Back to Journals » Nature and Science of Sleep » Volume 14
Differences in Predicted Therapeutic Outcome of Mandibular Advancement Determined by Remotely Controlled Mandibular Positioner in Canadian and Chinese Apneic Patients
Authors Li WY , Masse JF, Gakwaya S, Zhao Z, Wang W, Sériès F
Received 17 June 2022
Accepted for publication 28 August 2022
Published 8 September 2022 Volume 2022:14 Pages 1611—1622
DOI https://doi.org/10.2147/NSS.S377758
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Ahmed BaHammam
Wen-Yang Li,1 Jean-Francois Masse,2 Simon Gakwaya,3 Zhenjin Zhao,4 Wei Wang,1,* Frédéric Sériès3,*
1Institute of Respiratory and Critical Care, The First Hospital of China Medical University, Shenyang, People’s Republic of China; 2Faculté de Médecine Dentaire, Université Laval, Québec, QC, Canada; 3Unité de Recherche en Pneumologie, Centre de Recherche, Institut Universitaire de Cardiologie et de Pneumologie de Québec, Université Laval, Québec, QC, Canada; 4Institute of Orthodontics, The Stomatological Hospital of China Medical University, Shenyang, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Wei Wang, Institute of Respiratory Disease, The First Hospital of China Medical University, 155 Nanjing North Street, Heping District, Shenyang, 110001, People’s Republic of China, Tel: +86 83282532, Email [email protected] Frédéric Sériès, Centre de recherche, IUCPQ, 2725 chemin Sainte-Foy, Québec, QC, G1V 4G5, Canada, Email [email protected]
Background: In-lab mandibular protrusive titration using a remotely controlled mandibular positioner (RCMP) could predict the success rate of mandibular advancement device (MAD) and reliably determine the Optimal Protrusive Position (OPP) for obstructive sleep apnea (OSA) patients. The aim of this study was to compare MAD success rate using in-lab RCMP manual titration performed in Caucasian and Chinese OSA patients.
Methods: Manual RCMP titration was performed during an in-lab sleep study using the same procedure that had been previously reported in untreated Caucasian and Chinese OSA patients. Success rate was determined according to classical success criteria or to those previously used for RCMP titration.
Results: A total of 160 subjects were included in this study, and conclusive data were obtained from 141 (71 Chinese and 70 Caucasian OSA patients). Chinese patients were significantly younger, with lower BMI and more severe OSA disease than the Canadian counterparts. Among patients with predicted success, the OPP expressed in % of full protrusion position did not differ between the two ethnic groups. Chinese ethnicity, younger age and lower baseline AHI were significant determinants of RCMP success. In a multivariate analysis, only ethnicity and AHI were found to significantly account for success, the odds ratio for success in Chinese compared to Caucasians corrected for AHI being 3.7 and 4.6 depending on criteria used to define success.
Conclusion: Although the OSA disease was more severe in Chinese patients, the predicted success rate of MAD according to RCMP titration was higher in Chinese than in Caucasians. This study was registered on ClinicalTrials.gov (NCT03231254).
Keywords: obstructive sleep apnea, mandibular advancement device, remotely controlled mandibular positioner, optimal protrusive position
Preamble
Obstructive sleep apnea (OSA) is a common disorder characterized by repetitive partial or complete upper airway collapse causing sleep fragmentation and intermittent hypoxemia. It is associated with excessive daytime sleepiness, impaired cognitive performance, increased risk of motor vehicle accidents and reduced quality of life.1 It is also a strong and independent risk factor for cardiovascular and cerebrovascular disease,2–4 together with the associated all-cause mortality, collectively justifying the need for effective long-term treatment.
