Back to Journals » Therapeutics and Clinical Risk Management » Volume 13
ECMO as an effective rescue therapeutic for fulminant myocarditis complicated with refractory cardiac arrest
Authors Li YT , Yang LF, Chen ZG , Pan L, Duan MQ, Hu Y, Zhou CB, Guo YX
Received 31 July 2017
Accepted for publication 21 October 2017
Published 14 November 2017 Volume 2017:13 Pages 1507—1511
DOI https://doi.org/10.2147/TCRM.S147845
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Deyun Wang
Ya-Ting Li,1,* Li-Fen Yang,1,* Zhuang-Gui Chen,1,* Li Pan,1 Meng-Qi Duan,1 Yan Hu,2 Cheng-bin Zhou,3 Yu-Xiong Guo2
1Pediatric Intensive Care Unit, Department of Pediatrics, The Third Affiliated Hospital of Sun Yat-sen University, Guangzhou, 2Pediatric Intensive Care Unit, Department of Pediatrics, Guangdong General Hospital, Guangdong Academy of Medical Sciences, Guangzhou, 3Department of Cardiovascular Surgery, Guangdong Cardiovascular Institute, Guangdong Provincial Key Laboratory of South China Structural Heart Disease, Guangdong General Hospital, Guangdong Academy of Medical Sciences, Guangzhou, People’s Republic of China
*These authors contributed equally to this work
Abstract: Fulminant myocarditis (FM) is a life-threatening disease in children. With a rapid, progressive course of deterioration, it causes refractory cardiorespiratory failure even with optimal clinical intervention. We present the case of a 9-year-old girl with FM complicated by cardiogenic shock, malignant arrhythmia, and refractory cardiac arrest. She received effective cardiopulmonary resuscitation, therapeutic hypothermia, and other supportive treatments. However, the patient rapidly worsened into pulseless ventricular tachycardia and refractory cardiac arrest. Therefore, we performed extracorporeal membrane oxygenation (ECMO) to establish spontaneous circulation after the failure of standard resuscitation measures. The girl recovered with intact cardiac and neurocognitive functions after continued ECMO treatment for 221 hours. Therefore, ECMO is an effective rescue therapeutics for FM, especially when complicated with refractory cardiac arrest.
Keywords: cardiac arrest, children, extracorporeal membrane oxygenation, fulminant myocarditis
Introduction
Fulminant myocarditis (FM) is characterized by sudden onset after flu-like illness and rapidly progressive, resulting in fatal arrhythmias, severe congestive heart failure, and cardiogenic shock with a high risk of circulatory collapse.1–3 FM had a very high mortality ranging from 25% to 75% without immediate mechanical circulatory support.4 In cases of cardiogenic shock or cardiac arrest refractory to usual resuscitative therapies such as vasopressors or intra-aortic balloon pumps (IABPs), left ventricular assist devices have been used as a supportive therapy in FM but are often bulky and invasive.5–7 In contrast, a more readily accessible intervention, extracorporeal membrane oxygenation (ECMO), has been shown to be an effective life-saving strategy in adult patients with FM.7–9 Unfortunately, optimal clinical intervention is not effective in many cases of children with FM. In this report, we present the case of a 9-year-old girl with FM that rapidly progressed to cardiac arrest, unresponsive to advanced life support or medical treatment, but survived with the application of ECMO.
