Back to Journals » Clinical Interventions in Aging » Volume 12
Drug prescription appropriateness in the elderly: an Italian study
Authors Allegri N, Rossi F, Del Signore F, Bertazzoni P, Bellazzi R, Sandrini G, Vecchi T, Liccione D, Pascale A , Govoni S
Received 24 March 2016
Accepted for publication 8 July 2016
Published 10 February 2017 Volume 2017:12 Pages 325—333
DOI https://doi.org/10.2147/CIA.S109125
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Richard Walker
Nicola Allegri,1 Federica Rossi,2 Federica Del Signore,2 Paolo Bertazzoni,3 Roberto Bellazzi,4 Giorgio Sandrini,5 Tomaso Vecchi,1 Davide Liccione,1 Alessia Pascale,6 Stefano Govoni6
1Department of Brain and Behavioral Sciences, University of Pavia, 2Pavia and Vigevano Neuropsychological Center for Alzheimer’s Disease, 3”Bertazzoni” Pharmacy General Partnership, Vigevano, 4Nephrology and Dialysis Ward, Civil Hospital of Vigevano, 5C. Mondino National Institute of Neurology Foundation, IRCCS, 6Department of Drug Sciences, Section of pharmacology, University of Pavia, Pavia, Italy
Purpose: Correct drug prescription in the elderly is a difficult task that requires careful survey of the current pharmacological therapies. In this article, we reviewed the drug prescriptions provided to 860 persons aged 65 years or over, residing in a small city of Lombardy, Italy.
Methods: Subjects were recruited from a local nursing home, the Pavia and Vigevano Neuropsychological Center for Alzheimer’s Disease, general practitioners’ offices, and the local University of the Third Age. For each patient, the amount of potentially inappropriate prescriptions (PIPs), sedative and anticholinergic load (SL and AL, respectively), and drug–drug interactions were evaluated.
Results: Widespread polypharmacy, giving rise to 10.06% of PIPs in the whole collection of prescriptions, was observed. In particular, PIPs mainly concern drugs acting at the central nervous system level, mostly benzodiazepines and antipsychotics. Moreover, approximately one-fourth of the subjects had an elevated SL and approximately one-tenth a high AL. Drug–drug interactions were frequent (266 requiring medical attention), up to five for each single patient. Of concern was the underuse of antidementia drugs: only 20 patients received a cholinesterase inhibitor or memantine, although 183 patients were potentially suitable for this treatment.
Conclusion: These results demonstrate the need to develop novel strategies aimed at improving the quality of drug prescription.
Keywords: drug prescription, elderly, Beers criteria, drug–drug interactions, sedative load
Corrigendum for this paper has been published
Introduction
Drug prescription inappropriateness represents an issue of major interest in clinical practice, since it increases the prevalence of drug-induced harms, with consequences related to patients’ health1 and cost of care.2 This is particularly true for elderly people, whose frailty makes the choice of the correct drug prescription even more difficult.3
Appropriateness can be assessed by measuring explicit (criterion-based) or implicit (judgment-based) outcomes.4,5 In this regard, international explicit criteria have been developed to assess the pertinence of medications in the elderly, such as the Beers criteria, which was recently updated by the American Geriatrics Society,6–8 or the STOPP (Screening tool of older person’s prescriptions)/START (Screening tool to alert doctors to right treatment).9 These measures mainly take into account the relationship between a particular drug and the specific medical case. However, it should be stressed that the evaluation of prescription appropriateness in the elderly represents a complex issue10 that should also include other clinical dimensions, such as the amount of prescribed drugs,11 drug–drug interactions,12 and the potential sedative and anticholinergic effects of the prescribed drugs.13,14
In this context, the aim of the present work was to evaluate the appropriateness of drug prescription using the Beers criteria and including the assessment of the aforementioned aspects in a group of 860 persons, aged ≥65 years, residing in a small city of Lombardy, Italy.
