Back to Journals » Cancer Management and Research » Volume 11
Digital Immune-Related Gene Expression Signatures In High-Grade Serous Ovarian Carcinoma: Developing Prediction Models For Platinum Response
Authors Mairinger F , Bankfalvi A, Schmid KW , Mairinger E , Mach P, Walter RFH , Borchert S , Kasimir-Bauer S, Kimmig R, Buderath P
Received 18 June 2019
Accepted for publication 28 September 2019
Published 12 November 2019 Volume 2019:11 Pages 9571—9583
DOI https://doi.org/10.2147/CMAR.S219872
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Eileen O'Reilly
Fabian Mairinger,1 Agnes Bankfalvi,1 Kurt Werner Schmid,1 Elena Mairinger,2 Pawel Mach,3 Robert FH Walter,2 Sabrina Borchert,1 Sabine Kasimir-Bauer,3 Rainer Kimmig,3 Paul Buderath3
1Institute for Pathology, University Hospital Essen, Essen, Germany; 2Ruhrlandklinik, West German Lung Centre, University Hospital Essen, University of Duisburg-Essen, Essen, Germany; 3Department of Gynecology and Obstetrics, University Hospital Essen, Essen, Germany
Correspondence: Paul Buderath
Department of Gynecology and Obstetrics, University Hospital Essen, Hufelandstraße 55, Essen 45122, Germany
Tel +49 201 723 2442
Email [email protected]
Purpose: Response to platinum-based therapy is a major prognostic factor in high-grade serous ovarian cancer (HGSOC). While the exact mechanisms of platinum-resistance remain unclear, evidence is accumulating for a connection between the organism’s immune-response and response to platinum. However, predictive tools are missing. This study was performed to examine the putative role of the genetic tumor immune-microenvironment in mediating differential chemotherapy response in HGSOC patients.
Patients and methods: Expression profiling of 770 immune-related genes was performed in tumor tissues from 23 HGSOC cases. Tumors were screened for prognostic and predictive biomarkers using the NanoString nCounter platform for digital gene expression analysis with the appurtenant PanCancer Immune Profiling panel. As validation cohort, gene expression data (RNA Seq) of 303 patients with epithelial ovarian carcinoma (EOC) were retrieved from the The Cancer Genome Atlas (TCGA) database. Different scoring-systems were computed for prediction of risk-of-resistance to cisplatin, disease-free survival (DFS) and overall survival (OS).
Results: Validated on the TCGA-dataset, the developed scores identified 11 significantly differentially expressed genes (p <0.01**) associated with platinum response. HSD11B1 was highly significantly associated with lower risk of recurrence and 7 targets were found highly significantly influencing OS time (p <0.01**).
Conclusion: Our results suggest that response to platinum-based therapy and DFS in ovarian HGSOC is associated with distinct gene-expression patterns related to the tumor immune-system. We generated predictive scoring systems which proved valid when applied to a set of 303 EOC patients.
Keywords: ovarian cancer, tumor microenvironment, predictive biomarkers, platinum-resistance, HGSOC, gene expression signatures
Introduction
Epithelial ovarian cancer (EOC) is the deadliest gynecologic malignancy with a worldwide annual incidence of approximately 240,000 and is responsible for 152,000 cancer-related deaths every year.1 In Germany, 7380 women were diagnosed with EOC in 2012. EOC is responsible for 3.3% of all malignant neoplasms and 5.6% of all cancer deaths in women in Germany.2 As characteristic early symptoms are missing, most patients are diagnosed at an advanced stage. The most common ovarian cancer subtype is high-grade serous ovarian carcinoma (HGSOC). Standard treatment consists of radical cytoreductive surgery and adjuvant platinum-based chemotherapy. However, the relative 5-year survival remains poor and despite optimal cytoreductive surgery and standard-of-care chemotherapy, the majority of patients will ultimately relapse due to the development of platinum resistance.2
Platinum-analogs are genotoxic compounds inducing DNA damage which in turn leads to TP53-induced cell cycle arrest and apoptosis.3 It is, however, uncertain whether the failure of this mechanism alone is responsible for impaired therapy response. Several studies have therefore attempted to understand the basis of poor treatment response.4–7
The platinum-free interval to relapse has been identified as a predictive factor for response to subsequent platinum therapy.8 Thus, resistance to first-line platinum therapy limits therapeutic options for the relapsed disease significantly, making this patient cohort the most challenging to treat.
Several mechanisms leading to platinum resistance have been identified such as reduced intracellular cisplatin accumulation due to alterations in transmembrane transport, activation of cell growth-promoting and DNA-damage repair pathways, aberrant DNA methylation, enhanced epithelial-to-mesenchymal transition and reduced endocytosis of cisplatin.9,10
However, there are no established tools yet for predicting response to platinum therapy. Therefore, there is an unmet need for prognostic and predictive factors helping to identify patients at high risk for platinum resistance in order to select the adequate therapy for each individual.