From a clinical point of view, besides various types of therapy that can normalize breathing at night including weight loss, continuous positive airway pressure and upper airway surgery, oral appliances such as mandibular advancement device (MAD) have emerged as one acceptable therapy of OSA. According to recommendations of the American Academy of Sleep Medicine, MAD is accepted as a first-line therapy in patients with mild-to-moderate OSA and in more severe OSA patients who fail treatment attempts with CPAP therapy.5 The remotely controlled mandibular positioner (RCMP) device (MATRx, Zephyr Sleep Technologies Inc., Calgary, Canada) can be used to determine the ability of mandibular protrusion to normalize breathing at night with a specificity and sensitivity in predicting treatment response. Using a temporary dental appliance during a conventional polysomnographic study, the device allows for a step-by-step protrusion aimed at abolishing breathing disturbances particularly in REM sleep while supine and determining the optimal protrusion position (OPP).6
Many factors such as gender, obesity, skeletal malformations, soft tissue crowding and physiologic traits such as loop gain and arousal threshold can contribute to the occurrence of OSA. Ethnicity with its genetic, environmental and cultural components can interact with the recognized risk factors for OSA individually or in combination (ie obesity and craniofacial features in Caucasian and Asian populations).7–9 Therefore, the anatomical/physiologic factors involved in the occurrence of upper airway closure may differ according to ethnicity and could result in differences in MAD success rate between these two ethnic populations.10,11 Therefore, taking advantages of the standardization of the mandibular advancement titration technique using the RCMP, we designed a prospective study to compare the predicted therapeutic outcome of MAD between the two ethnic populations. We hypothesize that the predicted success rate will be different between two ethnicities.
Methods
Subjects
Participants were respectively recruited from the IUCPQ Sleep Clinic (Quebec, Canada) and the first affiliated hospital of CMU (Shenyang, China), which receives the majority of referrals in Quebec City and Shenyang City. Data from consecutive patients eligible for MAD according to the following clinical criteria were collected: 18 to 75 years of age, recent diagnosis of OSA (AHI > 15/h), mean oxygen saturation during sleep >90%, mandibular range of motion >5 mm, adequate dentition according to the dentists with ≥10 upper and 10 lower teeth. MAD was not considered when body mass index ≥40 kg/m2, presence of severe pharyngeal obstruction on physical examination (pharyngeal grade IV), nasal obstruction incompatible with exclusive nasal breathing and >50% of observed sleep apnea scored as central. Patients with temporomandibular disorders (TMD) were not considered in this study.
This study complied with the Declaration of Helsinki. Revision of medical files was approved by IUCPQ and CMU hospital internal review board. All subjects from Canada and China that were enrolled in this study went through the same research protocol (see study design below) and the informed consent was obtained from all the study participants prior to study commencement. Our study was registered on ClinicalTrials.gov (NCT03231254).
Procedures
RCMP titration was completed according to procedures described in former publications.6 Briefly, apneic patients eligible for MAD were referred to the dentist who confirmed treatment eligibility. After fitting the trays to the participant’s upper and lower teeth, the dentist assessed the scale readings for full retrusion and protrusion position (FPP) and habitual bite position (resting position). These readings quantified the participants’ mandibular range of motion and were used to determine the resting and maximal advancement levels to be provided to the PSG technologist for the overnight RCMP titration study. This one was carried out under PSG monitoring in the sleep laboratory, patients’ mandible being progressively advanced without disturbing sleep until obstructive respiratory events were eliminated. The titration procedure was completed according to previous described methods.6 The technician was provided with a pre-defined titration protocol including “up” and “down” adjustment of the device, together with their experienced judgement, to protrude mandible so as to eliminate obstructive respiratory events, similar to positive pressure adjustments during CPAP titration.6 The polysomnographic study was scored by a certified technologist using the Version 2.3 of the American Academy of Sleep Medicine Manual for the Scoring of Sleep and Associated Events.12 A minimum of 4 hours of technically satisfactory recording during sleep was needed to establish conclusive predictive data.
Characterization of Patients Following RCMP Titration
RCMP Titration Results According to Conventional Success Criteria
Predictive response to MAD therapy was assessed individually according to the AHI achieved at the optimal protrusion level during the RCMP titration night using two different success criteria. The first one used a conventional success criteria, where predicted success corresponded to AHI < 10/h with a reduction of AHI ≥ 50% compared to baseline for the total sleep time recorded at optimal advancement. The optimal protrusive position (OPP) is the lowest protrusive position at which such AHI values are achieved.
