Back to Journals » OncoTargets and Therapy » Volume 13
Down-Regulation of Hypoxia-Inducible Factor-1α and Downstream Glucose Transporter Protein-1 Gene by β-elemene Enhancing the Radiosensitivity of Lung Adenocarcinoma Transplanted Tumor
Authors Wu W , Hu Z, Zhao Q , Zhang X, Zhang H, Wang H, Xue W, Yu L, Duan G
Received 12 August 2020
Accepted for publication 29 October 2020
Published 13 November 2020 Volume 2020:13 Pages 11627—11635
DOI https://doi.org/10.2147/OTT.S275956
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Leo Jen-Liang Su
Wenbo Wu,1,2,* Zhonghui Hu,1,3,* Qingtao Zhao,1,* Xiaopeng Zhang,1 Hua Zhang,1 Huien Wang,1 Wenfei Xue,1 Lei Yu,1 Guochen Duan4
1Department of Thoracic Surgery, Hebei General Hospital, Shijiazhuang, People’s Republic of China; 2Graduate School of Hebei North University, Zhangjiakou, People’s Republic of China; 3Graduate School of Hebei Medical University, Shijiazhuang, People’s Republic of China; 4Department of Thoracic Surgery, Hebei Children’s Hospital, Shijiazhuang, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Guochen Duan
Department of Thoracic Surgery, Hebei Children’s Hospital, No. 133, Jianhua Main Street South, Yuhua District, Shijiazhuang 050000, People’s Republic of China
Tel +86 13513380703
Email [email protected]
Purpose: To study the effect of β-elemene on the radiosensitivity of A549 cell xenograft tumor and potential mechanisms by which β-elemene regulates the expression of hypoxia-inducible factor-1α (HIF-1α) and glucose transporter protein-1 (GLUT-1).
Methods: Using an A549 cell transplantation tumor model with male nude mice, we studied the effect of β-elemene on the radiosensitivity of non-small cell lung cancer (NSCLC). The expression of HIF-1α and GLUT-1 was detected by real-time PCR, Western blotting and immunohistochemistry. The relationship between the radiosensitivity of β-elemene and the expression of HIF-1α and GLUT-1 was analyzed.
Results: β-elemene and radiotherapy intervened in the growth of transplanted tumors in varying degrees. The enhancement factor (EF=2.44> 1) was calculated; β-elemene at 45 mg/kg had the most significant enhanced effect on radiosensitivity. When β-elemene was used in combination with radiation, the expression of HIF-1α and GLUT-1 was significantly decreased, and there was a positive correlation between the two genes.
Conclusion: β-elemene exhibits a radiosensitizing effect on A549 cell xenograft tumor. The underlying molecular mechanism is probably associated with the down-regulation of HIF-1α and GLUT-1 expression, suggesting that β-elemene may directly or indirectly inhibit the expression of HIF-1α and GLUT-1. There is a positive significant correlation between expression of HIF-1α and GLUT-1. HIF-1α and downstream GLUT-1 could be used as a new target for the radiosensitization of NSCLC.
Keywords: β-elemene, radiosensitivity, transplanted tumor, HIF-1α, GLUT-1
Introduction
In the past half-century, the morbidity and mortality of cancer are increasing, and cancer has become the leading cause of death in China.1 Radiotherapy is one of the major methods of the treatment of malignant tumors, which has been widely used in lung cancer treatment. In recent years, the combination of radiation and sensitizers has become a hot topic in lung cancer research. Radiotherapy such as ionizing radiation target and kill tumor tissue, but normal tissue can also be damaged, resulting in toxicity.2 Therefore, to reach a higher therapeutic effect at lower radiation doses will be beneficial to the treatment of cancer patients.
Elemene, extracted from Rhizoma Zedoariae, is a terpene compound with an immunomodulatory effect, and the radiosensitization of β-elemene has been confirmed as the main anti-tumor component of elemene.3 Although it has multiple effects such as the inhibition of tumor angiogenesis, promotion of tumor cell G-M phase arrest, induction of tumor cell apoptosis, and repairment of Deoxyribonucleic acid (DNA) double-strand damage,4–6 its sensitizing mechanism still remains poorly understood.