In the last decades, the role of the immune system has been intensively studied in various cancers, including EOC, with the presence of tumor-infiltrating lymphocytes (TIL) being positively correlated with progression-free survival (DFS) and overall survival (OS).11–13 On the other hand, immunosuppressive factors also play a significant role in EOC. Regulatory T-cells (Tregs) have been shown to create an immunosuppressive environment in EOC patients, resulting in poorer survival.14 High mRNA and protein levels of FOXP3, a surface protein predominantly expressed in Treg cells, have been reported to be negative prognostic factors for PFS and OS in these patients.15
The significance of the expression of immunoregulatory proteins such as PD-1 and PD-L1 in EOC has gained increasing attention in the scientific community in recent years.16 Anti-PD-1 and anti-PD-L1 antibodies have been successfully used in patients with EOC in preclinical studies17,18 and are currently evaluated in clinical trials.
However, the connection between platinum sensitivity and the patient’s immune-reaction as prognostic factors in HGSOC has not been understood yet. Mariya et al showed that the absence of TILs was an independent predictor for platinum resistance, indicating a possible correlation between the mechanisms of platinum resistance and those of immune-escape.19 Similarly, Lo et al reported an increase in CD3+, CD8+ and PD1+ TILs after neo-adjuvant platinum-based treatment in EOC patients in case TILs were present before therapy. In TIL-negative tumors, no increase could be observed following platinum-based therapy.20
The aim of the present study was to identify prognostic and predictive patterns of mRNA-expression in a small test cohort of platinum-resistant and platinum-sensitive HGSOC cases using the PanCancer Immune Profiling Panel of the NanoString nCounter platform. Considering the information stated above regarding the relevance of the organism’s immune-response for platinum-sensitivity and therapy outcome, we focused our analysis on a panel of 770 immune-related genes. Our hypothesis was that, as the effectivity of the immune-answer is highly relevant for the course of disease, the genetic regulation of immune-related processes might allow a prediction of therapy response and prognosis.
Materials And Methods
Patient Cohort And Study Design
In this retrospective study, ovarian tumor tissues of 24 patients with the primary diagnosis of HGSOC were investigated. Patients were treated at the University Hospital of Essen between 2005 and 2010. Inclusion criteria were: Diagnosis of HGSOC, primary cytoreductive surgery, completion of an adjuvant platinum-based chemotherapy consisting of 6 cycles, no simultaneous additional anti-cancer treatment including study medication, availability of tumor tissue, absence of simultaneous malignant diseases, availability of follow-up. All patients from the selected period of time who met these criteria were included in the study.
Mean age at diagnosis was 62 years (23–84 years, SD 13.08). OS ranged from 11 to 126 months (mean: 46.17 months, SD 32.68) DFS ranged from 4 to 80 months (mean: 20.26 months, SD 19.99). By the end of the observation period (October 2016), 20 of 24 patients (83.3%) had experienced a recurrence and 19 (79.1%) had died.
Platinum resistance was defined as recurrence within 6 months after the last cycle of adjuvant platinum-based chemotherapy.
Patients and tumor characteristics are shown in Tables 1 and 2. An overview about the study design is depicted in Figure 1.
 |
Table 1 Cohort Characteristics. Demographic Data |
 |
Table 2 Cohort Characteristics. Oncologic Details |
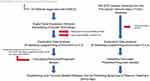 |
Figure 1 Methodical flowchart about the study design. |
Tumor Tissues
Routinely formalin-fixed and paraffin-embedded (FFPE) tumor tissues were retrieved from the archives of the Institute of Pathology of the University Hospital Essen (Germany). The initial diagnoses were re-evaluated by an experienced pathologist (AB) on hematoxylin and eosin (H&E) stained sections.
Validation Cohort
Gene expression data (RNA Seq) of 303 EOC patients were retrieved from the The Cancer Genome Atlas (TCGA) database (National Cancer Institute, National Human Genome Research institute, Bethesda, MD, US).
Ethics
The study conforms to the principles outlined in the declaration of Helsinki and was approved by the local Ethics Committee of the Medical Faculty of the University Duisburg-Essen (protocol no. 16-6916-BO). All patients provided written informed consent for the use of tissue samples in future research.
RNA Extraction And RNA Integrity Assessment
FFPE sections were prepared by using the “Microm HM340E” microtome (Thermo Fisher Scientific, Massachusetts, USA). Cut tissue slides were stored at −20°C until use for RNA isolation as this procedure resulted in higher RNA yields.
Two 10 µm sections of each FFPE block were used for semi-automatic isolation of RNA using the Maxwell purification system (Maxwell RSC RNA FFPE Kit, AS1440, Promega, Wisconsin, USA). The purification was performed according to the manufacturer’s instructions. RNA was eluted in 50µL RNase-free water and stored at −80°C.
