Back to Journals » International Journal of Chronic Obstructive Pulmonary Disease » Volume 14
Downregulated level of insulin in COPD patients during AE; role beyond glucose control?
Authors Ruan W, Yan C, Zhu H, Wang S , Jia X , Shao L, Xu Z, Ying K
Received 5 December 2018
Accepted for publication 11 June 2019
Published 15 July 2019 Volume 2019:14 Pages 1559—1566
DOI https://doi.org/10.2147/COPD.S197164
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Chunxue Bai
Wenjing Ruan,1 Chao Yan,1 Huiqi Zhu,1 Shuwen Wang,1 Xuan Jia,1 Lingyan Shao,1 Zhihao Xu,2 Kejing Ying1
1Department of Respiratory Diseases, Sir Run Run Shaw Hospital, School of Medicine, Zhejiang University, Hangzhou, People’s Republic of China; 2Department of Respiratory Diseases, The Fourth Affiliated Hospital, School of Medicine, Zhejiang University, Yiwu, People’s Republic of China
Objective: The purpose of this study was to explore the insulin level in the serum of chronic obstructive pulmonary disease (COPD) patients during acute exacerbation (AE).
Methods: The study population consisted of 22 acute exacerbation chronic obstructive pulmonary disease (AECOPD) patients, 20 COPD patients and 20 healthy controls. Fasting blood glucose, insulin and serum lipid levels were measured. After the patients recovered from AE, the insulin and glucose levels were also analyzed.
Results: Insulin level, glucose level and homeostasis model assessment of insulin resistance (HOMA-IR) of AECOPD patients were higher than healthy controls (7.19±6.02 vs 3.28±1.09 μIU/mL, P<0.05, 126.61±50.92 vs 96.21±12.66 mg/dL, P<0.05, 2.66±2.72 vs 0.78±0.26, P<0.05). For stable COPD patients, the insulin level, glucose level and HOMA-IR were 6.52±2.56 μIU/mL, 95.58±11.44 mg/dL, and 1.52±0.53, respectively. The triglyceride (TG) level, total cholesterol (CHOL) level and low-density lipoprotein cholesterol (LDL-CHOL) level were decreased in AECOPD patients (0.78±0.33 vs 1.05±0.35 mmol/L, P<0.05, 3.88±0.72 vs 4.49±0.7 mmol/L, P<0.05, 2.01±0.59 vs 2.59±0.58 mmol/L, P<0.05). When the patients had recovered from AE, the insulin levels increased (10.67±6.22 vs 7.12±6.19 μIU/mL, P<0.05) and the glucose levels decreased (122.69±41.41 vs 134.08±53.19 mg/dL, P>0.05).
Conclusion: A high insulin level and a high HOMA-IR status in COPD patients were demonstrated. Downregulated levels of insulin during AE compared with the convalescent state were detected, while the variation in the glucose level was not as great as expected, indicating a potentially important role for insulin in AECOPD.
Keywords: COPD, insulin, acute exacerbation
Introduction
Chronic obstructive pulmonary disease (COPD) is characterized by persistent airflow limitation and is usually progressive. Exacerbations contribute to the overall severity in the individual patient. Acute exacerbation of chronic obstructive pulmonary disease (AECOPD) is a growing clinical problem responsible for a substantial worldwide health burden. The diagnosis of AE depends on the acute change in the patient’s symptoms, including productive cough and dyspnea. The key mechanism behind AE is acute inflammation, including airway inflammation and systemic inflammation. The major medications for AECOPD are bronchodilators, corticosteroids and antibiotics. The broad-spectrum anti-inflammatory steroids carry a risk of side-effects. Two-thirds of patients admitted to intensive care units with AECOPD are treated with high doses of corticosteroids that are associated with poorer outcomes and more frequent adverse effects.1 Treatment of patients suffering from AECOPD is still challenging, especially when steroids and other current agents are less effective.
There is an urgent need to explore the pathophysiological mechanism of AECOPD, which will give future clues for new anti-inflammation target therapies. It has been reported that many molecules are upregulated during the AE process. Chen et al conducted a systematic review. Interleukin-6, C-reactive protein (CRP), and tumor necrosis factor-alpha were the most frequently studied biomarkers.2 In our previous studies, we found that levels of serum insulin-like growth factor binding protein 7 (IGFBP7) level was raised during AECOPD. Similar to the expression pattern of CRP, the IGFBP7 levels reduced after convalescence from AE.3 IGFBP7 belongs to IGFBP superfamily, which are a group of homologous proteins representing an important link between insulin and insulin-like growth factor (IGF) systems. IGFBP7 binds IGF at least 5- to 25-fold lower, and insulin 500-fold higher than conventional IGFBP1-6.4 Ye et al demonstrated that insulin-like growth factor 1 (IGF1) level was decreased in AECOPD serum.3,5 Insulin, IGF1 and IGFBP form the important insulin/IGF1/IGFBP network. However, the role of insulin in AECOPD was not clear.
