Back to Journals » Clinical Interventions in Aging » Volume 16
Economic Evaluations of Interventions to Optimize Medication Use in Older Adults with Polypharmacy and Multimorbidity: A Systematic Review
Authors Laberge M , Sirois C, Lunghi C , Gaudreault M, Nakamura Y, Bolduc C, Laroche ML
Received 3 February 2021
Accepted for publication 31 March 2021
Published 5 May 2021 Volume 2021:16 Pages 767—779
DOI https://doi.org/10.2147/CIA.S304074
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Prof. Dr. Nandu Goswami
Maude Laberge,1– 3 Caroline Sirois,2– 5 Carlotta Lunghi,3,6 Myriam Gaudreault,7 Yumiko Nakamura,8 Carolann Bolduc,4 Marie-Laure Laroche9,10
1Department of Operations & Decision Systems, Faculty of Administration, Université Laval, Quebec City, Quebec, Canada; 2Vitam, Centre de recherche en santé durable-Université Laval, Quebec, Canada; 3Centre de recherche du CHU de Québec-Université Laval, Quebec City, Quebec, Canada; 4Faculty of Pharmacy, Université Laval, Quebec City, Quebec, Canada; 5Institut National de santé publique du Québec, Quebec City, Quebec, Canada; 6Department of Health Sciences, Université du Québec à Rimouski, Rimouski, Quebec, Canada; 7Faculty of Administration, Université Laval, Quebec City, Quebec, Canada; 8College of Medicine, University of Florida, Gainesville, FL, USA; 9Centre de Pharmacovigilance, de Pharmacoépidémiologie et d’information sur les médicaments, Service de Pharmacologie, Toxicologie et Pharmacovigilance, CHU de Limoges, Limoges, France; 10Laboratoire Vie-Santé, Faculté de Médecine, Université de Limoges, Limoges, France
Correspondence: Maude Laberge
Department of Operations & Decision Systems, Faculty of Administration, Université Laval, 2325 rue de la Terrasse, Bureau 2519, Quebec City, Québec, G1V 0A6, Canada
Tel +1 418-656-2131 ext 407670
Fax +1 418-656-2180
Email [email protected]
Purpose: To conduct a systematic review of the economic impact of interventions intended at optimizing medication use in older adults with multimorbidity and polypharmacy.
Methods: We searched Ovid-Medline, Embase, CINAHL, Ageline, Cochrane, and Web of Science, for articles published between 2004 and 2020 that studied older adults with multimorbidity and polypharmacy. The intervention studied had to be aimed at optimizing medication use and present results on costs.
Results: Out of 3,871 studies identified by the search strategy, eleven studies were included. The interventions involved different provider types, with a majority described as a multidisciplinary team involving a pharmacist and a general practitioner, in the decision-making process. Interventions were generally associated with a reduction in medication expenditure. The benefits of the intervention in terms of clinical outcomes remain limited. Five studies were cost-benefit analyses, which had a net benefit that was either null or positive. Cost-utility and cost-effectiveness analyses resulted in incremental cost-effectiveness ratios that were generally within the willingness-to-pay thresholds of the countries in which the studies were conducted. However, the quality of the studies was generally low. Omission of key cost elements of economic evaluations, including intervention cost and payer perspective, limited interpretability.
Conclusion: Interventions to optimize medication use may provide benefits that outweigh their implementation costs, but the evidence remains limited. There is a need to identify and address barriers to the scaling-up of such interventions, starting with the current incentive structures for pharmacists, physicians, and patients.
Keywords: polypharmacy, economic evaluation, potentially inappropriate medication, cost-effectiveness, cost-utility, cost-benefit
Introduction
Population aging comes with an increase in older individuals living with multiple chronic conditions leading to a rise in the number of prescribed medications. Polypharmacy, the consumption of multiple medications simultaneously,1 is associated with the risk of adverse drug events (ADEs), hospital admissions, the reduction of patients’ functional capacity and quality of life,2–6 and the prescription of potentially inappropriate medications (PIM). PIMs, although unstandardized in definition, generally refer to the prescription of medications that lead to higher risk relative to clinical benefit or reasonable alternative therapies.4,7 The term may refer to incorrect dosing or indication, prolonged medication use, and presence of interactions, among other suboptimal practices.7,8 A recent review of systematic reviews suggests that interventions to decrease the prescriptions of PIMs are effective, even if the effect size was moderate.7 Commonly employed interventions include medication reviews, wherein recommendations are made from a systematic assessment of medications, and pharmaceutical interventions, which incorporate formal clinical practice of pharmacists.7 Yet, the costs of conducting interventions to optimize medications in older people with polypharmacy have to be offset by the added value for patients and for health care systems for the interventions to be scaled up. To the best of our knowledge, no systematic review analyzed the economic evaluations of such interventions.
