Back to Journals » Vascular Health and Risk Management » Volume 11
Effectiveness and tolerability of treatment intensification to basal–bolus therapy in patients with type 2 diabetes on previous basal insulin-supported oral therapy with insulin glargine or supplementary insulin therapy with insulin glulisine: the PARTNER observational study
Authors Pfohl M, Siegmund T, Pscherer S, Pegelow K, Seufert J
Received 12 February 2015
Accepted for publication 16 July 2015
Published 6 November 2015 Volume 2015:11 Pages 569—578
DOI https://doi.org/10.2147/VHRM.S82720
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Daniel Duprez
Martin Pfohl,1 Thorsten Siegmund,2 Stefan Pscherer,3 Katrin Pegelow,4 Jochen Seufert5
1Medizinische Klinik I, Evangelisches Bethesda-Klinikum GmbH, Duisburg, Germany; 2Städtisches Klinikum München GmbH, Klinikum Bogenhausen, III. Medizinische Abteilung, München, Germany; 3Klinisches Diabeteszentrum Südostbayern, Innere Medizin – Diabetologie, Traunstein, Germany; 4Sanofi-Aventis Deutschland GmbH, Berlin, Germany; 5Medizinische Universitätsklinik, Klinik für Innere Medizin II, Abteilung für Endokrinologie und Diabetologie, Freiburg, Germany
Background: Due to the progressive nature of type 2 diabetes mellitus (T2DM), antidiabetic treatment needs to be continuously intensified to avoid long-term complications. In T2DM patients on either basal insulin-supported oral therapy (BOT) or supplementary insulin therapy (SIT) presenting with HbA1c values above individual targets for 3–6 months, therapy should be intensified. This study investigated effectiveness and tolerability of an intensification of BOT or SIT to a basal–bolus therapy (BBT) regimen in T2DM patients in daily clinical practice.
Methods: This noninterventional, 8-month, prospective, multicenter study evaluated parameters of glucose control, occurrence of adverse events (eg, hypoglycemia), and acceptance of devices in daily clinical practice routine after 12 and 24 weeks of intensifying insulin therapy to a BBT regimen starting from either preexisting BOT with insulin glargine (pre-BOT) or preexisting SIT with ≥3 daily injections of insulin glulisine (pre-SIT).
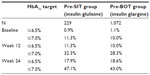
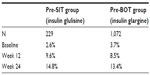
Results: A total of 1,530 patients were documented in 258 German medical practices. A total of 1,301 patients were included in the full analysis set (55% male, 45% female; age median 64 years; body mass index median 30.8 kg/m2; pre-BOT: n=1,072; pre-SIT: n=229), and 1,515 patients were evaluated for safety. After 12 weeks, HbA1c decreased versus baseline (pre-BOT 8.67%; pre-SIT 8.46%) to 7.73% and 7.66%, respectively (Δ mean -0.94% and -0.80%; P<0.0001). At week 24, HbA1c was further reduced to 7.38% and 7.30%, respectively (Δ mean –1.29% and -1.15%; P<0.0001), with a mean reduction of fasting blood glucose values in both treatment groups by more than 46 mg/dL. An HbA1c goal of ≤6.5% was reached by 17.9% (pre-BOT) and 18.6% (pre-SIT), and an HbA1c ≤7.0% by 46.1% (pre-BOT) and 43.0% (pre-SIT) of patients. During 24 weeks, severe as well as serious hypoglycemic events were rare (pre-BOT: n=5; pre-SIT: n=2; pretreated with both insulins: n=1).
Conclusion: Intensifying glargine-based BOT or glulisine-based SIT to a BBT regimen in poorly controlled T2DM patients in daily routine care led to marked improvements of glycemic control and was well tolerated.
Keywords: type 2 diabetes, clinical practice, BBT, insulin glargine, insulin glulisine, basal–bolus therapy
Background
Type 2 diabetes mellitus (T2DM) is a chronic progressive disease, and thus, antidiabetic treatment regimens need to be continuously intensified to avoid long-term complications.1–3 Therefore, introduction of insulin therapy at the right stage of the treatment algorithm represents an important hallmark for adequate glycemic control.4,5 Insulin therapy in T2DM is often started as a combination therapy, either by adding a basal insulin to the existing oral antihyperglycemic treatment (basal insulin-supported oral therapy [BOT]) or by adding a short-acting insulin at mealtimes while maintaining oral agents, eg, metformin (supplementary insulin therapy [SIT]).2 In patients with T2DM and HbA1c values above their individual treatment target after insulin pretreatment for 3–6 months (BOT or SIT), insulin therapy should be intensified.6,7 The 10-year follow-up data of the UKPD study as well as a meta-analysis on the impact of antihyperglycemic therapy on macrovascular events in T2DM patients convincingly demonstrated the benefit of adequate glycemic control in reducing long-term diabetic complications, eg, myocardial infarction.8,9
While the use of conventional insulin therapy (CT) is now declining, the efficacy of basal–bolus insulin therapy (BBT) has repeatedly been demonstrated.2,10 With this BBT, the patients achieve greater flexibility in their daily activities and diet, with consecutive improvements of quality of life. Glycemic control by BBT using insulin glargine and insulin glulisine is superior to CT, which has been demonstrated in clinical trials and in the outpatient setting. Moreover, patient satisfaction was markedly improved after switching from inadequately controlled CT to a basal–bolus combination of insulin glargine and insulin glulisine.11,12
The PARTNER (noninterventional, open, prospective, observational study in T2DM subjects to observe the efficacy of a BBT regimen with insulin glargine in combination with short-acting insulin analog and a BBT regime with insulin glulisine in combination with long-acting basal insulin in terms of HbA1c under daily routine in Germany) observational study was conducted to document outcomes of an intensification of insulin therapy in T2DM patients pretreated with insulin (BOT or SIT), who had elevated HbA1c values. This treatment intensification was in accordance with the guidelines of the German Diabetes Association (Deutsche Diabetes Gesellschaft; DDG) of 20092 as well as 201313 and is part of daily clinical practice. The German guideline13 also recommends the early use of insulin, starting with low effective doses and stepwise dose escalation from BOT or SIT toward a BBT to reach the individual HbA1c target that may range from ≤6.5% to much higher levels, especially in vulnerable elderly patients.