Insulin is a peptide hormone produced by beta cells of the pancreatic islets. It is the main anabolic hormone of the body. Insulin participates in not only glucose control, but also many other physiological and pathophysiological processes, including lipid metabolism and the inflammation. We hypothesize that insulin functions in the AE process of COPD. In this study, we wanted to determine the level of insulin in AECOPD patients, as well as the levels of glucose and lipid. Through this approach, it is hoped to achieve a greater clarification of the role of the insulin/IGF1/IGFBP7 network in AECOPD.
Materials and methods
Definition of AECOPD
COPD was diagnosed using the Global Initiative for Chronic Obstructive Lung Disease (GOLD) criterion of a post-bronchodilator FEV1/FVC ratio of 70% of that predicted. Exacerbations were defined as a change in baseline dyspnea, cough or sputum production that was greater than the normal day-to-day variation and sufficient to warrant a change in management. Selected COPD patients were excluded if they were diagnosed with active pulmonary tuberculosis, cancer, interstitial lung disease, or clinically significant bronchiectasis.
Study population
From December 2011 to August 2014, twenty-two hospitalized AECOPD patients were enrolled, of which 4 patients were combined with diabetes. Twenty age- and sex-matched stable COPD patients and twenty age- and sex-matched healthy subjects were also enrolled, of which no one were combined with diabetes. Healthy control subjects were recruited from the general population and had normal pulmonary function (FEV1/FVC 70% and FEV1 80% of predicted). All patients gave informed consent to participate in the present study, which was approved by the Medical Ethical Committee of the Sir Run Run Shaw Hospital. Written, informed patient consent was obtained, and the study was conducted in accordance with the Declaration of Helsinki.
Study design
A venous blood sample was obtained from patients with AECOPD within 24 h of hospitalization. Follow-up venous blood samples were obtained on the discharge day following recovery from the exacerbation. Decisions to discharge were based on GOLD guidelines,6 according to which a subject could leave the hospital when he was able to use inhaled bronchodilators, needed inhalation therapy with short-acting β agonists no more frequently than every 4 h, was able to walk across the room (if previously ambulatory), was able to eat and sleep without being awakened by dyspnea, had been clinically stable for 12–24 h, and had understood completely the correct use of medications, and when the subject and the physician were confident that the subject could manage successfully at home.3 All decisions for treatment and discharge were made by the attending physicians of the department, and data on duration of stay were recorded on discharge.
Blood sampling
After a 12 hr overnight fast, all venous samples were collected in evacuated blood collection tubes containing Ethylene Diamine Tetraacetic Acid (EDTA), centrifuged (1500 g, 20 min) within 60 min of collection, and the supernatants stored at −70 °C until required. The separated plasma sample was aliquoted into sterile microcentrifuge tubes (500 μl per vial) and were then stored at −80 °C.
Biochemical measurements
Insulin was detected by chemiluminescence immunoassay (Beckman Coulter DIX800, USA). Glucose was analyzed by the hexokinase method, triglycerides (TG) by the glycerol phosphate oxidase method, total cholesterol (CHOL) by the CHOD-PAP method, low-density lipoprotein cholesterol (LDL-CHOL) by surfactant assay, high density lipoprotein cholesterol (HDL-CHOL) by CAT assay. They were detected by Abbott Architect c16000, USA.
Statistical analyses
Quantitative data were expressed as means±standard deviations (SD). Differences between subject groups were measured by one-way analysis of variance (ANOVA) with post-hoc analysis performed using Tamhane’s test. For the comparison of insulin and glucose level on admission and at discharge, paired Student’s t test was used. P-values <0.05 were deemed to indicate statistical significance. Correlations between continuous variables were assessed by the Spearman partial correlation coefficient (rs). All statistical analyses were performed using SPSS for Windows, version 20.0 (SPSS Inc, Chicago, IL, USA).
Results
Baseline characteristics of AECOPD patients and control subjects
The study included 22 AECOPD patients, 20 stable COPD patients and 20 healthy controls. Baseline characteristics of the patients and the controls were summarized in Table 1.
 |
Table 1 Baseline characteristics of the patients and the controls |
Insulin level, glucose level and homeostasis model assessment of insulin resistance (HOMA-IR) of AECOPD patients were higher than healthy controls
Eighteen AECOPD patients without diabetes were enrolled. Insulin levels in AECOPD patients and COPD patients were higher than that in healthy controls (7.19±6.02 and 6.52±2.56 vs 3.28±1.09 μIU/mL, P<0.05). The glucose levels in AECOPD patients were also higher than that in stable COPD and healthy controls (126.61±50.9 vs 95.58±11.44 and 96.21±12.66 mg/dL, P<0.05). HOMA-IR=fast glucose (mmol/L)×fast insulin (μIU/mL)/22.5. The HOMA-IR in AECOPD patients and stable COPD patients were higher than that in healthy controls(2.66±2.72 and 1.52±0.53 vs 0.78±0.26, P<0.05). The HbA1c levels of the three groups were all below 6.4% (5.77±0.43%, 5.45±0.63%, 5.47±0.41%, Figure 1).
