Back to Journals » Therapeutics and Clinical Risk Management » Volume 18
Diagnostic Value of Serum Levels of IL-22, IL-23, and IL-17 for Idiopathic Pulmonary Fibrosis Associated with Lung Cancer
Authors Zhang Q, Tong L, Wang B, Wang T, Ma H
Received 17 November 2021
Accepted for publication 28 March 2022
Published 19 April 2022 Volume 2022:18 Pages 429—437
DOI https://doi.org/10.2147/TCRM.S349185
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Deyun Wang
Qian Zhang, Lihong Tong, Bing Wang, Ting Wang, Hongxia Ma
Pneumology Department, The Traditional Chinese Medicine Hospital Affiliated to Xinjiang Medical University, Urumqi City, People’s Republic of China
Correspondence: Hongxia Ma, Pneumology Department, The Traditional Chinese Medicine Hospital Affiliated to Xinjiang Medical University, Urumqi City, The Xinjiang Uygur Autonomous Region, 830000, People’s Republic of China, Email [email protected]
Background: Correlations between idiopathic pulmonary fibrosis (IPF) and lung cancer have been discussed in many previous studies. In the present work, we evaluated the potential diagnostic value of serum levels of Th17 cell-related cytokines for the detection of lung cancer-associated IPF.
Methods: Fifty-six patients who had been diagnosed with lung cancer-associated IPF were enrolled, and 59 patients with lung cancer but without IPF were also enrolled, and 60 healthy volunteers were served as the control group. The expression of IL-22, IL-23, and IL-17 was evaluated by enzyme-linked immunosorbent assay (ELISA) kits.
Results: We observed that IL-22, IL-23 as well as IL-17 were significantly increased in the serum of lung cancer patients associated IPF; moreover, the results of ROC curve showed that the expression of IL-22, IL-23 and IL-17 can distinguish the lung cancer patients with lung cancer-associated IPF group; finally, the expression of IL-22, IL-23 and IL-17 was positively correlated with the degree of differentiation and metathesis of the tumor.
Conclusion: In conclusion, we first reported that IL-22, IL-23 and IL-17 were elevated in the serum of patients with lung cancer-associated IPF, and our data may provide novel evidence for the prevention and treatment of lung cancer-associated IPF.
Keywords: idiopathic pulmonary fibrosis, diagnosis, IL-22, IL-23, IL-17, lung cancer
Graphical Abstract:
Introduction
Idiopathic pulmonary fibrosis (IPF) is known as an inflammation-associated lung disease. It has been characterized by the long-term inflammatory condition and infiltration of immune cells, as well as the aberrant collagen deposition, which leads to the remodeling of the lung.1–3 Although the pathogenesis of IPF remains unclear, the results of previous studies suggested that IPF may initiate from a lesion that locates at the alveolus, and the long-term inflammatory condition could further lead to damages on the structure within the alveolar, including aberrant apoptosis of type I epithelial cells, over-proliferation of the type II cells, as well as the thickening of small airways and the artery walls.4–6 It is believed that damage to the alveolar parenchymal tissue could consequentially induce the fibrotic condition in the alveolar walls and remodeling of the lung tissue.
Based on the results of previous studies, idiopathic pulmonary fibrosis was considered as an important risk factor for the progression of lung cancer.7–9 It has been estimated that the incidence rate of lung cancer associated IPF patients ranges from about 10% to 40%. Unfortunately, lung cancer has also become a major contributor to IPF related-death among the above patients10–12 and the prognosis for patients with IPF associated lung cancer remains poor.
CD4+ T helper cells (CD4+ T cells) were known as regulators of many immune responses. CD4+ T cells were first divided into two subgroups, Th1 and Th2, based on the cytokines they expressed. In recent years, the investigation of Th17, a new subgroup of CD4+ T cells, has become a hot field. Th17 cells were known to produce interleukin-17, as well as other cytokines, for example, IL-22 and IL-21.13,14 IL-17 is known as a cytokine that participates in many immune diseases, for example, autoimmune diseases. The secretion of IL-17 is affected by several factors, for example, interleukin-23 (IL-23). IL-23 is mainly produced by dendritic cells as well as macrophages.15 Previous studies have suggested that IL-23 and IL-17 may play important roles in different types of cancers, including lung cancer. On the other hand, the aberrant expressions of the above cytokines were also found in patients with idiopathic pulmonary fibrosis; however, whether Th-17 related cytokines may serve as a biomarker for the early diagnosis of patients with combined idiopathic pulmonary fibrosis and lung cancer remains unclear.