The purpose of our study was to conduct a systematic review of economic evaluations of interventions intended at optimizing medication use in older adults with multimorbidity and polypharmacy to determine if such interventions are cost-effective.
Methods
Using the PICO framework,9 we identified the population of interest as older adults with multimorbidity and polypharmacy. Multimorbidity was defined as the presence of at least two chronic conditions in a patient.10,11 Although there is no consensus on the definition of polypharmacy, it is generally considered when a patient is prescribed at least five medications.1 We, therefore, applied this common definition in our inclusion criteria when polypharmacy was not explicitly defined by the authors. When authors clearly defined polypharmacy (eg use of 10+ medications), we kept their definition. The intervention was aimed at optimizing medication use, the comparator being a group of individuals with similar characteristics but not receiving the intervention. Interventions were interpreted as optimizing medication use if they sought to evaluate patient medication lists in order to optimize prescription appropriateness and/or minimize medication-related problems (MRPs) and adverse drug events (ADEs).
Our systematic review is specific to studies including a full economic evaluation, that is a cost-effectiveness analysis (CEA), cost-utility analysis (CUA), or cost-benefit analysis (CBA). The outcome varied depending on the type of economic evaluation without any restrictive criteria on our side. While all three methods identify and measure the costs of at least two alternatives (for instance, an intervention and a control), they differ in how the outcomes are measured. In a CEA, a specific outcome related to the intervention is identified and measured in natural units for each alternative. For example, a study may measure cost-savings related to ADE avoided as a result of medication optimization.12,13 The CUA is a special case of a CEA in which the effect is measured in quality-adjusted life years (QALY). In a CBA, each outcome is given a monetary value. For example, a CBA may examine reduction in medication costs or medical expenditures due to PIM reduction.14–16
Research Strategy
With the support of a librarian, we identified all the terms associated with the elements of the PICO and the combinations possible. Details of the terminology are provided in the Appendix. The search was conducted in March 2018 and updated in May 2020 on the following databases: Ovid-Medline, Embase, CINAHL, Ageline, Cochrane, and Web of Science. We hand-searched references of all identified systematic reviews as well as included articles to single out additional potentially relevant articles.
Eligibility Criteria
To be included, studies needed to meet the following criteria: the study population was composed of at least 80% of older adults (defined as being aged 65 and over) with multimorbidity and having polypharmacy; the study included an economic evaluation of an intervention to optimize medication use; the article was published between 2004 and 2020; and the article was published in English, French, German, Italian, Spanish or Dutch, languages understood by our team. Because the definition of polypharmacy changed over the last decades, we restricted the search from 2004 to 2020 to limit heterogeneity in the definitions used between the studies. The lower limit of 2004 was chosen in the other systematic reviews performed by our team.17
The following criteria were used to exclude studies: the study population did not consist of older adults, the study targeted a population with polypharmacy within a specific condition (for instance, polypharmacy described as the use of multiple psychotropic medications in schizophrenia), the study did not assess an intervention, costs were not measured. We nevertheless included studies in which the intervention was on a specific medication class optimization, without identifying a specific condition. Studies that did not include a complete economic evaluation (CEA, CUA, or CBA) were excluded. We also excluded systematic reviews, study protocols and conference posters.
Study Selection
The first phase consisted of identifying articles eligible for full-review. First, one of the authors (MG) removed all the duplicates in the list. Then, four authors scanned titles and abstracts (CB, CS, MG, ML), with each title/abstract being independently assessed by two authors. Each author indicated whether an article should be included or excluded using the defined criteria. The authors discussed any discordance to reach a consensus on the list of articles. In the second phase, each article was independently fully read by at least two authors to confirm inclusion (CS, CL, MG, ML, YN). If, upon full review, the article met one of the exclusion criteria, it was removed, and the reason for the exclusion was noted. If multiple articles covered the same intervention, we selected the one deemed the most relevant based on the study objectives and inclusion/exclusion criteria and was usually the most recent. The list of references from systematic reviews and included articles were hand-searched. Titles and abstracts of articles that were not in the initial search were reviewed by two authors (YN and ML) using the same process as described above.