The following questions were addressed in the PARTNER observational study:
- Are there differences between the patients in the two pretreatment groups (insulin glargine-based BOT regimen and insulin glulisine-based SIT regimen)?
- How effective were these pretreatments?
- How effective was the intensification in each group?
- How satisfied were the patients with the relatively new injection devices SoloStar® (a disposable insulin pen), ClikStar®, and TactiPen® (two reusable insulin pens), respectively?
Methods
This noninterventional, observational study was conducted together with resident physicians specialized in the treatment of T2DM patients, who either received insulin glargine (Lantus®) or insulin glulisine (Apidra®) according to the labels in a BOT or SIT regimen for at least 3 months, but still lacked adequate glycemic control. The participating physicians were selected to produce an even geographical distribution all over Germany and represented more than 6% of diabetologists associated with the German Society of Diabetes (DDG). Since only very few exclusion criteria had been defined for eligible patients, the site as well as the patient sample evaluated in this study are considered representative for the current medical standard of care of patients with advanced T2DM requiring therapy intensification in Germany. The injection devices SoloStar®, ClikStar®, or TactiPen® (all pens: Sanofi-Aventis, Frankfurt, Germany) were used, and satisfaction with these devices was documented as a putative therapeutic adherence factor. Each center was asked to document up to six T2DM patients (three in each group) consecutively enrolled when found eligible. No diagnostic measures or treatment methods were stipulated, but remained in the sole responsibility of the participating physicians. In all cases, the patients’ physicians had made the decision to intensify the insulin therapy before initiation of the study.
Each physician received a documentation binder containing the corresponding study documents. The study included three patient visits: a screening documentation with initiation of the BBT at baseline and two subsequent measurements and documentations at 3 and 6 months (approximately after 12 and 24 weeks), respectively.
Patients and ethical issues
Eligible T2DM patients had to be ≥18 years, receiving either insulin glargine in a BOT regimen or insulin glulisine in a SIT regimen for at least 3 months with HbA1c ≥6.5%, and for whom their physician had decided to intensify the insulin treatment with a BBT regimen. Patients must not have had a known malignant disease in medical history, known alcohol or drug abuse, incapability to comprehend the content of this study, or a contraindication for the study drugs. They also had to be capable of performing self-monitored blood glucose assessments.
This study was conducted in accordance with the principles laid down by the 18th World Medical Assembly (Helsinki, 1964) and all subsequent amendments. Prior to any study activity, the study protocol was approved by the Ethics Committee at the Chamber of Physicians in Berlin. Patients documented their willingness for participation by signing the informed consent form.
Study endpoints
The primary study endpoint was the mean change in HbA1c from baseline after ~12 and 24 weeks on a BBT regimen in T2DM patients pretreated with insulin glargine (pre-BOT) or insulin glulisine (pre-SIT).
The main secondary study endpoints were 1) the percentage of patients achieving a target HbA1c of ≤6.5% or ≤7.0% at baseline and under BBT treatment; 2) mean change in fasting blood glucose (FBG); 3) the percentage of patients achieving the target FBG of ≤5.6 mmol/L (≤100 mg/dL) at baseline and under BBT treatment; 4) mean daily dose (U/day) and dose per kg body weight (BW; U/kg BW) of insulin glargine (BOT) or insulin glulisine (SIT) during pretreatment and after intensification to a BBT; 5) ratio of insulin glargine dosage and total prandial insulin dosage/day at study end (pre-BOT) and of insulin glulisine dosage and total basal insulin dosage/day at study end (pre-SIT), respectively; 6) change in BW from baseline and percentage of patients that maintained, reduced, or increased their BW from baseline after ~12 and 24 weeks on BBT; and 7) percentage distribution of the injection devices SoloStar®, ClikStar®, and TactiPen® and percentage distribution of satisfaction parameters for given pen properties in each group. In addition, blood pressure (BP) and blood lipids were measured.
Safety criteria comprised the incidence of confirmed symptomatic hypoglycemia with a blood glucose ≤70 mg/dL (≤3.9 mmol/L) or severe confirmed hypoglycemia with a blood glucose ≤56 mg/dL (≤3.1 mmol/L) during the BBT, as well as the incidence of adverse events (AEs), including serious adverse events (SAEs).