RCMP Titration Results According to Remmers Success Criteria
Success analysis was also completed using criteria established by Remmers et al, where predicted success was deemed when ≤1 respiratory event did occur per 5 min of supine REM (or lateral in side sleepers) at optimal protrusion level.6 Predicted failure was anticipated when >1 respiratory event did occur per 5 min of supine REM (or lateral in side sleepers) at the maximal protrusion level. If less than 5 min of REM sleep supine or lateral (for side-sleepers) were recorded, the response was classified as inconclusive.
Inter-Laboratory Concordance Analysis
In order to compare between centers inter-rater variability, a blind scoring was completed on 600 epochs randomly selected from tracings of 30 patients of each center to assess concordance in scoring of sleep and breathing abnormalities.
Statistical Analysis
In the absence of reliable data from the literature that could be used to estimate differences in MAD success between the two populations, the sample size was determined according to the results of a pilot study completed in our first 39 patients (16 Chinese, 23 Canadians). It was found that 160 subjects were needed to identify a significant difference in MAD predicted success between the two populations with a 90% study power and a 0.05 alpha error. Nominal variables were expressed with frequencies and percentage (%) and were analysed using Fisher’s exact test. Continuous variables were analyzed using one-way ANOVA. For some variables, one-way ANOVA was performed on separate residual variances due to heterogeneity in the covariance structure. Continuous variables expressed with median and interquartile range were analyzed using the Wilcoxon rank-sum test. A logistic regression analysis that identified variables independently associated with success was performed. For the multivariable model building, the variables were selected only if they maximized the within sample prediction rates. Following model building, the Hosmer–Lemeshow test was performed to assess the goodness-of-fit of the model. The level of scoring agreement between the two centers was assessed by the concordance correlation coefficient using the weighted Kappa with 95% intervals.
As to the cumulative occurrence of predicted success curve, an interval-censored analysis was performed with a Weibull distribution to investigate the distribution of patients predicted to be successful as a function of OPP. For all statistical analyses, the results were considered significant with P-values <0.05. All analyses were performed with the use of SAS software, version 9.4 (SAS Institute Inc., Cary, NC).
Results
A patient flow chart is presented in Figure 1. A total of 160 subjects were included in this study. Two Chinese and four Caucasian participants failed to complete the titration study due to discomfort with the temporary oral appliance. In the 154 patients who completed the titration study, conclusive data were obtained from 141 (71 ethnically Chinese and 70 ethnically Caucasians) OSA patients. In 13 patients, the RCMP titration study was inconclusive due to insufficient REM sleep in eleven participants (six Chinese and five Caucasians) and incomplete mandibular protrusion in two other Canadian subjects. The concordance correlation coefficient was 0.69 (0.63–0.75 95% CI) for sleep scoring and 0.61 (0.55–0.66 95% CI) for scoring of breathing disturbances. The inter-laboratory analysis showed a strong reproducibility (kappa 0.78 for sleep staging – 95% CI: 0.73–0.84 -, 0.83 for identification of SDB – 95% CI: 0.79–0.88 -, and 0.79 for identification of arousals – 95% CI: 0.74–0.85 -, p < 0.001).
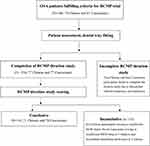 |
Figure 1 Flow chart of patients included in the study. |
The demographic characteristics and disease severity of subjects with conclusive RCMP titration study are presented in Table 1. Gender significantly differed between the two populations (83% and 63% males in Chinese and Caucasians, respectively, p=0.008). When compared with the Caucasian counterparts, Chinese patients were significantly younger [F (1139) = 35.63, p< 0.0001], with lower BMI [F (1119) = 8.17, p = 0.005] and higher AHI [F (1114) = 12.96, p = 0.0005]. The AI, REM AHI and ODI were also significantly higher in Chinese than Caucasian subjects. The resting mandibular position significantly differed between the Chinese (5.4 ± 2.0 mm) and Caucasian subjects (8.6 ± 2.4 mm) [F (1139) = 75.07, p< 0.0001]. The FPP in Chinese (14.6 ± 2.9 mm) was also significantly lower than in Caucasians (16.5 ± 2.4 mm) [F (1139) = 18.07, p<0.0001]. These differences were also observed when considering all the subjects who completed the RCMP titration (N = 154).