Data Extraction
A table of variables was developed for data extraction with the following elements: authors, year of publication, title, study design, time horizon, number of patients, study setting (primary care clinics, pharmacies, long term care facilities, etc.), country, intervention characteristics, clinical outcomes of the intervention, costs elements measured (direct medical, direct non-medical, indirect), perspective (hospital, health care system, patient, pharmacist, society), and results of the economic evaluation (incremental cost-effectiveness ratio [ICER], net benefit, or cost-benefit ratio). Data were extracted and reviewed by at least two authors independently with a third author when there was uncertainty (CL, CS, MG, ML, YN).
At least two authors independently assessed the quality of each study retained and discussed discrepancies, with a third author providing input when needed to reach consensus (CL, CS, ML, MLL, YN). The Quality of Health Economics Studies (QHES) instrument18 was used for the assessment. In this instrument, a number of points are attributed for fulfilling certain criteria; evaluators can only give 0 or full points.
Data Synthesis
Studies were organized based on intervention and economic evaluation themes. Intervention themes included setting, type of intervention to optimize medication use (PIM identification tool, medication reviews), provider types involved, and incorporation of patient-provider discussions in decision-making. We determined the type of economic evaluation based on costs and outcomes measured in each study. We independently determined whether each study qualified as a CEA, CUA, or CBA, and discrepancies were discussed. Quantitative analyses were limited due to heterogeneity of study design and economic evaluations.
Results
Figure 1 shows the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA)19 flow diagram (Figure 1). A total of 3,871 distinct articles were identified from the search strategy across six databases. After including four articles identified through systematic review references, 68 articles were eligible for full-text review. Of the total full text articles, eleven studies (including three from the 2020 update) met the inclusion criteria. The characteristics of the studies are summarized in Table 1. The studies came from different countries [Spain, n=2; Australia, n=1; Ireland, n=2; Netherlands, n=2; Northern Ireland, n=1; Taiwan, n=1; and United Kingdom, n=1, United States=1]. The settings were distributed between primary care clinics (n=4),12,14,15,20 nursing homes (n=1),21 pharmacies (n=3),22–24 one hospital,13 one academic medical centre,16 and one study included three different types of clinical sites.25 The most common study design was cluster randomized controlled trial (n=6),13,15,20,21,23,24 or randomized controlled trial (n=3).12,14,16 One study conducted a before-and-after analysis22 and one was a cohort study with a control group.25
 |
Table 1 Characteristics of Included Studies |
 |
Figure 1 Flow diagram for screening and selection processes. Updated search conducted in May 2020 resulted in 1,145 new records screened, 36 full-text articles assessed, and three studies included in the review. Notes: Adapted from: Moher D, Liberati A, Tetzlaff J, Altman DG, The PRISMA Group (2009). Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. 2009;6(7):e1000097. doi:10.1371/journal.pmed1000097.37 Copyright: © 2009 Moher et al Creative Commons Attribution License. For more information, visit www.prisma-statement.org. |
Intervention Characteristics
Characteristics of the interventions are reported in Tables 1 and 2. The stated purpose of interventions was variable. Seven studies examined interventions explicitly aimed at identifying PIMs.12–14,16,20–22 Five studies included interventions with a stated goal of optimizing prescribing to address MRPs or ADEs.13,16,23–25 One study evaluated medications reviews as a form of monitoring “prescription-related points of concern”, such as benzodiazepines use in older adults.15
 |
Table 2 Select Characteristics of Intervention |
All interventions included a medication review performed by a pharmacist except for one study in which the intervention consisted of pharmacogenetic testing coupled with a clinical decision support tool.25 The interventions involved different provider types; some described multidisciplinary teams, while others had providers as informal collaborators. The pharmacists’ recommendations were most often discussed with a general practitioner (GP) (n=8),12,14–16,20–22,25 or/and specialists (geriatricians, cardiologists, nephrologist) (n=1).16 The Screening Tool of Older Persons Prescriptions/Screening Tool to Alert doctors to Right Treatment (STOPP/START),13 and the Good-Palliative-Geriatric Practice (GP-GP) tools were mentioned as means to identify PIMs.14,22 Seven studies also mentioned the use of an algorithm that was either developed by the team and explained, using existing tools, or not further defined.12,15,16,20,21,23–25 The duration of the interventions and the study period varied between four25 and 12 months,14,16,20,21,24 with the exception of one study in which the time horizon was the 10 days following a patient’s discharge.13
Intervention Clinical Outcomes
