Back to Journals » OncoTargets and Therapy » Volume 12
Differential Expression And Effects Of Peroxiredoxin-6 On Drug Resistance And Cancer Stem Cell-Like Properties In Non-Small Cell Lung Cancer
Authors Xu J, Su Q, Gao M, Liang Q, Li J, Chen X
Received 4 April 2019
Accepted for publication 21 October 2019
Published 2 December 2019 Volume 2019:12 Pages 10477—10486
DOI https://doi.org/10.2147/OTT.S211125
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr William C. Cho
Jun Xu,1,* Qiang Su,2,* Mingxia Gao,3 Qingsong Liang,1 Junfeng Li,1 Xu Chen1
1Department of Cardiothoracic Surgery, Nanchong Central Hospital, The Second Clinical Medical College, North Sichuan Medical College, Nanchong, Sichuan 637000, People’s Republic of China; 2Department of Pharmacy, Nanchong Central Hospital, The Second Clinical Medical College, North Sichuan Medical College, Nanchong, Sichuan, People’s Republic of China; 3Department of Ultrasound, Nanchong Central Hospital, The Second Clinical Medical College, North Sichuan Medical College, Nanchong, Sichuan, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Xu Chen
Department of Cardiothoracic Surgery, Nanchong Central Hospital, The Second Clinical Medical College, North Sichuan Medical College, #97 of Renmin South Road, Nanchong City, Sichuan 637000, People’s Republic of China
Tel +86 139 9089 8058
Email [email protected]
Objective: Cancer stem-like cells (CSC) are thought to be involved in the cisplatin resistance of tumors. This study was designed to investigate the effect of PRDX6 on CSCs present in cisplatin-resistant non-small cell lung cancer (NSCLC) tumors.
Materials and methods: CD133+/ABCG2+ H1299 CSCs and A549 CSCs were isolated. The IC50 values for cisplatin in treatment of CSCs were detected using the CCK8 assay. Then the isolated cells were identified using CD133. Wnt/β-catenin expression was evaluated by Western blot assays. Specimens of tumor and adjacent para-carcinoma tissue were collected from 30 NSCLC patients and examined by immunohistochemistry (IHC), qRT-PCR, and Western blotting to determine and compare their levels of PRDX6 and CD133 expression. Finally, siRNA-mediated silencing of PRDX6 was employed with both types of CSCs to determine the impact of PRDX6 on CD133 enrichment by flow cytometry, cell viability, and sphere formation ability.
Results: High levels of PRDX6 and CD133 expression were detected in samples of tumor tissue from NSCLC patients, and expression of PRDX6 and CD13 presented a positive relationship. Increasing levels of cisplatin resistance and upregulated levels of PRDX6, ABCG2, Wnt, and β-catenin expression were detected in CD133+/ABCG2+ H1299 and A549 CSCs. Transfection with siRNA targeting PRDX6 changed these cellular characteristics by decreasing the levels of PRDX6, ABCG2, Wnt, and β-catenin expression. We further demonstrated that exogenous silencing of PRDX6 effectively inhibited the sphere formation ability of CSCs and re-sensitized them to cisplatin.
Conclusion: Our results strongly suggest that PRDX6 promotes cisplatin resistance in human lung cancer cells by promoting the stem-like properties of cancer cells. Our findings also suggest PRDX6 as a target for treating cisplatin resistant NSCLC.