Methods
As a part of a larger epidemiological study, neuropsychologists administered an anonymous questionnaire regarding prescribed medicines to 860 subjects. The study participants were divided into three groups: 1) belonging to a local nursing home (NH patients – n=142); 2) coming to the local memory clinic (MC; Pavia and Vigevano Neuropsychological Center – Alzheimer Section) for a neurological examination (MC patients – n=572); 3) volunteer subjects (VS patients – n=146), mainly recruited at the local University of the Third Age, at general practitioners’ offices or in other community centers. The questionnaire included the age of the patient (in years), sex, Mini-Mental State Examination (MMSE) score, current illnesses and treatments. MMSE was assessed according to the Folstein and McHugh method,15 as adapted for the Italian population16 (range 0–30; normal range >24). The results on drug use were correlated with the demographic and clinical data. The demographic characteristics, which referred to both the whole sample and the subgroups, are reported in Table 1. Figures 1 and 2 give a more detailed picture of the heterogeneity, in terms of age and MMSE, of the participants coming for a neurological examination to the local MC.
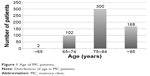  | Figure 1 Age of MC patients. |
Appropriateness of the prescription
Appropriateness of the prescription was analyzed by the use of the Beers criteria.8 In case of molecules available on the Italian market, but not included in the American Geriatrics Society list of medications, the drug was evaluated (ie, in terms of class, pharmacokinetics) and classified by the team of pharmacologists.
When applying the Beers criteria, specific care was taken regarding the diagnosis of the patient.
Sedative load definition
The sedative load (SL) was calculated for each subject according to a published model.13 Medications were classified as follows: Group 1, primary sedatives; Group 2, medications with sedation as a prominent side effect or preparations with a sedating component; Group 3, medications with sedation as a potential adverse effect; Group 4, all the other medications with no known sedative properties.
Anticholinergic load definition
The AL was calculated according to the scoring system of the Anticholinergic Drug Scale.17 On the Anticholinergic Drug Scale, drugs are rated from 0 to 3, with level zero indicating no anticholinergic activity, and level 3 indicating an elevated anticholinergic activity. The anticholinergic scores relative to each drug taken by the patient were summed up to obtain the total score.
Drug interactions
Drug interactions were evaluated using the Drug Interactions Checker program available at the website (www.drugs.com/drug_interactions.html) and further checked by a pharmacologist employing the Stockley’s Drug Interactions18 international source.
Statistics
Drugs were classified according to the Anatomical Therapeutic Chemical (ATC) code, data were collected in an ad hoc created database and analyzed using the SPSS software version 15.0 (SPSS Inc., Chicago, IL, USA).19 Mean and standard deviation were calculated for each variable. For some variables of interest, an analysis of variance (ANOVA) was performed. A P-value <0.05 was considered statistically significant. Moreover, to explore the relationship between some of the considered characteristics, a bivariate analysis based on Pearson’s r correlation was carried out.
Results
A total of 5,087 prescriptions was recorded for the 860 subjects examined in the study, corresponding to 472 molecules, with cardiovascular and central nervous system (CNS) drugs representing most of the sample (31.1% and 21.2%, respectively). Considering the single subgroups, cardiovascular drugs were most frequently prescribed to MC and VS subjects (32.8% and 38.8%, respectively), while NH patients were mainly prescribed CNS and gastrointestinal drugs (27.3% and 26.4%, respectively).
The average number of prescriptions per patient in the whole sample was 5.91 (Table 1). To this regard, a significant difference was found among the three groups (F[2,857] =41,593; P<0.001), and post hoc analyses specifically revealed that, as expected, NH patients take a greater amount of drugs (7.89) compared with MC (5.81) and VS (4.36) subjects.
Within the range of age investigated (65–100 years), age did not correlate with the amount of prescriptions, neither in the whole sample (r=−0.006; P=0.864), nor in NH and VS subjects (r=−0.143, P=0.089 and r=0.045, P=0.588, respectively), although a negative correlation was observed in MC patients (r=−0.123, P<0.05).
Not surprisingly, in the whole sample, the subjects who have a higher number of diseases also received more prescriptions (r=0.480; P<0.001), and this was true even when considering each subgroup (NH: r=0.477, MC: r=0.489, VS: r=0.593; P<0.001 for all).