RNA concentration was measured using a Qubit 2.0 fluorometer (Life Technologies, California, USA) appertaining the RNA broad-range assay. RNA integrity was assessed using a Fragment Analyzer (Advanced Analytical Inc., Ames, IA, USA) appertaining DNF-489 standard sensitivity RNA analysis kit.
Digital Gene Expression Analysis
Gene expression patterns were screened for prognostic and predictive biomarkers using the NanoString nCounter platform for digital gene expression analysis with the appurtenant PanCancer Immune Profiling panel, consisting of 770 genes mediating immune response as well as 30 reference genes. Hybridizations were performed using the high-sensitivity protocol on the nCounter Prep-Station. The post-hybridization processing was performed by using the nCounter MAX/FLEX System (NanoString) and the cartridge was scanned on the Digital Analyzer (NanoString). The cartridge was read with maximum sensitivity (555 FOV). 100 ng sample input were used for each reaction.
NanoString Data Processing
NanoString data processing was done with the R statistical programming environment (v3.4.2). Considering the counts obtained for positive control probe sets, raw NanoString counts for each gene were subjected to a technical factorial normalization, carried out by subtracting the mean counts plus two-times standard deviation from the CodeSet inherent negative controls. Subsequently, a biological normalization using the included mRNA reference genes was performed.
Additionally, all counts with p>0.05 after one-sided t-test versus negative controls plus 2x standard deviations were interpreted as not expressed to overcome basal noise.
Statistical Analysis
Statistical and graphical analyses were performed with the R statistical programming environment (v3.4.2).
The Shapiro–Wilks test was applied to test for normal distribution of the data. For dichotomous variables, either the Wilcoxon Mann–Whitney rank sum test (non-parametric) or two-sided Student’s t-test (parametric) was used. For ordinal variables with more than two groups, either the Kruskal–Wallis test (non-parametric) or ANOVA (parametric) was used to detect group differences.21
Double dichotomous contingency tables were analyzed using Fisher’s exact test. To test dependence on ranked parameters with more than two groups the Pearson’s Chi-squared test was used. Correlations between methylation levels and spread to regional lymph nodes, methylation analysis between genes as well as age of the patients were tested by using the Spearman’s rank correlation test.22 Pathway analysis was based on the KEGG database and was performed using the “pathview” package of R. Differences were specified by –log2 fold change between means (if parametric) or medians (if non-parametric) of compared groups.
Due to the multiple statistical tests, the p-values were adjusted by using the false discovery rate (FDR). The level of statistical significance was defined as p≤0.05 after adjustment.23
Quality control of run data was first performed in basic by mean-vs-variances plotting to find outliers in target or sample level. True differences were calculated by correlation matrices analysis.
To further specify the different candidate patterns, both unsupervised and supervised clustering, to overcome commonalities, as well as principal component analysis, to overcome differences, were performed. Sensitivity and specificity of markers were determined from receiver operating characteristic (ROC) curves illustrating their performance to discriminate the studied groups.24,25 The bootstrap procedure (1000 iterations) was used for internal validation of the estimates in the ROC analyses.
The best candidates-genes were selected and binarized (0, 1; with 1 equals better chance of event) by their respective cut-offs and finally summed up. The resulting scores were compared with respect to sensitivity and specificity. Adaption of profiles for diagnostic purposes was explored with the supervised machine learning tools “Classification and Regression Tree Algorithm” (CART) as implemented in the “rpart” library of R (according to Therneau and Atkinson) using a leave-one-out cross-validation. Additionally, conditional interference trees (CTree) as implemented in the “party” library of R were modeled, leading to non-parametric class of tree-structured regression models.
Results
General Overview
Analysis of digital gene expression was successful in all 24 samples. The mean gene expression count after biological and technical normalization procedures was 832.8 (median: 172.1; minimum: 1.0; maximum: 117,258.8). Seventy-three genes could be determined as not significantly expressed, resulting in a signature of 679 significantly expressed genes. Twenty-four of 30 reference genes have been used for calculation of the normalization factor.
An overview about all p-values is given in suppl. Table 1.
Risk-Of-Platinum-Resistance
After initial analysis of correlations to resistance to platinum-compounds, 11 highly significant (p<0.01, **) (KLRC1, TCF7, CD274, HSD11B1, COLEC12, PDGFC, FCF1, BMI1, TNFRSF9, ATG10, EWSR1ʺ) and further 23 significant candidate-genes (p<0.05, *) could be identified. Six of them were up- and five downregulated in resistant tumors (Figure 2).