Keywords: PRDX6, CSCs, cisplatin-resistance, NSCLC, cancer stem-like cell
Introduction
Non-small cell lung cancer (NSCLC) accounts for ~80% of all lung cancers and has a dismal 5-year patient survival rate of 15%. Furthermore, ~66% of NSCLC cancer patients initially present with stage IV disease.1,2 In recent years, the 5-year survival rate of NSCLC patients has not substantially increased, and remains as low as 20%, especially among patients with stage III/IV disease.3 Although new therapies have benefitted patients with predefined subclasses of carcinoma, cisplatin-based chemotherapy remains the standard treatment for NSCLC. However, cisplatin resistance to targeted therapy, which can result from multiple factors, is a major issue affecting the efficacy of NSCLC treatments.4,5 Many previous studies revealed that a combination of factors, including accelerated drug clearance, activation of alternative proliferation signaling pathways, and suppression of apoptotic pathways, may be involved in cisplatin resistance. Recent studies have indicated that some unique populations of cells are capable of surviving tumor treatments, and those cells are designated as cancer stem cells (CSCs), due to their stem cell-like characteristics, self-renewal ability, and multi-potency.6–8
As a special population of undifferentiated cells that contribute to the pathogenesis and progression of tumors, CSCs have been found in a variety of cancers, including myeloid leukemia, glioblastoma, gastric, and epithelial cancers.9,10 Due to their stem cell properties, CSCs have the capacity for multipotency, unlimited self-renewal, and proliferation with a natural tolerance to chemotherapy that result from their decreased cell cycling and enhanced expression of proteins associated with DNA repair and resistance to apoptosis.11 Various alleged stem cell markers, selective for human stem cells and their counterparts in tumors, have been used to identify and isolate CSCs; these markers include CD133 (prominin-1), a five-transmembrane glycoprotein,12 and ATP-binding cassette superfamily G member 2 (ABCG-2).13 ABCG2 is always co-expressed with CD133, and is accepted as a drug resistance marker due to its ability to confer the side population’s phenotype.14,15 Accordingly, the identification of some oncogenic factors that result in a persistent activation of CSCs is essential for further elucidating NSCLC pathobiology, as well as for developing novel effective therapies.
Peroxiredoxins (PRDXs) comprise a newly discovered class of non-selenium-dependent peroxidase proteins that are widely distributed in various organisms.16 PRDX a type of antioxidant enzyme, is thought to catalyze redox reactions and maintain the balance of hydrogen peroxide in cells.17 Presently, PRDX1-PRDX6 have been found to contain a 1-Cys PRDX group and 2-Cys PRDX group. PRDX1-5 belongs to the 2-Cys group, and PRDX6 belongs to the 1-Cys PRDX group.18,19 Besides helping to protect cells against oxidative stress (OS), PRDX6 uniquely possesses calcium independent phospholipase A2 (PLA2) activity and glutathione peroxidase activity that can help to prevent oxidative stress.20 Moreover, numerous studies have proven that PRDX6 plays essential roles in tumor maintenance and cell survival by protecting cells from OS-induced apoptosis.21,22 Recent studies have also confirmed that PRDX6 can attenuate cisplatin-induced apoptosis.23 In contrast, silencing of PRDX6 expression was shown to result in peroxide-induced cell death.24 Meanwhile, PRDX6 was also shown to promote the invasion and metastasis of lung cancer cells by activating the Akt pathway.25 However, the role played by PRDX6 in NSCLC, and its mechanism of action, remain unclear.
In this study, we examined the levels of PRDX6 and CD133 expression in NSCLC cells and tissues, the correlation between cisplatin resistance and PRDX6 expression, and further demonstrated the effect of PRDX6 on CSC maintenance in NSCLC. Our results suggest that downregulation of PRDX6 expression might be a potential biomarker and represent a strategy for treating NSCLC patients.
Materials And Methods
Patient Characteristics, Clinical Features, And Tissue Harvest
Between December 2016 and December 2017, this study enrolled a total of 60 pathologically diagnosed NSCLC patients, including 30 patients with resistance to cisplatin. No patient had a radiological record of disease development. The protocol for this study was approved by the Ethics Committee of Nanchong Central Hospital, and a signed Informed Consent form was obtained from each participant prior to study enrollment. Tumor tissues and adjacent tissues were harvested and immediately stored at −80°C or fixed in 4% paraformaldehyde for use in further experiments. The study was performed in accordance with the ethical principles of the Declaration of Helsinki.
Immunohistochemistry Analysis (IHC)
IHC methods were used to detect PRDX6 in tumor and para-carcinoma tissues. The tissues were fixed overnight in 4% paraformaldehyde, and then embedded in paraffin and sliced into 5 μm sections with a microtome (Cat. #HM325, Thermo, USA). After deparaffinization and hydration, the sections were incubated overnight at 4°C with a primary antibody against PRDX6 (Abcam, Cambridge, UK). The immunostained tissue sections were then photographed 3 times at ×400 magnification.