Hypoxia is one of the basic characteristics of the solid tumor microenvironment. Studies have shown that hypoxia is the initiating factor of tumor malignant transformation, which can promote its growth, infiltration and metastasis, and at the same time induce tumor cells to be resistant to radiotherapy and chemotherapy.7,8 Hypoxia-inducible factor-1 (HIF-1), discovered in 1992,9 is a cytokine that is widely present in human and animal cells under hypoxic conditions. It is expressed in a large quantity of tumors as an important role in information, metastasis, apoptosis and drug resistance.10 HIF-1 is composed of α and β subunit. The expression level of HIF-1α protein is highly regulated by intracellular oxygen concentration, while HIF-1β is not affected by oxygen concentration. In the microenvironment of tumor hypoxia, tumor cells maintain their oxygen and metabolic stability through the high expression of HIF-1α, and promote their own growth and metastasis. HIF-1α may be an important sign of rapid tumor proliferation, invasion and deterioration.11 Therefore, reducing the expression of HIF-1α has become a major focus of current non-small cell lung cancer (NSCLC) treatment research.
As the most important member of glucose transporter, glucose transporter protein-1 (GLUT-1) contains 492 amino acids and which consists of 12 hydrophobic transmembrane α-helical domains, two charged inner and outer regions.12 The expression of GLUT-1 in malignant tumors is significantly higher than that in benign tissues,13 Moreover, the expression of GLUT-1 is increased from the early stage of cell carcinogenesis, which is closely related to the occurrence and development of many malignant tumors, and can be used as an early diagnostic marker.14,15 A meta-analysis of 1,665 patients with NSCLC showed that the expression of GLUT-1 was negatively correlated with survival rate, and its overexpression could be used as a malignant biomarker with worse prognosis in NSCLC.16 GLUT-1 is one of the main downstream target genes of HIF-1α.17 HIF-1α can not only increase the production of non-mitochondrial pathway adenosine triphosphate (ATP) by regulating GLUT-1 but also bind to the promoter of Pyruvate Dehydrogenase Kinase 3 (PDK3) and induce its expression, weaken the mitochondrial respiratory chain and increase glycolysis, indicating that HIF-1α/GLUT-1 pathway is important in glycolysis of tumor cells.18
In a hypoxic microenvironment, the GLUT-1 gene binds to the DNA binding site on HIF-1α, and GLUT-1 transcription and expression are significantly increased.19,20 Moreover, the high expression of GLUT-1 can increase the radiation resistance of tumor by cooperating with HIF-1α.21 These observations suggest that inhibiting the expression of HIF-1α and downstream GLUT-1 may be a new target for radiosensitization of β-elemene. In this study, we investigated the effects of β-elemene on the growth of transplanted tumor of human lung adenocarcinoma A549 cell in nude mice under different intervention conditions, and studied the anti-tumor mechanism of radiosensitization of β-elemene.
Materials and Methods
Cell Line and Culture
The human lung cancer A549 cell line was purchased from Procell Life Science& Technology Co., Ltd, Wuhan, China. A549 cells were cultured as a monolayer in dulbecco’s modified eagle medium (DMEM) (HyClone, AE29427345) with 10% fetal bovine serum (FBS) (Every Green, 11011–8611) at 37°C in a humidified atmosphere with 5% CO2 in the CO2 incubator (Panasonic, MCO-18AC). The medium was removed when the cells covered 80–90% of the bottom of the glass culture flacks (CORNING, 430639). Cells were rinsed twice by phosphate buffer saline (PBS) and digested by lysis buffer containing 0.25% trypsin (Solarbio, T8150). The digestion was finished by adding the medium with FBS when cells shrank and turned round observed with an invert microscope (OLYMPUS, IX71). After centrifugation with Centrifuge (Eppendorf, 5415D) and resuspension with DMEM containing 10% FBS, the cells were subcultured in 2–4 flasks. Cells in the exponential growth phase were rinsed by DMEM and resuspended by PBS after digestion with 0.25% trypsin and centrifugation. Concentration of cells was adjusted to 5×106/mL.
Animals and Tumor Model
Male BALB/c-nude mice aged 4–6 weeks with a body weight of 16–20 g were purchased from the Beijing Vital River Laboratory Animal Technology Co. Ltd, Beijing, China. The animals were housed under controlled conditions with a room temperature of 21–23°C and humidity of 40–60%. All the animals were allowed to freely access food and water in specific pathogen-free conditions. All animals (n=20) were injected with human lung cancer cell A549 (200μL, 5×106/mL) on the lateral side of the right hind limb. The facilities and the protocol for the experiments were consistent with the regulations of animal use for biomedical experiments issued by the Ministry of Science and Technology of China and approved by the Ethics Review Committee for Animal Experimentation of Hebei Medical University.