Top ten prescriptions
Table 2 reports ten drugs that were most frequently prescribed while Table 3 shows the complete distribution of the medications in the whole sample according to their relative ATC class. In the whole sample, almost one-third of the patients were taking acetylsalicylic acid (35.3%), mainly for secondary prevention of cardiovascular and cerebrovascular events. Furosemide and ramipril held the second (22.4%) and third (14.2%) place, respectively. Proton pump inhibitors were also frequently used; indeed, combining esomeprazole and omeprazole, 220 prescriptions were reached, corresponding to 25.5% of patients. No psychotropic drugs were present within the top ten prescriptions. Similar data were obtained when analyzing each subgroup of patients. However, it is noteworthy to mention that psychotropic drugs were prescribed in the top ten prescriptions in NH patients, where quetiapine held the fifth place, and in VS subjects, where lorazepam held the ninth place. This finding is confirmed by the data referring to the complete distribution of medications according to their relative ATC class, where psychotropic drugs (the main prescribed drugs acting on the nervous system) held the second place (Table 3).
Potentially inappropriate drug prescriptions
Table 4 reports the PIPs recorded, according to the different drug classes, both in the whole population and in each subgroup. Irrespective of the drug classes, the total number of PIPs was 538, corresponding to 10.6% of the total prescriptions. Percentages were quite similar when considering the NH and MC groups (11% and 10%, respectively); conversely, VS subjects were found to have lower number of PIPs (56, corresponding to 8.8%). Reporting PIPs as percentage of the total prescriptions, however, somewhat “dilutes” the impact of PIPs due to the high number of prescriptions. When expressing the data as percentage of patients coping with at least one PIP, similar results were noticed in the whole sample (44.4%) and in MC patients (44.5%). Very different was the case of NH patients, where this percentage reached 57.7%, and VS subjects, where the value dropped down to 30.8%. In particular, lorazepam was one of the most frequent PIP, both in the whole sample and subgroups, as well as many other benzodiazepines (summing up all the benzodiazepines, they constituted 40.2% of all PIPs). Although some of the considered benzodiazepines were not listed in the tables reported in the Beers criteria (American Geriatrics Society, 2012), from a general point of view, benzodiazepines of intermediate or long half-lives should be avoided for the treatment of insomnia, agitation, or delirium in the elderly. Nevertheless, whenever a diagnosis of anxiety or a risk of convulsions was present, the benzodiazepine prescription was considered as appropriate. All the prescriptions relating to triazolam were considered inappropriate as this compound, in Italy, is used only as a hypnotic. Ticlopidine and doxazosin were also very frequently prescribed, both in whole sample and subgroups (in total, 48 patients received doxazosin and 60 received ticlopidine). Antipsychotics were inappropriately prescribed 97 times in patients with dementia for the treatment of behavioral symptoms before trying nonpharmacological therapies, as suggested by Beers criteria and European Federation of the Neurological Societies’ guidelines.8–20 Even if not expressed in Beers criteria, it is noteworthy that 26 patients did not receive a gastroprotective drug when indicated. Moreover, on grouping PIPs according to their ATC code, a prevalence of drugs belonging to the CNS system was found (347; 67.8% of all PIPs), followed by the cardiovascular system (85; 16.6% of all PIPs), blood and hematopoietic organs (60; 11.7% of all PIPs), respiratory system (10; 1.9% of all PIPs), gastrointestinal system (8; 1.6% of all PIPs), and musculoskeletal system (2; 0.4% of all PIPs). Finally, it should be mentioned that the Beers criteria, in addition to PIPs, list a series of drugs to be used with caution in the elderly. In this regard, we found that, in the whole sample, ~11% of prescriptions referred to drugs that needed to be used with caution in the elderly (Table 4). In particular, the cases most frequently detected in the present survey were: cardioaspirine for people aged ≥80 years (184), selective serotonin reuptake inhibitors (152), and vasodilators (77).
