Back to Journals » Infection and Drug Resistance » Volume 10
Drug resistance in influenza A virus: the epidemiology and management
Authors Hussain M, Galvin HD, Haw TY, Nutsford AN, Husain M
Received 1 March 2017
Accepted for publication 28 March 2017
Published 20 April 2017 Volume 2017:10 Pages 121—134
DOI https://doi.org/10.2147/IDR.S105473
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Suresh Antony
Mazhar Hussain, Henry D Galvin,* Tatt Y Haw,* Ashley N Nutsford,* Matloob Husain
Department of Microbiology and Immunology, University of Otago, Dunedin, New Zealand
*These authors contributed equally to this work
Abstract: Influenza A virus (IAV) is the sole cause of the unpredictable influenza pandemics and deadly zoonotic outbreaks and constitutes at least half of the cause of regular annual influenza epidemics in humans. Two classes of anti-IAV drugs, adamantanes and neuraminidase (NA) inhibitors (NAIs) targeting the viral components M2 ion channel and NA, respectively, have been approved to treat IAV infections. However, IAV rapidly acquired resistance against both classes of drugs by mutating these viral components. The adamantane-resistant IAV has established itself in nature, and a majority of the IAV subtypes, especially the most common H1N1 and H3N2, circulating globally are resistant to adamantanes. Consequently, adamantanes have become practically obsolete as anti-IAV drugs. Similarly, up to 100% of the globally circulating IAV H1N1 subtypes were resistant to oseltamivir, the most commonly used NAI, until 2009. However, the 2009 pandemic IAV H1N1 subtype, which was sensitive to NAIs and has now become one of the dominant seasonal influenza virus strains, has replaced the pre-2009 oseltamivir-resistant H1N1 variants. This review traces the epidemiology of both adamantane- and NAI-resistant IAV subtypes since the approval of these drugs and highlights the susceptibility status of currently circulating IAV subtypes to NAIs. Further, it provides an overview of currently and soon to be available control measures to manage current and emerging drug-resistant IAV. Finally, this review outlines the research directions that should be undertaken to manage the circulation of IAV in intermediate hosts and develop effective and alternative anti-IAV therapies.
Keywords: influenza A virus, drug resistance, M2 ion channel inhibitors, neuraminidase inhibitors, oseltamivir, zanamivir
Introduction
Influenza virus is one of the most successful, persistent, and unpredictable human pathogens. Influenza continues to cause regular seasonal epidemics, unpredictable pandemics, and frequent and deadly zoonotic outbreaks worldwide. Influenza virus transmits through aerosol and causes an acute febrile respiratory disease in humans, commonly known as “flu”, which is particularly severe in young children, elderly, and immunocompromised individuals. The burden of influenza virus on global human population and economy is significant.1–23 According to the World Health Organization (WHO) estimate, influenza virus annually causes 1 billion cases of flu, 3–5 million cases of severe illness, and 300,000 to 500,000 deaths worldwide. The annual influenza vaccination program alternating in the Northern and Southern Hemispheres is the major tool to prevent or control seasonal influenza epidemics. In addition, two classes of antiviral drugs, 1) M2 ion channel inhibitors (amantadine and rimantadine) and 2) neuraminidase (NA) inhibitors (NAIs; oseltamivir, zanamivir, peramivir, and laninamivir), have been approved to treat influenza virus infections. However, like antibiotic resistance, emergence of antiviral drug resistance in influenza virus is a major concern. Consequently, NAIs are the only class of anti-influenza drugs currently in use as most of the circulating influenza viruses have acquired resistance to M2 ion channel inhibitors.24 Nevertheless, many circulating influenza viruses have also acquired the resistance to NAIs. This review highlights the emergence and epidemiology of drug-resistance mutations in M2 and NA with focus on influenza A virus (IAV), the prototypic and most significant member of the Orthomyxoviridae family.
Natural history and epidemiology of IAV
IAV virion particles exhibit both spherical and filamentous morphology and possess the negative-sense, segmented and single-stranded RNA genome. Each of the eight IAV gene segments encodes at least one major viral protein. However, some IAV segments encode more than one viral protein through mechanisms including leaky ribosomal scanning, alternative splicing, ribosomal frameshifting, and use of alternative start codon.25,26 So far, IAV has been reported to encode at least 17 viral proteins, although not all IAV subtypes encode every protein. IAV is an enveloped virus, and each virion contains ~300 hemagglutinin (HA) and ~40 NA glycoprotein spikes on the surface.27,28 The HA is the receptor-binding protein and facilitates IAV entry to host cell, whereas NA facilitates the release of newly produced virions from the host cell.28 A third protein, M2 that forms an ion channel and is critically involved in virus entry, is also embedded in the viral envelope, which is derived from the host cell plasma membrane.28 Underneath the envelope is a rigid layer comprised of matrix protein 1 (M1), which maintains the shape and integrity of IAV virion.29 M1 also interacts with the cytoplasmic domains of IAV envelope proteins and viral ribonucleoprotein (vRNP) core. The vRNP core is primarily composed of viral genome, nucleoprotein (NP), and polymerase complex, which consists of PA, PB1, and PB2 proteins.28
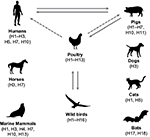
IAV has global presence and a broad host range that includes humans, seals, horses, pigs, dogs, cats, and birds (Figure 1). The aquatic birds, such as waterfowl and shorebirds, are the reservoir host of IAV.30 IAV is subtyped based on the type and antigenicity of its surface glycoproteins, HA and NA. So far, 18 HA and 11 NA subtypes have been described, of which 16 HA and 9 NA have been found to circulate in avian species, whereas 2 HA and 2 NA subtypes have been detected in bats (Figure 1).31,32 However, the bat IAV subtypes, H17N10 and H18N11, are remarkably different from other IAV subtypes prompting suggestion that these bat viruses should be labeled as influenza-like viruses.33
The interspecies transmission of IAV occurs and is common as well as significant between humans and pigs and poultry and pigs, while it is sporadic in others (Figure 1). The ability of IAV to transmit between species is determined by its capability to change specificity to target species. IAV is also well adapted to promote antigenic diversity by using two particular mechanisms known as antigenic drift and antigenic shift.34 Antigenic drift causes mutations in HA and NA resulting in antigenic variants, which can reinfect a host and avoid the pre-existing immunity.34 The error-prone nature of viral RNA polymerase is the major contributor to antigenic drift, which along with frequent reassortment and natural selection is the main cause of recurring seasonal influenza epidemics.35 These epidemics are capable of lasting at least 6 to 12 weeks, with observed infection rates of 10–30% in adults and 20–50% in children.36 On the other hand, antigenic shift is the reassortment of gene segments between two different parental viruses within the same host, giving rise to a novel pandemic IAV. The H1N1 subtype, which caused the first recorded IAV pandemic in 1918, was originated from the reassortment between a human H1 subtype and an avian N1 subtype.37 The next IAV pandemic of 1957 was caused by an H2N2 subtype, which originated when circulating 1918 H1N1 subtype reassorted with an avian H2N2 subtype.38 Subsequent IAV pandemic in 1968 was caused by the H3N2 subtype. This subtype arose when circulating 1957 H2N2 subtype reassorted with an avian H3 subtype.38 The most recent IAV pandemic in 2009 was caused by a swine-origin H1N1 subtype, which originated from the sequential reassortment events between human H3N2, swine H1N1 and avian H1N2 subtypes of North American and Eurasian lineages.38 A pandemic IAV has the potential to spread quickly and infect ~50% of the global human population within a short period of time. Therefore, all four pandemics combined resulted in the deaths of millions of people worldwide. Lately, several pure avian-origin IAV subtypes (e.g., H5N1, H7N9, and H10N8) have been found to cause deadly outbreaks in humans. Many of these IAV subtypes cause a disease that is clinically distinct and more severe than the disease caused by a human IAV subtype and results in a significantly high mortality rate (~35–50%).39 Fortunately, none of these avian IAV subtypes have acquired the capability of direct human-to-human transmission by aerosol; they mainly spread through a direct contact. The spread of avian IAV to humans is limited by differing receptor specificities.40 IAV utilizes carbohydrate moiety, sialic acids present on host cell surface as its receptor. The HA of human IAV subtypes specifically binds to sialic acids with α-2,6-linkages, whereas the HA of avian IAV subtypes binds to sialic acids with α-2,3-linkages.40 The upper respiratory tract in humans predominantly contains sialic acid with α-2,6-linkages. However, sialic acids with α-2,3-linkages are found in the lower respiratory tract of humans, meaning that avian IAV subtypes are capable of infecting humans upon exposure. Interestingly, both α-2,6- and α-2,3-linkages are found in swine upper respiratory tract; therefore, pigs are regarded as the “mixing vessels” for the generation of human and avian IAV reassortant subtypes.41
Antiviral drugs and drug resistance in IAV
Adamantanes: M2 ion channel inhibitors
The adamantanes, amantadine (Symmetrel), and rimantadine (Flumadine) were the first-approved class of anti-IAV drugs.42 Adamantanes target the M2 protein of IAV to exert their antiviral function.43,44 The M2 is a tetrameric, type III integral membrane protein, and the single transmembrane domain of M2 forms a 4-helix bundle that acts as a pH-sensitive gated ion channel in the IAV envelope.45–49 IAV enters the host cell via receptor-mediated endocytosis and uncoats through HA-mediated fusion of viral envelope with endosomal membrane.28 Prior to the fusion, the low pH in endosome activates and opens the M2 ion channel allowing the entry of protons and causing the internal acidification of virion. This event leads to the dissociation of M1 protein from vRNP core and subsequent release of the latter into host cell cytoplasm, initiating the IAV replication.50 The adamantanes bind to M2 channel pore and block proton conductance either directly or allosterically, consequently inhibiting the vRNP release and IAV replication.51–54 To acquire resistance to adamantanes, IAV mutated several amino acids (L26F, V27A, A30T/V, S31N, G34E, and L38F) in M2 transmembrane domain that line the channel pore (V27, A30, and G34) or are involved in the tetramer helix–helix packing (L26, S31, and L38), leading to increase in pore size and hydrophilicity of the channel or destabilization of helix–helix assembly and narrowing of the pore size, respectively.51–53,55–58