Cell Culture And Treatment
A549 and H1299 cells were obtained from the American Type Culture Collection (ATCC, Manassas, VA, USA). A549 cells were cultured for ~24 h in high glucose-DMEM medium (Hyclone, Logan, Utah, USA); H1299 cells were cultured for 24 h in Roswell Park Memorial Institute (RMPI-1640) medium (Hyclone). All media contained 10% fetal bovine serum (FBS, Hyclone) and 1% penicillin/streptomycin (P1400, Solarbio, Beijing, China). Both A549 and H1299 cells were grown in a humidified atmosphere with 5% CO2 at 37°C.
Following dilution into single cell suspensions and being seeded into the wells of different plates (1 x 105 cells/mL), the cells were transfected with or without PRDX6-siRNA (1 nM, Genepharma, Shanghai) for 72 h, with the use of Lipofectamine 2000 (Invitrogen, Carlsbad, CA, USA). Subsequently, the cellular proteins were harvested at specified times. Cells transfected with PRDX6-siRNA-NC served as control cells.
Immunomagnetic Separation (IMS)
Cells were rinsed in PBS and resuspended at a density of 1 × 107 cells/mL. Next, 300 mL of cell suspension was incubated with FcR blocking reagent (100 mL) and CD133-PE antibody (100 mL) for 30 min at 4°C in the dark. An immunoglobulin G-PE antibody served as a negative control. The cell suspension (500 mL) was rinsed twice with PBS and used to determine the percentage of CD133+ cells by flow cytometry (Becton Dickinson, Franklin Lakes, NJ, USA). The CD133-positive (CD133+) and CD133-negative (CD133−) cells were re-suspended in serum-free RMPI media. Flow cytometry was used to determine the percentages of CD133+ cell subpopulations prior to and following the separation process.
Immunofluorescence Staining
After separation, cell spheres were plated onto coverslips in RPMI 1640 medium containing 10% FBS and let sit for ~ 24 h before any further use. After 48 h of transfection, the SW620 cells were fixed with 4% PFA for 20 min, and then incubated with 0.3% Triton X-100 for 10 min at room temperature. Next, the treated cells were blocked with 5% goat serum for 30 min at 37°C, and then incubated overnight with the anti-F-actin IgG (1:2000, Biosensis, Australia) at 4°C; after which, they were incubated with goat anti-IgG conjugated to Cy3 (dilution, 1:400; Jackson ImmunoResearch, West Grove, PA, USA) for 1 h at 37°C. Finally, DAPI (1:1000; Sigma-Aldrich, Inc., MO, USA) was used to counterstain the nuclei, and the cells were observed and photographed under an inverted fluorescence microscope (Olympus). Cells in the negative control group were incubated with PBS rather than the primary antibody.
Cell Proliferation Assay
Cells were plated into individual wells and treated with different concentrations of cis-platinum for 24 h. Next, 100 uL of CCK8 solution (Dojindo, Japan) was pipetted into each well and incubated at 37°C for an additional 1 h. The absorbance of each well was measured at 450 nm with a microplate reader.
Real-Time Reverse Transcription-PCR (qRT-PCR)
Total cellular RNA was isolated with an RNA Isolation Kit and UNIQ-10 column (Sangon, Shanghai, China). Next, 1 μg of total RNA was reverse transcribed in a 20 μL volume of reaction solution with the use of a GoScriptTM Reverse Transcription Kit (Promega, Madison, WI, USA).
A 2 μL aliquot of cDNA was subjected to qRT-PCR performed with a TaKaRa Ex Taq RT-PCR Version 2.1 kit (TaKaRa, Shiga, Japan). The relative levels of gene expression were normalized to those for GAPDH and analyzed using the 2−ΔΔCt method. The PCR primers used for PRDX6, CD133, ABCG-2, and GAPDH were as follows: PRDX6: F: 5ʹ-AAGCTAGCGCCCTAGCGCGATCGCGAGG-3ʹ, R: 5ʹ-AAACGCGATCGATATCGACCCCATCGAC-3ʹ; CD133: F: 5ʹ-GGCGCCTATAGCTAGCTAGCGCGAT-3ʹ, R: 5ʹ-CGCGATCGATGATATCGCGCATA-3ʹ; GAPDH: F: 5ʹ-CGGAGTCAACGGATTTGGTCGTAT-3ʹ, R: 5ʹ-AGCCTTCTCCATGGTGGTGAAGAC-3ʹ.