This study was designed to test the hypothesis that one or more early serum biomarkers for idiopathic pulmonary fibrosis combined with lung cancer in the general Chinese population, recruited patients who signed informed consent while staying in hospital and was carried out according to the Declaration of Helsinki guidelines, and authorized by the ethics committee. Our data may provide novel evidence for the prevention and treatment of lung cancer patients with idiopathic pulmonary fibrosis.
Materials and Methods
Patients and Clinical Information
In this study, we enrolled 59 patients who had been diagnosed with lung cancer with idiopathic pulmonary fibrosis. Fifty-six patients with lung cancer but without idiopathic pulmonary fibrosis were also enrolled, and 60 healthy volunteers were served as the control group. The clinical information of the patients is shown in Table 1. The inclusion and exclusion criteria were as follows:
 |
Table 1 Clinical Information of the Patients |
Inclusion Criteria
1) Outpatients or inpatients aged 30 ~ 80 years, who meet the diagnostic criteria of lung cancer complicated with pulmonary interstitial fibrosis and sign informed consent.
2) No serious α 1-antitrypsin deficient patients;
3) Patients who did not take hormones and other immunosuppressants within one month and did not receive desensitization treatment.
Exclusion Criteria
1) Patients with severe respiratory failure (PaO2 < 40 mmhg, with or without PaO2 > 90 mmHg), pulmonary encephalopathy or artificial mechanical ventilation (noninvasive or invasive) who cannot cooperate with the information collection;
2) Patients with other serious chronic respiratory diseases, such as lung cancer, active tuberculosis and pulmonary fibrosis;
3) Patients with malignant tumors; acute or persistent chronic infectious diseases of any part; autoimmune diseases; hypertension, diabetes, heart failure and other chronic diseases that may affect the results;
4) Psychiatric patients and patients who cannot provide accurate medical history.
For each patient, the serum was collected and stored in −80°C until needed. This study has been approved by the ethics committee of The Traditional Chinese Medicine Hospital Affiliated to Xinjiang Medical University (No. 2021XE0184-1).
Enzyme-Linked Immunosorbent Assay
The serum levels of IL-22, IL-23, and IL-17 were measured using enzyme-linked immunosorbent assay (ELISA) kit (all purchased from Biocalvin, Suzhou, China) according to the manufacturer’s protocol.
Statistical Analysis
Statistical analysis was performed using SPSS 19.0. The qualitative data were compared with a χ2 test, and if the quantitative data fit the normal distribution, Student’s t-test was performed; if the quantitative data that did not fit the normal distribution, the Mann–Whitney test or Kruskal–Wallis test was performed. The Spearman and Pearson analysis were performed to investigate the correlation between the two variables. p < 0.05 has been considered to be statistically significant.
Results
Clinical Information of the Patients
Fifty-nine non-small cell lung cancer patients and 56 patients with combined lung cancer and idiopathic pulmonary fibrosis, as well as 60 healthy controls were included in the study. The clinical information of the patients is shown in Table 1.
Comparison of the Serum Levels of IL-17, IL-22, and IL-23 Among Different Groups
Next, we compared the serum levels of healthy controls, lung cancer patients, and lung cancer patients with idiopathic pulmonary fibrosis using ELISA methods. As shown in Figure 1, the serum level of IL-17 was significantly increased in the lung cancer group in comparison with the control and lung cancer group (p < 0.01), and on the other hand, the serum levels of IL-17 were markedly increased in lung cancer combined with idiopathic pulmonary fibrosis group compared with the lung cancer group (p < 0.01). Furthermore, both the serum levels of IL-22 (Figure 2, p < 0.01) and IL-23 (Figure 3, p < 0.01) were elevated in lung cancer patients compared with the controls, and the serum levels of both IL-22 (Figure 2, p < 0.01) and IL-23 (Figure 3, p < 0.01) were significantly up-regulated in lung cancer combined with idiopathic pulmonary fibrosis group compared with the lung cancer group.