Epidemiology of adamantane-resistant IAV
Amantadine was first of the two adamantanes to be approved for clinical use in 1966 followed by rimantadine in 1993.43,56,59 Initially, both drugs were highly successful in inhibiting and preventing the IAV infection with an efficacy rate of up to 90%.60–62 The resistance of IAV to adamantanes was first detected during 1980 epidemic.63 However, the resistance to both drugs in seasonal IAV subtypes was rare with only 1–2% frequency until 200059,64–66 but has risen dramatically since then.67 From 2000 to 2004, the resistance to adamantane among IAV H3N2 subtype isolates across Asia increased from 1.1% to 27%.67 The numbers from China (73.8%), Hong Kong (69.6%), and Taiwan (22.7%) were the main contributors to this 27% increase in Asia.67 However, during the same period, the increase in H3N2 subtypes resistant to adamantanes in Europe, North America, and South America was only 4.7%, 3.9%, and 4.3%, respectively, whereas no resistant virus was detected in the Oceania region. Nevertheless, during 2000–2004, the overall resistance to adamantanes among H3N2 subtypes rose to 12.3% globally.67 In majority of the H3N2 isolates (98·2%), the adamantane resistance was due to the S31N mutation while the L26F, V27A, and A30T mutations accounted for the rest (1.8%).67 On the other hand, only 0.3% H1N1 subtypes were resistant to adamantanes during this period. Similarly, during 2004–2005 season, only 15% of the H3N2 and 4.1% of the H1N1 global isolates were resistant to adamantanes.68 However, from 2005 onward, the resistance to adamantanes started to increase almost exponentially. Remarkably, during 2005–2006 season, 90.6% of the H3N2 and 15.6% of the H1N1 global isolates were adamantane resistant.68 The Asia, Europe, North and South America, and Oceania regions contributed almost equally to these statistics, although no significant data were available from Africa. Astonishingly, 100% of the H3N2 isolates from South Korea, Taiwan, Japan, Hong Kong, and China were resistant to adamantanes.68 Similarly, in the USA, up to 96.4% of the H3N2 isolates and up to 25% of the H1N1 isolates were adamantane resistant.68,69 These data prompted the Centers for Disease Control and Prevention (CDC), USA, to issue an advisory against the use of adamantanes to treat IAV infections.70 Again, in 90–98% of the isolates, the resistance-conferring mutation was S31N in both H1N1 and H3N2 subtypes. Similarly, the swine-origin IAV H1N1 subtype that caused the pandemic in 2009 also contained the S31N mutation in M2, hence, was resistant to adamantanes.71,72 Furthermore, the avian IAV subtypes H5N1 and H7N9 that emerged and caused severe zoonotic infections in humans in 2003 and 2013, respectively, also possess the S31N mutation in M2, hence, were resistant to adamantanes.39,73
As of 2013, ~45% of all IAV subtypes circulating in the world were resistant to adamantanes.74 Particularly, over 69% of the H1, 43% of the H3, 28% of the H5, 12% of the H7, and 23% of the H9 subtypes carried adamantane resistance-conferring mutations in M2. However, the adamantane-resistant mutations were rare in H2, H4, H6, H10, and H11 subtypes, and no such mutations were identified in H8 and H12–16 subtypes. Interestingly, all three bat-origin H17N10 isolates were also adamantane resistant. The adamantane-resistant H1 and H3 subtypes were mainly found in humans and swine and were widely distributed in the world. However, most of the adamantane-resistant H1 and H3 subtypes were detected in the Americas (52% and 56%), followed by Asia (26% and 34%), Europe (19% and 4.6%), Oceania (1.8% and 1.8%), and Africa (0.7% and 2.5%). In the Americas, USA had the largest distribution of adamantane-resistant H1 and H3 subtypes, whereas China, Singapore, and Hong Kong led the numbers in Asia and UK followed by Spain in Europe. In contrast, majority of the adamantane-resistant H4–H11 subtypes were detected in avian species and were mainly distributed in Asia. Particularly, the distribution of adamantane-resistant H5, H6, H7, and H9 subtypes in Asia was at 91%, 100%, 67%, and 100%, respectively. Vietnam had the highest number of adamantane-resistant H5 subtypes followed by Thailand, China, and Indonesia. On the other hand, China had the largest distribution of adamantane-resistant H7 and H9 subtypes followed by Hong Kong. The next largest distribution of the adamantane-resistant H7 (31.3%) and H5 (6.4%) subtypes was found in Americas (exclusively USA) and Africa (primarily Egypt), respectively.74
A vast majority of adamantane-resistant IAV subtypes (95%) contained the S31N mutation.74,75 Particularly, over 96% of the H1, 93% of the H3, 83% of the H5, 86% of the H7, and 87% of the H9 subtypes harbored the S31N mutation. Other mutations (L26F, V27A, A30T/V, G34E, and L38F) were sporadic, but over 11.8% of the H5 and 9.8% of the H9 subtypes contained V27A mutation. In addition, 2.8% of the H1, 5.4% of the H3, 2.6% of the H5, and 10.4% of the H7 viruses possessed the double mutation V27A+S31N and few of the H1, H3, and H5 viruses had the double mutation L26F+S31N. Furthermore, S31N was the most common mutation in human (98%), avian (88%), and swine (77%) IAV subtypes distantly followed by V27A in the same species. Interestingly, over 20% of the swine IAV subtypes had the double mutation V27A+S31N, frequency of which is believed to be rising in swine IAV.76 Predictably, S31N mutation was distributed worldwide with majority of the distribution in Americas followed by Asia, Europe, Africa, and Oceania. However, V27A, A30T and L26F mutations were more prevalent in Asia, with majority V27A distribution in Indonesia followed by China. The most common double mutation was V27A+S31N with major distribution in the Americas (mainly USA) followed by Europe and Asia. The next most common double mutation was L26F+S31N with similar geographical distribution.74
Temporally, the resistance to adamantanes in IAV subtypes H1 and H3 increased consistently in many countries from 2001 and spiked in 2009. Particularly, in Hong Kong, UK, and Germany, the frequency of adamantane-resistant H1N1 subtypes has increased consistently since 2001, and in China and USA since 2005 and 2006, respectively. Similarly, China, Hong Kong, Taiwan, and USA had a consistent increase in adamantane-resistant H3N2 subtypes since 2000, and Vietnam, Taiwan, and Malaysia since 2004.74 The most likely reason for this dramatic increase in IAV resistance to adamantanes is the widespread usage of these drugs to treat IAV infections. However, increase in resistance has also been noted in countries where adamantanes use was low, indicating the stable circulation of the fit mutant viruses, which may have emerged without drug pressure and have replication potential and virulence similar to the wild type.59,77–80 It has been proposed that S31N mutation independently emerged multiple times and introduced at least 11 times in H3N2 subtypes during 1997–2007. It was the seventh introduction in H3N2 during 2003 in Hong Kong that subsequently reassorted with another H3N2 subtype in 2005 to acquire a novel HA and gave rise to the “N-lineage”.75,79,81 The N-lineage then acquired other fitness-enhancing mutations elsewhere in the genome and spread globally; hence, all currently circulating adamantane-resistant H3N2 subtypes belong to a single lineage.79 The emergence of adamantane resistance in H1N1 subtypes was delayed and temporally different to H3N2 subtypes.68,69,74 It is believed that adamantane resistance in H1N1 subtypes did not arise through reassortment with a resistant H3N2 subtype, but through a separate introduction and spread process.68,82
NAIs
NAIs were the second approved and are the only currently used class of anti-influenza drugs. As the name suggests, NAIs target the IAV surface protein NA to exert their antiviral function.83 NA possesses the sialidase enzyme activity that cleaves the cell surface sialic acid to which the newly formed IAV progeny is attached.28,84,85 This action releases the IAV progeny from infected cells that go on to infect naive cells and spread the infection. In addition, NA sialidase activity also facilitates the movement of IAV particles through sialic acid-rich human respiratory tract. The concept of an inhibitor of the NA sialidase activity as an antiviral agent was envisaged as early as 1948, not long after the discovery of a receptor-destroying activity on influenza virus surface.83 The first such inhibitors were tested during 1966–1976 but exhibited low specificity and potency.83,86–88 However, the determination of the three-dimensional crystal structure and catalytic sites of NA led to the rational design of first two potent NAIs, now known as zanamivir (Relenza) and oseltamivir (Tamiflu).89–92 The NA is a mushroom-shaped, tetrameric glycoprotein, which is anchored in IAV envelope with exposed globular head.83 The enzyme active site is situated in the head and consists of 8 functional amino acids (R118, D151, R152, R224, E276, R292, R371, and Y406 – N2 numbering throughout) and 11 structural amino acids (E119, R156, W178, S179, D/N198, I222, E227, H274, E277, D293, and E425) that are conserved in almost all IAV subtypes. The functional amino acids form the catalytic core and directly contact the sialic acid, whereas the structural amino acids form the active site framework.93,94 The NAIs are sialic acid or transition state structural analogs that compete with cell surface sialic acid–viral NA interactions and inhibit the enzymatic reaction and release of the newly formed IAV progeny, consequently inhibiting the spread of the IAV and further infection.83,91 However, similar to M2, IAV has mutated several amino acids, notably E119V, I222V, H274Y, R292K, and N294S, in or around NA active site to acquire the resistance to NAIs.95,96 Many of these mutations alter the architecture of NA active site and reduce the binding of NAIs by many fold.95,97–103
Epidemiology of NAI-resistant IAV
Initial clinical studies showed that confirmed cases of IAV infection can be treated with both zanamivir and oseltamivir, and if administered within 36 to 48 h of the onset of clinical symptoms, both drugs reduced the duration of illness by up to 3 days in all ages.104–106 The prompt initiation of treatment after the onset of clinical symptoms was key in reducing the duration of illness proportionally.107 Accordingly, both zanamivir and oseltamivir also prevented the IAV infection by 70–90% when used as a prophylaxis before or after the exposure to close contacts infected with IAV.104,106 In July 1999, zanamivir was the first NAI approved for the prophylaxis and treatment of IAV infection in humans followed by oseltamivir in October 1999.108 Two related NAIs, peramivir109,110 and laninamivir,111–113 have also been recently approved in multiple countries and Japan, respectively.