Xenograft Treatment
On the 10th day after inoculation, tumor volumes reached the required size (around 75 mm3). Twenty nude mice were randomly divided into four groups: normal saline group (control group), β-elemene group (elemene group), β-elemene combined with radiotherapy group (combination group) and normal saline combined with radiotherapy group (radiotherapy group), with five mice in each group. β-elemene dose was 45 mg/kg. For radiotherapy, nude mice were anesthetized with chloral hydrate and fixed on a foam plate. The tumors of hind limbs were exposed in the target area of radiotherapy. Radiation was delivered by 6 MeV electron beams from a linear accelerator (ELEKTA) at a single dose of 5 Gry. The tumor measurement started on the 4th day after radiotherapy. The longest diameter (a) and the shortest diameter (b) were measured every two days by vernier caliper purchased from Guilin Measuring & Cutting Tool Works, Guilin, China. The volume of the transplanted tumor was calculated by the approximate volume formula V=ab2/2.22
Sensitization Coefficient Calculation and Curative Effect Judgment
The volume doubling time of tumor was observed in each group. The absolute tumor growth retardation time (AGD) and standardized tumor growth delay time (NGD) were calculated: AGD = volume doubling days in radiotherapy group (TR) - volume doubling days in control group (TC); NGD= volume doubling days in combination group (TL) - volume doubling days in elemene group (TG). The enhancement factor (EF) >1 revealed the curative effect of the tumors.23
Real-Time PCR Analysis of HIF-1α and GLUT-1 mRNA
Total RNA from tumor tissues (five from each group) were isolated using Trizol reagent (Invitrogen, Cat#15596-026) according to the manufacturer’s instructions. Two μL of RNA was used, and the quality and concentration of RNA were measured with BioPhotometer (Eppendorf) (A260/A280 is between 1.72 and 2.01, A260/A230 is between 1.97 and 2.31). The TIANScript RT KIT (Cat#KR104-02) was obtained from TIANGEN BIOTECH Co., LTD, Beijing, China. The RT-PCR was carried out using SYBR Green SuperRealPreMix Plus (TIANGEN) and CFX connectTM (BIO RAD) as previously described.24–26 RT-PCR was performed for 40 cycles of 95°C for 15 s and 60°C for 1 min followed by thermal denaturation. The messenger RNA (mRNA) levels of HIF-1α and GLUT-1 were normalized by β-actin. The amplification results for RT-PCR were calculated by using the 2(-Delta Delta C(T)) Method.27 The primers used for HIF-1α were 5ʹ-AGTGTACCCTAACTAGCCG-3ʹ and 5ʹ-CACAAATCAGCACCAAGC-3ʹ, and the primers used for GLUT-1 were 5ʹ-CACTGTGCTCCTGGTTCTGTTCT-3ʹ and 5ʹ-TCGGGTGTCTTGTCACTTTGG-3ʹ.
Western Blot Analysis of HIF-1α and GLUT-1
The primary 100 mg of tumor tissue was rinsed with PBS and normal saline for 2–3 times, an appropriate amount of RIPA lysis buffer was purchased from Beijing Solarbio Science & Technology Co., Ltd. Beijing, China was added. The tissue was lysed on ice for 1 h after blown several times with a pipette to make the lysate fully contact with the cells. The lysates were centrifuged at 4°C for 10 min at 12000 rpm. The supernatant was removed and the protein concentration was measured. The sample loaded in equal amount was separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) was performed by the DYCZ-24DN electrophoresis apparatus (LIUYI Science & Technology Co., Ltd. Beijing, China), followed by transfer to poly-vinylidene fluoride (PVDF) membrane (Millipore, IPVH00010) (45 min for GLUT-1 and 80 min for HIF-1α). The membranes were carefully removed and blocked at room temperature for 2 h. The membranes were incubated with primary antibodies against GLUT-1 (1:1000) and HIF-1α (1:600) at 4°C overnight, then washed three times in PBST for 10 min each. After incubation with secondary antibodies which was diluted 3000 times with 1 × PBST, the membrane was washed with PBST for 10 min 3 times. After chemiluminescence (ECL) color development, the results were observed on Gis software. Anti-β-actin antibody and ECL kit were purchased from Zhongshan Goldenbridge Bio\Technology Co., Ltd., Beijing, China.