Characteristics of the patients receiving PIPs
Comparing the characteristics of the patients coping with PIPs versus those who did not receive a PIP, the mean age of the two groups was superimposable (80.4±7.3 and 80.2±7.4 years, respectively) and females seemed to receive more PIPs than males (46.3% vs 40.6%), even if the difference did not reach a statistical significance (F[1,858] =1.348; P=0.246). The probability of running the risk of receiving a PIP was strictly dependent on the number of prescriptions; indeed, within the group of patients coping with at least one PIP, a statistically significant correlation was found between the total sum of all prescriptions and the number of PIPs (r=0.400; P<0.001). Moreover, a significant correlation was observed between the number of illnesses and the probability of receiving a PIP (r=0.181; P<0.001); this result was also confirmed by data showing that the number of illnesses per patient was significantly different between patients who are receiving/not receiving at least one PIP (F[1,859] =20.897; P<0.001). In order to find the patients’ or drug’s characteristics that were more related to the inappropriate prescribing of a medication, various univariate analyses (ANOVA) were performed (Table 5), with the “number of PIPs” as dependent variable. Patients with a MMSE score ≤18 received more PIPs than the other subjects (F[1,855] =10.216; P<0.05), although no significant difference in the number of illnesses was observed (F[1,858] =3.103; P=0.078). A similar difference was found when examining the Geriatric Depression Scale scores: patients with a Geriatric Depression Scale score ≥8 considered depressed and received more PIPs than nondepressed individuals (F[1,591] =14.277; P<0.001). As expected, polypharmacy and the use of psychotropic drugs were all statistically significant, even considering antipsychotic, anxiolytic, and antidepressant drugs separately. No differences were found when considering age, sex, multiple comorbidities (even if this last parameter was close to significance: P=0.068, mirroring the weak correlation, previously described, between illnesses and the amount of PIPs) and using the admission to hospital or emergency department as independent variables.
Sedative load
The SL due to CNS-active drugs was calculated for each patient and the relative data are reported in Table 6. In the whole sample, 209 patients (24.1%) had a SL ≥3, which is considered elevated. The values were similar when considering MC and VS subgroups (22.5% and 21.0%, respectively), but they were quite different with respect to NH subjects, where 34.4% of patients had a SL ≥3. This finding is in agreement with the data on the SL per patient; indeed, a statistically significant more elevated SL was detected in NH patients (SL mean: 1.97) in comparison to the other subgroups (SL mean in MC patients: 1.47 and VS subjects: 1.51) (F[2,857] =5.155; P<0.05).
  | Table 6 SL and AL scores |
Anticholinergic load
The AL was calculated for each patient in the three groups and the relative data are reported in Table 6. A total of 10.7% of the whole sample, corresponding to 92 patients, had an AL ≥3, which was considered inappropriate.17 Interestingly, the majority of these patients belonged to the NH group (46 patients). A significant difference among the subgroups was obtained following an ANOVA performed with the “anticholinergic load” as dependent variable and “group” as factor (F[2,843] =19.464; P<0.001). Post hoc comparisons revealed a significant difference between NH patients and the other two subgroups, but not between MC and VS patients (mean AL in NH patients was 1.62; MC patients was 0.92; and VS subjects was 0.83).
Drug interactions
In addition to the previously described issues, we reasoned that increasing the amount of prescriptions might also lead to a more elevated possibility of drug–drug interactions. Indeed, several interactions of potential clinical interest were observed in the whole sample. We detected 266 interactions, even up to five in three patients, which would have required a change in the prescribed drugs and 3,475 interactions of minor concern, requiring attention. The most frequent potential interactions (shown in Table 7) were those between allopurinol and ramipril (25 cases), citalopram and esomeprazole (16 cases), amiodarone and furosemide (12 cases). In order to compare the three subgroups, an ANOVA was performed with the “number of severe interactions” as a dependent variable and “group” as a factor, obtaining statistically significant values (F[2,843] =3.354; P<0.05). Post hoc analyses, however, did not allow observation of a statistical significance among the different subgroups.
  | Table 7 Most frequent drug interactions |
Antidementia drugs
In the whole sample, only 20 patients received an acetylcholinesterase inhibitor (18) or memantine (two). Of these, eight were NH patients and 12 were MC patients. Examining the clinical diagnosis of each subject, 64 patients had Alzheimer’s disease or mixed dementia, which, according to recent guidelines,21 should have been treated with antidementia drugs; among these patients, only four received a pharmacological therapy for dementia. Moreover, 119 had a diagnosis of generic cognitive impairment, being therefore potential candidates for this type of treatment.