Western Blot Assay
A total protein isolation kit (Solarbio, Beijing, China) was used to extract the total proteins from tissues and cells. The protein concentration of each sample was estimated with a BCA protein assay kit (P0012, Beyotime, Shanghai, China). After separation by 10% SDS-PAGE electrophoresis, the target proteins were transferred onto polyvinylidene fluoride (PVDF) membranes (Millipore, Burlington, MA, USA), which were subsequently blocked with 5% non-fat milk for 2 h. The membranes were then incubated with primary antibodies overnight at 4°C, and then treated with a secondary antibody (peroxidase-conjugated) for 2 h. Finally, the immunostained proteins were visualized with X-ray film (Fujifilm). The primary antibodies included anti-PRDX6 (1:10000; Santa Cruz Biotechnology, Dallas, TX, USA), anti-CD133 (1:10,000; Abcam, Cambridge, UK), anti-ABCG-2 (1:4000; Abcam), anti-Wnt (1:1500; Abcam), anti-β-catenin (1:2000; Abcam), and anti-GAPDH (1:10,000; Santa Cruz Biotechnology).
Sphere Formation Assay
After 7 days of culture, spheres were seeded into 6-well plates and cultured in a serum-free medium supplemented with EGF (20 ng/mL, Sigma-Aldrich), 2% B27 (Invitrogen), and basic fibroblast growth factor (20 ng/mL, bFGF; BD).
Data Analysis
All statistical analyses were performed using SPSS Statistics for Windows, Version 17.0 (SPSS, Inc., Chicago, IL, USA), and results are expressed as the mean ± SD. Differences between groups were analyzed by using one-way ANOVA or student’s t-test. A P-value < 0.05 was considered statistically significant.
Results
CD133+ And ABCG2+ Subpopulations Presented Resistance To DDP
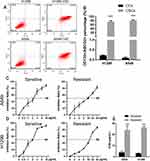
Prior to immunomagnetic selection by flow cytometry, our results showed that CD133+/ABCG2+ cells comprised 0.22% ± 0.03% of the H1299 cells and 0.10% ± 0.04%) of the A549 cells. After cell subpopulation enrichment, 74.89% ± 5.35% of the cells were CD133+/ABCG2+ in H1299-CSC, while 0.22 ± 0.03% were CD133 of cells in H1299. Similarly, 81.36 ± 3.02% of cells with CD133+/ABCG2+ in A549-SCS, while 0.10 ± 0.04% in A549 cell line (Figure 1A and B).
As shown in Figure 1C and D, for both the H1299 cell line and A549 cell line, the cisplatin IC50 values for the enriched H1299 and A549 stem-like cells were increased when compared to values for the non-enriched human lung cancer cells, suggesting that the enriched stem-like cells had a higher potential for cisplatin resistance (Figure 1E).
PRDX6 Expression Was Positively Related To CD133 In NSCLC Upregulation Of PRDX6 And CD133 In NSCLC Patients
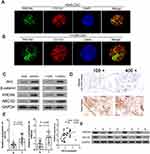
Immunofluorescence studies were performed to demonstrate the expression of PRDX6 and CD133 in A549 (Figure 2A) and H1299 (Figure 2B) cells, and both cell types showed more intense staining after enrichment. We also found that PRDX6 and ABCG2 expression, as well as the expression of several important signaling pathway proteins such as Wnt and β-catenin, were induced after immunomagnetic selection and the enrichment of human lung cancer cells (Figure 2C).
To investigate the PRDX6 expression in patients’ tissues, immunohistochemical assay was performed. Results of immunohistochemical assays showed that PRDX6 was expressed at higher levels in the tumor tissues than in the adjacent tissues (Figure 2D). Next, qRT-PCR analyses were performed to validate findings from the IHC assays. The qRT-PCR analyses suggested that both PRDX6 and CD133 mRNA levels were up-regulated in the tumor tissues when compared with those in the adjacent tissues (Figure 2E, left and middle panel). Interesting, we found that CD133, a stem cell marker, was positively related to PRDX6 (Figure 2E, right panel). Furthermore, similar results were obtained by Western blot analyses (Figure 2F).