The benefits of the intervention in terms of clinical outcomes remain limited. Only two studies specifically reported a decrease in PIM use,20,21 including one restricted on antipsychotics use.21 Two studies reported decreases in ADEs.13,24 Two studies reported recommendations for changes in medication,15,22 with one documenting those implemented during the intervention, but the changes between groups were no longer significant nine months after the intervention.15 Two studies that evaluated the decrease in the total number of medications did not obtain statistically significant results between groups.14,23 Further, no statistically significant difference was noted in the Duke’s severity of illness score, the number of hospital admissions or GP visits in the study by Sorensen et al.12 Hospital re-admissions were higher in the intervention group than they were in the control group in van der Heijden et al’s study.24 The number of hospitalizations and of emergency room visits were reduced in the study by Brixner et al, but outpatient visits increased, although not statistically significantly.25 Finally, mixed results were obtained on laboratory data in the study by Lin et al, where about half of the many tests performed were not statistically significant.16
Nine studies included the costs of the interventions. One included only the cost of a pharmacogenetic test.25 The other eight included the time of pharmacists, and of physicians.12–15,20,22–24 When included, non-medication expenditures, and specifically costs of hospitalizations, drove total patient expenditures down following the intervention.16,20,23,25 In one study, the factors driving the reduction in medication expenditure costs were identified as, first, the reduction in the total number of prescribed medications, second, the reduction of newer (more expensive) prescribed medications, and lastly, the change in dosages.14
Economic Evaluations
Five studies were cost-benefit analyses where the outcomes were medication expenditure14,15 or health care costs.16,23,25 They reported either a non-significant benefit15 or cost-benefit ratios between 2.3814 and 6.22.23 Cost-utility analyses with QALYs were conducted in two studies.20,22 Estimates of the cost per QALY gained varied from £11,885 to £32,466 in the UK22 and were €30,535 in Ireland.20 Cost-effectiveness analyses were conducted in five studies and used either the proportion of patients having a PIM,21 the number of PIM avoided20 or the number of ADE avoided.12,13,24 The cost per PIM avoided was estimated at €1,269 (95% CI=€-1,400-€6,302).20 The cost per ADE avoided varied between AUS $6912 and €8,270.24 Patterson et al found the intervention to be associated with non-significant lower cost as well as a reduction in patients having at least one PIM.21 Complete results are reported in Table 3.
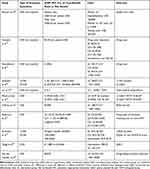 |
Table 3 Economic Evaluation Characteristics |
Quality of the Studies
The QHES score of each study is indicated in Table 4 and ranges from 51 to 99 out of 100. Only five studies stated the perspective.13,20,21,23,24 Sensitivity analyses were reported in six studies.14,20,21,23–25 Similarly, seven studies transparently displayed the components for the economic evaluation13,14,20–22,24,25 and six explained the potential sources of bias.14,15,20,21,24,25 The type of economic evaluation was not always explicit, and there were key cost elements that were often omitted, notably the cost of the intervention itself (physician time, training of providers, etc.).
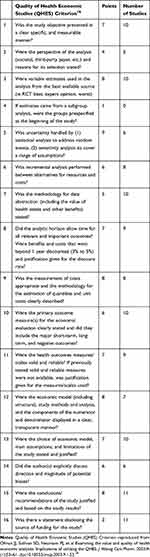 |
Table 4 Quality Assessment of Included Studies |
Discussion
Despite a rich literature on interventions to optimize medication uses in older individuals with polypharmacy, only eleven studies met the criteria of the presence of a full economic evaluation. Cost-effectiveness of the intervention may depend on contextual factors of the intervention and willingness-to-pay thresholds for a marginal improvement in medication use or a marginal reduction in ADEs. Due to heterogeneity in reported outcomes and suboptimal quality of the economic evaluations, we cannot draw a clear conclusion on the cost-effectiveness of interventions to optimize medication use.
Only four studies evaluated the cost per PIM avoided or ADE avoided as result of the interventions.13,20,21,24 Specifically, Gillespie et al included both a cost-effectiveness analysis (CEA) and a cost-utility analysis (CUA) with estimates of the cost per PIM avoided and per QALY gained (measured using the EQ5D-3L instrument), respectively.20 This study provided a more complete estimate of costs, including health care services utilization (visits with nurses and physicians in the primary care clinic as well as in other settings, hospital visits and admissions).20 The outcome in the study was also the QALY, and the intervention was found to be costlier and more effective. Its cost-effectiveness depends on the willingness-to-pay for each additional QALY, with a marginal cost here estimated at € 30,535.20 However, one could put such an ICER in perspective with treatments that have much higher ICER and that are covered in other jurisdictions. O’Brien et al estimated the ICER per ADE due to suboptimal prescribing at 5,358 and van der Heijden at 8,270. Both authors conclude that the interventions are not cost-effective. Yet, the cost-effectiveness depends on the willingness-to-pay for these outcomes. While a few jurisdictions have (implicit or explicit) thresholds for QALYs, such thresholds do not exist for ADEs.