Case presentation
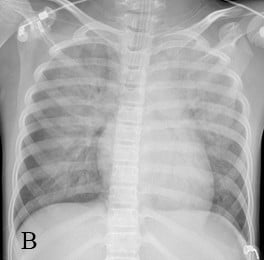
A previously healthy 9-year-old girl complained of sore throat for 2 days, with general fatigue, dizziness, and paleness for 6 hours. She was first taken to a local hospital on September 5, 2015, and was admitted to the hospital due to unresponsiveness to antibiotics. She was conscious, 28 kg in weight, and had low fever (38°C), heart rate 50–60 bpm, and hypotension (77/47 mmHg). Laboratory tests showed creatine kinase-MB (CKMB) 1,525 U/L, CKMB 170 U/L, alanine aminotransferase (ALT) 67 U/L, aspartate aminotransferase (AST) 241 U/L, and troponin I 137.06 μg/L, while electrocardiogram (ECG) demonstrated atrial fibrillation, aberrant ventricular conduction, and premature ventricular contraction. Chest X-ray was normal. High doses of methylprednisolone (20 mg/kg/d) and immunoglobulin (400 mg/kg/d) were administrated immediately. The blood pressure (BP) remained around 88/58 mmHg, heart rate (HR) 80–90 bpm, and saturation of peripheral oxygen (SPO2) 95%. Considering the critical conditions, she was transferred to our hospital with a diagnosis of acute myocarditis complicated by cardiogenic shock. Laboratory reevaluation showed CK 1,123 U/L, CKMB 126.6 U/L, AST 611 U/L, lactate dehydrogenase (LDH) 1,792 U/L, hydroxybutyrate dehydrogenase (HBDH) 1,011 U/L, pro-brain natriuretic peptide (Pro-BNP) 16,894 pg/mL, ALT 348 U/L, blood urea nitrogen (BUN) 17.17 μmol/L, creatinine 163 mmol/L, and ECG showed multiple induced ventricular premature repeatedly occurred. Chest X-ray demonstrated cardiomegaly (cardiothoracic ratio: 60%) (Figure 1A). Echocardiography showed globally reduced wall motion, a severely dilated ventricular cavity with wall thickening, atrioventricular valve regurgitation, and severe left ventricular dysfunction with ejection fraction (EF) of 37% and fractional shortening (FS) of 16%. Dopamine and dobutamine were continuously administered. The girl’s condition worsened with hemodynamic instability, respiratory failures, ventricular tachycardia (VT) with a heart rate of 158 bpm, and BP 65/50 mmHg 4 hours after administration. Intubation and synchronized cardio version were immediately conducted. But she soon went into a witnessed pulseless VT and cardiac arrest. Chest compressions and defibrillations were conducted multiple times. In addition, the patient also received multiple doses of intravenous adrenaline, lidocaine, and amiodarone. Unfortunately, the girl’s condition was progressively worse. Emergency echocardiography demonstrated a poorly contracting heart. Considering VT refractory to antiarrhythmic therapy, the patient was shifted to percutaneous veno-arterial ECMO (VA-ECMO) at 7 h after admission. Chest compressions were continued until placement of the right femoral arterial and venous cannulae. The total duration of cardiopulmonary resuscitation (CPR) lasted for nearly 180 mins. In addition, therapeutic hypothermia was induced and maintained at 33°C–35°C for 120 h after establishment of ECMO support. The ECMO device was equipped with a centrifugal pump and an artificial lung was used (Medtronic Bio-Medicus, Minneapolis, MN, USA). Blood circulation was performed from an 18-Fr tube (Bio-Medicus) inserted into the femoral artery to an 18-Fr tube inserted into the femoral vein. The ECMO flow started around 2.0 L/min and was adjusted according to the hemodynamic status (maintained mean airway pressure [MAP] at 50–70 mmHg and central venous pressure [CVP] at 5–12 mmHg). The target blood flow was 80–100 mL/kg/min, and low-dose heparin was infused to maintain activated clotting time of 160–200 s. The target oxygenation was a normal arterial partial pressure of carbon dioxide (PaCO2) and partial pressure of oxygen (PaO2). After ECMO deployment, the dose of inotropic agents was slowly tapered off and decreased to a minimum according to the tissue perfusion and hemodynamic status, and the respirator was set at “resting” parameters. On the 48th hour of ECMO deployment, echocardiography showed left ventricle (LV) end diastolic dimensions and left atrial (LA) end systolic dimensions were 32 and 38 mm, respectively. But predominant decrease of PaO2 and increase of renal function indicators (renal failure) also occurred. Chest X-ray showed aggravating pulmonary edema (Figure 1B). Conventional ventilation with 100% oxygen and 30 cm H2O Ppeak and 15 cm positive end-expiratory pressure (PEEP) H2O cannot maintain normal SPO2. ECG demonstrated aberrant VT. Continuous renal replacement therapy (CRRT) and high-frequency oscillatory ventilation (HFOV) were performed to improve oxygenation, extract water retention, and maintain electrolytes and acid–base balance. HFOV was converted to conventional mechanical ventilation (CMV) 2 days later. With the improvement in the chest radiograph (Figure 1C), she was successfully weaned off CMV and extubated after 3 days. After treatment with ECMO for 221 hours, the ECMO was weaned by decreasing the pump flow to 0.5 L/min when left ventricular ejection fraction (LVEF) increased to >45% with acceptable hemodynamic conditions. The total duration of CRRT lasted for 22 days until urine volume and cardiac function returned to normal. The patient was discharged from the hospital on the 35th day after admission when EF improved to 55% with normal chest radiograph, echocardiography results, and hemodynamics (Figure 2). More importantly, there was no neurological complication and cognitive function was normal, with a cerebral performance category score of 1.