PRDX6 Silencing Inhibited The Stem-Like Properties Of Lung Cancer Cells
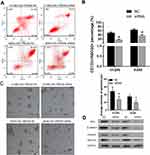
Knockdown of PRDX6 with siRNA was used to examine the role of PRDX6 in cisplatin resistance. After measured by flow cytometry, results showed CD133+/ABCG2+cells accounted for 40.46% ± 5.04% of total H1299 cells in NC and 16.07% ± 2.41% of total A549 cells after PRDX6 knockdown. Besides, that was 64.98 ± 4.88% and 36.47 ± 4.59%, respectively (Figure 3A and B).
Meanwhile, sphere formation assays were conducted to examine the role of PRDX6 in promoting cellular self-renewal, which is a critical trait of CSCs. The sphere formation efficiency of A549 CSCs was suppressed by siRNA, as indicated by a decrease in both spheroid diameters (Figure 3C). Forced depletion of PRDX6 dramatically also reduced the numbers of H1299 CSCs (Figure 3C).
Western blot assays also showed the effect of PRDX6 silencing. As shown in Figure 3D, the levels of PRDX6, ABCG2, and some important signaling pathway proteins such as Wnt and β-catenin were decreased in human lung CSCs transfected with PRDX6 siRNA.
Knockdown Of PRDX6 Suppressed Cisplatin Resistance In H1299 And A549 Cancer Stem-Like Cells
As shown in Figure 4, the cisplatin IC50 values for H1229 and A549 stem-like cells were significantly reduced when the cells were transfected with stable PRDX6 siRNA, suggesting that cisplatin resistance was inhibited by knockdown of PRDX6.
Discussion
Although significant advances have been made in using surgery, external radiation, and interventional radiology in the treatment of patients with NSCLC, the survival rate of those patients, and especially patients with stage III/IV disease, has plateaued in recent years. One reason for that plateau is the resistance of tumors to chemotherapeutic drugs.26 Therefore, it is important to increase the sensitivity of cancer cells to chemotherapy. Our results suggest that NSCLC patients with cisplatin-resistance might have increased levels of PRDX6, as well as more cancer stem-like cells with positive markers, which is consistent with previous research. We also found that the CSCs might increase their tolerance to chemotherapy by activating the Wnt signaling pathway and possess an enhanced sphere formation ability that can be reversed by PRDX6 silencing. These findings imply that silencing of PRDX6 can be used as a new therapeutic strategy for the clinical management of cisplatin-resistant NSCLC. They also suggest PRDX6 as a specific pharmacological target and reducing PRDX6 levels as a potential method of therapeutic intervention.
Ever since the discovery of a subpopulation of CSCs in solid tumors with enhanced tumorigenicity and chemoresistance, CSCs have been considered as a cause of treatment failure and tumor recurrence after chemotherapy. Ovarian CSCs can survive cisplatin treatment due to enhanced translesion DNA synthesis (TLS) resulting from Pol η-mediated enhancement of miR-93 expression.27 In breast cancer tumors, a distinct CSC population that potentially causes chemoresistance has been identified and implicated in the clonal evolution and expansion of cancer stem-like cells.28,29 Although CSCs have been studied in other diseases, further research needs to be conducted on the biology of lung CSCs. There is also a need to identify new therapeutic targets for specifically eradicating that cell population. Recently, scientists generated and characterized a panel of cisplatin-resistant NSCLC cell lines with a putative stem-like signature for use as a valuable research tool.30 Our study identified the protective effect of silencing PRDX6 in NSCLC CSCs in a cisplatin-resistance scenario in vitro, based on previous observations.
Several studies have found a link between the activation of PRDX family members and chemotherapy resistance. Overexpression of PRDX6 has been shown to promote lung tumor development by activating the JAK2/STAT3 pathway31 and increasing glutathione peroxidase31 and iPLA2 activity via upregulation of the AP-1 and JNK pathways.31,32 Moreover, it was found that the development of drug resistance was accompanied by a significant increase in PRDX6 expression in various cisplatin-resistant sublines, including human erythroleukemia K652 cells,33 human breast carcinoma MCF-7 cells,34 and human ovarian carcinoma SKOV-3 cells,23 further confirming the important contribution of redox-dependent mechanisms to the development of cisplatin resistance. Consistent with those findings, we have provided evidence that genetic silencing of PRDX6 or its enzymatic activity renders CSCs sensitive to chemical anti-cancer treatments and abrogates tumor cell sphere formation, laying the foundation for a promising therapeutic strategy. Endogenous and overexpressed PRDX6 was shown to reduce oxidative stress in cancer stem cells, as reflected by lower levels of oxidized phospholipids. This effect led to an accelerated malignant progression of existing tumors.35 ABCG2, which is highly expressed in CSC populations, exports Hoechst-33342 and certain cytotoxic drugs from the interior regions cells, as detected by a side population (SP) analysis.36 When combined with our results, this suggests that a positive correlation might be found between high rates of ABCG2/PRDX6 positivity and high populations of CSCs.