Studies do carry limitations that render any generalization of such results difficult. The economic evaluation profile of interventions to optimize medication use in older adults with polypharmacy depends on several factors in terms of context, cost elements included, and details of the intervention itself. For instance, in one study, implementation costs consisted of only the pharmacist salary, not taking the remuneration of physicians into account even though they were also involved in the intervention.23 Albeit, according to authors, the limited time involved would not make a significant difference. One study did estimate the average time of both the physician’s and the pharmacist’s involvement in the intervention and used the time to estimate costs and found that the intervention costs were more than compensated for with a reduction in medication costs, resulting in a net benefit.14
In the studies identified in this review, reduction in PIM and in ADE due to suboptimal prescribing were the main effect measured in 2 studies each. One found a negative ICER21 while the other three found that interventions, although effective, came with an ICER varying between €1,26920 and €8,270.24 When a cost-utility analysis is conducted, the estimates for the incremental cost per QALY suggest that the intervention is cost-effective if decision-makers are willing to pay £32,46622 in one case and €30,53520 in the other, for a QALY.
Perspectives and Outcomes of Interventions
Most studies did not rigorously follow the methods for economic evaluations as explained by Drummond et al26 and failed to report important elements. Such is the case for the perspective, which was not explicitly stated in most studies (n=7). Payers for different types of health services and medications were rarely mentioned. If we assume that medications for older adults are mostly paid by public health insurers, which also pay for other health services (hospitalizations, physician visits, etc.), the public health insurers would have an interest in decreasing costs related to all such services, versus shifting services or medication expenditure to another payer. Community pharmacists may have no stake in reducing medication use in patients from a strictly economic perspective (which, of course, differs from the clinical and ethical perspectives), whereas nursing homes or hospitals that are covering the costs within their own budget would. In all cases, it is likely that there are also costs for patients, in the form of co-pays for health care services and medications, yet the patient perspective was never included. Acknowledging the out-of-pocket costs for patients would add value to such interventions.
Another way of considering the issue of suboptimal medication use is through its impact on patients’ quality of life. Five studies measured quality of life and used the QALY as an outcome measure of the intervention.12,16,20,22,23 Although it is essential to consider the impact of suboptimal medication use in multimorbid older adults with polypharmacy on quality of life, the low average effect may reflect dilution from heterogeneity of the effect. One study examined cost-effectiveness of reducing prescriptions for some categories of medications and found that interventions to reduce long-term use of non-steroidal anti-inflammatory medications seemed to be the most likely to be cost-effective compared to benzodiazepines and proton pump inhibitors.27
Contextual Factors: Pharmacists’ and Physicians’ Incentives
On the context aspect, whether the intervention happens in the community or in an institutional setting represents an important difference. In an institutional setting, the pharmacists are generally salaried employees. In contrast, owners of pharmacies may be receiving dispensing fees and higher discounts when increasing the volumes of medications purchased.28 Hence, from an economic perspective, the incentives in such an environment would motivate the continued dispensing of medications, and not support a conversation with physicians to deprescribe medications. From the cost calculation of pharmacists’ remuneration in studies conducted in Spain, it appears that community pharmacists were paid by salary, and there was no indication as to the business model of community pharmacies.14,29,30 Aside from the intervention implemented in Spain, there was no or limited information as to the remuneration models of pharmacists and how different payment schemes could affect the effectiveness of such interventions. For instance, the Canadian province of Ontario implemented a fee-for-service type remuneration for pharmacists’ medication therapy management (MTM) services, including medication reconciliation. The uptake of MTM services may depend not only on the remuneration levels, but also on the capacity of a pharmacy to adapt and transform its business model.31 In Ontario, the uptake required changes to pharmacists’ practices32 that may have been stimulated by a bill reducing pharmacists’ income from generic medication manufacturers’ rebates.33