Due to the experience with rapid emergence of adamantane-resistant IAV, several in vitro and preclinical studies were performed to select and isolate NAI-resistant IAV. Only few drug-resistant IAV mutants were isolated from in vitro passage cultures as well as patients treated with both zanamivir and oseltamivir.114 These mutants predominantly had two mutations, E119G/A/D/V and R292K in NA.114 In addition, two resistant mutants containing the H274Y mutation in NA were also isolated from healthy volunteers experimentally infected with IAV H1N1 subtype and subsequently treated with oseltamivir.115 Therefore, to monitor IAV resistance to NAIs, a global Neuraminidase Inhibitor Susceptibility Network (NISN) was established in 1999.116 The main objectives of NISN were to set the guidelines to test sensitivity of NAIs and monitor the susceptibility and resistance to NAIs in clinical influenza virus isolates collected from various parts of the world. In their first study published in 2003, NISN concluded that all clinical IAV isolates collected during 1996–1999 were susceptible to NAIs.117,118 Furthermore, during first 3 years (1999–2002) of NAI usage, basically no resistant IAV was detected, except few isolates from untreated individuals that exhibited reduced susceptibility to oseltamivir,118,119 and a disproportionally high rate (18–27%) of oseltamivir resistance observed in both H1N1 and H3N2 isolates from children.120,121 Similarly, no resistance was detected during 2004–2005 season.122 In the following 2005–2006 and 2006–2007 seasons, the frequency of oseltamivir resistance in global H1N1 isolates was only 0.4% and 0.6%, respectively.123 Particularly in USA, no resistance was detected during 2005–2006 season, but during 2006–2007 season, it was detected to be ~0.9%. However, 2007–2008 season had a significant 7% increase in oseltamivir resistance in global H1N1 isolates, but no resistant H3N2 isolates were detected, and all 2007–2008 oseltamivir-resistant H1N1 isolates were sensitive to zanamivir.122 Nevertheless, 2.3% of the H1N1 isolates circulating between 2006 and early 2008 in Australasia and Southeast Asia exhibited resistance to zanamivir.124 During 2007–2008, several European countries also had ~20% increase in oseltamivir-resistant H1N1 subtypes.125–127 Particularly, Norway witnessed an unprecedented 65% increase in the circulation of oseltamivir-resistant H1N1 subtypes followed by France (46%), Belgium (37%), and rest of the Europe. According to the WHO, by mid-2008, 15% of the H1N1 isolates circulating worldwide were oseltamivir resistant. Remarkably, in 2008–2009 season, more than 90% of the circulating H1N1 subtypes globally were oseltamivir resistant.128,129 According to CDC (USA), until April 2009 (just before the emergence of 2009 pandemic H1N1 subtype), over 99% of the H1N1 isolates were resistant to oseltamivir; however, all were sensitive to zanamivir and none of the H3N2 isolates were resistant to oseltamivir. Similarly, during 2008–2009 season, 100% of the H1N1 isolates from Japan were oseltamivir resistant.130,131 Further, 86%, 32%, and 100% of the H1N1 subtypes isolated in Oceania, South East Asia, and South Africa, respectively, were oseltamivir resistant.132
In April 2009, a novel swine-origin IAV H1N1 subtype emerged and caused the first IAV pandemic of 21st century. Fortunately, the 2009 pandemic H1N1 subtype was sensitive to NAIs.71 Therefore, the only positive outcome of 2009 pandemic was that in the following seasons, the circulating prepandemic oseltamivir-resistant H1N1 subtype was replaced by the oseltamivir-sensitive pandemic H1N1 subtype.133 Almost all of the pandemic H1N1 global isolates collected between April 2009 and January 2010 were sensitive to NAIs, except an odd 0.7% and other few H1N1 subtypes isolated from local cases that were resistant to oseltamivir.129,133–137 In April 2011, only 1.6% of the global pandemic H1N1 isolates were oseltamivir resistant.138 Furthermore, only 3.2% of the pandemic H1N1 isolates collected between 2009 and 2012 from Asia, Africa, and Oceania were resistant to peramivir, but none to laninamivir.139 Therefore, more than 98% of the pandemic H1N1 and seasonal H3N2 isolates collected globally till May 2015 remain largely sensitive to all NAIs.140–142 Nevertheless, a high transmission (15–29%) of oseltamivir-resistant pandemic H1N1 virus in some local communities141,143–146 and the rapid emergence and transmission of resistant viruses in immunocompromised patients have been detected.147–151
Unlike adamantanes resistance, which initially emerged and was more widespread in IAV H3N2 subtypes, the NAI resistance first emerged and was predominant in H1N1 subtypes.75,152 Majority of avian IAV H5N1 subtypes (except some isolates from Indonesia and Vietnam that exhibit reduced sensitivity to oseltamivir) and H7N9 subtypes circulating in nature are largely susceptible to NAIs.39,153 Further, the resistance in circulating IAV was mainly against oseltamivir and peramivir, and not zanamivir, and it is primarily limited to human IAV of N1 subtype.75 The most common oseltamivir (and peramivir) resistance-conferring mutation in NA of the H1N1 and H5N1 subtypes was H274Y, whereas in H3N2 and H7N9 subtypes, the E119V and R292K mutations were more common (Table 1).122,125,127,130–133,138,141,142 Further, few of the H1N1 and H3N2 isolates carrying single mutations I222K/T/R, N142S, D198E, S246G/N, N294S, and G320E and double mutations T156I+D213G, I222T+S331R, I222R/V+H274Y, and S246N+H2724Y in their NA also exhibited reduced sensitivity to oseltamivir and peramivir and seldom to laninamivir (Table 1).140–142 In addition, the H1N1 subtypes with H274Y mutation also acquired secondary permissive mutations R193G, R221Q, V233M, V240I, D343N, D353G, and N368K in NA and T82K, K141E, R189K, and A193T in HA to improve their fitness.130,154–159 On the other hand, the resistance or reduced sensitivity to zanamivir in H1N1 and H3N2 isolates was conferred by the Q136K mutation in NA (Table 1).124,140,142,160
  | Table 1 Susceptibility of IAV subtypes with naturally acquired NA mutations to NAIs Notes: Bold indicates resistance; pH1N1, 2009 pandemic H1N1. #As per the WHO GISRS guidelines.140–142 Abbreviations: IAV, influenza A virus; NA, neuraminidase; NAI, neuraminidase inhibitor; NI, normal inhibition; RI, reduced inhibition; NT, not tested; HRI, highly reduced inhibition; GISRS, Global Influenza Surveillance and Response System. |
Although use of NAIs is high in some countries such as Japan and USA, the emergence of oseltamivir-resistant IAV is largely attributed to the spread of a fit H274Y mutant, both pre- and post-pandemic period of 2009. Except in Japan (where the use of oseltamivir dramatically increased in 2001–2002 and was at its peak during 2008–2009) and USA, little or no oseltamivir was in use in Europe, Australia, New Zealand, and Southeast Asian countries where H274Y mutant was also pre-dominantly circulating during 2008–2009.119,127,130,132 Furthermore, rapid transmission of 2009 pandemic H1N1 (H274Y) mutant has been detected in communities with little or no previous exposure to oseltamivir.144,145 The pre-pandemic oseltamivir-resistant IAV H1N1 lineage (A/Brisbane/59/2007), first detected in Europe, emerged without drug pressure and subsequently acquired permissive mutations in NA and spread globally.127,130–132,152,157,161 The NAI-sensitive 2009 pandemic H1N1 subtype displaced the pre-pandemic oseltamivir-resistant H1N1 lineage and remains largely NAI sensitive and is predominantly circulating at present. However, some oseltamivir- and peramivir-resistant 2009 H1N1 subtypes found circulating in local community clusters acquired two permissive NA mutations V240I and N368K, which improved their fitness.158,162 Concernedly, almost all currently circulating oseltamivir-sensitive 2009 H1N1 subtypes possess the V240I and N368K substitutions in NA.141 Therefore, similar to pre-pandemic H1N1 variants, a potential exists for the fit oseltamivir-resistant 2009 H1N1 variants to emerge and spread globally in the future.
It is somewhat intriguing that only some IAV subtypes (predominantly H1, H3, and H5) primarily circulating in birds, humans, and pigs – the three most important IAV hosts, have been reported to acquire the adamantane- or NAI-resistant mutations. This could be explained partly by the circulation and transmission frequency as well as epidemiological fitness of these subtypes in above hosts. One theory is that the acquisition of drug resistant, but potentially fitness-compromising mutations by these IAV subtypes under drug pressure in one host (humans) helps them acquire the fitness-enhancing permissive or epistatic mutations when transmitted to the other host (birds or pigs) under no drug pressure and a favorable immune environment. Consequently, such drug-resistant variants then become epidemiologically fit and prevail in multiple host populations.