Most interestingly, the signaling pathways for natural killer cell-mediated cytotoxicity, T- and B-cell receptor signaling and leukocyte transendothelial migration were strongly activated in the population of platinum-resistant tumors (Figure S1). Overall, the lymphoid lineage of hematopoietic cells was strongly activated in Pt-resistant cancers (Figure S2). As CD8 shows no significant upregulation, whereas CD3, CD25 and CD4 do, most of the tumor-infiltrating lymphocytes should be classified as Treg. Moreover, Th1/Th2 as well as Th17 cell differentiation was strongly affected in Pt-resistant tumors. The myeloid lineage showed a more heterogeneous picture, but it also has been activated in a gross of involved genes, eg, genes involved in the formation and differentiation of granulocytes.
Risk-Of-Recurrence
For DFS, only HSD11B1 showed a highly significant predictive value [p< 0.01 (**)]; 19 other genes had a lower level of significance [p<0.05 (*)]. Thereby, a high expression of HSD11B1 predicts a prolonged DFS compared to the low-expression subpopulation (Figure 3, upper lines).
No correlations or association could be found with the classical immune-related pathways by pathway enrichment analysis.
Risk-Of-Early-Death
COX regression modelling revealed 7 targets highly significantly influencing overall survival time (p<0.01, **) and further 34 candidates achieved a lower level of significance (p<0.05) (Figure 3, lower lines). Of those 7 genes, NUP107, CCL18, LY96, ATG5, SLAMF7 and CXCL9 expression indicate a prolonged, and SDHA a shortened overall survival rate.
None of the classical immune pathways was found associated with patients’ overall survival.
Generation Of Immune-Scores
Based on the above-stated results, different scoring-systems were applied for prediction of risk-of-resistance to platinum, disease/recurrence-free survival (DFS) as well as overall survival (OS) rate and duration.
To predict response to platinum therapy, three different score types were tested: Score1, as a binarized scoring system based on the sum of each single-binarized value (0,1), Score2 was based on the z-scores after scaling each target and Score3 was the sum of absolute-normalized counts. Score1 and Score2 led to a logistic regression model (Figure 4A), Score3 defined a clear cut-off between responders and non-responders. By analyzing the association between Score1 and a resistance/sensitivity to platinum-compounds, a most significant correlation could be calculated (p<0.0001) (Figure 4B). Applying Score1, Receiver-Operating-Characteristic (ROC) analysis revealed an area under the curve (AUC) of 0.98 when compared with specificity and sensitivity in predicting platinum-response, Score2 revealed and AUC of 0.99 and Score3 an AUC of 1.0. The logistic regression revealed a significance for Score1 of p=0.0078, for Score2 of p=0.0042. As Score3 is based on a clear cut-off, a singular gradient matrix has been generated and therefore no non-linear regression can be applied.
All platinum-resistance scores consisted of 11 genes and were based on the combination of KLRC1, TCF7, CD274, HSD11B1, COLEC12, PDGFC, FCF1, BMI1 as well as TNFRSF9. Of them, HSD11B1, PDGFC, FCF1, BMI1 and ATG10 high gene expression were associated with low risk for platinum resistance, whereas the others were inversely correlated.
In contrast, the scoring system generated for prediction of DFS (Recurrence (Rec)Score) was based on the expression of the four targets HSD11B1, DMBT1, IKBKG and CKLF. While HSD11B1, DMBT1 and CKLF were associated with longer survival, IKBKG gene expression was associated with shortened DFS. The RecScore was calculated as the sum of binarized digital expression values, leading to an exponential mathematical interrelation (p=0.0048, 3dF; Figure 4C). Thereby, the single scores separately differed in DFS time (Score (logrank) test: p=0.0195, Likelihood ratio test: p=0.0168, Wald test: p=0.0049, HR=9.4; Figure 4D)
The score for overall survival outcome prediction (SurvScore) was similarly structured and calculated like the RecScore and was based on a combination of the seven targets NUP107, CCL18, LY96, ATG5, SlAMF7, CXCL9 (prolonged-) as well as SDHA (shortened OS). Thus, the SurvScore single groups were significantly associated with patients’ survival time (p=0.0076, HR=7.9; Figure 4F). The SurvScore describes an exponential mathematical interrelation (p<0.0001, 5dF; Figure 4E).
Score-Validation With TCGA Data-Sets
Prognostic and predictive explanatory power of scores calculated within the trainings-group got validated using the provisional TCGA dataset for ovarian serous cystadenocarcinoma downloaded from the Broad Institute Firehouse (https://cancergenome.nih.gov).