Statistical analysis
The sample size estimate of 2,700 patients was based on a return (without dropout) of 90% (pre-BOT; n=1,215 of 1,350 patients) and 85% (pre-SIT; n=1,147 of 1,350 patients) patients, assuming a mean clinically significant absolute reduction in HbA1c of 0.4% with a standard deviation (SD) of 1.2% from baseline to endpoint. The estimated 95% confidence intervals (CIs) for the evaluable 1,215 patients (pre-BOT) and 1,147 patients (pre-SIT) were 0.333%–0.467% and 0.331%–0.469%, respectively. The probability was each 95% to detect at least one AE, occurring with a frequency of 1:406 in the pre-SIT cohort and 1:383 in the pre-BOT cohort, respectively.
Double data entry was done using Oracle Clinical version 4.5.3 software (Oracle Corporation, Redwood Shores, CA, USA). A comparison with the source data documentation was performed at ten sites (3.9% of all sites). All collected data were validated after the end of data capture by running check programs in Statistical Analysis System version 9.3 software (SAS Institute Inc., Cary, NC, USA). Because selected variables were missing in some patients, we considered only the subsets with available data, as indicated in the tables, for calculating proportions. Statistical analysis of all collected data was performed using descriptive measures according to a predefined statistical analysis plan. Continuous variables were described by patient numbers, mean ± SD, median, and range. For categorical variables, absolute, percentage, and adjusted percentage of frequencies were determined. In addition, 95% CIs were calculated. Paired two-tailed t-tests were applied to test for differences in values between baseline and after 12 and 24 weeks, respectively. All statistical analyses were performed separately for the total and each of the two treatment groups.
Sanofi-Aventis Deutschland GmbH was responsible for the distribution and collection of the documentation, including completeness and plausibility control, as well as for source data verification at ~3% of the centers and discrepancy management by clarification through queries to the centers.
Results
A total of 258 sites throughout Germany participated in this observational study from July 2011 to July 2012, which is a representative sample of German diabetologists (Table 1 for demographic and baseline data). Due to an unexpected slow recruitment of patients on treatment with insulin glulisine, at the end of the recruitment period, a total of 1,530 patients were screened for inclusion into this study, 834 males and 689 females aged 19–93 (median: 64) years, with a body mass index (BMI) range of 16.5–62.9 (median: 31) kg/m2. Approximately 1,301 of the 1,530 screened patients were included in the full analysis set (FAS; ie, all patients who were treated with BBT medication at least once and for whom valid baseline value and valid follow-up values [12 and 24 weeks] were available to analyze change in HbA1c), 1,515 patients in the safety analysis set (SAS; ie, all included patients who were treated with BBT medication at least once), and 1,403 patients in the enrolled analysis set (EAS; ie, those patients being clearly assigned to one pretreatment group and for whom HbA1c measures were available at first study visit).
At screening, 258 patients received insulin glulisine (16.9%), 1,198 insulin glargine (78.3%), and 44 patients both insulins (2.9%), while data were missing for 30 patients (2.0%). The most common oral antidiabetic drug in the FAS was metformin in 161/229 patients (pre-SIT) and in 844/1,072 patients (pre-BOT). No direct comparisons were made between pre-BOT and pre-SIT groups because patients were not randomized to the groups.
Relatively high rates of long-term diabetes complications were observed, likely due to a high proportion of patients with a long diabetes duration of >10 years (>40% of patients in both treatment groups). Complications were most frequently neuropathy (~40% of patients), cardiovascular disease and nephropathy (~20%), and retinopathy (~15%). Pretreatment duration with either insulin glargine or insulin glulisine ranged from 3 months to <1 year in ~34% of patients, from 1 to <3 years in 30% (pre-SIT) and 28% (pre-BOT), and from 3 to <6 years in 26% (pre-SIT) and 22% (pre-BOT). Pretreatment with insulin for more than 6 years was found in 5%–6% of all patients. Glycemic control was suboptimal (ie, HbA1c ≥6.5%) in all patients.
Intensification of insulin treatment for FAS patients was initiated at baseline and was continued until the 24-week visit for a total of 235 patients pretreated with insulin glulisine and 1,103 patients pretreated with insulin glargine. Preintensification daily doses (mean ± SD) of the short-acting insulin glulisine (36.44±24.61 U/day) were higher than for the long-acting insulin glargine (22.96±14.40 U/day). Consecutively, doses of these pretreatment insulins were not further recorded during BBT. Therefore, it was not possible to calculate ratios of the basal and prandial insulins during the BBT. At study end, the total daily doses of additional insulins in the BBT were higher in the pre-BOT group (33.62 U/day) than in the pre-SIT group (22.43 U/day). Insulin doses per kg BW of the additional insulins at study end were 0.24 U/kg (pre-SIT) and 0.36 U/kg (pre-BOT).
After cautious introduction of additional prandial or basal insulins at baseline in both groups to start a BBT, final assessment at the 24-week visit showed similar daily doses for these additional insulins as had been used for the respective pretreatment insulin class (short- or long-acting insulin) at baseline. This suggests a similar balance between short- and long-acting insulins in the two treatment groups during BBT. However, no final conclusions can be drawn as final values of the pretreatment insulin doses during the BBT were not recorded.