Immunohistochemistry Analysis of HIF-1α and GLUT-1
The tumor specimens were fixed in 4% formaldehyde, embedded in paraffin, and cut into 4μm sections for immunohistochemical analysis. The morphological characteristics of the transplanted tumor of human lung adenocarcinoma A549 cells were observed with H&E staining. Immunohistochemical staining of HIF-1αand GLUT-1 was performed by PV method as previously described.28 PV-6001 was purchased from Zhongshan Goldenbridge Bio\Technology Co., Ltd., Beijing, China. Primary antibodies against glut-1 and HIF-1α (BIO-HIGH Technology, Shijiazhuang, China). The staining procedure was carried out in strict accordance with the manufacturer’s instructions of the DAB kit (Maixin Biotech Co, Ltd. Fuzhou, China). The positive protein expression of HIF-1α and GLUT-1 staining in a yellow-brown color was observed under the microscope. The staining results were analyzed by Image-Pro Plus 6.0 software. The measurement area was selected, and the cumulative optical density and average light density were calculated for statistical analysis.
Statistical Analysis
Software SPSS21.0 and GraphPad Prism8.0 were used for statistical analysis of data. Normally distributed measurement data were expressed as the mean ± standard (x ± s), and the single-factor analysis of variance method was used for comparison among multiple groups. Pearson was used for correlation analysis of the mRNA and protein in the experiment. P<0.05 indicates that the difference is statistically significant.
Results
β-Elemene Has a Sensitizing Effect of Radiotherapy
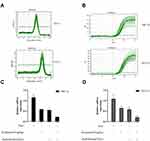
To determine whether β-elemene enhances the sensitivity, the corresponding intervention was carried out on 75 mm3 tumors as described in materials and methods. The tumor volume of each group was measured on the 4th day after radiotherapy as the initial volume, and the changes of transplanted tumor volume were monitored and recorded every other day, the growth curve of transplanted tumor in each group was drawn (Figure 1A), and the doubling time of transplanted tumor in each group was obtained (Figure 1B). As shown in Figure 1, β-elemene has a sensitizing effect (EF is 2.44>1).
β-Elemene and Radiotherapy Decrease the Expression of HIF-1α and GLUT-1 mRNA in Transplanted Tumor
The mRNA expression of HIF-1α and GLUT-1 in tumor samples of the control group and experimental group was measured by RT-PCR, melting curve and amplification curve of HIF-1α and GLUT-1 are shown in Figure 2A and B. Compared with the basal level of the control group, the expression of HIF-1α and GLUT-1 mRNA was significantly reduced in the experimental group (P<0.01) (Figure 2C and D). As shown in Figure 2, in comparison with the control group, the expression of HIF-1α and GLUT-1 mRNA in the elemene group were decreased by 1.98-fold and 1.69-fold, respectively, and the expression levels of HIF-1α and GLUT-1 mRNA in the radiotherapy group were decreased by 2.13-fold and 1.86-fold, respectively (P<0.01). It is worth noting that compared with the control group, HIF-1α mRNA and GLUT-1 mRNA were significantly inhibited in the combined group (5.48-fold and 5.40-fold lower, respectively, P<0.01).
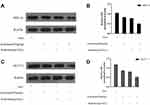
β-Elemene and Radiotherapy Decrease the Expression of HIF-1α and GLUT-1 Protein in Transplanted Tumor
The HIF-1α and GLUT-1 protein of transplanted tumors in each group were also studied by Western blot (Figure 3A and C). Compared with the basic protein expression level of the control group, the expression of HIF-1α and GLUT-1 protein in each experimental group was also inhibited (P<0.01), consistent with their mRNA expression level (Figure 2). Compared with the control group, the elemene group had lower expression of HIF-1α protein (1.25-fold) and GLUT-1 protein (1.39-fold), while the radiotherapy group had a reduction of both protein expression by 1.35-fold and 1.49-fold, respectively (Figure 3B and D). Moreover, the protein levels of HIF-1α and GLUT-1 were significantly decreased when β-elemene was combined with radiotherapy by 2.08-fold and 2.31-fold, respectively (P<0.01, Figure 3B and D).