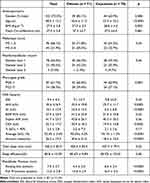 |
Table 1 Baseline Characteristics of Subjects with Conclusive RCMP Titration Study |
According to the classical success criteria, individual AHI values obtained at baseline and at OPP as a function of BMI in each ethnic population are displayed in Figure 2. It illustrates that there is a clear overlap in characteristics of patients of the anticipated success and failure groups. Subjects with predicted success had BMI values ranging from 20.3 to 33.0 kg/m2 and baseline AHI values ranging from 6.7/h to 73.3/h (top panel). Among the 47 Chinese and 30 Caucasian patients of this predicted success group, 25 (62.5%) Chinese and 16 (53.3%) Caucasians had BMI and/or baseline AHI values that exceeded those usually recommended for selecting candidates for oral appliance therapy (BMI < 30 kg/m2 and AHI < 30/h). Moreover, 6 (25.0%) Chinese and 15 (37.5%) of Caucasian subjects lying within the recommended guidelines were not identified as predicted success. According to the classical success criteria, 77 patients were predicted to have therapeutic success outcome (Table 2). Chinese ethnicity [47(61.0%) vs 24 (37.5%), p = 0.007], younger age (45.7 ± 11.8 y vs 50.8 ± 12.1 y, p = 0.01), lower baseline AHI (27.2 ± 15.3 events/h vs 34.8 ± 18.1events/h, p = 0.008) and lower resting bite position (6.6 ± 2.4 mm vs 7.6 ± 2.9, p = 0.03) were significant determinants of RCMP success. In the logistic regression model, only ethnicity (p = 0.0002) and AHI (p = 0.0004) was found to significantly account for predicted RCMP success. When corrected for AHI, the odds ratio [OR (95% CI)] for predicted success was 4.60 [2.04–10.39] in Chinese compared to Caucasians (Table 3).
 |
Table 2 Determinants of Predicted Outcomes According to Results of RCMP Titration |
 |
Table 3 Multivariate Regression Analysis Predicted Outcomes According to Results of RCMP Titration |
According to Remmers’ criteria, 56 patients were predicted to have therapeutic success outcome, while 85 resulted in anticipated failure (Table 2). Chinese ethnicity [34 (60.7%) vs 37 (43.5%), p = 0.058], younger age (45.0 ± 12.0 y vs 50.3 ± 11.8 y, p = 0.006), lower baseline AHI (23.9 ± 14.1 events/h vs 35.1 ± 17.3 events/h, p < 0.0001) and lower resting bite position (6.3 ± 2.6 mm vs 7.5 ± 2.7, p = 0.013) were significant determinants of RCMP success. In the logistic regression model, only ethnicity (p = 0.0012) and AHI (p < 0.0001) were found to significantly account for predicted RCMP success. When corrected for AHI, the odds ratio [OR (95% CI)] for predicted success was 3.76 (1.68–8.39) in Chinese compared to Caucasians (Table 3).
Among patients with predicted success according to Remmers’ criteria, the OPP expressed in % of FPP did not differ between the two ethnic groups (Chinese vs Caucasians: 76.4%±18.2% vs 79.8%±21.7%) (p > 0.1) (Figure 3A). The cumulative occurrence of predicted success as a function of OPP (expressed in % of FPP) is also analyzed in Figure 3B. No significant difference in cumulative predicted success rate was observed between the two ethnic groups (log rank, p > 0.05). Moreover, the cumulative success curves as a function of OPP were also analyzed according to BMI, age and AHI values stratified according to their respective median values and according to Mallampati score. Predicted success was found to occur at a lower OPP in those with BMI < 27.5 kg/m2 (log rank, p = 0.058), less crowded oropharyngeal (MS 1–2) (log rank, p = 0.04), younger age (<47 y) (log rank, p = 0.003) and less-severe disease (AHI <20/h) (log rank, p = 0.02) when compared with the others (Figure 4A–D). Similar results were also seen with the classical criteria analysis.