Evaluation of HbA1c concentrations
HbA1c decreased from baseline 8.67% to 7.73% (pre-BOT) and 8.46% to 7.66% (pre-SIT) at the 12-week visit and further to 7.38% and 7.30%, respectively, at the 24-week visit, resulting in a statistically significant mean difference of –0.94% and –0.80% versus baseline at 12 weeks and of –1.29% and –1.15%, respectively, at 24 weeks (all P<0.0001; Figure 1). The 95% CIs of HbA1c declined from baseline until week 24 from [8.58%; 8.77%] to [7.32%; 7.45%] (pre-BOT) and from [8.26%; 8.66%] to [7.19%; 7.42%] (pre-SIT). During intensification of insulin treatment, HbA1c decreased in most patients of both groups after 12 weeks already, and in even more patients until the 24-week visit, ie, in 185 (80.7%) and 191 (83.4%) patients (pre-SIT) and in 894 (83.3%) and 934 (87.1%) patients (pre-BOT), respectively. The proportion of patients with an increase in HbA1c was 15.7% (n=36) and 13.5% (n=31) at weeks 12 and 24 (pre-SIT) and slightly lower in the pre-BOT group with 12.2% (n=131) and 10.4% (n=112) at weeks 12 and 24, respectively. A small proportion of patients showed no changes in HbA1c: eight (3.5%; 12 weeks) and seven patients (3.0%; 24 weeks) in the pre-SIT group as well as 47 (4.4%; 12 weeks) and 26 patients (2.4%; 24 weeks) in the pre-BOT group.
HbA1c target achievement
A total of 17.9% and 47.1% of patients in the pre-SIT group and 18.6% and 43.0% in the pre-BOT group achieved the stringent BBT or less stringent BBT HbA1c target of ≤6.5% and ≤7.0%, respectively, at the study end after 24 weeks (Table 2). Subgroup analyses revealed that in patients with a BMI <30 kg/m2, HbA1c targets of ≤6.5% and ≤7.0% at the 24-week visit were achieved by 20.7% and 54.4% (n=101; pre-SIT group) and by 20.8% and 48.8% (n=466; pre-BOT group), respectively. Percentages were lower in patients with BMI ≥30 kg/m2, ie, 15.8% and 42.0% (n=126; pre-SIT group) and 16.9% and 38.9% (n=603; pre-BOT group), respectively. In the pre-SIT and the pre-BOT group, similar proportions of patients aged <65 years achieved the target of HbA1c ≤6.5% at 24 weeks (18.6%, n=24 and 19.4%, n=104) compared with patients aged ≥65 years (16.6%, n=16 and 17.5%, n=92). The respective figures for achieving an HbA1c of ≤7.0% were 46.5% (n=60) and 43.0% (n=230) for patients aged <65 years and 47.9% (n=46) and 43.2% (n=226) for those aged ≥65 years.
The proportions of females who achieved an HbA1c of ≤6.5% and ≤7.0% were 20.2% (n=20) and 45.5% (n=45) in the pre-SIT group and 15.6% (n=76) and 40.2% (n=195) in the pre-BOT group, respectively. The respective figures for males were 16.4% (n=21) and 49.2% (n=63; pre-SIT group) and 20.9% (n=122) and 45.2% (n=264; pre-BOT group), respectively. Overall, subgroup analyses pointed to better glycemic control in patients with shorter diabetes duration. For example, in the pre-SIT group, 20.7% (n=16) and 51.9% (n=40) of patients with a diabetes duration of 3 months to 1 year achieved the HbA1c limits of ≤6.5% and ≤7.0%, in contrast to 11.6% (n=7) and 36.6% (n=22) of patients with diabetes duration of 3–6 years, respectively. In the pre-BOT group, 21.6% (n=80) and 45.6% (n=169) of patients with a diabetes duration of 3 months to 1 year achieved the HbA1c limits of ≤6.5% and ≤7.0%, in contrast to 17.4% (n=41) and 39.5% (n=93) of patients with diabetes duration of 3–6 years, respectively.
Evaluation of FBG concentrations
Concomitant with the significant decrease in HbA1c, FBG was also reduced to a clinically relevant and statistically significant extent (all P<0.0001). In both groups, FBG was similarly lower at 12 and 24 weeks compared to baseline as shown in Figure 2 for absolute concentrations. The decreases at weeks 12 and 24 were −35 mg/dL (−1.9 mmol/L) and −46 mg/dL (−2.6 mmol/L) in the pre-SIT group, and −37 mg/dL (−2.1 mmol/L) and −47 mg/dL (−2.6 mmol/L) in the pre-BOT group, respectively.
All subgroup analyses for BMI, age, sex, and diabetes duration revealed similar, statistically significant FBG decreases (all P<0.0001). Patients aged <65 years generally exhibited tighter glycemic control. Patients with a BMI ≥30 kg/m2 and those with diabetes duration ≥6 years were better controlled with respect to FBG in the pre-SIT group, while in the pre-BOT group decreases in FBG were greatest in patients with diabetes duration of 3 to <6 years.