Management of drug-resistant IAV
The trio – vaccines, antiviral drugs, and surveillance – are key to control and eradicate viral pathogens, and the same is true for IAV.
Vaccines
The vaccines are at the forefront to prevent and manage the spread of drug-resistant IAV. Although a universal influenza vaccine is yet to be developed due to highly variable nature of its surface antigens HA and NA, vaccines specific to individual influenza viruses are available and have come a long way in terms of their composition and administration since first introduction in 1940s.163 Now, intramuscularly, intradermally, and intranasally administered subunit and live-attenuated vaccines are available against seasonal IAV as well as zoonotic IAV such as H5N1 and H7N9. Based on a worldwide surveillance program in both Northern and Southern hemispheres spearheaded by the WHO, seasonal IAV vaccines are annually reformulated, manufactured, and delivered for administration to global human population just before the start of flu season. Numerous individual and meta-analysis studies have found the seasonal IAV vaccines to be effective, albeit modestly in some instances, for both healthy and at-risk population.164–167 Nevertheless, influenza vaccine effectiveness is an important issue, and efforts are being made continuously to improve the efficacy of current vaccines and develop next-generation influenza vaccines. These include new vaccine formulations, development of universal influenza vaccine targeting the HA stalk domain, M2 ectodomain or inducing T-cell response, DNA vaccine against different influenza antigens, recombinant HA vaccine using baculovirus expression system (e.g., FluBlock) and other viral vectors (e.g., adenovirus and poxvirus), and influenza virus-like particles as vaccine.168–170 Furthermore, cell culture-based platforms are being used to prepare vaccines (e.g., Optaflu, Flucelvax, Preflucel, and Celvapan) to circumvent the issues faced with egg-based virus culture and vaccine delivery timeframe, a critical factor in the event of a pandemic or zoonotic outbreak.168
Antiviral drugs
In the absence of a universal influenza vaccine, antiviral drugs become the first line of defense against a pandemic and zoonotic IAV. The adamantane-resistant IAV with S31N mutation appears to have established itself in nature and it is unlikely that this mutation will ever be lost; hence, adamantanes have become practically obsolete as anti-IAV drugs. Luckily, majority of currently circulating IAV H1N1 and H3N2 subtypes are sensitive to NAIs, and primarily human IAV H1N1 subtypes have acquired the resistance to oseltamivir. Therefore, NAIs are still effective in treating the infections with adamantane-resistant IAV and newly emerging IAV, provided they are administered at least within 48 hours of the appearance of clinical symptoms. Furthermore, majority of the oseltamivir- and peramivir-resistant IAV are still largely sensitive to zanamivir and laninamivir (Table 1). Nevertheless, the emergence and global spread of the fit IAV variants resistant to all NAIs is a concern. Therefore, a variety of new antiviral agents targeting either existing targets M2 and NA or other IAV components HA, NP, NS1, and polymerase complex are being developed, and several of them have shown promising results in clinical trials. Particularly, a small-molecule inhibitor of HA, nitazoxanide, and polymerase inhibitors, VX-787 and S-033188 are undergoing Phase 3 and Phase 2 clinical trials, respectively. One of the influenza polymerase inhibitors, Favipiravir (T-705) has already been approved in Japan and is undergoing Phase 3 clinical trial in USA and Europe. Several other polymerase inhibitors (L-742001, Compounds “1”, “7”, and “367”, ASN2, ANA1) and NP inhibitors (Curcumin, Naproxen, Nucleozin, RK424) are at the experimental stages. In addition, therapeutic monoclonal antibodies are also being developed against IAV. Some of them (e.g., CR6261, CR8020, MHAA4549A, and VIS410) targeting the HA are in Phase 2 clinical trials.24,171–173 Furthermore, host factors that are involved in IAV replication and pathogenesis are also being explored as targets to develop anti-IAV strategies.24,172 Finally, viral RNA is also being targeted by antisense and short-interfering RNAs to develop the alternative anti-IAV therapeutics; however, this approach would have its own challenges and it will be interesting to know whether and how IAV develops resistance to it. Once more than one class of anti-IAV drugs is available, a combination therapy involving different classes of drugs could be more effective and beneficial in reducing the emergence of antiviral resistance in IAV.
Surveillance
IAV transmission in humans via aerosol and intercontinental spread by migratory birds makes surveillance a crucial player in the global management of IAV.174,175 The WHO Global Influenza Surveillance and Response System (GISRS) through its collaborating centers and reference laboratories in 113 member states conducts influenza virus surveillance and provides recommendations regarding the laboratory diagnosis, vaccines, antiviral susceptibility, and risk assessment. In addition, GISRS also provides global alerts on the emergence of novel influenza viruses. Therefore, GISRS serves as a single and timely source on worldwide status and management of the influenza virus, including drug-resistant IAV. In addition, CDC (USA) also puts out timely updates and advisories on influenza virus.
Future research directions
The broad host range of IAV and interspecies transmission are critical factors for its continuous circulation and evolution in nature (Figure 1).175 The intermediate hosts such as pigs, birds, and horses play a crucial role in maintaining the IAV in nature and its transfer to humans (Figure 1). Therefore, in addition to continuous surveillance and developing a universal vaccine and effective antivirals, an effective management of such hosts to restrict the circulation and generation of new and more virulent IAV variants is needed. Eradication of IAV from its zoonotic hosts using existing knowledge and approaches is practically impossible. There is a need to gain molecular and genetic understanding of why some animals (e.g., sheep and rabbits) are resistant to IAV infection and some (e.g., ducks) are resistant to IAV disease and identify the genes that confer such resistance. Then, by inserting those genes or using gene-editing methods (e.g., RNA interference and CRISPR-Cas), the intermediate hosts could potentially be made resistant to IAV infection. This will reduce and potentially eliminate IAV from the intermediate hosts and consequently its maintenance in nature and transfer to humans. Initial proof-of-concept studies in this direction have already begun to restrict IAV transmission or replication in transgenic animals.176–179
The current understanding of IAV biology in a host is mainly acquired using cell culture or animal models, because it is not possible to experimentally infect humans. Due to lack of the insight into IAV infection dynamics in human respiratory tract, many antiviral drugs fail to advance beyond experimental and clinical trial stage. Therefore, large animals such as pigs may be engineered to exhibit respiratory tract physiology akin to humans to make advances in this area. Finally, the relationship between the microbiota of healthy and at-risk humans and IAV pathogenesis needs to be understood to develop alternative treatments such as probiotics to treat IAV infections.
Acknowledgments
Matloob Husain has been funded by the Lottery Health Research, HS and JC Anderson Charitable Trust, and School of Biomedical Sciences (University of Otago). Mazhar Hussain, Henry D Galvin and Tatt Y Haw have been supported by the University of Otago Doctoral Scholarship.
Disclosure
The authors report no conflicts of interest in this work.
References
Peasah SK, Azziz-Baumgartner E, Breese J, Meltzer MI, Widdowson MA. Influenza cost and cost-effectiveness studies globally – a review. Vaccine. 2013;31(46):5339–5348. | ||
Cheng PY, Palekar R, Azziz-Baumgartner E, et al. Burden of influenza-associated deaths in the Americas, 2002-2008. Influenza Other Respir Viruses. 2015;9(suppl 1):13–21. | ||
Kaczmarek MC, Ware RS, Coulthard MG, McEniery J, Lambert SB. Epidemiology of Australian influenza-related paediatric intensive care unit admissions, 1997-2013. PLoS One. 2016;11(3):e0152305. | ||
Viboud C, Simonsen L, Fuentes R, Flores J, Miller MA, Chowell G. Global mortality impact of the 1957-1959 influenza pandemic. J Infect Dis. 2016;213(5):738–745. | ||