One of the main reasons for the unreasonable use of chemotherapy and occurrence of medicamentous adverse reactions is that CSCs are continually produced under conditions of disease progression and stabilization. Studies have suggested that disorders of highly conserved developmental pathways, including the mutant Wnt/β-catenin pathway, might regulate self-renewal in embryonic and adult stem cells. This regulation could promote CSC proliferation, metastasis, and chemoresistance.37,38 Furthermore, recent studies have shown that the Wnt/β-catenin pathway might participate in regulating stem cells. For example, proliferation nuclear antigen-associated factor (PAF) has the ability to promote self-renewal and heterogeneity.39 Consistent with our study results, aberrant activation of PRDX6 might be involved in dysregulation of the Wnt/β-catenin pathway in NSCLC, and associated with the maintenance of CSCs.
In this study, we identified two different stem-like cell lines with a high tolerance to cisplatin and proved that PRDX6 could accelerate cisplatin resistance in human lung cancer cells by enhancing stem-like properties. Our results suggest a potential new therapeutic strategy of targeting PRDX6 during NSCLC treatment.
Ethics Approval And Informed Consent
Informed consent was obtained from all subjects who participated in the study. The study was performed in accordance with the ethical principles of the Declaration of Helsinki.
Availability Of Data And Materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Disclosure
All authors declare that they have no conflict of interest in this work.
References
1. Yang X-Y, Zhang J, Yu X-L, Zheng GF, Zhao F, Jia XJ. Death-associated protein kinase promoter methylation correlates with clinicopathological and prognostic features in nonsmall cell lung cancer patients: A cohort study. J Cancer Res Ther. 2018;14(8):S65–S71. doi:10.4103/0973-1482.158197
2. Schreiner W, Dudek W, Lettmaier S, Fietkau R, Sirbu H. Long-term survival after salvage surgery for local failure after definitive chemoradiation therapy for locally advanced non-small cell lung cancer. Thorac Cardiovasc Surg. 2018;66(2):135–141. doi:10.1055/s-0037-1606597
3. Guilbault C, Garant A, Faria S, et al. Long-term outcomes of induction carboplatin and gemcitabine followed by concurrent radiotherapy with low-dose paclitaxel and gemcitabine for stage III non-small-cell lung cancer. Clin Lung Cancer. 2017;18(5):565–571. doi:10.1016/j.cllc.2017.02.003
4. Jiang YQ, Xu XP, Guo QM, et al. Reversal of cisplatin resistance in non-small cell lung cancer stem cells by Taxus chinensis var. Genet Mol Res. 2016;15(3). doi:10.4238/gmr.15038336
5. Zwitter M, Rajer M, Stanic K, et al. Intercalated chemotherapy and erlotinib for non-small cell lung cancer (NSCLC) with activating epidermal growth factor receptor (EGFR) mutations. Cancer Biol Ther. 2016;17(8):833–839. doi:10.1080/15384047.2016.1195049
6. Chiu W-T, Huang Y-F, Tsai H-Y, et al. FOXM1 confers to epithelial-mesenchymal transition, stemness and chemoresistance in epithelial ovarian carcinoma cells. Oncotarget. 2015;6(4):2349–2365. doi:10.18632/oncotarget.v6i4
7. Van Houdt WJ, Emmink BL, Pham TV, et al. Comparative proteomics of colon cancer stem cells and differentiated tumor cells identifies BIRC6 as a potential therapeutic target. Mol Cell Proteomics. 2011;10(12):M111 011353. doi:10.1074/mcp.M111.011353