For platinum-response, the exact same score (Score1) could be applied to the given data set. Interestingly, in contrast to the logistic regression generated within the training’s cohort, the samples showed a linear association (p=0.0006, R2=0.75) with risk-of resistance to platinum-based compounds. Moreover, when focusing on patients’ outcome by overall survival, a clear separation of HGSOC with high and low scores can be observed, leading to a 5y-OS rate of >20% when showing a score above 5 (Figure 5A)
For OS, only four of the seven original genes (SLAMF7, CXCL9, COLEC12, HSD11B1), forming the SurvScore, could be proven significantly expressed and also influencing patients’ OS. Nevertheless, even when differing from the original setting, a still significant association to estimated OS time could be found (non-linear least square regression, p=0.0222;). COX-regression did find significant associations between a high- and a low-score group and OS [Score (logrank) test: p=0.0370, Likelihood ratio test: p=0.0369, Wald test: p=0.0377, HR=1.4] (Figure 5B)
DFS could be represented by the RecScore using an exponential function, leading to nearly identical results and estimated DFS times (p=0.0032, 3dF). Thereby, the RecScore was significantly associated with patients’ survival when forming two groups [Score (logrank) test: p=0.0171, Likelihood ratio test: p=0.0205, Wald test: p=0.0178, HR=1.77] (Figure 5C).
Discussion
We hereby present a study focusing on a broad mRNA screening of anti-tumor immune response-associated targets aiming at understanding the mechanisms of platinum-based therapy-induced cytotoxicity and to identify possible predictive markers. Therefore, we used the NanoString nCounter system, a non-enzymatic digital detection method that can be used to quantitatively analyze mRNA, miRNA and copy number variations (CNV).26–29 Due to the enzyme-free processing, the nCounter method is particularly suitable for FFPE and archived tissue samples.27–29 We used the NanoString nCounter System in various previous studies to screen whole pathways, leading to a better understanding of biological processes and to identify prognostic, diagnostic and predictive biomarkers in clinical specimens27–29
As the main cause of platinum-based cytotoxicity is DNA damage, different DNA-damage repair systems stand in the focus of research for the prediction of platinum response,5,30 including nucleotide excision repair, mismatch repair,6 base-excision repair31 but also the recombination repair system.32 In addition, uptake and detoxification of heavy metals may play a crucial role in cellular response to platinum-compound.33–37 In this context, the role of the immune system is still mostly unclear.
Based on the expression profiles found in our HGSOC patients, we managed to generate scoring-systems for the prediction of platinum-resistance, DFS and OS. Validation on the TCGA data-set showed a very good concordance for platinum-resistance and DFS. These results suggest that the gene expression patterns found in our study sample might represent essential mechanisms in HGSOC which could help to predict response to adjuvant therapy as well as the duration of the therapeutic effect.
Our results show a strong association of platinum-resistance with the expression of genes related to i) T- and B-cell receptor signaling, ii) natural killer cell-mediated toxicity and iii) leukocyte transendothelial migration. Interestingly, the expression of these pathways seems to be upregulated in platinum-resistant tumors. This might seem to be contradictory to the observation that the presence and activity of TILs correlate with better prognosis in HGSOC. However, the up-regulation of CD3, CD4 and CD25 indicate a high proportion of Tregs among the T-cells, a population known to create an immunosuppressive environment which has been associated with worse prognosis.14
Another explanation for the significant up-regulation of lymphocyte-associated genes could be found considering the fact that the over-expression of mentioned genes does not allow any quantitative statements concerning the number of immune cells actually present at the tumor site. Therefore, the increased expression activity could also mirror a lack of active T- and B-cells in terms of a positive feedback mechanism. This seems especially likely considering the results of Mariya et al reporting the absence of TILs in platinum-resistant tumors.19 Considering the results of Lo et al, it can be expected that even following platinum-based chemotherapy, there will be no increase in TILs in these patients. It can be hypothesized that in the platinum-resistant subset of tumors, immunosuppressive mechanisms are at play in the tumor microenvironment, which prevent lymphocytes to infiltrate the tumor even despite massive up-regulation of the associated genes. In order to shed some light on these mechanisms, an evaluation of TILs in correlation with gene expression profiles could be the next step for further investigation.
For DFS, the observed effect could mainly be attributed to the HSD11B1 gene which codes for the microsomal enzyme hydroxysteroid 11-beta dehydrogenase. There are to our knowledge no available data on the biologic significance of HSD11B1 in ovarian cancer. However, single-nucleotide polymorphisms of the gene have been reported to play a role in the susceptibility for colorectal cancer.38 As HSD11B1 is mainly responsible for the regeneration of cortisol from cortisone, it can be hypothesized that differences in the corticoid metabolism in the tumor microenvironment contribute to different outcomes in EOC. However, the biologic significance of these observation has yet to be understood.
The scoring system for the prediction of OS failed to prove the same accuracy when applied to the TCGA data-set as when applied to our trainings data. This could be due to a number of different reasons.