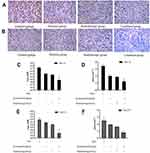
HIF-1α/GLUT-1 Staining by Immunohistochemistry
We further studied the expression of HIF-1α and GLUT-1 in each group of transplanted tumors by immunohistochemistry (Figure 4A and B). The staining results were analyzed by IPP software in which the data of integrated optical density (IOD) and average optical density (AOD) (Figure 4C–F) were consistent with those detected by the Western blot method. The expression of HIF-1α and GLUT-1 in each experimental group decreased in varying degrees compared to control groups (P<0.05). Compared with the elemene group and radiotherapy group, the expression of HIF-1α and GLUT-1 was also significantly lower in the combined group (P<0.05).
Correlation Analysis of mRNA/Protein Expression Between HIF-1α and GLUT-1
The results of RT-PCR and Western blot were analyzed by Pearson correlation analysis, showing that HIF-1α and GLUT-1 mRNA/protein expression were of positive significant correlation (r=0.940, P=0.000<0.01; r=0.946, P=0.000<0.01).
Discussion
β-elemene is the main anti-tumor component of elemene. Because of its broad-spectrum, low toxicity and high efficiency, β-elemene has been widely used in the treatment of many kinds of malignant tumors.29 At present, the accepted dose of radiotherapy for the transplanted tumor in nude mice is 45mg/kg, at which the radiosensitization effect of radiotherapy is moderate.30 In this study, the transplanted tumor model of lung adenocarcinoma A549 cells in mice was successfully established. The sensitization coefficient EF calculated demonstrates that β-elemene has played a sensitizing effect. Moreover, through the growth curve and the changes of transplanted tumor volume, both β-elemene and radiotherapy could effectively inhibit the growth of the transplanted tumors, and the combination of β-elemene and radiation could significantly inhibit the tumor growth. The observations indicate that β-elemene enhances the radiosensitivity of the transplanted tumor.
Previous studies have reported that GLUT-1 is highly expressed in most malignant tumors, but generally low in normal tissues. GLUT-1, as an internal sign of hypoxia,31 promotes the malignant proliferation, invasion, and migration of lung cancer and inhibits apoptosis.32 Moreover, GLUT-1 is regulated by HIF-1. The expression of HIF-1α protein increases in hypoxia and upregulates the expression of downstream gene GLUT-1 protein. Both of the genes are involved in biochemical reactions regulated by hypoxia to adapt to the microenvironment.33,34 Under hypoxia conditions, glycolysis metabolic factors such as GLUT-1 would cooperate with HIF-1α to increase tumor radiation resistance. In extrapulmonary tumors (cervical cancer,35 laryngeal cancer36 and breast cancer37), the expression of GLUT-1 is related to the tumor radiosensitivity, and inhibition of expression could enhance radiosensitivity. Moreover, there is a positive correlation between the expression of HIF-1α and GLUT-1 in rectal cancer,38 breast cancer,39 gastric cancer,40 laryngeal carcinoma,41 tongue cancers,42 and metastatic colorectal cancer.43 HIF-1α is the oxygen-sensitive HIF-1 subunit. In hypoxia microenvironment of the tumors, activated HIF-1α can not only directly bind to the downstream survivin promoter to positively regulate survivin transcription but also bind to the DNA binding site of the downstream GLUT-1 5ʹ enhancer of HIF-1, resulting in a large number of GLUT-1 mRNA transcriptions and expression.19,20,44 Previous studies have shown that β-elemene can down-regulate the expression of HIF-1α to inhibit the transcription and expression of downstream survivin and play an important role in radiosensitization. The above observations suggest that activating HIF-1a may exert a radioprotective effect in tumors. However, to the best of our knowledge, there has been no study reporting the radiosensitization of β-elemene on the transcription and expression of GLUT-1.
In this study, using RT-PCR, Western blot and immunohistochemical analysis, we found that the treatment with β-elemene or radiotherapy reduced the expression of HIF-1α and GLUT-1, which was further decreased when they are combined. This suggests that β-elemene directly or indirectly inhibits the expression of HIF-1α and GLUT-1, thus enhancing the radiosensitivity of the A549 transplantation tumor. We further studied the correlation between the expression of HIF-1α and GLUT-1, demonstrating a significant positive correlation between them (r>0.9, P<0.01). These results suggest the mechanism of radiosensitization of β-elemene is through the inhibition of the expression of downstream gene GLUT-1 by down-regulating the expression of HIF-1α, to exert its anti-tumor effect.