TG, CHOL, LDL-CHOL levels was decreased in AECOPD patients
With regard to serum lipids, levels of TG, CHOL, and LDL-CHOL were decreased in AECOPD patients compared with healthy controls (0.78±0.33 vs 1.05±0.35 mmol/L, P<0.05, 3.88±0.72 vs 4.49±0.7mmol/L, P<0.05, 2.01±0.59 vs 2.59±0.58mmol/L, P<0.05, respectively). The variation of HDL-CHOL levels reached no significance (1.27±0.37 vs 1.2±0.2 mmol/L, P>0.05, Figure 2). While for stable COPD patients, the TG, CHOL, LDL-CHOL and HDL-CHOL levels were 1.15±0.65 mmol/L, 3.51±0.69mmol/L, 1.67±0.42 mmol/L and 1.06±0.27 mmol/L, respectively.
In AECOPD patients, the insulin level increased after recovery
The blood samples of the 22 AECOPD patients were obtained both on the admission day of AE and on the discharge day after recovery. Serum insulin and glucose levels were measured. After recovery from AE, the insulin level increased (10.67±6.22 vs 7.12±6.19 μIU/mL, P<0.05) while the glucose levels decreased (122.69±41.41 vs 134.08±53.19 mg/dL, P>0.05 Figure 3). The variation in glucose level was not as great as expected.
Correlation of the amplitude of insulin variation with the amplitude of glucose variation during AE and after recovery
Using the data of insulin and glucose levels during AE and after recovery, we calculated the variation amplitude of insulin and glucose. We analyzed the correlation of the amplitude of insulin variation (recovery-AE) with the amplitude of glucose variation (AE-recovery). On the basis of the scatter diagram, we observed no significant linear correlation (r=−0.184, P=0.41, Figure 4).
Discussion
Different from hyperinsulinemia and hyperlipidemia in diseases like coronary heart disease and type 2 diabetes,7 we detected hyperinsulinemia and hypolipidemia in COPD patients, indicating the different pathological state of the disease. Insulin regulates the metabolism of carbohydrates, fats and proteins by promoting the absorption of carbohydrates. Also, insulin is one of the central homeostatic hormones with global effects outside the glucose and lipid metabolism pathways. Hyperinsulinemia has been demonstrated to have adverse effects on airway structure and function.8,9 In fact, the possible role of insulin in airway inflammation has already been demonstrated. Inhaled insulin in diabetic patients is associated with a decrease in forced expiratory volume in 1 second (FEV1). Patients with asthma or COPD were excluded from this therapy.10,11 The relation of hypolipemia with COPD has also been explored. A previous study has reported that low CHOL levels were significantly associated with increased mortality after AECOPD.12
We first reported that after recovery from AE, the serum insulin level significantly increased. The glucose level decreased a little, but the variation was not as great as expected. Out of expect, the correlation analysis resulted in negative correlation. The lipid level variation during AE and after convalescence was not significant (data not shown).The phenomenon indicated “the downregulated role” of insulin during AE was beyond glucose and lipid control. Interestingly, the direction and amplitude of the insulin’s variation was not the same for every patient. Future work is needed to explore this inconsistency.
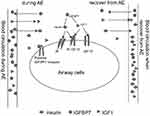
Our finding of the downregulated level of insulin during AE, together with the previous result of increased levels of IGFBP7 and decreased IGF1 levels in the serum of AECOPD patients,3,5 indicated the potential for an “insulin-IGF1-IGFBP7 flow” occurring during AECOPD (Figure 5). The insulin/IGFBP/IGF1 network may serve as a potential target for the development of compounds to better manage AECOPD patients. Interestingly, a previous study has discussed the therapeutic value of various strategies targeting the IGF1 and IGFBP3 signaling pathways for the management of asthma, another chronic airway disease.13 An IGFBP3 mutant that does not bind IGF1 but binds to IGFBP-3R and acts as an IGFBP-3R agonist thus enhances the IGFBP-3R–mediated anti-inflammatory responses.14
Our findings indicate a potentially important role of insulin in AE. According to our findings, we think that the future direction should be toward: 1) The mechanisms of how the insulin/IGF1/IGFBP7 network influences airway elements to produce structural and functional changes in COPD during AE. 2)Whether targeting the hyperinsulinemia and hypolipemia in COPD could reduce the AE frequency. 3) The therapeutic potential of targeting the insulin/IGFBP7/IGF1 network in AECOPD.