First of all, OS is not as directly related to tumor biology and response to therapy as DFS, as it is not only influenced by tumor-related deaths but also by comorbidities and side-effects of therapy, among others. Secondly, the sequence of therapies used for the, often, multiple recurrences vary widely between patients. As any line of therapy or combination of therapies is likely to influence survival, this makes the results hardly comparable between different populations. Moreover, the usage of different methods for RNA expression quantification (NanoString nCounter digital gene expression analysis versus RNA-sequencing in the TCGA dataset) have to be taken into account. Therefore, the (overall) high concordance between both training and validation cohort highlight the significance of the discovered underlying gene expression signature.
Besides the difference in the detection method between trainings- and validation cohort, a further limitation of these studies is the relatively small sample size used in the training cohort. Results of a complex field as host’s anti-cancer immune response rendered on 24 subjects must be weighted carefully. We used this approach as we could train our model on a homogenous, well-characterized cohort of HGSOC, and rendered reliability by validating on a big, diverse collective including different kinds of EOC. Also, the usage of the enzyme-free gene expression quantification method strengthens our results, as the method renders a digital signal by counting single barcodes, each referring to one single-detected mRNA, and is therefore independent of overall signal intensity, making the results more reliable.5,27,29,30,39–43
In conclusion, our results suggest that response to platinum-based therapy and DFS is associated with distinct gene-expression patterns related to the immune system. While the exact underlying mechanisms are not fully understood yet, we generated as set of predictive scoring systems which proved valid when applied to the TCGA data-set of 303 EOC patients. Forthcoming investigation has to elucidate the biological function and significance of these genetic patterns found. Validation of the newly developed scoring systems on larger series will help to answer the question if they can help to select high-risk patients in need for a modification of the standard adjuvant treatment in the future. Furthermore, these scores will have to be evaluated in a prospective setting in order to prove their value as predictive markers. As biomarkers for therapy stratification are missing in ovarian cancer, further research on the foundation of our results should be done to address this urgent issue.
Conclusion
Our results suggest that response to platinum-based therapy and DFS in ovarian HGSOC is associated with distinct gene-expression patterns related to the tumor immune-system. The predictive scoring systems generated by us proved valid when applied to a set of 303 EOC patients.
Acknowledgment
We gratefully thank the patients for participation in the study and for kindly providing the samples. We also acknowledge support by the Open Access Publication Fund of the University of Duisburg-Essen.
Disclosure
Prof. Sabine Kasimir-Bauer is a consultant for Qiagen. Prof. Rainer Kimmig reports personal fees from Astra Zeneca, Prostrakan, Riemser, and Teva outside the submitted work. The authors report no other conflicts of interest in this work.
References
1. Ervik M, Lam F, Ferlay J, Mery L, Soerjomataram I, Bray F. Cancer Today. Lyon, France: International Agency for Research on Cancer; 2016. Available from: http://gco.iarc.fr/today.
2. Heintz AP, Odicino F, Maisonneuve P, et al. Carcinoma of the ovary. FIGO 26th annual report on the results of treatment in gynecological cancer. Int J Gynaecol Obstet. 2006;95(Suppl 1):S161–S192. doi:10.1016/S0020-7292(06)60033-7
3. Koch M, Krieger ML, Stolting D, et al. Overcoming chemotherapy resistance of ovarian cancer cells by liposomal cisplatin: molecular mechanisms unveiled by gene expression profiling. Biochem Pharmacol. 2013;85(8):1077–1090. doi:10.1016/j.bcp.2013.01.028
