41467-019-14065-6
139. Zhang C, Wang B, Wang X, Sheng X, Cui Y. Sevoflurane inhibits the progression of ovarian cancer through down-regulating stanniocalcin 1 (STC1). Cancer Cell Int. 2019;19:339. doi:10.1186/s12935-019-1062-0
140. Xue F, Xu Y, Song Y, Zhang W, Li R, Zhu X. The effects of sevoflurane on the progression and cisplatinum sensitivity of cervical cancer cells. Drug Des Devel Ther. 2019;13:3919–3928. doi:10.2147/DDDT.S219788
141. Chen M, Zhou L, Liao Z, et al. Sevoflurane inhibited osteosarcoma cell proliferation and invasion via targeting miR-203/WNT2B/Wnt/β-catenin axis. Cancer Manag Res. 2019;11:9505–9515. doi:10.2147/CMAR.S225911
142. Hu J, Jiao H, Li Q, Li Q. Anesthetic effects of isoflurane and the molecular mechanism underlying isoflurane‑inhibited aggressiveness of hepatic carcinoma. Mol Med Rep. 2018;18(1):184–192. doi:10.3892/mmr.2018.8945
143. Elias KM, Kang S, Liu X, Horowitz NS, Berkowitz RS, Frendl G. Anesthetic selection and disease-free survival following optimal primary cytoreductive surgery for stage III epithelial ovarian cancer. Ann Surg Oncol. 2015;22(4):1341–1348. doi:10.1245/s10434-014-4112-9
144. Hurmath FK, Mittal M, Ramaswamy P, Umamaheswara Rao GS, Dalavaikodihalli Nanjaiah N. Sevoflurane and thiopental preconditioning attenuates the migration and activity of MMP-2 in U87MG glioma cells. Neurochem Int. 2016;94:32–38. doi:10.1016/j.neuint.2016.02.003
145. Bundscherer AC, Ullrich V, Malsy M, et al. Effects of volatile anesthetics on proliferation and viability of SW480 colon cancer cells in vitro. Anticancer Res. 2019;39(11):6049–6055. doi:10.21873/anticanres.13811
146. Cata JP, Keerty V, Keerty D, et al. A retrospective analysis of the effect of intraoperative opioid dose on cancer recurrence after non-small cell lung cancer resection. Cancer Med. 2014;3(4):900–908. doi:10.1002/cam4.236
147. Zhu J, Han S. Lidocaine inhibits cervical cancer cell proliferation and induces cell apoptosis by modulating the lncRNA-MEG3/miR-421/BTG1 pathway. Am J Transl Res. 2019;11(9):5404–5416.
148. Wall TP, Crowley PD, Sherwin A, Foley AG, Buggy DJ. Effects of lidocaine and src inhibition on metastasis in a murine model of breast cancer surgery. Cancers (Basel). 2019;11(10):1414. doi:10.3390/cancers11101414
149. Sun H, Sun Y. Lidocaine inhibits proliferation and metastasis of lung cancer cell via regulation of miR-539/EGFR axis. Artif Cells Nanomed Biotechnol. 2019;47(1):2866–2874. doi:10.1080/21691401.2019.1636807
150. Dong Q, Mao Z. The local anaesthetic lignocaine exhibits potent antilung cancer cell activity by inhibiting the phosphoinositide 3-kinases/mammalian target of rapamycin/mammalian target of rapamycin pathway. Pharmacology. 2019;104(3–4):139–146. doi:10.1159/000500743
151. Xia W, Wang L, Yu D, Mu X, Zhou X. Lidocaine inhibits the progression of retinoblastoma in vitro and in vivo by modulating the miR‑520a‑3p/EGFR axis. Mol Med Rep. 2019;20(2):1333–1342. doi:10.3892/mmr.2019.10363
152. Ye L, Zhang Y, Chen YJ, Liu Q. Anti-tumor effects of lidocaine on human gastric cancer cells in vitro. Bratisl Lek Listy. 2019;120(3):212–217. doi:10.4149/BLL_2019_036
153. Xing W, Chen DT, Pan JH, et al. Lidocaine induces apoptosis and suppresses tumor growth in human hepatocellular carcinoma cells in vitro and in a xenograft model in vivo. Anesthesiology. 2017;126(5):868–881. doi:10.1097/ALN.0000000000001528
154. Zhu Q, Zhu G, Xu W, Dan J, Xia R, Liu W. Bupivacaine inhibits angiogenesis through oxidative stress-dependent inhibition of Akt/mTOR and activation of AMPK. Fundam Clin Pharmacol. 2020. doi:10.1111/fcp.12554
155. Zhang H, Lin J, Hu T, Ren Z, Wang W, He Q. Effect of miR-132 on bupivacaine-induced neurotoxicity in human neuroblastoma cell line. J Pharmacol Sci. 2019;139(3):186–192. doi:10.1016/j.jphs.2019.01.014
156. Xuan W, Zhao H, Hankin J, Chen L, Yao S, Ma D. Local anesthetic bupivacaine induced ovarian and prostate cancer apoptotic cell death and underlying mechanisms in vitro. Sci Rep. 2016;6:26277. doi:10.1038/srep26277
157. Li YC, Wang Y, Li DD, Zhang Y, Zhao TC, Li CF. Procaine is a specific DNA methylation inhibitor with anti-tumor effect for human gastric cancer. J Cell Biochem. 2018;119(2):2440–2449. doi:10.1002/jcb.26407
158. Li C, Gao S, Li X, Ma L, Ma L. Procaine inhibits the proliferation and migration of colon cancer cells through inactivation of the ERK/MAPK/FAK pathways by regulation of RhoA. Oncol Res. 2018;26(2):209–217. doi:10.3727/096504017X14944585873622
159. Ying B, Huang H, Li H, Song M, Wu S, Ying H. Procaine inhibits proliferation and migration and promotes cell apoptosis in osteosarcoma cells by upregulation of microRNA-133b. Oncol Res. 2017;25(9):1463–1470. doi:10.3727/096504017X14878518291077
160. Ma Y, Ren Z, Ma S, et al. Morphine enhances renal cell carcinoma aggressiveness through promotes survivin level. Ren Fail. 2017;39(1):258–264. doi:10.1080/0886022X.2016.1256322
161. Li T, Chen L, Zhao H, et al. Both Bupivacaine and Levobupivacaine inhibit colon cancer cell growth but not melanoma cells in vitro. J Anesth. 2019;33(1):17–25. doi:10.1007/s00540-018-2577-6
162. Malsy M, Graf B, Bundscherer A. The effects of analgesics and local anesthetics on gene transcription mediated by NFATc2 and Sp1 in pancreatic carcinoma. Anticancer Res. 2019;39(9):4721–4728. doi:10.21873/anticanres.13654
163. Wang HL, Yan HD, Liu YY, et al. Intraoperative intravenous lidocaine exerts a protective effect on cell-mediated immunity in patients undergoing radical hysterectomy. Mol Med Rep. 2015;12(5):7039–7044. doi:10.3892/mmr.2015.4235
164. Piegeler T, Votta-Velis EG, Liu G, et al. Antimetastatic potential of amide-linked local anesthetics: inhibition of lung adenocarcinoma cell migration and inflammatory Src signaling independent of sodium channel blockade. Anesthesiology. 2012;117(3):548–559. doi:10.1097/ALN.0b013e3182661977
165. Li R, Xiao C, Liu H, Huang Y, Dilger JP, Lin J. Effects of local anesthetics on breast cancer cell viability and migration. BMC Cancer. 2018;18(1):666. doi:10.1186/s12885-018-4576-2
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