Discussion
Inappropriateness of prescription represents a complex topic and an issue of major concern for the health care system. In the light of paucity of studies investigating this phenomenon in the Italian population, in the present research, we analyzed the prescriptions of 860 elderly subjects living in a small town of Lombardy, Italy.
The investigation revealed that nearly half of them (44.4%) received at least one PIP; NH patients were more exposed, with 57.7% of them dealing with at least one or up to five PIPs. Our data are quite different from the ones obtained by other Italian and European studies. Concerning NH residents, in the study of Ruggiero et al,22 the prevalence was 35%. With regard to outpatients, Maio et al23 found a prevalence of 18%, while a retrospective cohort study24 reveals that 25.8% of outpatients received at least one prescription for any potentially inappropriate drugs; of these, 15.2% were found to receive prescriptions for medications always identified as inappropriate. In case of outpatients, however, the data were not directly comparable because of the use of different criteria for determining medication inappropriateness. Conversely, our results are very similar to the ones obtained by Davidoff et al,25 who applied the Beers criteria to a large population study performed in US, found that 42.6% of persons had dealt with at least one PIP. With regard to NH patients, on the other hand, the high amount of PIPs may be due to the elevated number of demented patients (89.7%), with 42.2% (corresponding to 60 patients) having a diagnosis of severe dementia. In all the subgroups, the most frequent inappropriate prescription concerned CNS-active drugs. This is largely due to the long-term use of benzodiazepines, which are considered inappropriate in the elderly, as they increase the risk of cognitive impairment and falls. However, another reason possibly explaining this phenomenon is the high number of demented patients treated with antipsychotic drugs before being treated with nonpharmacological therapies, as suggested by the international guidelines.8–20 Of concern are the data relative to SL and AL; to our knowledge, there are very few Italian studies examining these parameters in the elderly. Notably, both these parameters, if elevated, can have adverse consequences related to cognitive and physical functioning.13,14 One-fourth of our sample (24.1%) had an elevated SL score, in particular the NH subgroup, where there was a high consumption of psychotropic drugs. The percentage of patients with a high AL was, on the other hand, 10.7%. In addition to these indicators, we found 266 potentially relevant drug–drug interactions in our sample. Drug interactions are responsible for up to 2–3% of hospital admissions26 and for this reason many campaigns to improve appropriateness in general practice have been implemented during the last years.27 In our survey, medication underprescribing was observed in the case of antidementia drugs: only 20 (10.9%) out of 183 patients, who were potential candidates for this treatment, received a cholinesterase inhibitor or memantine. Our percentage is lower than the one reported by Gruber-Baldini et al,28 who found a prevalence of treated dementia patients of 24.7%. The reasons for undertreatment of dementia are unknown, but a negative attitude versus antidementia drugs is widespread among general practitioners. Multimorbidity, polypharmacy, agism, fear of adverse events, and economic problems may contribute to underprescription of antidementia drugs. Selected interventions may help to improve the quality of prescriptions, as suggested by Cherubini et al,29 not only for antidementia drugs, but also for drugs prescription in general. Indeed, adequate education and interventions within the domain of geriatric pharmacology can improve the recognition of drug-drug interaction and help to prevent adverse drug reactions. To this aim, various strategies can be adopted, such as the insertion of formal surveillance systems and built-in computer programs alerting in case of PIPs, and the involvement of a multidisciplinary geriatric team reviewing therapies. In addition, a number of educational actions can be taken within the hospitals. For example, as suggested by Petrovic et al30 for older adults, a global geriatric assessment may be helpful in improving the quality of prescribing.