4. Kelly RJ, Sharon E, Hassan R. Chemotherapy and targeted therapies for unresectable malignant mesothelioma. Lung Cancer. 2011;73(3):256–263. doi:10.1016/j.lungcan.2011.04.014
5. Walter RF, Vollbrecht C, Werner R, et al. Screening of pleural mesotheliomas for DNA-damage repair players by digital gene expression analysis can enhance clinical management of patients receiving platin-based chemotherapy. J Cancer. 2016;7(13):1915–1925. doi:10.7150/jca.16390
6. Ting S, Mairinger FD, Hager T, et al. ERCC1, MLH1, MSH2, MSH6, and betaIII-tubulin: resistance proteins associated with response and outcome to platinum-based chemotherapy in malignant pleural mesothelioma. Clin Lung Cancer. 2013;14(5):558–567. e553. doi:10.1016/j.cllc.2013.04.013
7. Mairinger FD, Werner R, Flom E, et al. miRNA regulation is important for DNA damage repair and recognition in malignant pleural mesothelioma. Virchows Arch. 2017. doi:10.1007/s00428-017-2133-z
8. Bolis G, Scarfone G, Luchini L, et al. Response to second-line weekly cisplatin chemotherapy in ovarian cancer previously treated with a cisplatin- or carboplatin-based regimen. Eur J Cancer. 1994;30A(12):1764–1768. doi:10.1016/0959-8049(94)e0125-n
9. Galluzzi L, Senovilla L, Vitale I, et al. Molecular mechanisms of cisplatin resistance. Oncogene. 2012;31(15):1869–1883. doi:10.1038/onc.2011.384
10. Shen DW, Pouliot LM, Hall MD, Gottesman MM. Cisplatin resistance: a cellular self-defense mechanism resulting from multiple epigenetic and genetic changes. Pharmacol Rev. 2012;64(3):706–721. doi:10.1124/pr.111.005637
11. Zhang L, Conejo-Garcia JR, Katsaros D, et al. Intratumoral T cells, recurrence, and survival in epithelial ovarian cancer. N Engl J Med. 2003;348(3):203–213. doi:10.1056/NEJMoa020177
12. Sato E, Olson SH, Ahn J, et al. Intraepithelial CD8+ tumor-infiltrating lymphocytes and a high CD8+/regulatory T cell ratio are associated with favorable prognosis in ovarian cancer. Proc Natl Acad Sci U S A. 2005;102(51):18538–18543. doi:10.1073/pnas.0509182102
13. Hamanishi J, Mandai M, Iwasaki M, et al. Programmed cell death 1 ligand 1 and tumor-infiltrating CD8+ T lymphocytes are prognostic factors of human ovarian cancer. Proc Natl Acad Sci U S A. 2007;104(9):3360–3365. doi:10.1073/pnas.0611533104
14. Curiel TJ, Coukos G, Zou L, et al. Specific recruitment of regulatory T cells in ovarian carcinoma fosters immune privilege and predicts reduced survival. Nat Med. 2004;10(9):942–949. doi:10.1038/nm1093
15. Milne K, Kobel M, Kalloger SE, et al. Systematic analysis of immune infiltrates in high-grade serous ovarian cancer reveals CD20, FoxP3 and TIA-1 as positive prognostic factors. PLoS One. 2009;4(7):e6412. doi:10.1371/journal.pone.0006412
16. Webb JR, Milne K, Kroeger DR, Nelson BH. PD-L1 expression is associated with tumor-infiltrating T cells and favorable prognosis in high-grade serous ovarian cancer. Gynecol Oncol. 2016;141(2):293–302. doi:10.1016/j.ygyno.2016.03.008
17. Brahmer JR, Tykodi SS, Chow LQ, et al. Safety and activity of anti-PD-L1 antibody in patients with advanced cancer. N Engl J Med. 2012;366(26):2455–2465. doi:10.1056/NEJMoa1200694
18. Hamanishi J, Mandai M, Ikeda T, et al. Safety and antitumor activity of anti-PD-1 antibody, nivolumab, in patients with platinum-resistant ovarian cancer. J Clin Oncol. 2015;33(34):4015–4022. doi:10.1200/JCO.2015.62.3397
19. Mariya T, Hirohashi Y, Torigoe T, et al. Prognostic impact of human leukocyte antigen class I expression and association of platinum resistance with immunologic profiles in epithelial ovarian cancer. Cancer Immunol Res. 2014;2(12):1220–1229. doi:10.1158/2326-6066.CIR-14-0101
20. Lo CS, Sanii S, Kroeger DR, et al. Neoadjuvant chemotherapy of ovarian cancer results in three patterns of tumor-infiltrating lymphocyte response with distinct implications for immunotherapy. Clin Cancer Res. 2017;23(4):925–934. doi:10.1158/1078-0432.CCR-16-1433
21. Tan JX, Dao FY, Lv H, Feng PM, Ding H. Identifying phage virion proteins by using two-step feature selection methods. Molecules. 2018;23(8). doi:10.3390/molecules23082000
22. Yin XH, Wang ZQ, Yang SZ, Jia HY, Shi M. Clinical observation of laparoscopic radical hysterectomy for cervical cancer. Int J Clin Exp Med. 2014;7(5):1373–1377.