8. Penfornis P, Cai DZ, Harris MR, et al. High CD49f expression is associated with osteosarcoma tumor progression: a study using patient-derived primary cell cultures. Cancer Med. 2014;3(4):796–811. doi:10.1002/cam4.2014.3.issue-4
9. Nabissi M, Morelli MB, Amantini C, et al. Cannabidiol stimulates Aml-1a-dependent glial differentiation and inhibits glioma stem-like cells proliferation by inducing autophagy in a TRPV2-dependent manner. Int J Cancer. 2015;137(8):1855–1869. doi:10.1002/ijc.29573
10. Corces MR, Chang HY, Majeti R. Preleukemic hematopoietic stem cells in human acute myeloid leukemia. Front Oncol. 2017;7:263. doi:10.3389/fonc.2017.00263
11. Choi SH, Lee SW, Ok M, Kim KS, Kim S, Ahn SH. Gene expression profiling of hepatocellular carcinoma derived cancer stem like cell under hypoxia. Yonsei Med J. 2017;58(5):925–933. doi:10.3349/ymj.2017.58.5.925
12. Gopal K, Gupta N, Zhang H, et al. Oxidative stress induces the acquisition of cancer stem-like phenotype in breast cancer detectable by using a Sox2 regulatory region-2 (SRR2) reporter. Oncotarget. 2016;7(3):3111–3127. doi:10.18632/oncotarget.v7i3
13. Zhao W, Luo Y, Li B, Zhang T. Tumorigenic lung tumorospheres exhibit stem-like features with significantly increased expression of CD133 and ABCG2. Mol Med Rep. 2016;14(3):2598–2606. doi:10.3892/mmr.2016.5524
14. Mao Q, Unadkat JD. Role of the breast cancer resistance protein (BCRP/ABCG2) in drug transport–an update. AAPS J. 2015;17(1):65–82. doi:10.1208/s12248-014-9668-6
15. Phiboonchaiyanan PP, Kiratipaiboon C, Chanvorachote P. Ciprofloxacin mediates cancer stem cell phenotypes in lung cancer cells through caveolin-1-dependent mechanism. Chem Biol Interact. 2016;250:1–11. doi:10.1016/j.cbi.2016.03.005
16. Lu D, Wang W, Liu J, et al. Peroxiredoxins in inflammatory liver diseases and ischemic/reperfusion injury in liver transplantation. Food Chem Toxicol. 2018;113:83–89. doi:10.1016/j.fct.2018.01.025
17. Rhee SG, Kil ISJARoB. Multiple functions and regulation of mammalian peroxiredoxins. Ann Rev Biochem. 2017;86(1):
18. Tu D-D, Zhou Y-L, Gu W-B, et al. Identification and characterization of six peroxiredoxin transcripts from mud crab Scylla paramamosain: the first evidence of peroxiredoxin gene family in crustacean and their expression profiles under biotic and abiotic stresses. Mol Immunol. 2018;93:223–235. doi:10.1016/j.molimm.2017.11.029
19. Ren L, Sun Y, Wang R, Xu T. Gene structure, immune response and evolution: comparative analysis of three 2-Cys peroxiredoxin members of miiuy croaker, Miichthys miiuy. Fish Shellfish Immunol. 2014;36(2):409–416. doi:10.1016/j.fsi.2013.12.014
20. Ho J-N, Lee SB, Lee S-S, et al. Phospholipase A2 activity of peroxiredoxin 6 promotes invasion and metastasis of lung cancer cells. Mol Cancer Ther. 2010;9(4):825–832. doi:10.1158/1535-7163.MCT-09-0904
21. Hirota Y, Acar N, Tranguch S, et al. Uterine FK506-binding protein 52 (FKBP52)–peroxiredoxin-6 (PRDX6) signaling protects pregnancy from overt oxidative stress. Proc Natl Acad Sci U S A. 2010;107(35):15577–15582. doi:10.1073/pnas.1009324107
22. Chhunchha B, Fatma N, Bhargavan B, Kubo E, Kumar A, Singh DP. Specificity protein, Sp1-mediated increased expression of Prdx6 as a curcumin-induced antioxidant defense in lens epithelial cells against oxidative stress. Cell Death Dis. 2011;2(11):e234.