 |
Figure 1 Comparison of the levels of IL-17 between control, lung cancer patients and lung cancer-associated IPF. ** P<0.01, *** P<0.001. |
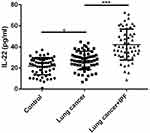 |
Figure 2 Comparison of the levels of IL-22 between control, lung cancer patients and lung cancer-associated IPF. *P<0.05, *** P<0.001. |
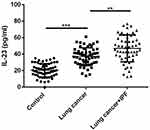 |
Figure 3 Comparison of the levels of IL-23 between control, lung cancer patients and lung cancer associated IPF. ** P<0.01, *** P<0.001. |
Serum Levels of IL-17, IL-22, and IL-23 as Diagnos tic Markers in Patients with Combined Lung Cancer and Idiopathic Pulmonary Fibrosis
Finally, the receiver-operating characteristic curve (ROC) has been drawn to show the sensitivity and specificity of IL-17, IL-22, and IL-23 to distinguish patients with combined lung cancer and idiopathic pulmonary fibrosis from lung cancer patients. As shown in Figures 4–6, the area under the curve (AUC) of IL-17 was 0.8229 (95% confidence interval (CI), 0.7502 to 0.8955), the IL-22 was 0.8081 (95% CI, 0.7275 to 0.8887), and the IL-23 was 0.7001 (95% CI, 0.6016 to 0.7986).
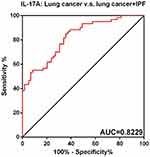 |
Figure 4 ROC of IL-17. |
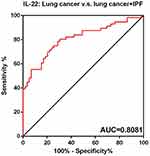 |
Figure 5 ROC of IL-22. |
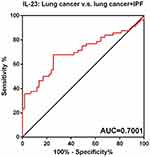 |
Figure 6 ROC of IL-23. |
Discussion
Idiopathic pulmonary fibrosis (IPF) represents a group of heterogeneous disorders. The burden of IPF is increasing worldwide and is associated with acute and chronic complications, leading to significant morbidity and mortality. Similar to IPF, the prevalence of lung cancer has also been increasing worldwide. However, the relationship between IPF and cancer has long been debated. Epidemiologic studies have demonstrated an increased risk of lung cancer in patients with IPF.9,16,17 The relevance of this finding is complicated by the fact that both IPF and cancer have similar risk factors (for example, age, family history, and smoking history), which might confound the results of these studies.18,19 Furthermore, some somatic alterations were shared by IPF-associated lung cancer and fibrosing lung tissue, for example, KRAS or TP53 mutations, were found to be higher in lung tissue samples among patients with IPF associated with lung cancer than those with IPF but without lung cancer.18–20 However, the relationship between IPF and lung cancer remains to be further investigated.
New-onset lung cancer in IPF is likely a paraneoplastic phenomenon, mediated by tumor secreted product(s).7,8,21 Since IPF has a high prevalence in lung cancer patients and develops at a time when the cancer may not even be visualized on imaging, patients with new-onset IPF offer a group of high-risk patients who could be screened for lung cancer. Further studies to discover the mediators of lung-associated IPF will not only improve our understanding of the pathophysiology of this phenomenon but could also offer potential biomarkers to screen patients for lung cancer.10,11,17
There are studies about whether IL-17 could serve as biomarkers for cancer; however, the results have been controversial. In the field of lung cancer studies, it has been reported that the expression level of IL-17 is elevated in the serum of patients, and the level of IL-17 in the serum of the patients may function as a prognostic marker,15,22–24 while on the other hand, some other studies had shown that the levels of IL-17 did not show differences between the patients and healthy controls. In the present study, we observed that the serum level of IL-17 was significantly upregulated in the serum of the patients in comparison with the healthy control group. Interestingly, we first observed that the serum level of IL-17 was significantly upregulated in patients with lung cancer compared with lung cancer patients with idiopathic pulmonary fibrosis, and the curve (AUC) of IL-17 was 0.8066 (95% confidence interval (CI), 0.7220 to 0.8912), suggesting that lung cancer patients with high served level of IL-17 may have the risk of idiopathic pulmonary fibrosis.
In addition, the roles of Th17 cells-related cytokines IL-22 and IL-23 in tumors have also been discussed. In the case of lung cancer, Bi et al proved that Interleukin-22 promotes lung cancer cell proliferation and migration via the IL-22R1/STAT3 and IL-22R1/AKT signaling pathways.25 Tufman et al proved that interleukin-22 is elevated in lavage from patients with lung cancer and other pulmonary diseases.26 Baird et al proved that IL-23 is pro-proliferative, epigenetically regulated, and modulated by chemotherapy in non-small cell lung cancer.27 Li et al observed that interleukin 23 regulates the proliferation of lung cancer cells in a concentration-dependent way in association with the interleukin-23 receptor.28 In the present study, we observed that both IL-22 and IL-23 were upregulated in the serum of patients with lung cancer compared with healthy controls, which were consistent with previous findings. Moreover, we first reported that the expression of both IL-22 and IL-23 were markedly increased in the serum of patients with combined lung cancer and idiopathic pulmonary fibrosis, compared with the lung cancer group, the AUC of IL-17 was 0.8066 (95% confidence interval (CI), 0.7220 to 0.8912), the IL-22 was 0.7818 (95% CI, 0.6952 to 0.8684), and the IL-23 was 0.7324 (95% CI, 0.6419 to 0.8230), suggesting that IL-22 and IL-23 are effective biomarkers to distinguish patients with combined lung cancer and idiopathic pulmonary fibrosis with the lung cancer patients.