Limitations
The present study has some limitations. First, the use of Beers’ criteria may be criticized as, according to some authors,31 they are too focused on drugs available in the US market rather than in Europe. Nevertheless, using the revised criteria, as published by the American Geriatrics Society,8 we encountered only a few cases that needed discussion. The second limitation is related to the fact that we collected the medications indicated by the participants at the time of answering the questionnaire, therefore we cannot be sure about their effective pharmacological therapy, due to possible mistakes in reporting the correct therapy. Nevertheless, this is the only way to collect data. The third limitation is related to the fact that the subgroups are not similar in terms of sample size, and this aspect makes it difficult to compare them. However, our results highlight the existence of key areas within the current health practices that may be potentially improved to favor a more correct medication prescription.
Compliance with ethical standards
The study was not submitted to the ethical committee as it was based on an anonymous questionnaire reporting only the age and sex of the patient. The survey was not aimed to change clinical management of the individual patients. In addition the questionnaires were not coded, therefore the collected data following the input in the database cannot be associated with the individual patients. Formal informed consent was obtained for all patients, for patients with dementia consent from a relative was obtained. All principles outlined in the Declaration of Helsinki were followed.
Acknowledgment
We thank Piacenza and Vigevano foundations for the financial support.
Disclosure
The authors report no conflicts of interest in this work.
References
Laroche ML, Charmes JP, Nouaille Y, Nouaille Y, Picard N, Merle L. Is inappropriate medication use a major cause of adverse drug reactions in the elderly? Br J Clin Pharmacol. 2007;63(2):177–186. | ||
Page RL, Ruscin JM. The risk of adverse drug events and hospital-related morbidity and mortality among older adults with potentially inappropriate medication use. Am J Geriatr Pharmacother. 2006;4(4): 297–305. | ||
Poudel A, Hubbard RE, Nissen L, Mitchell C. Frailty: a key indicator to minimize inappropriate medication in older people. QJM. 2013;106(10):969–975. | ||
O’Connor MN, Gallagher P, O’Mahony D. Inappropriate prescribing: criteria, detection and prevention. Drugs Aging. 2012;29(6): 437–452. | ||
Dimitrow MS, Airaksinen MS, Kivelä SL, Lyles A, Leikola SN. Comparison of prescribing criteria to evaluate the appropriateness of drug treatment in individuals aged 65 and older: a systematic review. JAGS. 2011;59(8):1521–1530. | ||
Beers MH, Ouslander JG, Rollingher I, Reuben DB, Brooks J, Beck JC. Explicit criteria for determining inappropriate medication use in nursing home residents. UCLA Division of Geriatric Medicine. Arch Intern Med. 1991;151(9):1825–1832. | ||
Fick DM, Cooper JW, Wade WE, Waller JL, Maclean JR, Beers MH. Updating the Beers criteria for potentially inappropriate medication use in older adults: results of a US consensus panel of experts. Arch Intern Med. 2003;163(22):2716–2724. | ||
American Geriatrics Society. American geriatric society updated Beers criteria for potentially inappropriate medication use in older adults: the American Geriatrics Society 2012 Beers Criteria Update Expert Panel. J Am Geriatr Soc. 2012;60(4):616–631. | ||
Gallagher P, Ryan C, Byrne S, Kennedy J, O’Mahony D. STOPP (Screening tool of older person’s prescriptions) and START (Screening tool to alert doctors to right treatment). Consensus validation. Int J Clin Pharmacol Ther. 2008;46(2):72–83. | ||
Hanlon JT, Lindblad CI, Hajjar ER, McCarthy TC. Update on drug-related problems in the elderly. Am J Geriatr Pharmacother. 2003;1(1):38–43. | ||