23. Pak JH, Choi WH, Lee HM, et al. Peroxiredoxin 6 overexpression attenuates cisplatin-induced apoptosis in human ovarian cancer cells. Cancer Invest. 2011;29(1):21–28. doi:10.3109/07357907.2010.535056
24. Walsh B, Pearl A, Suchy S, Tartaglio J, Visco K, Phelan SA. Overexpression of Prdx6 and resistance to peroxide-induced death in Hepa1-6 cells: prdx suppression increases apoptosis. Redox Rep. 2009;14(6):275–284. doi:10.1179/135100009X12525712409652
25. Lee SB, Ho J-N, Yoon SH, Kang GY, Hwang SG, Um HD. Peroxiredoxin 6 promotes lung cancer cell invasion by inducing urokinase-type plasminogen activator via p38 kinase, phosphoinositide 3-kinase, and Akt. Mol Cells. 2009;28(6):583–588. doi:10.1007/s10059-009-0152-6
26. Wong KM, Noonan S, O’Bryant C, Jimeno A. Alectinib for the treatment of ALK-positive stage IV non-small cell lung cancer. Drugs Today. 2015;51(3):161–170. doi:10.1358/dot.2015.51.3.2294597
27. Srivastava AK, Han C, Zhao R, et al. Enhanced expression of DNA polymerase eta contributes to cisplatin resistance of ovarian cancer stem cells. Proc Natl Acad Sci U S A. 2015;112(14):4411–4416. doi:10.1073/pnas.1421365112
28. Lindeman GJ, Visvader JE. Insights into the cell of origin in breast cancer and breast cancer stem cells. Asia Pac J Clin Oncol. 2010;6(2):89–97. doi:10.1111/j.1743-7563.2010.01279.x
29. Shafee N, Smith CR, Wei S, et al. Cancer stem cells contribute to cisplatin resistance in Brca1/p53-mediated mouse mammary tumors. Cancer Res. 2008;68(9):3243–3250. doi:10.1158/0008-5472.CAN-07-5480
30. Barr MP, Gray SG, Hoffmann AC, et al. Generation and characterisation of cisplatin-resistant non-small cell lung cancer cell lines displaying a stem-like signature. PLoS One. 2013;8(1):e54193. doi:10.1371/journal.pone.0054193
31. Yun H-M, Park K-R, Park MH, et al. PRDX6 promotes tumor development via the JAK2/STAT3 pathway in a urethane-induced lung tumor model. Free Radic Biol Med. 2015;80:136–144. doi:10.1016/j.freeradbiomed.2014.12.022
32. Yun H-M, Park MH, Kim DH, et al. Loss of presenilin 2 is associated with increased iPLA2 activity and lung tumor development. Oncogene. 2014;33(44):5193–5200. doi:10.1038/onc.2014.128
33. Kalinina EV, Berezov TT, Shtil’ AA, Glazunova VA, Novichkova MD, Nurmuradov NK. Expression of peroxiredoxin 1, 2, 3, and 6 genes in cancer cells during drug resistance formation. Bull Exp Biol Med. 2012;153(6):879–882. doi:10.1007/s10517-012-1849-7
34. Glorieux C, Dejeans N, Sid B, Beck R, Calderon PB, Verrax J. Catalase overexpression in mammary cancer cells leads to a less aggressive phenotype and an altered response to chemotherapy. Biochem Pharmacol. 2011;82(10):1384–1390. doi:10.1016/j.bcp.2011.06.007
35. Rolfs F, Huber M, Gruber F, et al. Dual role of the antioxidant enzyme peroxiredoxin 6 in skin carcinogenesis. Cancer Res. 2013;73(11):3460–3469. doi:10.1158/0008-5472.CAN-12-4369
36. Teng L, Lei H-M, Sun F, et al. Autocrine glutamatergic transmission for the regulation of embryonal carcinoma stem cells. Oncotarget. 2016;7(31):49552–49564. doi:10.18632/oncotarget.v7i31
37. Aminuddin A, Ng PY. Promising druggable target in head and neck squamous cell carcinoma: Wnt signaling. Front Pharmacol. 2016;7:244. doi:10.3389/fphar.2016.00244
38. Jang G-B, Kim J-Y, Cho S-D, et al. Blockade of Wnt/β-catenin signaling suppresses breast cancer metastasis by inhibiting CSC-like phenotype. Sci Rep. 2015;5:12465. doi:10.1038/srep12465
39. Sun R, Liu Y, Li S-Y, et al. Co-delivery of all-trans-retinoic acid and doxorubicin for cancer therapy with synergistic inhibition of cancer stem cells. Biomaterials. 2015;37:405–414. doi:10.1016/j.biomaterials.2014.10.018
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.