Our studies have limitations. First, the results should be verified with larger sample size in future studies. In this study, dozens of samples represent part of the experimental results and lots of assumptions is still remained to be confirmed. We would like to calculate the sample-size under a single sample clinical diagnostic test as α=0.05, type of two-sided test, 1-β=0.8, and produce a result with the minimum required number of sample data is 62 cases.29 However, the study of biomarkers follows the rules of multiplex analysis, multi-biomarker and multi-validation, as well as a rule of thumb, we would like to take at least 134 cases for the future experimental verification process.
Second, this study was based on Chinese Han population, so whether IL-17, IL-22, and IL-23 have diagnostic value for patients with combined lung cancer and idiopathic pulmonary fibrosis in other races still needs to be validated.
In conclusion, we proved for the first time that IL-17, IL-22, and IL-23 were significantly upregulated in patients with combined lung cancer and idiopathic pulmonary fibrosis, and our results provided novel evidence that IL-17, IL-22 and IL-23 were sensitive biomarkers for the early diagnosis of patients with combined lung cancer and idiopathic pulmonary fibrosis.
Ethics Statement
This study has been approved by the ethics committee of The Traditional Chinese Medicine Hospital Affiliated to Xinjiang Medical University (No. 2021XE0184-1).
Funding
This work was supported by the TCM Syndrome Differentiation and Genomics Study of Pulmonary Interstitial Fibrosis in Lung Cancer Based on Targeted Deep Sequencing (2018Q049).
Disclosure
The authors report no conflicts of interest in this work.
References
1. Le Garrec M, Rousseau C, Thepault F, Kerjouan M, Sale A, Jouneau S. Prevalence, predicting factors and impact of internet use by patients with idiopathic pulmonary fibrosis: a cross-sectional observational study. Respir Med Res. 2021;79:100818. doi:10.1016/j.resmer.2021.100818
2. Behr J, Prasse A, Kreuter M, et al. Pirfenidone in patients with progressive fibrotic interstitial lung diseases other than idiopathic pulmonary fibrosis (RELIEF): a double-blind, randomised, placebo-controlled, phase 2b trial. Lancet Respir Med. 2021;9(5):476–486.
3. Biondini D, Cocconcelli E, Bernardinello N, et al. Prognostic role of MUC5B rs35705950 genotype in patients with idiopathic pulmonary fibrosis (IPF) on antifibrotic treatment. Respir Res. 2021;22(1):98. doi:10.1186/s12931-021-01694-z
4. Ghang B, Nam SH, Lee J, et al. Correction to: risk of progression of idiopathic pulmonary fibrosis to connective tissue disease: a long-term observational study in 527 patients. Clin Rheumatol. 2021;40(6):2525.
5. Rahaghi F, Belperio JA, Fitzgerald J, et al. Delphi consensus recommendations on management of dosing, adverse events, and comorbidities in the treatment of idiopathic pulmonary fibrosis with nintedanib, clinical medicine insights. Circ Respir Pulm Med. 2021;15:11795484211006050.
6. Fedi A, Keddache S, Quetant S, et al. Concurrence of 1- and 3-min sit-to-stand tests with the 6-min walk test in idiopathic pulmonary fibrosis. Respiration. 2021;100:1–9.