Discussion
Our results demonstrate that using a standardized in-laboratory oral appliance titration procedure, Chinese subjects are more prone to reach therapeutic success than Canadians. Such findings were observed taking into account for variables known as conventional determinants of MAD success.
To our knowledge, this study is the first to compare the predicted therapeutic outcome of MAD from well-characterized samples of ethnically Caucasian and Asian OSA patients from Canada and China. In this study, the RCMP procedure was used during an in-lab standardized mandibular protrusive titration protocol to predict the MAD therapeutic outcome and also prospectively determines the optimal protrusion position for participants predicted to be therapeutically successful with MAD therapy. In the absence of available results from the literature, our sample size was determined according to the results of a pilot study conducted in 39 patients. To our knowledge, the present cohort is the largest one using RCMP prospectively as a mean to predict MAD outcomes. Noteworthy, two different criteria were used to determine the predicted outcome of MAD therapy. When compared with the conventional criteria, the Remmers’ criteria were stricter and the outcome was determined during or immediately after the titration procedure. A key finding of this study is that Chinese ethnicity, younger age and lower baseline AHI were significant determinants of RCMP success with both Remmers’ and classical success criteria. In a multivariate analysis, only ethnicity and AHI were found to significantly account for success independently of the success criteria, with the odds ratio for success in Chinese compared to Caucasians corrected for AHI being 3.7 and 4.6 using Remmers’ and classical criteria, respectively.
It is well recognized that the ability of MAD to normalize breathing at night is less than what is seen with CPAP.13 Furthermore, important variations in success rates have been reported in Asian populations (31–75%).14,15 However, methodological issues – small sample size, confining subjects to severe OSA patients,16 lack of one-night titration using PSG or using non-adjustable OA following not standardized titration procedure could account for such variability.17,18 Furthermore, these studies frequently used appliances that are not validated as effective treatment in OSA. In this regard, the RCMP device used in this study was used to overcome the influence of MAD type and titration procedure on its anticipated success by completing an identical standardized in-laboratory manual titration procedure in both ethnic populations aimed at accurately identifying MAD favourable candidates.6,19–21
In accordance with previous results of the literature, the current study confirmed that the severity of OSA at baseline is the best predictor of the treatment success with MAD regardless of ethnicity.22,23 However, for a given subject, anticipated success can be seen in some patients with severe OSA in both ethnic groups. In addition, the cumulative occurrence of RCMP success is more pronounced with lower OPP when the patients were younger (<47y), with less severe OSA (AHI <20/h) or less crowded oropharyngeal area (MS 1–2). Consistently, age is a predictive variable for OA treatment outcome.24–26 This age-related reduction of MAD efficacy was ethnic independent and could be attributed to the decreased pharyngeal cross-sectional area and upper airway muscle activity, as well as the adaptation of the mandibular shape with aging that ultimately lead to the increment of the pharyngeal collapsibility.27,28 In addition, our findings are also in line with the previous observations from Japanese OSA patients demonstrating the unfavorable effect of high MS on the clinical outcome of MAD treatment.29,31
It is noteworthy that our study stressed the importance of studying response to RCMP using ethnicity as a phenotype. In the current study, Chinese patients had more severe OSA disease. This is expected since, for the same degree of obesity, Asian ethnicity exhibits more craniofacial bony restrictions compared to Caucations.8 In the current study, although the mean AHI was higher by 9.8 events/h in Chinese patients, the predicted success rate of MAD according to the RCMP titration was 17.2–23.5% higher than in Canadian patients according to the different success criteria. Multivariate analysis confirmed that ethnicity is a strong predictor of MAD success with odds ratio for success in Chinese compared to Caucasians corrected for AHI being 3.7 and 4.6 using Remmers’ and classical criteria, respectively. From a pathophysiologic point of view, differences in anticipated MAD therapeutic success may suggest that OSA disease in Asians is driven much more by the relative contribution of their anatomical predisposition (lower upper airway gain) and to a lesser extent by non-anatomical causes (lower loop gain) compared to Caucasians.8,23,30 Indeed, the higher frequency of obstructive apneas observed in Chinese patients (higher propensity for complete upper airway occlusion) may relate to more severe anatomical compromise in this ethnic group.