Limitations
The present study has several limitations that require consideration. The number of samples was small because of the current coronavirus epidemic which has brought great inconvenience to our experiment. Therefore, we will increase the sample size in the follow-up studies and focus on the correlation between HIF-lα and GLUT-1 in further in-depth research.
Conclusion
β-elemene decreases radiation-induced expression of HIF-1α and GLUT-1 mRNA/protein, which may enhance radiosensitization. The mechanism involves the down-regulation of HIF-1α and its downstream GLUT-1 gene, which is probably a new target for radiosensitization for the therapy of NSCLC.
Acknowledgments
This work was supported by the Hebei Medical Applied Technology tracking Project (No. G2018008), the Key projects of Medical Science Research in Hebei Province (No. 20180151) and the Scientific Research Program of traditional Chinese Medicine (No. 2019139).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Wei W, Zeng H, Zheng R, et al. Cancer registration in China and its role in cancer prevention and control. Lancet Oncol. 2020;21(7):e342–e349. doi:10.1016/S1470-2045(20)30073-5
2. De Ruysscher D, Niedermann G, Burnet NG, Siva S, Lee AWM, Hegi-Johnson F. Radiotherapy toxicity. Nat Rev Dis Primers. 2019;5(1):13. doi:10.1038/s41572-019-0064-5
3. Zhai B, Zhang N, Han X, et al. Molecular targets of β-elemene, a herbal extract used in traditional Chinese medicine, and its potential role in cancer therapy: a review. Biomed Pharmacother. 2019;114:108812. doi:10.1016/j.biopha.2019.108812
4. Zou K, Liu C, Zhang Z, Zou L. The effect of elemene on lung adenocarcinoma A549 cell radiosensitivity and elucidation of its mechanism. Clinics (Sao Paulo). 2015;70(8):556–562. doi:10.6061/clinics/2015(08)05
5. Wu Z, Wang T, Zhang Y, et al. Anticancer effects of β-elemene with hyperthermia in lung cancer cells. Exp Ther Med. 2017;13(6):3153–3157. doi:10.3892/etm.2017.4350
6. Lee RX, Li QQ, Reed E. β-elemene effectively suppresses the growth and survival of both platinum-sensitive and -resistant ovarian tumor cells. Anticancer Res. 2012;32(8):3103–3113.
7. Choudhry H, Harris AL. Advances in hypoxia-inducible factor biology. Cell Metab. 2018;27(2):281–298. doi:10.1016/j.cmet.2017.10.005
8. Xie H, Simon MC. Oxygen availability and metabolic reprogramming in cancer. J Biol Chem. 2017;292(41):16825–16832. doi:10.1074/jbc.R117.799973
9. Semenza GL, Wang GL. A nuclear factor induced by hypoxia via de novo protein synthesis binds to the human erythropoietin gene enhancer at a site required for transcriptional activation. Mol Cell Biol. 1992;12(12):5447–5454. doi:10.1128/MCB.12.12.5447
10. Wan J, Chai H, Yu Z, et al. HIF-1α effects on angiogenic potential in human small cell lung carcinoma. J Exp Clin Cancer Res. 2011;30(1):77. doi:10.1186/1756-9966-30-77
11. Bhattarai D, Xu X, Lee K. Hypoxia-inducible factor-1 (HIF-1) inhibitors from the last decade (2007 to 2016): a “structure-activity relationship” perspective. Med Res Rev. 2018;38(4):1404–1442. doi:10.1002/med.21477
12. Jurcovicova J. Glucose transport in brain - effect of inflammation. Endocr Regul. 2014;48(1):35–48. doi:10.4149/endo_2014_01_35
13. Důra M, Němejcová K, Jakša R, et al. Expression of Glut-1 in malignant melanoma and melanocytic nevi: an immunohistochemical study of 400 cases. Pathol Oncol Res. 2019;25(1):361–368.