FBG target achievement
As listed in Table 3, FBG control (≤100 mg/dL) was achieved in smaller proportions of patients at week 24 with 14.8% (pre-SIT group) and 13.4% (pre-BOT group) as in those achieving the HbA1c target of ≤6.5% (~18%–19%). However, the lower number of patients with good FBG control may have been biased by a higher percentage of missing data for FBG at study end (3.5%–7.9%) than for HbA1c (0%). Subanalyses for BMI, age, sex, and diabetes duration did not reveal any significant differences in achievement of the FBG target. Results for the elderly were similar to those for younger patients in both groups. Stricter control appeared to be achieved for women in the pre-SIT group, but for men in the pre-BOT group. On the basis of results of rather small subgroups, it appears that patients with long diabetes duration showed a greater benefit from intensification of a therapy based on BOT pretreatment, while those with shorter diabetes duration tended to benefit from an intensified SIT-based pretreatment.
Changes in BW
In the pre-SIT group, the mean BW at baseline was 92.54 kg (n=228; 95% CI: 89.88–95.20 kg). In the pre-BOT group, the mean BW at baseline was 92.07 kg (n=1,071; 95% CI: 90.95–93.19 kg). There were essentially no changes in BW in either group during BBT (mean weight change after 24 weeks versus baseline: 0.16 kg [n=224; 95% CI: −0.25 to 0.68 kg] in the pre-SIT group and 0.26 kg [n=1,054; 95% CI: −0.07 to 0.58 kg] in the pre-BOT group).
Assessment of satisfaction with the pens
As assessed at baseline, SoloStar® was the pen device most frequently used and also had the highest rating for satisfaction, followed by TactiPen®. Only a few patients used the ClikStar® pen. Besides ranking of the patient’s satisfaction with the pen in use, several features (ie, look/design, handling, dose adjustment, effort for dose injection, and size) of the respective pen could be marked by the participating physician, if particularly positive for the respective patient. Most patients (82%–90%) in both groups were satisfied with the handling of the pens. More than half of the patients were also satisfied with the dose adjustment and the effort for dose injection of the pen they used. The feature “size” was not important for the majority of patients, irrespective of the pen in use. Very few patients (1–25) in either group reported problems with their device; therefore, no conclusions can be drawn from these limited data.
Changes in BP and blood lipids
BP values were available for >92.5% of patients in both groups (SAS, n=1,515). BP slightly decreased during the observation period, from an average at baseline of 138/81 mmHg in the pre-SIT and 139/82 mmHg in the pre-BOT group, respectively, to an average of 135/79 and 135/80 mmHg at 24 weeks, respectively. Mean changes were −3.12/−2.07 and −2.30/−1.75 mmHg, respectively, with 95% CIs for the change at 24 weeks of [−5.10; −1.14/ −3.41; −0.73 mmHg] for the pre-SIT group and [−4.21; −2.44/−2.33; −1.16 mmHg] for the pre-BOT group.
Blood lipids values were available for <50% of patients at both visits after baseline. In the pre-BOT group, the 95% CIs for subjects with data at 24 weeks indicate that total cholesterol changed by a mean of −9.82 [−13.6; −6.04] mmol/L and LDL by −5.98 [–8.83; −3.13] mmol/L, while HDL slightly increased by 1.49 [0.51; 2.48] mmol/L. In the smaller pre-SIT group, however, the mean change for total cholesterol was only −3.24 [−10.0; 3.54] mmol/L, for LDL −2.35 [−8.02; 3.32] mmol/L, and for HDL 1.85 [−0.03; 3.73] mmol/L. Similarly, triglyceride concentrations showed a pronounced change of −30.72 [–39.7; −21.7] mmol/L in the pre-BOT group, but only a trend toward lower values in the pre-SIT group (−10.21 [−25.4; 5.00] mmol/L).
Safety
Safety analyses evaluated AEs within the 1,515 patients of the SAS, including a patient subset who received both treatments (pre-SIT and pre-BOT) already at baseline. FBG and HbA1c values within the SAS did not show any noteworthy difference compared to the data reported for the FAS population shown above.
Overall, there were 24 AEs reported for 24 patients. Among those, 14 were reported as SAEs. The most frequent AE was hypoglycemia (n=9), which was reported as an SAE in eight patients (two in the pre-SIT group [0.8%], two in the pre-BOT group [0.42%], and one in a patient with pretreatment of both drugs). One patient in the pre-BOT group had a nonserious hypoglycemia. Also, in eight patients, severe hypoglycemia was observed (two in the pre-SIT group [0.8%], five in the pre-BOT group [0.42%], and one in a patient with pretreatment of both drugs). Another patient in the pre-BOT group reported a nonsevere hypoglycemia. All cases of hypoglycemia were considered treatment related by the sponsor.
In the pre-BOT group, two cases of arthralgia and one hypersensitivity reaction were also considered treatment related by the sponsor. Other AEs in the pre-SIT group were hypertensive encephalopathy (n=1), epicondylitis (n=1), and arthralgia (n=1), which were not classified as being treatment related by the sponsor. The 14 SAEs included four cases of death unrelated to treatment; two of these patients died from natural course of the disease, one patient died from neoplasm, and one from myocardial infarction (pre-BOT group). The SAEs included one case of pancreatic cancer (pre-BOT group) and one case of hypertensive encephalopathy (pre-SIT group).
Based on an analysis of the safety set, three (1.21%) patients in the pre-SIT group discontinued treatment with insulin glargine until visit 2 (week 12) and another two patients (0.81%) until visit 3 (week 24). For the pre-BOT group, insulin glulisine discontinuation rates were similarly low with 1.26% (n=15) at visit 2 and further 0.67% (n=8) at visit 3. Only one (2.27%) of the patients already on BBT at enrollment had discontinued insulin glargine/glulisine at visit 3. However, these figures may include patients still on BBT after changing to another insulin.