Although pharmacists have an important role in identifying suboptimal medication use, physicians, as prescribers, have an equally important role. Physicians may not consider the costs to patients or to the healthcare system of their prescriptions, and they have no incentive to review their patients’ prescriptions to remove any prescriptions that may not have any benefit to patients. Some of them also receive benefits from the pharmaceutical industry to increase prescription.34,35
Methodological Limitations of the Included Studies
The studies included in this review had a number of limitations. First, the cost elements included varied among studies and the total cost of the intervention was not always estimated. Without such information, it is difficult to assess the value of the interventions and their potential for scaling up. Second, the design in some studies limited the validity of the results. For instance, one study did the randomization at the patient level with physicians treating both control and intervention patients, which may have led to contamination.14
Most studies had a small sample size, which does not enable estimating how patient characteristics could affect the cost-effectiveness of an intervention. Yet, from an economic perspective, it is questionable whether pharmacists should review all patients’ medications, or if such an intervention is only cost-effective at a given level of patient complexity. This is illustrated in the study by Brixner et al25 who conducted CBA separately on median and average costs. Using median costs resulted in a net loss while using average costs resulted in a net benefit. The results suggest that the higher reductions in expenditures happen in the tail, ie in patients with very high expenditure. Therefore, there is a need to investigate further the characteristics of the patients for whom such interventions are more likely to be cost-effective so that payers could determine selection criteria for patients that would be eligible.36
A factor that is seldom considered in the interpretation of the results is the self-selection of health care providers (mainly pharmacists and physicians) into studies. Not only was there attrition of participation in one study,12 but researchers reported having difficulty recruiting providers to participate in the studies, suggesting that the interest is limited and that scaling-up such interventions would prove challenging. In addition, pharmacists and physicians participating into studies could differ from non-participants, which could lead to overestimating the benefits of the interventions. Moreover, although RCTs are considered the gold standard, results in real context may differ from those of the RCT.
Strengths and Limitations
One strength of this review is the fact that we applied an in-depth search strategy following the PICO framework in different databases to identify all the suitable studies. Nevertheless, we cannot exclude that relevant studies may have been missed, especially if they were published only in languages other than those spoken by the authors of this review. We considered all studies that included an economic evaluation of an intervention aimed at optimizing medication use in the context of polypharmacy, including all kinds of interventions and without restricting the definition of polypharmacy, referring to the way it was defined by the authors. This means that we did not evaluate the relevance of the interventions and the correctness of the definitions of polypharmacy. It is indeed known that, especially for polypharmacy, different definitions are proposed across the studies.1
Conclusion
Our review revealed very few studies with a full economic evaluation on interventions to optimize medication use, which is particularly surprising when considering the high number of studies in which such interventions are conducted. Whether interventions to optimize medication use are cost-effective may depend on a number of contextual factors. More importantly, it requires that the intervention be scalable in its given context.
The quality of the studies was not optimal. One of the striking elements of the studies identified is that none includes the patient perspective. The burden of prescription medications is increasing for patients. As such, optimizing medication use may have benefits not only to health care systems, but also to patients, and not only in terms of health care status.
Future research is needed with more thoughtful estimates of total intervention cost and rigorous methodology applied to economic evaluations of interventions in order to assess the economic impact of such intervention in large-scale environments.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Funding
There was no funding received for this specific study. CS and ML are each recipients of a Research Scholar from the Fonds de recherche du Québec – Santé. The funding agencies had no role in any part of the study.
Disclosure
Dr Caroline Sirois reports grants from Fonds de recherche du Québec, during the conduct of the study. The authors declare that they have no other potential conflicts of interest for this work.