14. Yu M, Yongzhi H, Chen S, et al. The prognostic value of GLUT1 in cancers: a systematic review and meta-analysis. Oncotarget. 2017;8(26):43356–43367. doi:10.18632/oncotarget.17445
15. Zhang B, Xie Z, Li B. The clinicopathologic impacts and prognostic significance of GLUT1 expression in patients with lung cancer: a meta-analysis. Gene. 2019;20(689):76–83. doi:10.1016/j.gene.2018.12.006
16. Tan Z, Yang C, Zhang X, Zheng P, Shen W. Expression of glucose transporter 1 and prognosis in non-small cell lung cancer: a pooled analysis of 1665 patients. Oncotarget. 2017;8(37):60954–60961. doi:10.18632/oncotarget.17604
17. Chang J, Yang B, Zhou Y, et al. Acute methylmercury exposure and the hypoxia-inducible factor-1α signaling pathway under normoxic conditions in the rat brain and astrocytes in vitro. Environ Health Perspect. 2019;127(12):127006. doi:10.1289/EHP5139
18. Huang CY, Kuo WT, Huang YC, Lee TC, Yu LC. Resistance to hypoxia-induced necroptosis is conferred by glycolytic pyruvate scavenging of mitochondrial superoxide in colorectal cancer cells. Cell Death Dis. 2013;4(5):e622. doi:10.1038/cddis.2013.149
19. Hayashi M, Sakata M, Takeda T, et al. Induction of glucose transporter 1 expression through hypoxia-inducible factor 1alpha under hypoxic conditions in trophoblast-derived cells. J Endocrinol. 2004;183(1):145–154. doi:10.1677/joe.1.05599
20. Vannucci SJ, Reinhart R, Maher F, et al. Alterations in GLUT1 and GLUT3 glucose transporter gene expression following unilateral hypoxia-ischemia in the immature rat brain. Brain Res Dev Brain Res. 1998;107(2):255–264.
21. Shen LF, Zhao X, Zhou SH, et al. In vivo evaluation of the effects of simultaneous inhibition of GLUT-1 and HIF-1α by antisense oligodeoxynucleotides on the radiosensitivity of laryngeal carcinoma using micro 18F-FDG PET/CT. Oncotarget. 2017;8(21):34709–34726. doi:10.18632/oncotarget.16671
22. Chen Q, Chen R, Dong Y. Inhibitory effect of endostar combined with radiotherapy on gastric cancer animal models. World J Surg Oncol. 2020;18(1):165. doi:10.1186/s12957-020-01937-1
23. Li G, Xie B, Li X, et al. Downregulation of peroxiredoxin-1 by β-elemene enhances the radiosensitivity of lung adenocarcinoma xenografts. Oncol Rep. 2015;33(3):1427–1433. doi:10.3892/or.2015.3732
24. Li Y, Wang K, Chen L, Zhu X, Zhou J. Quantification of mRNA levels using real-time polymerase chain reaction (PCR). Methods Mol Biol. 2016;1406:73–79.
25. Tsai PC, Bake S, Balaraman S, et al. MiR-153 targets the nuclear factor-1 family and protects against teratogenic effects of ethanol exposure in fetal neural stem cells. Biol Open. 2014;3(8):741–758. doi:10.1242/bio.20147765
26. Kang J, Kim E, Kim W, et al. Rhamnetin and cirsiliol induce radiosensitization and inhibition of epithelial-mesenchymal transition (EMT) by miR-34a-mediated suppression of Notch-1 expression in non-small cell lung cancer cell lines. J Biol Chem. 2013;288(38):27343–27357. doi:10.1074/jbc.M113.490482
27. Seyedabadi S, Saidijam M, Najafi R, et al. Assessment of CEP55, PLK1 and FOXM1 expression in patients with bladder cancer in comparison with healthy individuals. Cancer Invest. 2018;36(8):407–414. doi:10.1080/07357907.2018.1514504
28. Guo AT, Song X, Wei LX, Zhao P. Histological origin of pseudomyxoma peritonei in Chinese women: clinicopathology and immunohistochemistry. World J Gastroenterol. 2011;17(30):3531–3537. doi:10.3748/wjg.v17.i30.3531
29. Xie Q, Li F, Fang L, Liu W, Gu C. The antitumor efficacy of β-elemene by changing tumor inflammatory environment and tumor microenvironment. Biomed Res Int. 2020;2020:6892961. doi:10.1155/2020/6892961
30. Li G, Xie B, Li X, et al. Down-regulation of survivin and hypoxia-inducible factor-1 α by β-elemene enhances the radiosensitivity of lung adenocarcinoma xenograft. Cancer Biother Radiopharm. 2012;27(1):56–64. doi:10.1089/cbr.2011.1003
31. Jóźwiak P, Lipińska A. Rola transportera glukozy 1 (GLUT1) w diagnostyce i terapii nowotworów [The role of glucose transporter 1 (GLUT1) in the diagnosis and therapy of tumors]. Postepy Hig Med Dosw (Online). 2012;4(66):165–174.