“Lost to follow-up” was defined as patients without documented visit date, BW, and HbA1c value as primary endpoint at visits 2 and 3. Thus, 15 patients were considered lost to follow-up, including three patients (1.21%) in the pre-SIT group, eleven (0.92%) in the pre-BOT group, and one (2.27%) on BBT already at study start.
Discussion
The PARTNER study was an open, prospective, noninterventional study in T2DM patients with inadequately controlled blood glucose on insulin plus oral antidiabetic drugs pretreatment to evaluate the effectiveness and safety of intensifying a BOT pretreatment with insulin glargine (pre-BOT) or a prandial SIT pretreatment with insulin glulisine (pre-SIT) to a BBT by adding a bolus insulin at least TID and a basal insulin, respectively, to the pretreatment insulin under daily clinical practice conditions in Germany.
HbA1c as the primary study endpoint showed a statistically significant reduction at the 12- and 24-week visits, impressively demonstrating that BBT represents a valuable escalation strategy for both pretreatment arms. Within subgroups, patients younger than 65 years exhibited lower HbA1c values. The results for males were slightly more favorable than those for females. Patients with a BMI ≥30 kg/m2 had a similar decrease in HbA1c as patients with BMI <30 kg/m2. There was a trend for lower HbA1c in patients with shorter diabetes duration.
The absolute 95% CIs for the HbA1c indicate that at baseline >95% of patients were above, but after 24 weeks of BBT >95% of patients were within the HbA1c target range between 6.5% and 7.5%, a range that is widely accepted.6,7,13 Daikeler et al12 showed a mean decrease in HbA1c by −1.0% for 1,447 type 1 diabetes mellitus (T1DM) patients and by −1.2% for 5,695 T2DM patients within 6 months for a similar BBT regimen also utilizing insulin glargine and insulin glulisine, representing results that are very close to the mean observed differences of −1.29% (pre-BOT) and −1.15% (pre-SIT), respectively, in this study. Results from other studies in T2DM patients based on daily clinical practice are also in good accordance with those reported herein.14–17 Daikeler et al12 considered the combination of insulin glargine and insulin glulisine a safe and effective therapeutic option for T1DM and T2DM patients, also improving substantially patient reported outcomes. Similar conclusions were drawn for T1DM patients from a trial in poorly controlled T1DM patients using a BBT regimen and showing a mean reduction of HbA1c from 8.1%±1.4% at baseline to 7.1%±0.9% after 3 months of treatment (n=2,437) and further to 6.9%±0.9% after 24 weeks extension period (n=1,342); 97% of these patients received insulin glargine, and all of them were started on insulin glulisine at baseline visit.18 Using a BBT regimen with insulin glargine and insulin glulisine has also demonstrated significantly superior glycemic control versus a premix insulin regimen in a T2DM population with a substantial duration of insulin pretreatment.11
As secondary study endpoints, the percentage of patients achieving the target HbA1c of ≤6.5% and ≤7.5%, respectively, increased to nearly half of the patients after intensifying insulin treatment to a BBT regimen in both pretreatment arms. However, study duration may have been too short to uncover the true rate of long-term glycemic control and possible differences between the two study groups of strongly differing size. Moreover, patients of the much larger pre-BOT group were on average somewhat older than the pre-SIT group (median: 64 versus 62 years); thus, physicians may have had a higher target HbA1c more frequently accepted in that group.
In both treatment groups, FBG was also markedly and statistically significantly reduced. The decrease in FBG was also statistically significant in all subgroup analyses. The proportion of patients achieving target FBG ≤5.6 mmol/L (≤100 mg/dL) similarly increased between baseline and week 24 in both pretreatment arms. Subgroup analyses revealed no clear pattern in patients achieving FBG control. The decrease of FBG (−49.2±41.6 mg/dL) was nearly identical in a much larger T2DM population (n=5,695) after switching to a BBT regimen with insulin glargine and insulin glulisine in another observational study.12
Among other secondary study endpoints, data on periprandial glucose profiles were too rarely documented to provide a meaningful evaluation herein. On average, no relevant change in BW was observed in either group. It is also important to know that the injection device, most frequently SoloStar®, only rarely caused handling or dosing problems in this mainly elderly population. Up to 90% of the study population was satisfied with the device in use, which may support patient adherence and help to achieve and maintain optimized glycemic control.
Safety analyses included 24 reported AEs with four cases of death, which were unrelated to treatment. Only six hypoglycemic AEs (five SAEs) related to treatment in the pre-BOT group, two hypoglycemic AEs (two SAEs) related to treatment in the pre-SIT group as well as one hypoglycemic AE (one SAE) related to treatment in a patient pretreated with insulin glargine and insulin glulisine were documented, presenting an overall low rate of hypoglycemia in the PARTNER trial, thus confirming the safety of treatment intensification to BBT with insulin glargine and insulin glulisine.