Maher RL, Hanlon J, Hajjar ER, McCharty TC. Clinical consequences of polypharmacy in elderly. Expert Opin Drug Saf. 2014;13(1):57–65. | ||
Hines LE, Murphy JE. Potentially harmful drug–drug interactions in the elderly: a review. Am J Geriatr Pharmacother. 2011;9(6):364–377. | ||
Linjakumpu T, Hartikainen S, Klaukka T, Koponen H, Kivela SL, Isoaho R. A model to classify the sedative load of drugs. Int J Geriatr Psychiatry. 2003;18(6):542–544. | ||
Fox C, Smith T, Maidment I, et al. Effect of medications with anti-cholinergic properties on cognitive function, delirium, physical function and mortality: a systematic review. Age Ageing. 2014;43(5): 604–615. | ||
Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12(3):189–198. | ||
Measso G, Cavarzeran F, Zappalà G, et al. Il Mini Mental State Examination: studio normativo di un campione random della popolazione italiana. Dev Neuropsychol. 1993;9(2):77–85. | ||
Carnahan RM, Lund BC, Perry PJ, Pollock BJ, Culp KR. The anticholinergic drug scale as a measure of drug-related anticholinergic burden. Associations with serum anticholinergic activity. J Clin Pharmacol. 2006;46(12):1481–1486. | ||
Baxter K. Stockley’s Drug Interactions, 9th ed. London: Pharmaceutical Press; 2010. | ||
IBM SPSS. Statistical Package for Social Science 15.0 for Windows. Chicago, IL. 2006. | ||
Waldemar G, Dubois B, Emre M, et al. Recommendations for the diagnosis and management of Alzheimer’s disease and other disorders associated with dementia: EFNS guideline. Eur J Neurol. 2007;14(1):e1–e26. | ||
Bond M, Rogers G, Peters J, et al. The effectiveness and cost-effectiveness of donepezil, galantamine, rivastigmine and memantine for the treatment of Alzheimer’s disease (review of technology appraisal no. 111): a systematic review and economic model. Southampton: Health Technol Assess. 2012;16(21):1–470. | ||
Ruggiero C, Lattanzio F, Dell’Aquila G, Gasperini B, Cherubini A. Inappropriate drug prescriptions among older nursing home residents: the Italian perspective. Drugs Aging. 2009;26(1):15–30. | ||
Maio V, Yuen EJ, Novielli K, Smith KD, Louis DZ. Potentially inappropriate medication prescribing for elderly outpatients in Emilia Romagna, Italy. Drugs Aging. 2006;23(11):915–924. | ||
Herrera MS, Del Canale MD, Brianti MD, Fabi MD, Louis MS. Improving the appropriateness of prescribing medicine in the elderly: a comprehensive approach in the Local Health Unit of Parma, Italy. Population Health Matters (Formerly Health Policy Newsletter). 2010;23(2):3. | ||
Davidoff AJ, Miller GE, Sarpong EM, Yang E, Brandt N, Fick D. Prevalence of potentially inappropriate medication use in older adults using the 2012 Beers criteria. JAGS. 2015;63(3):486–500. | ||
Tragni E, Casula M, Pieri V, et al. Prevalence of the prescription of potentially interacting drugs. PLoS One. 2013;8(10):e78827. | ||
Raschi E, Piccinni C, Signoretta V, et al. Clinically important drug-drug interactions in poly-treated elderly outpatients: a campaign to improve appropriateness in general practice. Br J Clin Pharmacol. 2015;80(6):1411–1420. | ||
Gruber-Baldini AL, Stuart B, Zuckerman IH, Simoni-Wastila L, Miller R. Treatment of dementia in community-dwelling and Institutionalized Medicare Beneficiaries. JAGS. 2007;55(10):1508–1516. | ||
Cherubini A, Corsonello A, Lattanzio F. Underprescription of beneficial medicines in older people: causes, consequences and prevention. Drugs Aging. 2012;29(6):463–475. | ||
Petrovic M, van der Cammen T, Onder G. Adverse drug reactions in older people: detection and prevention. Drugs Aging. 2012;29(6):453–462. | ||
Lam MP, Cheung BM. The use of STOPP/START criteria as a screening tool for assessing the appropriateness of medications in the elderly population. Expert Rev Clin Pharmacol. 2012;5(2):187–189. |
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.