Cooper BS, Kotirum S, Kulpeng W, et al. Mortality attributable to seasonal influenza A and B infections in Thailand, 2005-2009: a longitudinal study. Am J Epidemiol. 2015;181(11):898–907. | ||
Wu P, Goldstein E, Ho LM, et al. Excess mortality associated with influenza A and B virus in Hong Kong, 1998-2009. J Infect Dis. 2012;206(12):1862–1871. | ||
Lee VJ, Yap J, Ong JB, et al. Influenza excess mortality from 1950-2000 in tropical Singapore. PLoS One. 2009;4(12):e8096. | ||
Huo X, Chen LL, Hong L, et al. Economic burden and its associated factors of hospitalized patients infected with A (H7N9) virus: a retrospective study in Eastern China, 2013-2014. Infect Dis Poverty. 2016;5(1):79. | ||
Haas J, Braun S, Wutzler P. Burden of influenza in Germany: a retrospective claims database analysis for the influenza season 2012/2013. Eur J Health Econ. 2016;17(6):669–679. | ||
Kessaram T, Stanley J, Baker MG. Estimating influenza-associated mortality in New Zealand from 1990 to 2008. Influenza Other Respir Viruses. 2015;9(1):14–19. | ||
Khieu TQ, Pierse N, Telfar-Barnard LF, Huang QS, Baker MG. Estimating the contribution of influenza to hospitalisations in New Zealand from 1994 to 2008. Vaccine. 2015;33(33):4087–4092. | ||
Kyeyagalire R, Tempia S, Cohen AL, et al. Hospitalizations associated with influenza and respiratory syncytial virus among patients attending a network of private hospitals in South Africa, 2007-2012. BMC Infect Dis. 2014;14:694. | ||
Hirve S, Krishnan A, Dawood FS, et al. Incidence of influenza-associated hospitalization in rural communities in western and northern India, 2010-2012: a multi-site population-based study. J Infect. 2015;70(2):160–170. | ||
Jules A, Grijalva CG, Zhu Y, et al. Influenza-related hospitalization and ED visits in children less than 5 years: 2000-2011. Pediatrics. 2015;135(1):e66–e74. | ||
Yu H, Huang J, Huai Y, et al. The substantial hospitalization burden of influenza in central China: surveillance for severe, acute respiratory infection, and influenza viruses, 2010-2012. Influenza Other Respir Viruses. 2014;8(1):53–65. | ||
Savy V, Ciapponi A, Bardach A, et al. Burden of influenza in Latin America and the Caribbean: a systematic review and meta-analysis. Influenza Other Respir Viruses. 2013;7(6):1017–1032. | ||
Wilson N, Nhung N, Higgins A, Kvizhinadze G, Baker MG, Blakely T. A national estimate of the hospitalisation costs for the influenza (H1N1) pandemic in 2009. N Z Med J. 2012;125(1365):16–20. | ||
Chadha MS, Hirve S, Dawood FS, et al. Burden of seasonal and pandemic influenza-associated hospitalization during and after 2009 A(H1N1)pdm09 pandemic in a rural community in India. PLoS One. 2013;8(5):e55918. | ||
Nair H, Brooks WA, Katz M, et al. Global burden of respiratory infections due to seasonal influenza in young children: a systematic review and meta-analysis. Lancet. 2011;378(9807):1917–1930. | ||
Young-Xu Y, van Aalst R, Russo E, Lee JK, Chit A. The annual burden of seasonal influenza in the US veterans affairs population. PLoS One. 2017;12(1):e0169344. | ||
Zhou H, Thompson WW, Viboud CG, et al. Hospitalizations associated with influenza and respiratory syncytial virus in the United States, 1993-2008. Clin Infect Dis. 2012;54(10):1427–1436. | ||
Chiu SS, Chan KH, So LY, Chen R, Chan EL, Peiris JS. The population based socioeconomic burden of pediatric influenza-associated hospitalization in Hong Kong. Vaccine. 2012;30(10):1895–1900. | ||
Gasparini R, Amicizia D, Lai PL, Panatto D. Clinical and socioeconomic impact of seasonal and pandemic influenza in adults and the elderly. Hum Vaccin Immunother. 2012;8(1):21–28. | ||
Webster RG, Govorkova EA. Continuing challenges in influenza. Ann N Y Acad Sci. 2014;1323:115–139. | ||
Vasin AV, Temkina OA, Egorov VV, Klotchenko SA, Plotnikova MA, Kiselev OI. Molecular mechanisms enhancing the proteome of influenza A viruses: an overview of recently discovered proteins. Virus Res. 2014;185:53–63. | ||
Yamayoshi S, Watanabe M, Goto H, Kawaoka Y. Identification of a novel viral protein expressed from the PB2 segment of influenza A virus. J Virol. 2015;90(1):444–456. | ||
Harris A, Cardone G, Winkler DC, et al. Influenza virus pleiomorphy characterized by cryoelectron tomography. Proc Natl Acad Sci U S A. 2006;103(50):19123–19127. | ||
Nayak DP, Balogun RA, Yamada H, Zhou ZH, Barman S. Influenza virus morphogenesis and budding. Virus Res. 2009;143(2):147–161. | ||
Shtykova EV, Baratova LA, Fedorova NV, et al. Structural analysis of influenza A virus matrix protein M1 and its self-assemblies at low pH. PLoS One. 2013;8(12):e82431. | ||
Webster RG, Bean WJ, Gorman OT, Chambers TM, Kawaoka Y. Evolution and ecology of influenza A viruses. Microbiol Rev. 1992;56(1):152–179. | ||
Tong S, Li Y, Rivailler P, et al. A distinct lineage of influenza A virus from bats. Proc Natl Acad Sci U S A. 2012;109(11):4269–4274. | ||
Tong S, Zhu X, Li Y, et al. New world bats harbor diverse influenza A viruses. PLoS Pathog. 2013;9(10):e1003657. | ||
Wu Y, Wu Y, Tefsen B, Shi Y, Gao GF. Bat-derived influenza-like viruses H17N10 and H18N11. Trends Microbiol. 2014;22(4):183–191. | ||
Hope-Simpson RE, Golubev DB. A new concept of the epidemic process of influenza A virus. Epidemiol Infect. 1987;99(1):5–54. | ||
Rambaut A, Pybus OG, Nelson MI, Viboud C, Taubenberger JK, Holmes EC. The genomic and epidemiological dynamics of human influenza A virus. Nature. 2008;453(7195):615–619. | ||
Neuzil KM, Hohlbein C, Zhu Y. Illness among schoolchildren during influenza season: effect on school absenteeism, parental absenteeism from work, and secondary illness in families. Arch Pediatr Adolesc Med. 2002;156(10):986–991. | ||
Worobey M, Han GZ, Rambaut A. Genesis and pathogenesis of the 1918 pandemic H1N1 influenza A virus. Proc Natl Acad Sci U S A. 2014;111(22):8107–8112. | ||
Neumann G, Noda T, Kawaoka Y. Emergence and pandemic potential of swine-origin H1N1 influenza virus. Nature. 2009;459(7249):931–939. | ||
Husain M. Avian influenza A (H7N9) virus infection in humans: epidemiology, evolution, and pathogenesis. Infect Genet Evol. 2014;28:304–312. | ||
Bouvier NM, Palese P. The biology of influenza viruses. Vaccine. 2008;26(suppl 4):D49–D53. | ||
Ito T, Couceiro JN, Kelm S, et al. Molecular basis for the generation in pigs of influenza A viruses with pandemic potential. J Virol. 1998;72(9):7367–7373. | ||
Hayden FG, Cote KM, Douglas RG Jr. Plaque inhibition assay for drug susceptibility testing of influenza viruses. Antimicrob Agents Chemother. 1980;17(5):865–870. | ||
Hay AJ, Wolstenholme AJ, Skehel JJ, Smith MH. The molecular basis of the specific anti-influenza action of amantadine. EMBO J. 1985;4(11):3021–3024. | ||
Hay AJ, Zambon MC, Wolstenholme AJ, Skehel JJ, Smith MH. Molecular basis of resistance of influenza A viruses to amantadine. J Antimicrob Chemother. 1986;18(suppl B):19–29. | ||
Sugrue RJ, Hay AJ. Structural characteristics of the M2 protein of influenza A viruses: evidence that it forms a tetrameric channel. Virology. 1991;180(2):617–624. | ||
Pinto LH, Holsinger LJ, Lamb RA. Influenza virus M2 protein has ion channel activity. Cell. 1992;69(3):517–528. | ||
Pinto LH, Dieckmann GR, Gandhi CS, et al. A functionally defined model for the M2 proton channel of influenza A virus suggests a mechanism for its ion selectivity. Proc Natl Acad Sci U S A. 1997;94(21):11301–11306. | ||
Tang Y, Zaitseva F, Lamb RA, Pinto LH. The gate of the influenza virus M2 proton channel is formed by a single tryptophan residue. J Biol Chem. 2002;277(42):39880–39886. | ||
Ma C, Polishchuk AL, Ohigashi Y, et al. Identification of the functional core of the influenza A virus A/M2 proton-selective ion channel. Proc Natl Acad Sci U S A. 2009;106(30):12283–12288. | ||
Pinto LH, Lamb RA. The M2 proton channels of influenza A and B viruses. J Biol Chem. 2006;281(14):8997–9000. | ||
Stouffer AL, Acharya R, Salom D, et al. Structural basis for the function and inhibition of an influenza virus proton channel. Nature. 2008;451(7178):596–599. | ||
Schnell JR, Chou JJ. Structure and mechanism of the M2 proton channel of influenza A virus. Nature. 2008;451(7178):591–595. | ||
Cady SD, Schmidt-Rohr K, Wang J, Soto CS, Degrado WF, Hong M. Structure of the amantadine binding site of influenza M2 proton channels in lipid bilayers. Nature. 2010;463(7281):689–692. | ||
Kozakov D, Chuang GY, Beglov D, Vajda S. Where does amantadine bind to the influenza virus M2 proton channel? Trends Biochem Sci. 2010;35(9):471–475. | ||
Astrahan P, Kass I, Cooper MA, Arkin IT. A novel method of resistance for influenza against a channel-blocking antiviral drug. Proteins. 2004;55(2):251–257. | ||
Belshe RB, Smith MH, Hall CB, Betts R, Hay AJ. Genetic basis of resistance to rimantadine emerging during treatment of influenza virus infection. J Virol. 1988;62(5):1508–1512. | ||