In the current study, the habitual occlusal position and maximal voluntary protrusion position of the lower mandible (determined by the scales on RCMP trays) were both lower in Chinese patients than in Canadians. This is in accordance with the shorter mandibular length observed in Chinese population.7 Among patients with predicted success, no difference in OPP was found between Chinese and Canadian subjects. Our OPP data were higher than that reported in Remmers’ former study (median OPP being 68% of the maximal titration protrusive range).6 Such difference might be ascribed to the different traits (ie sex, age, BMI and disease severity) characterizing population enrolled in the two studies. Notably, in the current study, the demographic characteristics and OSA severity are expected to contribute differentially to OPP in two ethnic groups.
When interpreting the present results, several limitations have to be considered. Enrolled patients were continuously recruited from the flow of patients who were candidates for MAD as a treatment of OSA and had an RCMP titration. In this context, enrolled subjects could not be matched for potential confounding factors such as sex, BMI, age and AHI. Besides, the difference in socioeconomic status, local culture and environmental factors between Canadians and Chinese could also have influenced the modalities of recruitment according to inclusion criteria and then population characteristics. For instance, Chinese from the northeast of China are prone to more severe periodontitis and tooth loss when compared to the Caucasians.29,32 Thus, the poor dental condition in China emerged as the main obstacle for recruiting older Chinese subjects. At least, RCMP titration was used to identify MAD predicted success but no comparison was made on final polysomnographic results obtained with the effective MAD treatment. Nevertheless, although it was difficult to generalize our findings given differences in age and disease severity between two groups, this study was the first looking at ethnic differences regarding the effectiveness of MAD treatment. On the other hand, the analysis of MAD therapeutic outcomes would be severely biased by the fact that 1) patients with anticipated RCMP failure usually do not proceed to MAD treatment, 2) it would be impossible to use a standardized oral appliance model in both populations, 3) loss to follow-up may be seen during the course of home-titration procedure. In this instance, the RCMP device allowed us to standardize the titration procedure and avoid interaction with above-mentioned important confounders.
Collectively, we successfully conducted and compared the predicted therapeutic outcome of MAD from well-characterized samples of ethnically Caucasian and Asian OSA patients from Canada and China, though it was difficult to exactly match the age, BMI, and AHI across two ethnicity groups. Our results demonstrate that even considering the influence of factors known to influence MAD efficiency to normalize sleep breathing disturbances, the predicted success rate of MAD according to the RCMP titration was superior in Chinese than Canadians, with similar OPP between the two ethnics. Further investigation using imaging tools and comprehensive analysis, together with the result of the final MAD therapeutic outcome, will help to further identify the interaction of the anatomical factors on the MAD therapeutic efficiencies across ethnic groups.
Data Sharing Statement
Individual participant data that support the findings of this study after deidentification are available on request from the corresponding author. Study protocol and statistical analysis plan are available immediately following publication. Requests for material should be made to the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Acknowledgments
Research time devoted to assess inter-rater concordance in Quebec was covered by a grant from the Fondation de l’IUCPQ. Remotely controlled mandibular positioner devices were courtesy of Zephyr Sleep Technologies Inc., Calgary, Canada. We thank all the efforts of staff dedicated in this project from IUCPQ and the first hospital of CMU.
Funding
National Natural Science Foundation of China. (NO.81900091); 2.Project of Education Department of Liaoning Province. (NO. ZF2019030).
Disclosure
The abstract of this paper was presented at the 2018 APSS Conference (in Baltimore) as a poster presentation with interim findings. The poster’s abstract was published in “Poster Abstracts” in Sleep, Volume 40, Issue suppl_1, 28 April 2017, Page A220, name “DIFFERENCES IN PREDICTED THERAPEUTIC OUTCOME AND OPTIMAL PROTRUSION POSITION OF ORAL APPLIANCE DETERMINED DURING PSG WITH REMOTELY CONTROLLED MANDIBULAR POSITIONER BETWEEN CANADIAN AND CHINESE OSA PATIENTS”. https://academic.oup.com/sleep/article/40/suppl_1/A220/3781917
The authors report no conflicts of interest in this work.