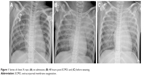  | Figure 1 Series of chest X-rays: (A) on admission; (B) 48 hours post-ECMO; and (C) before weaning. |
  | Figure 2 Timeframe of the case. |
Discussion
We presented the case of a 9-year-old girl with FM, who rapidly worsened into pulseless VT and refractory cardiac arrest. We performed ECMO to establish spontaneous circulation due to persistent circulatory collapse that was refractory to multiple antiarrhythmic therapy and prolonged CPR for nearly 180 min. Fortunately, the girl recovered with intact cardiac and neurocognitive functions after continued ECMO treatment for 221 h. As an effective treatment of mechanical support in adult, ECMO can provide immediate hemodynamic support for patients with refractory heart failure. However, to date, the application of ECMO for children in China is still uncommon due to less available practices. To our knowledge, the girl was the first one with FM to survive a maximal duration of circulatory arrest prior to ECMO initiation as per the available reports. This suggests that prolonged effective CPR and building up ECMO transport system to transfer FM patients to centers that have experience in ECMO may be helpful for those who otherwise would have a small chance of survival through to hospital discharge. With the advantage of easy and rapid set-up in 20–30 min without requiring sternotomy, ECMO is the only suitable mechanical support available for use in CPR situations.7 In recent reports of ECMO support in children with FM, the whole survival rate was 59.6%–67%.8,10 In contrast, the mortality rate was up to 50% without ECMO support.11 So ECMO provides a good chance of myocardial recovery and survival if instituted in time. But to date, the optimal timing of ECMO application has not been established yet. It is still very difficult to decide when to initiate ECMO support in pediatrics. Literature listed arrhythmia, circulation collapse, and end-organ failure as indications for the need of ECMO support.12 Clinically, not all patients with arrhythmia or end-organ failure require prompt ECMO support. ECMO was used in 16.7% and 7.4% of children in Taiwan and US, respectively, due to acute myocarditis.13,14 According to the previous studies,15–17 the following were indications for the ECMO therapy: 1) low blood pressure (systolic blood pressure <60 mmHg for children, and <50 mmHg for infants) despite an inotropic equivalent (IE, mg/kg/min = dopamine + dobutamine +15× milrinone +100 * epinephrine +100 * norepinephrine +100 * isoproterenol), >40 mg/kg/min support, 2) cardiac and pulmonary failure refractory to medical therapy, 3) cardiac arrest requiring continuous CPR, and 4) uncontrolled arrhythmia including VT and ventricular fibrillation. We conclude that children with FM requiring continuous CPR should definitely be considered for ECMO support, just as the case reported above. The worst survival rate is for those who experience cardiac arrest. In two studies where ECMO was utilized for FM only after cardiopulmonary arrest, the rates of survival to hospital discharge significantly decreased to 34.1%18 and 42.5%, respectively.19 A logistic regression analysis revealed that the probability of survival was ~0.5, 0.3, or 0.1 when the duration of CPR was 30, 60, or 90 mins, respectively.18 It suggested that ECMO has the potential to become the first line of treatment of mechanical support for FM, especially complicated with cardiac arrest. To decrease the risks of further deterioration of myocardium in addition to neurological damage and multi-organ failure, prompt diagnosis, intensive care, and backup with ECMO support by a multidisciplinary team are pivotal to survival for pediatric patients with FM.
Conclusion
With the advantage of rapid set-up, ECMO is an effective rescue therapeutic for FM complicated with refractory cardiac arrest, and is suitable for use in CPR situations.
Ethics approval and consent to participate
This study was performed after approval by the Ethics Committee of Guangdong General Hospital and the Ethics Committee of the Third Affiliated Hospital of Sun Yat-sen University. Written informed consent was obtained from the patient’s guardian for the publication of this case report and any accompanying images.
Acknowledgments
The authors would like to acknowledge Dr Yue-Yu Sun and Dr Shu-Shui Wang, who were the main physicians in the team that successfully managed the case reported, for their support and input with regard to the manuscript.