Thomaston JL, DeGrado WF. Crystal structure of the drug-resistant S31N influenza M2 proton channel. Protein Sci. 2016;25(8):1551–1554. | ||
Pielak RM, Schnell JR, Chou JJ. Mechanism of drug inhibition and drug resistance of influenza A M2 channel. Proc Natl Acad Sci U S A. 2009;106(18):7379–7384. | ||
Suzuki H, Saito R, Masuda H, Oshitani H, Sato M, Sato I. Emergence of amantadine-resistant influenza A viruses: epidemiological study. J Infect Chemother. 2003;9(3):195–200. | ||
Dolin R, Reichman RC, Madore HP, Maynard R, Linton PN, Webber-Jones J. A controlled trial of amantadine and rimantadine in the prophylaxis of influenza A infection. N Engl J Med. 1982;307(10):580–584. | ||
Sears SD, Clements ML. Protective efficacy of low-dose amantadine in adults challenged with wild-type influenza A virus. Antimicrob Agents Chemother. 1987;31(10):1470–1473. | ||
Reuman PD, Bernstein DI, Keefer MC, Young EC, Sherwood JR, Schiff GM. Efficacy and safety of low dosage amantadine hydrochloride as prophylaxis for influenza A. Antiviral Res. 1989;11(1):27–40. | ||
Heider H, Adamczyk B, Presber HW, Schroeder C, Feldblum R, Indulen MK. Occurrence of amantadine- and rimantadine-resistant influenza A virus strains during the 1980 epidemic. Acta Virol. 1981;25(6):395–400. | ||
Hayden FG, Belshe RB, Clover RD, Hay AJ, Oakes MG, Soo W. Emergence and apparent transmission of rimantadine-resistant influenza A virus in families. N Engl J Med. 1989;321(25):1696–1702. | ||
Belshe RB, Burk B, Newman F, Cerruti RL, Sim IS. Resistance of influenza A virus to amantadine and rimantadine: results of one decade of surveillance. J Infect Dis. 1989;159(3):430–435. | ||
Ziegler T, Hemphill ML, Ziegler ML, et al. Low incidence of rimantadine resistance in field isolates of influenza A viruses. J Infect Dis. 1999;180(4):935–939. | ||
Bright RA, Medina MJ, Xu X, et al. Incidence of adamantane resistance among influenza A (H3N2) viruses isolated worldwide from 1994 to 2005: a cause for concern. Lancet. 2005;366(9492):1175–1181. | ||
Deyde VM, Xu X, Bright RA, et al. Surveillance of resistance to adamantanes among influenza A(H3N2) and A(H1N1) viruses isolated worldwide. J Infect Dis. 2007;196(2):249–257. | ||
Bright RA, Shay DK, Shu B, Cox NJ, Klimov AI. Adamantane resistance among influenza A viruses isolated early during the 2005-2006 influenza season in the United States. JAMA. 2006;295(8):891–894. | ||
Centers for Disease Control and Prevention (CDC). High levels of adamantane resistance among influenza A (H3N2) viruses and interim guidelines for use of antiviral agents--United States, 2005-06 influenza season. MMWR Morb Mortal Wkly Rep. 2006;55(2):44–46. | ||
Novel Swine-Origin Influenza A (H1N1) Virus Investigation Team, Dawood FS, Jain S, et al. Emergence of a novel swine-origin influenza A (H1N1) virus in humans. N Engl J Med. 2009;360(25):2605–2615. | ||
Rungrotmongkol T, Intharathep P, Malaisree M, et al. Susceptibility of antiviral drugs against 2009 influenza A (H1N1) virus. Biochem Biophys Res Commun. 2009;385(3):390–394. | ||
World Health Organization Global Influenza Program Surveillance Network. Evolution of H5N1 avian influenza viruses in Asia. Emerg Infect Dis. 2005;11(10):1515–1521. | ||
Dong G, Peng C, Luo J, et al. Adamantane-resistant influenza a viruses in the world (1902-2013): frequency and distribution of M2 gene mutations. PLoS One. 2015;10(3):e0119115. | ||
Garcia V, Aris-Brosou S. Comparative dynamics and distribution of influenza drug resistance acquisition to protein m2 and neuraminidase inhibitors. Mol Biol Evol. 2014;31(2):355–363. | ||
Durrant MG, Eggett DL, Busath DD. Investigation of a recent rise of dual amantadine-resistance mutations in the influenza A M2 sequence. BMC Genet. 2015;16(suppl 2):S3. | ||
Sweet C, Hayden FG, Jakeman KJ, Grambas S, Hay AJ. Virulence of rimantadine-resistant human influenza A (H3N2) viruses in ferrets. J Infect Dis. 1991;164(5):969–972. | ||
Furuse Y, Suzuki A, Oshitani H. Large-scale sequence analysis of M gene of influenza A viruses from different species: mechanisms for emergence and spread of amantadine resistance. Antimicrob Agents Chemother. 2009;53(10):4457–4463. | ||
Simonsen L, Viboud C, Grenfell BT, et al. The genesis and spread of reassortment human influenza A/H3N2 viruses conferring adamantane resistance. Mol Biol Evol. 2007;24(8):1811–1820. | ||
Abed Y, Goyette N, Boivin G. Generation and characterization of recombinant influenza A (H1N1) viruses harboring amantadine resistance mutations. Antimicrob Agents Chemother. 2005;49(2):556–559. | ||
Nelson MI, Simonsen L, Viboud C, Miller MA, Holmes EC. The origin and global emergence of adamantane resistant A/H3N2 influenza viruses. Virology. 2009;388(2):270–278. | ||
Barr IG, Hurt AC, Deed N, Iannello P, Tomasov C, Komadina N. The emergence of adamantane resistance in influenza A(H1) viruses in Australia and regionally in 2006. Antiviral Res. 2007;75(2):173–176. | ||
Gubareva LV, Kaiser L, Hayden FG. Influenza virus neuraminidase inhibitors. Lancet. 2000;355(9206):827–835. | ||
Gottschalk A. Neuraminidase: the specific enzyme of influenza virus and Vibrio cholerae. Biochim Biophys Acta. 1957;23(3):645–646. | ||
Palese P, Tobita K, Ueda M, et al. Characterization of temperature sensitive influenza virus mutants defective in neuraminidase. Virology. 1974;61(2):397–410. | ||
Edmond JD, Johnston RG, Kidd D, et al. The inhibition of neuraminidase and antiviral action. Br J Pharmacol Chemother. 1966;27(2):415–426. | ||
Meindl P, Bodo G, Palese P, et al. Inhibition of neuraminidase activity by derivatives of 2-deoxy-2,3-dehydro-N-acetylneuraminic acid. Virology. 1974;58(2):457–463. | ||
Palese P, Compans RW. Inhibition of influenza virus replication in tissue culture by 2-deoxy-2,3-dehydro-N-trifluoroacetylneuraminic acid (FANA): mechanism of action. J Gen Virol. 1976;33(1):159–163. | ||
Varghese JN, Laver WG, Colman PM. Structure of the influenza virus glycoprotein antigen neuraminidase at 2.9 A resolution. Nature. 1983;303(5912):35–40. | ||
Colman PM, Varghese JN, Laver WG. Structure of the catalytic and antigenic sites in influenza virus neuraminidase. Nature. 1983;303(5912):41–44. | ||
von Itzstein M, Wu WY, Kok GB, et al. Rational design of potent sialidase-based inhibitors of influenza virus replication. Nature. 1993;363(6428):418–423. | ||
Kim CU, Lew W, Williams MA, et al. Influenza neuraminidase inhibitors possessing a novel hydrophobic interaction in the enzyme active site: design, synthesis, and structural analysis of carbocyclic sialic acid analogues with potent anti-influenza activity. J Am Chem Soc. 1997;119(4):681–690. | ||
Colman PM. Influenza virus neuraminidase: structure, antibodies, and inhibitors. Protein Sci. 1994;3(10):1687–1696. | ||
Shtyrya YA, Mochalova LV, Bovin NV. Influenza virus neuraminidase: structure and function. Acta Naturae. 2009;1(2):26–32. | ||
Collins PJ, Haire LF, Lin YP, et al. Structural basis for oseltamivir resistance of influenza viruses. Vaccine. 2009;27(45):6317–6323. | ||
McKimm-Breschkin JL. Influenza neuraminidase inhibitors: antiviral action and mechanisms of resistance. Influenza Other Respir Viruses. 2013;7(suppl 1):25–36. | ||
Russell RJ, Haire LF, Stevens DJ, et al. The structure of H5N1 avian influenza neuraminidase suggests new opportunities for drug design. Nature. 2006;443(7107):45–49. | ||
Collins PJ, Haire LF, Lin YP, et al. Crystal structures of oseltamivir-resistant influenza virus neuraminidase mutants. Nature. 2008;453(7199):1258–1261. | ||
Wang NX, Zheng JJ. Computational studies of H5N1 influenza virus resistance to oseltamivir. Protein Sci. 2009;18(4):707–715. | ||
Li Q, Qi J, Wu Y, et al. Functional and structural analysis of influenza virus neuraminidase N3 offers further insight into the mechanisms of oseltamivir resistance. J Virol. 2013;87(18):10016–10024. | ||
Huang L, Cao Y, Zhou J, et al. A conformational restriction in the influenza A virus neuraminidase binding site by R152 results in a combinational effect of I222T and H274Y on oseltamivir resistance. Antimicrob Agents Chemother. 2014;58(3):1639–1645. | ||
Samson M, Abed Y, Desrochers FM, et al. Characterization of drug-resistant influenza virus A(H1N1) and A(H3N2) variants selected in vitro with laninamivir. Antimicrob Agents Chemother. 2014;58(9):5220–5228. | ||
Wu Y, Gao F, Qi J, et al. Resistance to mutant group 2 influenza virus neuraminidases of an oseltamivir-zanamivir hybrid inhibitor. J Virol. 2016;90(23):10693–10700. | ||
Oxford JS, Bossuyt S, Balasingam S, Mann A, Novelli P, Lambkin R. Treatment of epidemic and pandemic influenza with neuraminidase and M2 proton channel inhibitors. Clin Microbiol Infect. 2003;9(1):1–14. | ||