References
1. Young T, Palta M, Dempsey J, Skatrud J, Weber S, Badr S. The occurrence of sleep-disordered breathing among middle aged adults. N Engl J Med. 1993;328(17):1230–1235. doi:10.1056/NEJM199304293281704
2. Malhotra A, White DP. Obstructive sleep apnoea. Lancet. 2002;360(9328):237–245. doi:10.1016/S0140-6736(02)09464-3
3. Peppard PE, Young T, Palta M, Skatrud J. Prospective study of the association between sleep-disordered breathing and hypertension. N Engl J Med. 2000;342(19):1378–1384. doi:10.1056/NEJM200005113421901
4. Bradley TD, Floras JS. Obstructive sleep apnoea and its cardiovascular consequences. Lancet. 2009;373(9657):82–93. doi:10.1016/S0140-6736(08)61622-0
5. Ramar K, Dort LC, Katz SG, et al. Clinical practice guideline for the treatment of obstructive sleep apnea and snoring with oral appliance therapy: an update for 2015. J Clin Sleep Med. 2015;11(7):773–827. doi:10.5664/jcsm.4858
6. Remmers J, Charkhandeh S, Grosse J, Topor Z, Brant R, Santosham P. Remotely controlled mandibular protrusion during sleep predicts therapeutic success with oral appliances in patients with obstructive sleep apnea. Sleep. 2013;36(10):1517–1525. doi:10.5665/sleep.3048
7. Schorr F, Kayamori F, Hirata RP, et al. Different craniofacial characteristics predict upper airway collapsibility in Japanese-Brazilian and white men. Chest. 2016;149(3):737–746. doi:10.1378/chest.15-0638
8. Lee RW, Vasudavan S, Hui DS, et al. Differences in craniofacial structures and obesity in Caucasian and Chinese patients with obstructive sleep apnea. Sleep. 2010;33(8):1075–1080. doi:10.1093/sleep/33.8.1075
9. Sutherland K, Lee RW, Cistulli PA. Obesity and craniofacial structure as risk factors for obstructive sleep apnoea, impact of ethnicity. Respirology. 2012;17(2):213–222. doi:10.1111/j.1440-1843.2011.02082.x
10. Watanabe T, Isono S, Tanaka A, Tanzawa H, Nishino T. Contribution of body habitus and craniofacial characteristics to segmental closing pressures of the passive pharynx in patients with sleep-disordered breathing. Am J Respir Crit Care Med. 2002;165(2):260–265. doi:10.1164/ajrccm.165.2.2009032
11. Tsuiki S, Isono S, Ishikawa T, Yamashiro Y, Tatsumi K, Nishino T. Anatomical balance of the upper airway and obstructive sleep apnea. Anesthesiology. 2008;108(6):1009–1015. doi:10.1097/ALN.0b013e318173f103
12. Ruehland WR, O’Donoghue FJ, Pierce RJ, et al. The 2007 AASM recommendations for EEG electrode placement in polysomnography: impact on sleep and cortical arousal scoring. Sleep. 2011;34(1):73–81. doi:10.1093/sleep/34.1.73
13. Aarab G, Lobbezoo F, Heymans MW, Hamburger HL, Naeije M. Long-term follow-up of a randomized controlled trial of oral appliance therapy in obstructive sleep apnea. Respiration. 2011;82(2):162–168. doi:10.1159/000324580
14. Byun JI, Kim D, Ahn SJ, et al. Efficacy of oral appliance therapy as a first-line treatment for moderate or severe obstructive sleep apnea: a Korean prospective multicenter observational study. J Clin Neurol. 2020;16(2):215–221. doi:10.3988/jcn.2020.16.2.215
15. Lee CH, Mo JH, Choi IJ, et al. The mandibular advancement device and patient selection in the treatment of obstructive sleep apnea. Arch Otolaryngol Head Neck Surg. 2009;135(5):439–444. doi:10.1001/archoto.2009.31
16. Lam B, Sam K, Lam JC, Lai AY, Lam CL, Ip MS. The efficacy of oral appliances in the treatment of severe obstructive sleep apnea. Sleep Breath. 2011;15(2):195–201. doi:10.1007/s11325-011-0496-y