32. Zhao H, Sun J, Shao J, et al. Glucose transporter 1 promotes the malignant phenotype of non-small cell lung cancer through integrin β1/Src/FAK signaling. J Cancer. 2019;10(20):4989–4997. doi:10.7150/jca.30772
33. Seleit I, Bakry OA, Al-Sharaky DR, Ragab RAA, Al-Shiemy SA. Evaluation of hypoxia inducible factor-1α and glucose transporter-1 expression in non melanoma skin cancer: an immunohistochemical study. J Clin Diagn Res. 2017;11(6):EC09–EC16.
34. Fujino M, Aishima S, Shindo K, et al. Expression of glucose transporter-1 is correlated with hypoxia-inducible factor 1α and malignant potential in pancreatic neuroendocrine tumors. Oncol Lett. 2016;12(5):3337–3343. doi:10.3892/ol.2016.5092
35. Huang XQ, Chen X, Xie XX, et al. Co-expression of CD147 and GLUT-1 indicates radiation resistance and poor prognosis in cervical squamous cell carcinoma. Int J Clin Exp Pathol. 2014;7(4):1651–1666.
36. Yan SX, Luo XM, Zhou SH, et al. Effect of antisense oligodeoxynucleotides glucose transporter-1 on enhancement of radiosensitivity of laryngeal carcinoma. Int J Med Sci. 2013;10(10):1375–1386. doi:10.7150/ijms.6855
37. Zhao F, Ming J, Zhou Y, Fan L. Inhibition of Glut1 by WZB117 sensitizes radioresistant breast cancer cells to irradiation. Cancer Chemother Pharmacol. 2016;77(5):963–972. doi:10.1007/s00280-016-3007-9
38. Vitoratou DI, Tolia M, Liakos P, et al. Clinical value of significance of hypoxia inducible factor-1α, glucose transporter-1 and carbonic anhydrase IX in rectal cancer after preoperative chemoradiotherapy. J BUON. 2019;24(2):456–463.
39. Vermeulen MA, van Deurzen CH, Schroder CP, Martens JW, van Diest PJ. Expression of hypoxia-induced proteins in ductal carcinoma in situ and invasive cancer of the male breast. J Clin Pathol. 2020;73(4):204–208. doi:10.1136/jclinpath-2019-206116
40. Jung JH, Im S, Jung ES, Kang CS. Clinicopathological implications of the expression of hypoxia-related proteins in gastric cancer. Int J Med Sci. 2013;10(9):1217–1223. doi:10.7150/ijms.6054
41. Wu XH, Chen SP, Mao JY, Ji XX, Yao HT, Zhou SH. Expression and significance of hypoxia-inducible factor-1α and glucose transporter-1 in laryngeal carcinoma. Oncol Lett. 2013;5(1):261–266. doi:10.3892/ol.2012.941
42. Vasconcelos MG, Vasconcelos RG, Pereira de Oliveira DHet al., Distribution of hypoxia-inducible factor-1α and glucose transporter-1 in human tongue cancers. J Oral Maxillofac Surg. 2015;73(9):1753–1760. doi:10.1016/j.joms.2015.03.013
43. Iorio J, Lastraioli E, Tofani L, et al. hERG1 and HIF-2α behave as biomarkers of positive response to bevacizumab in metastatic colorectal cancer patients. Transl Oncol. 2020;13(3):100740. doi:10.1016/j.tranon.2020.01.001
44. Wang YF, Ma SR, Wang WM, et al. Inhibition of survivin reduces HIF-1α, TGF-β1 and TFE3 in salivary adenoid cystic carcinoma. PLoS One. 2014;9(12):e114051. doi:10.1371/journal.pone.0114051
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.