Whitley RJ, Hayden FG, Reisinger KS, et al. Oral oseltamivir treatment of influenza in children. Pediatr Infect Dis J. 2001;20(2):127–133. | ||
Moscona A. Neuraminidase inhibitors for influenza. N Engl J Med. 2005;353(13):1363–1373. | ||
Aoki FY, Macleod MD, Paggiaro P, et al. Early administration of oral oseltamivir increases the benefits of influenza treatment. J Antimicrob Chemother. 2003;51(1):123–129. | ||
Moss RB, Davey RT, Steigbigel RT, Fang F. Targeting pandemic influenza: a primer on influenza antivirals and drug resistance. J Antimicrob Chemother. 2010;65(6):1086–1093. | ||
Babu YS, Chand P, Bantia S, et al. BCX-1812 (RWJ-270201): discovery of a novel, highly potent, orally active, and selective influenza neuraminidase inhibitor through structure-based drug design. J Med Chem. 2000;43(19):3482–3486. | ||
McLaughlin MM, Skoglund EW, Ison MG. Peramivir: an intravenous neuraminidase inhibitor. Expert Opin Pharmacother. 2015;16(12):1889–1900. | ||
Yamashita M, Tomozawa T, Kakuta M, Tokumitsu A, Nasu H, Kubo S. CS-8958, a prodrug of the new neuraminidase inhibitor R-125489, shows long-acting anti-influenza virus activity. Antimicrob Agents Chemother. 2009;53(1):186–192. | ||
Kubo S, Tomozawa T, Kakuta M, Tokumitsu A, Yamashita M. Laninamivir prodrug CS-8958, a long-acting neuraminidase inhibitor, shows superior anti-influenza virus activity after a single administration. Antimicrob Agents Chemother. 2010;54(3):1256–1264. | ||
Kashiwagi S, Watanabe A, Ikematsu H, Uemori M, Awamura S; Laninamivir Prophylaxis Study Group. Long-acting neuraminidase inhibitor laninamivir octanoate as post-exposure prophylaxis for influenza. Clin Infect Dis. 2016;63(3):330–337. | ||
McKimm-Breschkin JL. Resistance of influenza viruses to neuraminidase inhibitors – a review. Antiviral Res. 2000;47(1):1–17. | ||
Gubareva LV, Kaiser L, Matrosovich MN, Soo-Hoo Y, Hayden FG. Selection of influenza virus mutants in experimentally infected volunteers treated with oseltamivir. J Infect Dis. 2001;183(4):523–531. | ||
Zambon M, Hayden FG; Global Neuraminidase Inhibitor Susceptibility Network. Position statement: global neuraminidase inhibitor susceptibility network. Antiviral Res. 2001;49(3):147–156. | ||
McKimm-Breschkin J, Trivedi T, Hampson A, et al. Neuraminidase sequence analysis and susceptibilities of influenza virus clinical isolates to zanamivir and oseltamivir. Antimicrob Agents Chemother. 2003;47(7):2264–2272. | ||
Hurt AC, Barr IG, Hartel G, Hampson AW. Susceptibility of human influenza viruses from Australasia and South East Asia to the neuraminidase inhibitors zanamivir and oseltamivir. Antiviral Res. 2004;62(1):37–45. | ||
Monto AS, McKimm-Breschkin JL, Macken C, et al. Detection of influenza viruses resistant to neuraminidase inhibitors in global surveillance during the first 3 years of their use. Antimicrob Agents Chemother. 2006;50(7):2395–2402. | ||
Kiso M, Mitamura K, Sakai-Tagawa Y, et al. Resistant influenza A viruses in children treated with oseltamivir: descriptive study. Lancet. 2004;364(9436):759–765. | ||
Stephenson I, Democratis J, Lackenby A, et al. Neuraminidase inhibitor resistance after oseltamivir treatment of acute influenza A and B in children. Clin Infect Dis. 2009;48(4):389–396. | ||
Sheu TG, Deyde VM, Okomo-Adhiambo M, et al. Surveillance for neuraminidase inhibitor resistance among human influenza A and B viruses circulating worldwide from 2004 to 2008. Antimicrob Agents Chemother. 2008;52(9):3284–3292. | ||
Escuret V, Frobert E, Bouscambert-Duchamp M, et al. Detection of human influenza A (H1N1) and B strains with reduced sensitivity to neuraminidase inhibitors. J Clin Virol. 2008;41(1):25–28. | ||
Hurt AC, Holien JK, Parker M, Kelso A, Barr IG. Zanamivir-resistant influenza viruses with a novel neuraminidase mutation. J Virol. 2009;83(20):10366–10373. | ||
Lackenby A, Hungnes O, Dudman SG, et al. Emergence of resistance to oseltamivir among influenza A(H1N1) viruses in Europe. Euro Surveill. 2008;13(5):8026. | ||
Hauge SH, Dudman S, Borgen K, Lackenby A, Hungnes O. Oseltamivir-resistant influenza viruses A (H1N1), Norway, 2007-08. Emerg Infect Dis. 2009;15(2):155–162. | ||
Meijer A, Lackenby A, Hungnes O, et al. Oseltamivir-resistant influenza virus A (H1N1), Europe, 2007-08 season. Emerg Infect Dis. 2009;15(4):552–560. | ||
Dharan NJ, Gubareva LV, Meyer JJ, et al. Infections with oseltamivir-resistant influenza A(H1N1) virus in the United States. JAMA. 2009;301(10):1034–1041. | ||
Okomo-Adhiambo M, Sleeman K, Ballenger K, et al. Neuraminidase inhibitor susceptibility testing in human influenza viruses: a laboratory surveillance perspective. Viruses. 2010;2(10):2269–2289. | ||
Baranovich T, Saito R, Suzuki Y, et al. Emergence of H274Y oseltamivir-resistant A(H1N1) influenza viruses in Japan during the 2008-2009 season. J Clin Virol. 2010;47(1):23–28. | ||
Matsuzaki Y, Mizuta K, Aoki Y, et al. A two-year survey of the oseltamivir-resistant influenza A(H1N1) virus in Yamagata, Japan and the clinical effectiveness of oseltamivir and zanamivir. Virol J. 2010;7:53. | ||
Hurt AC, Ernest J, Deng YM, et al. Emergence and spread of oseltamivir-resistant A(H1N1) influenza viruses in Oceania, South East Asia and South Africa. Antiviral Res. 2009;83(1):90–93. | ||
Gubareva LV, Trujillo AA, Okomo-Adhiambo M, et al. Comprehensive assessment of 2009 pandemic influenza A (H1N1) virus drug susceptibility in vitro. Antivir Ther. 2010;15(8):1151–1159. | ||
Leung TW, Tai AL, Cheng PK, et al. Detection of an oseltamivir-resistant pandemic influenza A/H1N1 virus in Hong Kong. J Clin Virol. 2009;46(3):298–299. | ||
Centers for Disease Control and Prevention (CDC). Oseltamivir-resistant 2009 pandemic influenza A (H1N1) virus infection in two summer campers receiving prophylaxis – North Carolina, 2009. MMWR Morb Mortal Wkly Rep. 2009;58(35):969–972. | ||
Centers for Disease Control and Prevention (CDC). Oseltamivir-resistant novel influenza A (H1N1) virus infection in two immunosuppressed patients – Seattle, Washington, 2009. MMWR Morb Mortal Wkly Rep. 2009;58(32):893–896. | ||
Dapat C, Kondo H, Dapat IC, et al. Neuraminidase inhibitor susceptibility profile of pandemic and seasonal influenza viruses during the 2009-2010 and 2010-2011 influenza seasons in Japan. Antiviral Res. 2013;99(3):261–269. | ||
Hurt AC, Chotpitayasunondh T, Cox NJ, et al. Antiviral resistance during the 2009 influenza A H1N1 pandemic: public health, laboratory, and clinical perspectives. Lancet Infect Dis. 2012;12(3):240–248. | ||
Leang SK, Kwok S, Sullivan SG, et al. Peramivir and laninamivir susceptibility of circulating influenza A and B viruses. Influenza Other Respir Viruses. 2014;8(2):135–139. | ||
Meijer A, Rebelo-de-Andrade H, Correia V, et al. Global update on the susceptibility of human influenza viruses to neuraminidase inhibitors, 2012-2013. Antiviral Res. 2014;110:31–41. | ||
Takashita E, Meijer A, Lackenby A, et al. Global update on the susceptibility of human influenza viruses to neuraminidase inhibitors, 2013-2014. Antiviral Res. 2015;117:27–38. | ||
Hurt AC, Besselaar TG, Daniels RS, et al. Global update on the susceptibility of human influenza viruses to neuraminidase inhibitors, 2014-2015. Antiviral Res. 2016;132:178–185. | ||
Chen LF, Dailey NJ, Rao AK, et al. Cluster of oseltamivir-resistant 2009 pandemic influenza A (H1N1) virus infections on a hospital ward among immunocompromised patients – North Carolina, 2009. J Infect Dis. 2011;203(6):838–846. | ||
Takashita E, Kiso M, Fujisaki S, et al. Characterization of a large cluster of influenza A(H1N1)pdm09 viruses cross-resistant to oseltamivir and peramivir during the 2013-2014 influenza season in Japan. Antimicrob Agents Chemother. 2015;59(5):2607–2617. | ||
Hurt AC, Hardie K, Wilson NJ, et al. Characteristics of a widespread community cluster of H275Y oseltamivir-resistant A(H1N1)pdm09 influenza in Australia. J Infect Dis. 2012;206(2):148–157. | ||
Moore C, Galiano M, Lackenby A, et al. Evidence of person-to-person transmission of oseltamivir-resistant pandemic influenza A(H1N1) 2009 virus in a hematology unit. J Infect Dis. 2011;203(1):18–24. | ||
Tramontana AR, George B, Hurt AC, et al. Oseltamivir resistance in adult oncology and hematology patients infected with pandemic (H1N1) 2009 virus, Australia. Emerg Infect Dis. 2010;16(7):1068–1075. | ||
Chan PA, Connell NT, Gabonay AM, et al. Oseltamivir-resistant 2009-2010 pandemic influenza A (H1N1) in an immunocompromised patient. Clin Microbiol Infect. 2010;16(10):1576–1578. | ||
Memoli MJ, Hrabal RJ, Hassantoufighi A, Eichelberger MC, Taubenberger JK. Rapid selection of oseltamivir- and peramivir-resistant pandemic H1N1 virus during therapy in 2 immunocompromised hosts. Clin Infect Dis. 2010;50(9):1252–1255. | ||