17. Shen HL, Wen YW, Chen NH, Liao YF. Craniofacial morphologic predictors of oral appliance outcomes in patients with obstructive sleep apnea. J Am Dent Assoc. 2012;143(11):1209–1217. doi:10.14219/jada.archive.2012.0066
18. Zhou J, Liu YH. A randomised titrated crossover study comparing two oral appliances in the treatment for mild to moderate obstructive sleep apnoea/hypopnoea syndrome. J Oral Rehabil. 2012;39(12):914–922. doi:10.1111/joor.12006
19. Dort LC, Hadjuk E, Remmers JE. Mandibular advancement and obstructive sleep apnoea, a method for determining effective mandibular protrusion. Eur Respir J. 2006;27(5):1003–1009. doi:10.1183/09031936.06.00077804
20. Tsai WH, Vazquez JC, Oshima T, et al. Remotely controlled mandibular positioner predicts efficacy of oral appliances in sleep apnea. Am J Respir Crit Care Med. 2004;170(4):366–370. doi:10.1164/rccm.200310-1446OC
21. Kastoer C, Dieltjens M, Op de Beeck S, Braem MJ, Van de Heyning PH, Vanderveken OM. Remotely controlled mandibular positioning during drug-induced sleep endoscopy toward mandibular advancement device therapy: feasibility and protocol. J Clin Sleep Med. 2018;14(8):1409–1413. doi:10.5664/jcsm.7284
22. Marklund M, Verbraecken J, Randerath W. Non-CPAP therapies in obstructive sleep apnoea: mandibular advancement device therapy. Eur Respir J. 2012;39(5):1241–1247. doi:10.1183/09031936.00144711
23. Edwards BA, Andara C, Landry S. Upper-airway collapsibility and loop gain predict the response to oral appliance therapy in patients with obstructive sleep apnea. Am J Respir Crit Care Med. 2016;194(11):1413–1422. doi:10.1164/rccm.201601-0099OC
24. Ng AT, Darendeliler MA, Petocz P, Cistulli PA. Cephalometry and prediction of oral appliance treatment outcome. Sleep Breath. 2012;16(1):47–58. doi:10.1007/s11325-011-0484-2
25. Mostafiz W, Dalci O, Sutherland K, et al. Influence of oral and craniofacial dimensions on mandibular advancement splint treatment outcome in patients with obstructive sleep apnea. Chest. 2011;139(6):1331–1339. doi:10.1378/chest.10-2224
26. Chan AS, Lee RW, Cistulli PA. Dental appliance treatment for obstructive sleep apnea. Chest. 2007;132(2):693–699. doi:10.1378/chest.06-2038
27. Li WY, Gakwaya S, Saey D, Sériès F. Assessment of tongue mechanical properties using different contraction tasks. J Appl Physiol. 2017;123(1):116–125.
28. Eikermann M, Jordan AS, Chamberlin NL, et al. The influence of aging on pharyngeal collapsibility during sleep. Chest. 2007;131(6):1702–1709. doi:10.1378/chest.06-2653
29. Kassebaum NJ, Smith AGC, Bernabé E, et al.; GBD 2015 Oral Health Collaborators. Global, regional, and national prevalence, incidence, and disability-adjusted life years for oral conditions for 195 countries, 1990–2015: a systematic analysis for the global burden of diseases, injuries, and risk factors. J Dent Res. 2017;96(4):380–387. doi:10.1177/0022034517693566
30. O’Driscoll DM, Landry SA, Pham J, et al. The physiological phenotype of obstructive sleep apnea differs between Caucasian and Chinese patients. Sleep. 2019;42(11):zsz186. doi:10.1093/sleep/zsz186
31. Tsuiki S, Ito E, Isono S, et al. Oropharyngeal crowding and obesity as predictors of oral appliance treatment response to moderate obstructive sleep apnea. Chest. 2013;144(2):558–563. doi:10.1378/chest.12-2609
32. GBD 2017 Disease and Injury Incidence and Prevalence Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2018;392(10159):1789–1858. doi:10.1016/S0140-6736(18)32279-7
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.