References
1. Sirois C, Domingues NS, Laroche M-L, et al. Polypharmacy definitions for multimorbid older adults need stronger foundations to guide research, clinical practice and public health. Pharmacy. 2019;7(3):126. doi:10.3390/pharmacy7030126
2. Brown JD, Hutchison LC, Li C, Painter JT, Martin BC. Predictive validity of the beers and screening tool of older persons’ potentially inappropriate prescriptions (STOPP) criteria to detect adverse drug events, hospitalizations, and emergency department visits in the United States. J Am Geriatr Soc. 2016;64(1):22–30. doi:10.1111/jgs.13884
3. Hill-Taylor B, Sketris I, Hayden J, Byrne S, O’Sullivan D, Christie R. Application of the STOPP/START criteria: a systematic review of the prevalence of potentially inappropriate prescribing in older adults, and evidence of clinical, humanistic and economic impact. J Clin Pharm Ther. 2013;38(5):360–372. doi:10.1111/jcpt.12059
4. Kaufmann C, Tremp R, Hersberger K, Lampert M. Inappropriate prescribing: a systematic overview of published assessment tools. Eur J Clin Pharmacol. 2014;70(1):1–11. doi:10.1007/s00228-013-1575-8
5. van der Stelt C, Windsant-van den Tweel AV, Egberts A, et al. The association between potentially inappropriate prescribing and medication-related hospital admissions in older patients: a nested case control study. Drug Saf. 2016;39(1):79–87. doi:10.1007/s40264-015-0361-1
6. Black CD, Thavorn K, Coyle D, Bjerre LM. The health system costs of potentially inappropriate prescribing: a population-based, retrospective cohort study using linked health administrative databases in Ontario, Canada. PharmacoEconomics. 2020;4:27–36. doi:10.1007/s41669-019-0143-2
7. Santos NSD, Marengo LL, Moraes F da S, Barberato Filho S. Interventions to reduce the prescription of inappropriate medicines in older patients. Rev Saude Publica. 2019;53:7. doi:10.11606/S1518-8787.2019053000781
8. Hepler C, Strand L. Opportunities and responsibilities in pharmaceutical care. Am J Hosp Pharm. 1990;47(3):533–543.
9. Institute of Medicine (U.S.). Finding what works in health care standards for systematic reviews. National Academies Press; 2011. Available from: http://www.nap.edu/catalog.php?record_id=13059.
10. Le Reste JY, Nabbe P, Rivet C, et al. The European general practice research network presents the translations of its comprehensive definition of multimorbidity in family medicine in ten European languages. PLoS One. 2015;10(1):e0115796. doi:10.1371/journal.pone.0115796
11. Xu X, Mishra GD, Jones M. Evidence on multimorbidity from definition to intervention: an overview of systematic reviews. Ageing Res Rev. 2017;37(C):53–68. doi:10.1016/j.arr.2017.05.003
12. Sorensen L, Stokes JA, Purdie DM, Woodward M, Elliott R, Roberts MS. Medication reviews in the community: results of a randomized, controlled effectiveness trial. Br J Clin Pharmacol. 2004;58(6):648–664. doi:10.1111/j.1365-2125.2004.02220.x
13. O’Brien GL, O’Mahony D, Gillespie P, et al. Cost-effectiveness analysis of a physician-implemented medication screening tool in older hospitalised patients in Ireland. Drugs Aging. 2018;35(8):751–762. doi:10.1007/s40266-018-0564-0
14. Campins L, Serra-Prat M, Palomera E, Bolibar I, Martinez MA, Gallo P. Reduction of pharmaceutical expenditure by a drug appropriateness intervention in polymedicated elderly subjects in Catalonia (Spain). Gac Sanit. 2017;33(2):106–111. doi:10.1016/j.gaceta.2017.09.002
15. Denneboom W, Dautzenberg MGH, Grol R, De Smet PAGM. Treatment reviews of older people on polypharmacy in primary care: cluster controlled trial comparing two approaches. Br J Gen Pract J R Coll Gen Pract. 2007;57(542):723–731.
16. Lin HW, Lin CH, Chang CK, et al. Economic outcomes of pharmacist-physician medication therapy management for polypharmacy elderly: a prospective, randomized, controlled trial. J Formos Med Assoc. 2018;117(3):235–243. doi:10.1016/j.jfma.2017.04.017
17. Sirois C, Laroche M-L, Guénette L, Kröger E, Cooper D, Émond V. Polypharmacy in multimorbid older adults: protocol for a systematic review. Syst Rev. 2017;6(1). doi:10.1186/s13643-017-0492-9
18. Ofman JJ, Sullivan SD, Neumann PJ, et al. Examining the value and quality of health economic analyses: implications of utilizing the QHES. J Manag Care Pharm. 2003;9(1):53–61. doi:10.18553/jmcp.2003.9.1.53
19. Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med. 2009;151(4):264. doi:10.7326/0003-4819-151-4-200908180-00135