23. Long WL, Liang P, Liu S, Zuo, Y. Transcriptome comparisons of multi-species identify differential genome activation of mammals embryogenesis. IEEE Access. 2019;7:7794–7802. doi:10.1109/ACCESS.2018.2889809
24. Yang L, Wang J, Wang H, Lv Y, Zuo Y, Jiang W. Analysis and identification of toxin targets by topological properties in protein-protein interaction network. J Theor Biol. 2014;349:82–91. doi:10.1016/j.jtbi.2014.02.001
25. Yang L, Wang J, Wang H, et al. Analysis and identification of essential genes in humans using topological properties and biological information. Gene. 2014;551(2):138–151. doi:10.1016/j.gene.2014.08.046
26. Waggott D, Chu K, Yin S, Wouters BG, Liu FF, Boutros PC. NanoStringNorm: an extensible R package for the pre-processing of NanoString mRNA and miRNA data. Bioinformatics. 2012;28(11):1546–1548. doi:10.1093/bioinformatics/bts188
27. Mairinger FD, Walter RF, Werner R, et al. Activation of angiogenesis differs strongly between pulmonary carcinoids and neuroendocrine carcinomas and is crucial for carcinoid tumourgenesis. J Cancer. 2014;5(6):465–471. doi:10.7150/jca.9235
28. Walter RF, Werner R, Ting S, et al. Identification of deregulation of apoptosis and cell cycle in neuroendocrine tumors of the lung via NanoString nCounter expression analysis. Oncotarget. 2015. doi:10.18632/oncotarget.3992
29. Walter RF, Mairinger FD, Werner R, et al. SOX4, SOX11 and PAX6 mRNA expression was identified as a (prognostic) marker for the aggressiveness of neuroendocrine tumors of the lung by using next-generation expression analysis (NanoString). Future Oncol. 2015;11(7):1027–1036. doi:10.2217/fon.15.18
30. Mairinger FD, Werner R, Flom E, et al. miRNA regulation is important for DNA damage repair and recognition in malignant pleural mesothelioma. Virchows Arch. 2017;470(6):627–637. doi:10.1007/s00428-017-2133-z
31. Ollikainen T, Linnainmaa K, Kinnula VL. DNA single strand breaks induced by asbestos fibers in human pleural mesothelial cells in vitro. Environ Mol Mutagen. 1999;33(2):153–160.
32. Helleday T, Petermann E, Lundin C, Hodgson B, Sharma RA. DNA repair pathways as targets for cancer therapy. Nat Rev Cancer. 2008;8(3):193–204. doi:10.1038/nrc2342
33. Andrews PA, Murphy MP, Howell SB. Metallothionein-mediated cisplatin resistance in human ovarian carcinoma cells. Cancer Chemother Pharmacol. 1987;19(2):149–154. doi:10.1007/bf00254568
34. Surowiak P, Kaplenko I, Spaczynski M, Zabel M. The expression of metallothionein (MT) and proliferation intensity in ovarian cancers treated with cisplatin and paclitaxel. Folia Morphol (Warsz). 2003;62(4):493–495.
35. Choi CH, Cha YJ, An CS, et al. Molecular mechanisms of heptaplatin effective against cisplatin-resistant cancer cell lines: less involvement of metallothionein. Cancer Cell Int. 2004;4(1):6. doi:10.1186/1475-2867-4-6
36. Surowiak P, Materna V, Maciejczyk A, et al. Nuclear metallothionein expression correlates with cisplatin resistance of ovarian cancer cells and poor clinical outcome. Virchows Arch. 2007;450(3):279–285. doi:10.1007/s00428-006-0362-7
37. Mairinger FD, Schmeller J, Borchert S, et al. Immunohistochemically detectable metallothionein expression in malignant pleural mesotheliomas is strongly associated with early failure to platin-based chemotherapy. Oncotarget. 2018;9(32):22254–22268. doi:10.18632/oncotarget.24962
38. Wang J, Gao Y, Wang L, et al. A variant (rs932335) in the HSD11B1 gene is associated with colorectal cancer in a Chinese population. Eur J Cancer Prev. 2013;22(6):523–528. doi:10.1097/CEJ.0b013e3283656346
39. Walter RF, Mairinger FD, Werner R, et al. Folic-acid metabolism and DNA-repair phenotypes differ between neuroendocrine lung tumors and associate with aggressive subtypes, therapy resistance and outcome. Oncotarget. 2016;7(15):20166–20179. doi:10.18632/oncotarget.7737
40. Walter RF, Vollbrecht C, Christoph D, et al. Massive parallel sequencing and digital gene expression analysis reveals potential mechanisms to overcome therapy resistance in pulmonary neuroendocrine tumors. J Cancer. 2016;7(15):2165–2172. doi:10.7150/jca.16925
41. Walter RF, Vollbrecht C, Werner R, et al. microRNAs are differentially regulated between MDM2-positive and negative malignant pleural mesothelioma. Oncotarget. 2016;7(14):18713–18721. doi:10.18632/oncotarget.7666
42. Walter RF, Werner R, Ting S, et al. Identification of deregulation of apoptosis and cell cycle in neuroendocrine tumors of the lung via NanoString nCounter expression analysis. Oncotarget. 2015;6(28):24690–24698. doi:10.18632/oncotarget.3992
43. Walter RF, Werner R, Vollbrecht C, et al. ACTB, CDKN1B, GAPDH, GRB2, RHOA and SDCBP were identified as reference genes in neuroendocrine lung cancer via the nCounter technology. PLoS One. 2016;11(11):e0165181. doi:10.1371/journal.pone.0165181
44. Yang L, Wang S, Zhang Q, et al. Clinical significance of the immune microenvironment in ovarian cancer patients. Mol Omics. 2018;14(5):341–351. doi:10.1039/c8mo00128f
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.