Hypoglycemia was associated with increased cardiovascular risk and mortality in a number of multicenter trials. This issue has been extensively discussed in reviews by Goto et al19 and Hanefeld et al.20 As a consequence of these trials, the European Association for Cardiovascular Prevention and Rehabilitation included an explicit warning in their recent guideline (2012) to strictly avoid hypoglycemia in diabetic patients.21
In the ORIGIN trial22 that was designed to assess the effect of sufficient basal insulin on the reduction of cardiovascular events in comparison to standard of care, an HbA1c of 6.2% was reached after treatment with insulin glargine. This therapy had a neutral effect on cardiovascular outcomes and cancers. The incidence of a first episode of nonsevere symptomatic hypoglycemia that was confirmed by a self-measured glucose level of ≤54 mg/dL (≤3.0 mmol/L) was 9.83 and 2.68 per 100 person-years in the insulin glargine and standard-care groups. Severe hypoglycemic episodes were three- to fourfold less frequent as compared with the ACCORD and VADT studies.23
In the EARLY study, only seven of 1,438 T2DM patients presented with a severe hypoglycemia with an overall rate of 2.45% symptomatic hypoglycemias under basal insulin therapy added to maximally tolerated metformin doses.4 In the 52-week GINGER study,11 a basal–bolus (n=153) and a twice-daily premixed insulin regimen (n=157) were compared. Mean overall hypoglycemic events with basal–bolus and premix were 13.99 and 18.54 events per patient year, respectively. Finally, in an observational trial in 1,447 T1DM and 5,695 T2DM patients who were switched to a combination of insulin glargine and glulisine, statistically significant reductions in HbA1c by 1.0% and 1.2% were achieved within 6 months in T1DM and T2DM patients, respectively; the corresponding rates of any hypoglycemia were 17 (1.0%) and 41 events (0.9%), respectively.12 Thus, results from this study showed a similar or even lower rate of hypoglycemia. Overall, the combination of insulin glargine and glulisine can be considered safe.
Systolic and diastolic BP statistically significantly decreased in both treatment groups. As also reported for different insulin treatments in T2DM patients,22 this type of BBT involving glargine and glulisine has also been proven to have a very satisfactory safety profile, with a very low rate of severe hypoglycemia and without related cardiac implications as a major consequence of hypoglycemia.20
Conclusion
The PARTNER observational study, conducted in German daily routine practice, demonstrated important and clinically relevant improvements in glycemic control when intensifying a BOT regimen with insulin glargine or a SIT regimen with insulin glulisine to BBT. This improvement in glycemic control was accompanied by a very low rate of side effects. Overall, safety analyses indicated that the BBT was well tolerated, with few severe hypoglycemic episodes or other SAEs. The improved glycemic control was accompanied by a small, but statistically significant reduction in systolic and diastolic BP in both treatment groups.
Therefore, the results of this study further underline effectiveness and safety of insulin glargine and insulin glulisine as adequate combination partners in a BBT regimen in T2DM, and add confidence for the effective and safe use of this therapy in daily practice, which is supported by a substantial body of evidence from former observational studies.12,14–16
Acknowledgments
Editorial support was provided by Joachim Linke, MD, MSc, of creative clinical research and was funded by Sanofi-Aventis Deutschland GmbH, Berlin, Germany. The study was funded by Sanofi-Aventis Deutschland GmbH, Berlin, Germany.
Author contributions
All authors contributed toward data analysis, drafting and revising the paper and agree to be accountable for all aspects of the work. MP, TS, SP, JS, and KP revised it critically for important intellectual content. All authors approved the final version for publication.
Disclosure
MP has been on speaker’s bureaus for Berlin Chemie AG, Boehringer-Ingelheim, Fresenius, Eli Lilly and Co, Novartis, Novo Nordisk, and Sanofi. TS has been on speaker’s bureaus for Abbott, AstraZeneca, Bristol-Myers Squibb, Berlin Chemie AG, Boehringer-Ingelheim, Eli Lilly and Co, Medtronic, Merck Sharp and Dohme, Novartis, Novo Nordisk, and Sanofi. SP has been on speaker’s bureaus for Astra Zeneca, Berlin Chemie AG, Eli Lilly and Co, Novo Nordisk, Novartis, and Sanofi. KP is an employee of Sanofi-Aventis Deutschland GmbH, Berlin, Germany. JS has been on speaker’s bureaus for AstraZeneca, Bristol-Myers Squibb, Bayer Healthcare, Berlin Chemie AG, Eli Lilly and Co, GlaxoSmithKline, Lifescan Inc., Merck Sharp and Dohme, Novartis, Novo Nordisk, Pfizer Inc., Sanofi, and Takeda. The authors have no other conflicts of interest in this work.