20. Gillespie P, Clyne B, Raymakers A, Fahey T, Hughes CM, Smith SM. Reducing potentially inappropriate prescribing for older people in primary care: cost-effectiveness of the Opti-script intervention. Int J Technol Assess Health Care. 2017;33(4):494–503. doi:10.1017/S0266462317000782
21. Patterson SM, Hughes CM, Cardwell C, Lapane KL, Murray AM, Crealey GE. A cluster randomized controlled trial of an adapted U.S. model of pharmaceutical care for nursing home residents in Northern Ireland (Fleetwood Northern Ireland study): a cost-effectiveness analysis. J Am Geriatr Soc. 2011;59(4):586–593. doi:10.1111/j.1532-5415.2011.03354.x
22. Twigg MJ, Wright D, Barton GR, Thornley T, Kerr C. The four or more medicines (FOMM) support service: results from an evaluation of a new community pharmacy service aimed at over-65s. Int J Pharm Pract. 2015;23(6):407–414. doi:10.1111/ijpp.12196
23. Malet-Larrea A, Goyenechea E, Gastelurrutia M, et al. Cost analysis and cost-benefit analysis of a medication review with follow-up service in aged polypharmacy patients. Eur J Health Econ. 2017;18(9):1069–1078. doi:10.1007/s10198-016-0853-7
24. van der Heijden AA, de Bruijne MC, Nijpels G, Hugtenburg JG. Cost-effectiveness of a clinical medication review in vulnerable older patients at hospital discharge, a randomized controlled trial. Int J Clin Pharm. 2019;1–9. doi:10.1007/s11096-019-00825-3
25. Brixner D, Biltaji E, Bress A, et al. The effect of pharmacogenetic profiling with a clinical decision support tool on healthcare resource utilization and estimated costs in the elderly exposed to polypharmacy. J Med Econ. 2016;19(3):213–228. doi:10.3111/13696998.2015.1110160
26. Drummond M, Sculpher MJ, Claxton K, Stoddart GL, Torrance GW. Methods for the economic evaluation of health care programmes. Oxford University Press; Accès via ProQuest Ebook Central - La consultation est limitée à un usager à la fois; 2015. Available from: https://ebookcentral.proquest.com/lib/ulaval/detail.action?docID=4605509.
27. Moriarty F, Cahir C, Bennett K, Fahey T. Economic impact of potentially inappropriate prescribing and related adverse events in older people: a cost-utility analysis using Markov models. BMJ Open. 2019;9(1):e021832. doi:10.1136/bmjopen-2018-021832
28. Silversides A. Pharmacies receiving massive rebates from generic drug-makers. CMAJ Can Med Assoc J. 2006;175(4):342. doi:10.1503/cmaj.060904
29. Malet-Larrea A, Goyenechea E, Garcia-Cardenas V, et al. The impact of a medication review with follow-up service on hospital admissions in aged polypharmacy patients. Br J Clin Pharmacol. 2016;82(3):831–838. doi:10.1111/bcp.13012
30. Parody Rúa E, Segu Tolosa JL. Efectividad y estimación de costes en una intervención sobre problemas relacionados con los medicamentos en atención primaria. Aten Primaria. 2005;35(9):472–477. doi:10.1157/13075471
31. Feletto E, Wilson LK, Roberts AS, Benrimoj S. Flexibility in community pharmacy: a qualitative study of business models and cognitive services. Pharm World Sci. 2010;32(2):130–138. doi:10.1007/s11096-009-9355-3
32. Dolovich L, Gagnon A, McAiney CA, Sparrow L, Burns S. Initial pharmacist experience with the Ontario-based MedsCheck Program. Can Pharm J. 2008;141(6):339–345.e1. doi:10.3821/1913-701X-141.6.339
33. Lynas K. Beyond bill 102: Ontario government reviews provincial drug system. Can Pharm J. 2009;142(5):212. doi:10.3821/1913-701X-142.5.212
34. DeJong C, Aguilar T, Tseng C-W, Lin GA, Boscardin WJ, Dudley RA. Pharmaceutical industry–sponsored meals and physician prescribing patterns for medicare beneficiaries. JAMA Intern Med. 2016;176(8):1114–1122. doi:10.1001/jamainternmed.2016.2765
35. Fleischman W, Agrawal S, King M, et al. Association between payments from manufacturers of pharmaceuticals to physicians and regional prescribing: cross sectional ecological study. BMJ. 2016;354:i4189. doi:10.1136/bmj.i4189
36. Trygstad TK, Christensen DB, Wegner SE, Sullivan R, Garmise JM. Analysis of the North Carolina long-term care polypharmacy initiative: a multiple-cohort approach using propensity-score matching for both evaluation and targeting. Clin Ther. 2009;31(9):2018–2037. doi:10.1016/j.clinthera.2009.09.006
37. Moher D, Liberati A, Tetzlaff J, Altman DG; The PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6(7):e1000097. doi:10.1371/journal.pmed1000097
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