This research was supported by the National Natural Science Foundation of China (grant number 81470219) and Science and Technology Projects Foundation of Guangdong Province (grant number 2014A020212120).
Disclosure
The authors report no conflicts of interest in this work.
References
Feldman AM, McNamara D. Myocarditis. N Engl J Med. 2000;343(19):1388–1398. | ||
Amabile N, Fraisse A, Bouvenot J, Chetaille P, Ovaert C. Outcome of acute fulminant myocarditis in children. Heart. 2006;92(9):1269–1273. | ||
Simpson KE, Canter CE. Acute myocarditis in children. Expert Rev Cardiovasc Ther. 2011;9(6):771–783. | ||
Rady HI, Zekri H. Prevalence of myocarditis in pediatric intensive care unit cases presenting with other system involvement. J Pediatr (Rio J). 2015;91(1):93–97. | ||
Cheng R, Hachamovitch R, Kittleson M, et al. Clinical outcomes in fulminant myocarditis requiring extracorporeal membrane oxygenation: a weighted meta-analysis of 170 patients. J Card Fail. 2014;20(6):400–406. | ||
Asaumi Y, Yasuda S, Morii I, et al. Favourable clinical outcome in patients with cardiogenic shock due to fulminant myocarditis supported by percutaneous extracorporeal membrane oxygenation. Eur Heart J. 2005;26(20):2185–2192. | ||
Chen YS, Yu HY, Huang SC, et al. Experience and result of extracorporeal membrane oxygenation in treating fulminant myocarditis with shock: what mechanical support should be considered first? J Heart Lung Transplant. 2005;24(1):81–87. | ||
Hsu KH, Chi NH, Yu HY, et al. Extracorporeal membranous oxygenation support for acute fulminant myocarditis: analysis of a single center’s experience. Eur J Cardiothorac Surg. 2011;40(3):682–688. | ||
Mirabel M, Luyt CE, Leprince P, et al. Outcomes, long-term quality of life, and psychologic assessment of fulminant myocarditis patients rescued by mechanical circulatory support. Crit Care Med. 2011;39(5):1029–1035. | ||
Wilmot I, Morales DL, Price JF, et al. Effectiveness of mechanical circulatory support in children with acute fulminant and persistent myocarditis. J Card Fail. 2011;17(6):487–494. | ||
Kodama M, Oda H, Okabe M, Aizawa Y, Izumi T. Early and long-term mortality of the clinical subtypes of myocarditis. Jpn Circ J. 2001;65(11):961–964. | ||
Kato S, Morimoto S, Hiramitsu S, et al. Risk factors for patients developing a fulminant course with acute myocarditis. Circ J. 2004;68(8):734–739. | ||
Enteroviruses I, Parvovirus B, Cytomegalovirus A. Predictors of extracorporeal membrane oxygenation support for children with acute myocarditis. Biomed Res Int. 2017;2017:2510695. | ||
Klugman D, Berger JT, Sable CA, He J, Khandelwal SG, Slonim AD. Pediatric patients hospitalized with myocarditis: a multi-institutional analysis. Pediatric Cardiol. 2010;31(2):222–228. | ||
Lee EY, Lee HL, Kim HT, Lee HD, Park JA. Clinical features and short-term outcomes of pediatric acute fulminant myocarditis in a single center. Korean J Pediatr. 2014;57(11):489–495. | ||
Fleming GM, Gurney JG, Donohue JE, Remenapp RT, Annich GM. Mechanical component failures in 28,171 neonatal and pediatric extracorporeal membrane oxygenation courses from 1987 to 2006. Pediatr Crit Care Med. 2009;10(4):439–444. | ||
Gupta P, Robertson MJ, Rettiganti M, et al. Impact of timing of ECMO initiation on outcomes after pediatric heart surgery: a multi-institutional analysis. Pediatr Cardiol. 2016;37(5):971–978. | ||
Chen YS, Yu HY, Huang SC, et al. Extracorporeal membrane oxygenation support can extend the duration of cardiopulmonary resuscitation. Crit Care Med. 2008;36(9):2529–2535. | ||
Chung SY, Sheu JJ, Lin YJ, et al. Outcome of patients with profound cardiogenic shock after cardiopulmonary resuscitation and prompt extracorporeal membrane oxygenation support. A single-center observational study. Circ J. 2012;76(6):1385–1392. |
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