References
Köster I, von Ferber L, Ihle P, Schubert I, Hauner H. The cost burden of diabetes mellitus: the evidence from Germany – the CoDiM Study. Diabetologia. 2006;49(7):1498–1504. | |
Matthaei S, Bierwirth R, Fritsche A, et al. Medical antihyperglycaemic treatment of type 2 diabetes mellitus: update of the evidence-based guideline of the German Diabetes Association. Exp Clin Endocrinol Diabetes. 2009;117(9):522–557. | |
Owens DR. Clinical evidence for the earlier initiation of insulin therapy in type 2 diabetes. Diabetes Technol Ther. 2013;15(9):776–785. | |
Hanefeld M, Fleischmann H, Landgraf W, Pistrosch F. EARLY study: early basal insulin therapy under real-life conditions in type 2 diabetics. Diabetes Stoffw Herz. 2012;21(2):91–97. | |
Chon S, Oh S, Kim SW, Kim JW, Kim YS, Woo JT. The effect of early insulin therapy on pancreatic beta-cell function and long-term glycemic control in newly diagnosed type 2 diabetic patients. Korean J Intern Med. 2010;25(3):273–281. | |
Inzucchi SE, Bergenstal RM, Buse JB, et al. Management of hyperglycemia in type 2 diabetes, 2015: a patient-centered approach. Update to a position statement of the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes Care. 2015;38(1):140–149. | |
Landgraf R, Kellerer M, Fach E, et al. Praxisempfehlungen DDG/DGIM. Diabetologie Stoffwechsel. 2013;8 (Suppl 2):S146–S158. | |
Holman RR, Paul SK, Bethel, MA, Matthews DR, Neil HA. 10-year follow-up of intensive glucose control in type 2 diabetes. N Engl J Med. 2008;359(15):1577–1589. | |
Stettler C, Allemann S, Jüni P, et al. Glycemic control and macrovascular disease in types 1 and 2 diabetes mellitus: metaanalysis of randomized trials. Am Heart J. 2006;152(1):27–38. | |
Ryden L, Standl E, Bartnik M, et al. Guidelines on diabetes, prediabetes, and cardiovascular disease: executive summary. Eur Heart J. 2007;28(1):88–136. | |
Fritsche A, Larbig M, Owens D, Häring H-U; on behalf of the GINGER Study Group. Comparison between a basal-bolus and a premixed insulin regimen in individuals with type 2 diabetes – results of the GINGER study. Diabetes Obes Metab. 2010;12(2):115–123. | |
Daikeler R, Högy B, Donaubauer B. Effects of an intensified insulin therapy (BBT) with insulin glargine and insulin glulisine on patient reported outcomes (PRO) in diabetes mellitus. Diabetologie Stoffwechsel. 2007;2(5):308–314. | |
Bundesärztekammer (BäK), Kassenärztliche Bundesvereinigung (KBV), Arbeitsgemeinschaft der Wissenschaftlichen Medizinischen Fachgesellschaften (AWMF). Nationale VersorgungsLeitlinie Therapie des Typ-2-Diabetes – Langfassung, 1. Auflage. Version 2. 2013, [last changed: April 2014]. Available from: http://www.versorgungsleitlin ien.de/themen/diabetes2/dm2_Therapie. Accessed July 25, 2015. | |
Siegmund T, Weber S, Blankenfeld H, Oeffner A, Schumm-Draeger PM. Comparison of insulin glargine versus NPH insulin in people with type 2 diabetes mellitus under outpatient-clinic conditions for 18 months using a basal-bolus regimen with a rapid-acting insulin analogue as mealtime insulin. Exp Clin Endocr Diabetes. 2007;115(6):349–353. | |
Donaubauer B, Ferlinz K, Schreiber S. Combining insulin glulisine with basal insulin glargine provides improved glycaemic control in patients with type 2 diabetes in everyday clinical practice. Diabet Med. 2008;25 (Suppl 1):48, Abstr P42. | |
Schreiber SA, Landgraf W, Böhler S. Efficacy and tolerability of insulin glargine in BBT. Diabetes Stoffw Herz. 2009;18(2):91–98. | |
Owens DR, Luzio SD, Sert-Langeron C, Riddle MC. Effects of initiation and titration of a single pre-prandial dose of insulin glulisine while continuing titrated insulin glargine in type 2 diabetes: a 6-month ‘proof of concept’ study. Diabetes Obes Metab. 2011;13(11):1020–1027. | |
Schreiber SA, Fiesselmann A, Bornstein SR, Landgraf W. Insulin analogues insulin glargine and insulin glulisine in type 1 diabetes. Diabetes Stoffw Herz. 2011;20(2):69–77. | |
Goto A, Arah OA, Goto M, Terauchi Y, Noda M. Severe hypoglycaemia and cardiovascular disease: systematic review and meta-analysis with bias analysis. BMJ. 2013;347:f4533. doi: 10.1136/bmj.f4533. Available from: http://www.isdbweb.org/documents/file/5293c692ca507.pdf. Accessed July 25, 2014. | |
Hanefeld M, Duetting E, Bramlage P. Cardiac implications of hypoglycaemia in patients with diabetes – a systematic review. Cardiovasc Diabetol. 2013;12:135. doi: 10.1186/1475-2840-12-135. Available from: http://www.cardiab.com/content/pdf/1475-2840-12-135.pdf. Accessed July 25, 2014. | |
Perk J, De Backer G, Gohlke H, et al. European guidelines on cardiovascular disease prevention in clinical practice (version 2012). The Fifth Joint Task Force of the European Society of Cardiology and Other Societies on Cardiovascular Disease Prevention in Clinical Practice (constituted by representatives of nine societies and by invited experts) Developed with the special contribution of the European Association for Cardiovascular Prevention and Rehabilitation (EACPR). Eur Heart J. 2012;33:1635–1701. | |
Gerstein HC, Bosch J, Dagenais GR, et al. Basal insulin and cardiovascular and other outcomes in dysglycemia. N Engl J Med. 2012;367(4):319–328. | |
Hanefeld M, Bramlage P. Insulin Use early in the course of type 2 diabetes mellitus: the origin trial. Curr Diab Rep. 2013;13:342–349. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.