Campanini G, Piralla A, Rovida F, et al. First case in Italy of acquired resistance to oseltamivir in an immunocompromised patient with influenza A/H1N1v infection. J Clin Virol. 2010;48(3):220–222. | ||
Harvala H, Gunson R, Simmonds P, et al. The emergence of oseltamivir-resistant pandemic influenza A (H1N1) 2009 virus amongst hospitalised immunocompromised patients in Scotland, November-December, 2009. Euro Surveill. 2010;15(14):19536. | ||
Hurt AC. The epidemiology and spread of drug resistant human influenza viruses. Curr Opin Virol. 2014;8:22–29. | ||
To KK, Ng KH, Que TL, et al. Avian influenza A H5N1 virus: a continuous threat to humans. Emerg Microbes Infect. 2012;1(9):e25. | ||
Ginting TE, Shinya K, Kyan Y, et al. Amino acid changes in hemagglutinin contribute to the replication of oseltamivir-resistant H1N1 influenza viruses. J Virol. 2012;86(1):121–127. | ||
Bloom JD, Gong LI, Baltimore D. Permissive secondary mutations enable the evolution of influenza oseltamivir resistance. Science. 2010;328(5983):1272–1275. | ||
Abed Y, Pizzorno A, Bouhy X, Boivin G. Role of permissive neuraminidase mutations in influenza A/Brisbane/59/2007-like (H1N1) viruses. PLoS Pathog. 2011;7(12):e1002431. | ||
Duan S, Govorkova EA, Bahl J, et al. Epistatic interactions between neuraminidase mutations facilitated the emergence of the oseltamivir-resistant H1N1 influenza viruses. Nat Commun. 2014;5:5029. | ||
Butler J, Hooper KA, Petrie S, et al. Estimating the fitness advantage conferred by permissive neuraminidase mutations in recent oseltamivir-resistant A(H1N1)pdm09 influenza viruses. PLoS Pathog. 2014;10(4):e1004065. | ||
Abed Y, Pizzorno A, Bouhy X, Boivin G. Permissive changes in the neuraminidase play a dominant role in improving the viral fitness of oseltamivir-resistant seasonal influenza A(H1N1) strains. Antiviral Res. 2015;114:57–61. | ||
Dapat C, Suzuki Y, Saito R, et al. Rare influenza A (H3N2) variants with reduced sensitivity to antiviral drugs. Emerg Infect Dis. 2010;16(3):493–496. | ||
Zaraket H, Saito R, Suzuki Y, et al. Genetic makeup of amantadine-resistant and oseltamivir-resistant human influenza A/H1N1 viruses. J Clin Microbiol. 2010;48(4):1085–1092. | ||
Petrie SM, Butler J, Barr IG, McVernon J, Hurt AC, McCaw JM. Quantifying relative within-host replication fitness in influenza virus competition experiments. J Theor Biol. 2015;382:259–271. | ||
Barberis I, Myles P, Ault SK, Bragazzi NL, Martini M. History and evolution of influenza control through vaccination: from the first monovalent vaccine to universal vaccines. J Prev Med Hyg. 2016;57(3):E115–E120. | ||
Breteler JK, Tam JS, Jit M, Ket JC, De Boer MR. Efficacy and effectiveness of seasonal and pandemic A (H1N1) 2009 influenza vaccines in low and middle income countries: a systematic review and meta-analysis. Vaccine. 2013;31(45):5168–5177. | ||
Osterholm MT, Kelley NS, Sommer A, Belongia EA. Efficacy and effectiveness of influenza vaccines: a systematic review and meta-analysis. Lancet Infect Dis. 2012;12(1):36–44. | ||
Demicheli V, Jefferson T, Al-Ansary LA, et al. Vaccines for preventing influenza in healthy adults. Cochrane Database Syst Rev. 2014;3:CD001269. | ||
Cowling BJ, Feng S, Finelli L, Steffens A, Fowlkes A. Assessment of influenza vaccine effectiveness in a sentinel surveillance network 2010-13, United States. Vaccine. 2016;34(1):61–66. | ||
Wong SS, Webby RJ. Traditional and new influenza vaccines. Clin Microbiol Rev. 2013;26(3):476–492. | ||
Krammer F, Palese P. Advances in the development of influenza virus vaccines. Nat Rev Drug Discov. 2015;14(3):167–182. | ||
Dunkle LM, Izikson R. Recombinant hemagglutinin influenza vaccine provides broader spectrum protection. Expert Rev Vaccines. 2016;15(8):957–966. | ||
Naesens L, Stevaert A, Vanderlinden E. Antiviral therapies on the horizon for influenza. Curr Opin Pharmacol. 2016;30:106–115. | ||
McKimm-Breschkin JL, Fry AM. Meeting report: 4th ISIRV antiviral group conference: novel antiviral therapies for influenza and other respiratory viruses. Antiviral Res. 2016;129:21–38. | ||
Yen HL. Current and novel antiviral strategies for influenza infection. Curr Opin Virol. 2016;18:126–134. | ||
Alexander DJ. A review of avian influenza in different bird species. Vet Microbiol. 2000;74(1–2):3–13. | ||
Donatelli I, Castrucci MR, De Marco MA, Delogu M, Webster RG. Human-animal interface: the case for influenza interspecies transmission. Adv Exp Med Biol. Epub 2016 Sep 28. | ||
Lyall J, Irvine RM, Sherman A, et al. Suppression of avian influenza transmission in genetically modified chickens. Science. 2011;331(6014):223–226. | ||
Ruiz-Hernandez R, Mwangi W, Peroval M, et al. Host genetics determine susceptibility to avian influenza infection and transmission dynamics. Sci Rep. 2016;6:26787. | ||
Wang S, Chen C, Yang Z, Chi X, Zhang J, Chen JL. Targeted disruption of influenza A virus hemagglutinin in genetically modified mice reduces viral replication and improves disease outcome. Sci Rep. 2016;6:23746. | ||
Lee HJ, Lee HC, Han JY. Germline modification and engineering in avian species. Mol Cells. 2015;38(9):743–749. | ||
Boltz DA, Douangngeun B, Phommachanh P, et al. Emergence of H5N1 avian influenza viruses with reduced sensitivity to neuraminidase inhibitors and novel reassortants in Lao People’s Democratic Republic. J Gen Virol. 2010;91(pt 4):949–959. | ||
Ilyushina NA, Seiler JP, Rehg JE, Webster RG, Govorkova EA. Effect of neuraminidase inhibitor-resistant mutations on pathogenicity of clade 2.2 A/Turkey/15/06 (H5N1) influenza virus in ferrets. PLoS Pathog. 2010;6(5):e1000933. | ||
Tamura D, Sugaya N, Ozawa M, et al. Frequency of drug-resistant viruses and virus shedding in pediatric influenza patients treated with neuraminidase inhibitors. Clin Infect Dis. 2011;52(4):432–437. | ||
Nguyen HT, Fry AM, Loveless PA, et al. Recovery of a multidrug-resistant strain of pandemic influenza A 2009 (H1N1) virus carrying a dual H275Y/I223R mutation from a child after prolonged treatment with oseltamivir. Clin Infect Dis. 2010;51(8):983–984. | ||
Pizzorno A, Abed Y, Bouhy X, et al. Impact of mutations at residue I223 of the neuraminidase protein on the resistance profile, replication level, and virulence of the 2009 pandemic influenza virus. Antimicrob Agents Chemother. 2012;56(3):1208–1214. | ||
Hurt AC, Lee RT, Leang SK, et al. Increased detection in Australia and Singapore of a novel influenza A(H1N1)2009 variant with reduced oseltamivir and zanamivir sensitivity due to a S247N neuraminidase mutation. Euro Surveill. 2011;16(23):19884. | ||
Takashita E, Fujisaki S, Kishida N, et al. Characterization of neuraminidase inhibitor-resistant influenza A(H1N1)pdm09 viruses isolated in four seasons during pandemic and post-pandemic periods in Japan. Influenza Other Respir Viruses. 2013;7(6):1390–1399. | ||
Okomo-Adhiambo M, Sleeman K, Lysen C, et al. Neuraminidase inhibitor susceptibility surveillance of influenza viruses circulating worldwide during the 2011 Southern Hemisphere season. Influenza Other Respir Viruses. 2013;7(5):645–658. | ||
Okomo-Adhiambo M, Nguyen HT, Abd Elal A, Sleeman K, Fry AM, Gubareva LV. Drug susceptibility surveillance of influenza viruses circulating in the United States in 2011-2012: application of the WHO antiviral working group criteria. Influenza Other Respir Viruses. 2014;8(2):258–265. | ||
Nguyen HT, Trujillo AA, Sheu TG, et al. Analysis of influenza viruses from patients clinically suspected of infection with an oseltamivir resistant virus during the 2009 pandemic in the United States. Antiviral Res. 2012;93(3):381–386. | ||
Nguyen HT, Sheu TG, Mishin VP, Klimov AI, Gubareva LV. Assessment of pandemic and seasonal influenza A (H1N1) virus susceptibility to neuraminidase inhibitors in three enzyme activity inhibition assays. Antimicrob Agents Chemother. 2010;54(9):3671–3677. | ||
Le QM, Kiso M, Someya K, et al. Avian flu: isolation of drug-resistant H5N1 virus. Nature. 2005;437(7062):1108. | ||
Yen HL, Herlocher LM, Hoffmann E, et al. Neuraminidase inhibitor-resistant influenza viruses may differ substantially in fitness and transmissibility. Antimicrob Agents Chemother. 2005;49(10):4075–4084. | ||
Pizzorno A, Bouhy X, Abed Y, Boivin G. Generation and characterization of recombinant pandemic influenza A(H1N1) viruses resistant to neuraminidase inhibitors. J Infect Dis. 2011;203(1):25–31. | ||
Earhart KC, Elsayed NM, Saad MD, et al. Oseltamivir resistance mutation N294S in human influenza A(H5N1) virus in Egypt. J Infect Public Health. 2009;2(2):74–80. |
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.