Abbreviations list
COPD, chronic obstructive pulmonary disease; AE, acute exacerbation; IGFBP7, insulin-like growth factor binding protein 7; IGF1, insulin-like growth factor 1; TG, triglycerides; CHOL, total cholesterol; LDL-CHOL, low-density lipoprotein cholesterol; HDL-CHOL, high-density lipoprotein cholesterol; HOMA-IR, homeostasis model assessment of insulin resistance.
Acknowledgments
This work was supported by the Zhejiang Provincial Natural Science Foundation of China (LY17H160007, LY16H010003). We thank Dr Brian Eyden (Manchester, UK) for assistance with English language expression.
Author contributions
WR designed the experiments, collected the clinical data, and drafted the manuscript; CY analyzed the data; HZ, SW, and XJ collected the serum samples; LS and ZX collected clinical data; KY conceived and designed the experiments. All authors contributed to data analysis, drafting and revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Kiser TH, Allen RR, Valuck RJ, Moss M, Vandivier RW. Outcomes associated with corticosteroid dosage in critically ill patients with acute exacerbations of chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2014;189(9):1052–1064. doi:10.1164/rccm.201401-0058OC
2. Chen YW, Leung JM, Sin DD. A systematic review of diagnostic biomarkers of COPD exacerbation. PLoS One. 2016;11(7):e0158843. doi:10.1371/journal.pone.0158843
3. Ruan W, Wu M, Shi L, et al. Serum levels of IGFBP7 are elevated during acute exacerbation in COPD patients. Int J Chron Obstruct Pulmon Dis. 2017;12:1775–1780. doi:10.2147/COPD.S132652
4. Yamanaka Y, Wilson EM, Rosenfeld RG, Oh Y. Inhibition of insulin receptor activation by insulin-like growth factor binding proteins. J Biol Chem. 1997;272(49):30729–30734. doi:10.1074/jbc.272.49.30729
5. Ye M, Yu H, Yu W, et al. Evaluation of the significance of circulating insulin-like growth factor-1 and C-reactive protein in patients with chronic obstructive pulmonary disease. J Int Med Res. 2012;40(3):1025–1035. doi:10.1177/147323001204000321
6. Vestbo J, Hurd SS, Agusti AG, et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease: GOLD executive summary. Am J Respir Crit Care Med. 2013;187(4):347–365. doi:10.1164/rccm.201204-0596PP
7. Araki E, Yamashita S, Arai H, et al. Effects of pemafibrate, a novel selective pparalpha modulator, on lipid and glucose metabolism in patients with type 2 diabetes and hypertriglyceridemia: a randomized, double-blind, placebo-controlled, phase 3 trial. Diabetes Care. 2018;41(3):538–546. doi:10.2337/dc17-1589
8. Singh S, Bodas M, Bhatraju NK, et al. Hyperinsulinemia adversely affects lung structure and function. Am J Physiol Lung Cell Mol Physiol. 2016;310(9):L837–L845. doi:10.1152/ajplung.00091.2015
9. Dekkers BG, Schaafsma D, Tran T, Zaagsma J, Meurs H. Insulin-induced laminin expression promotes a hypercontractile airway smooth muscle phenotype. Am J Respir Cell Mol Biol. 2009;41(4):494–504. doi:10.1165/rcmb.2008-0251OC
10. Siekmeier R, Scheuch G. Inhaled insulin–does it become reality? J Physiol Pharmacol. 2008;59(Suppl 6):81–113.
11. Guevara CA. Inhaled insulin for diabetes mellitus. N Engl J Med. 2007;356(20):2106–2107. author reply 2108.
12. Fruchter O, Yigla M, Kramer MR. Lipid profile and statin use: the paradox of survival after acute exacerbation of chronic obstructive pulmonary disease. Am J Med Sci. 2015;349(4):338–343. doi:10.1097/MAJ.0000000000000435
13. Lee H, Kim SR, Oh Y, Cho SH, Schleimer RP, Lee YC. Targeting insulin-like growth factor-I and insulin-like growth factor-binding protein-3 signaling pathways. A novel therapeutic approach for asthma. Am J Respir Cell Mol Biol. 2014;50(4):667–677. doi:10.1165/rcmb.2013-0397TR
14. Lee YC, Jogie-Brahim S, Lee DY, et al. Insulin-like growth factor-binding protein-3 (IGFBP-3) blocks the effects of asthma by negatively regulating NF-kappaB signaling through IGFBP-3R-mediated activation of caspases. J Biol Chem. 2011;286(20):17898–17909. doi:10.1074/jbc.M111.231035
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.