7. Kai Y, Matsuda M, Fukuoka A, et al. Remarkable response of non-small cell lung cancer to nintedanib treatment in a patient with idiopathic pulmonary fibrosis. Thorac Cancer. 2021;12(9):1457–1460. doi:10.1111/1759-7714.13935
8. Hata A, Nakajima T, Matsusaka K, et al. Genetic alterations in squamous cell lung cancer associated with idiopathic pulmonary fibrosis. J Clin Investig. 2021;148(12):3008–3018. doi:10.1002/ijc.33499
9. Song MJ, Kim SY, Park MS, Kang MJ, Lee SH, Park SC. A nationwide population-based study of incidence and mortality of lung cancer in idiopathic pulmonary fibrosis. Sci Rep. 2021;11(1):2596. doi:10.1038/s41598-021-82182-8
10. Kanaji N, Shimizu J, Sakai K, et al. Clinical features of patients with small cell lung cancer and idiopathic pulmonary fibrosis treated with chemotherapy or chemoradiotherapy. Ther Adv Respir Dis. 2020;14:1753466620963866. doi:10.1177/1753466620963866
11. Leng D, Yi J, Xiang M, Zhao H, Zhang Y. Identification of common signatures in idiopathic pulmonary fibrosis and lung cancer using gene expression modeling. BMC Cancer. 2020;20(1):986. doi:10.1186/s12885-020-07494-w
12. Yamamoto Y, Yano Y, Kuge T, et al. Safety and effectiveness of pirfenidone combined with carboplatin-based chemotherapy in patients with idiopathic pulmonary fibrosis and non-small cell lung cancer: a retrospective cohort study. Thorac Cancer. 2020;11(11):3317–3325. doi:10.1111/1759-7714.13675
13. Behram M, Oglak SC, Baskiran Y, et al. Maternal serum IL-22 concentrations are significantly upregulated in patients with preterm premature rupture of membranes. Ginekol Pol. 2021. doi:10.5603/GP.a2021.0036
14. Nehmar R, Fauconnier L, Alves-Filho J, et al. Aryl hydrocarbon receptor(Ahr)-dependent Il-22 expression by type 3 innate lymphoid cells control of acute joint inflammation. J Cell Mol Med. 2021;25(10):4721–4731. doi:10.1111/jcmm.16433
15. Walker EM, Slisarenko N, Gerrets GL, et al. Dysregulation of IL-17/IL-22 Effector functions in blood and gut mucosal gamma delta T cells correlates with increase in circulating leaky gut and inflammatory markers during cART-treated chronic SIV infection in macaques. Front Immunol. 2021;12:647398. doi:10.3389/fimmu.2021.647398
16. Duchemann B, Pluvy J, Crestani B, Zalcman G, Nunes H. Immune checkpoint blockade for patients with lung cancer and idiopathic pulmonary fibrosis. Eur J Cancer. 2021;145:179–182. doi:10.1016/j.ejca.2020.12.016
17. Lee HY, Lee J, Lee CH, Han K, Choi SM. Risk of cancer incidence in patients with idiopathic pulmonary fibrosis: a nationwide cohort study. Respirology. 2021;26(2):180–187. doi:10.1111/resp.13911
18. Otsubo K, Iwama E, Ijichi K, et al. Paired genetic analysis by next-generation sequencing of lung cancer and associated idiopathic pulmonary fibrosis. Cancer Sci. 2020;111(7):2482–2487. doi:10.1111/cas.14488
19. Shiratori T, Tanaka H, Tabe C, et al. Effect of nintedanib on non-small cell lung cancer in a patient with idiopathic pulmonary fibrosis: a case report and literature review. Thorac Cancer. 2020;11(6):1720–1723. doi:10.1111/1759-7714.13437
20. Rubio K, Castillo-Negrete R, Barreto G. Non-coding RNAs and nuclear architecture during epithelial-mesenchymal transition in lung cancer and idiopathic pulmonary fibrosis. Cell Signal. 2020;70:109593. doi:10.1016/j.cellsig.2020.109593
21. Iida Y, Gon Y, Nakanishi Y, et al. Genomic analysis between idiopathic pulmonary fibrosis and associated lung cancer using laser-assisted microdissection: a case report. Thorac Cancer. 2021;12(9):1449–1452. doi:10.1111/1759-7714.13924
22. Lin X, Tawch S, Wong HT, et al. Nrf2 through aryl hydrocarbon receptor regulates IL-22 response in CD4(+) T cells. J Immunol. 2021;206(7):1540–1548. doi:10.4049/jimmunol.1900656
23. Dubenko OE, Chyniak OS, Potapov OO. Levels of proinflammatory cytokines Il-17 and Il-23 in patients with Alzheimer’s disease, mild cognitive impairment and vascular dementia. Wiad Lek. 2021;74(1):68–71. doi:10.36740/WLek202101113
24. Ogongo P, Tezera LB, Ardain A, et al. Tissue resident-like CD4+ T cells secreting IL-17 control Mycobacterium tuberculosis in the human lung. J Clin Invest. 2021;131(10). doi:10.1172/JCI142014
25. Bi Y, Cao J, Jin S, et al. Interleukin-22 promotes lung cancer cell proliferation and migration via the IL-22R1/STAT3 and IL-22R1/AKT signaling pathways. Mol Cell Biochem. 2016;415(1–2):1–11. doi:10.1007/s11010-016-2663-8
26. Tufman A, Huber RM, Volk S, et al. Interleukin-22 is elevated in lavage from patients with lung cancer and other pulmonary diseases. BMC Cancer. 2016;16(1):409. doi:10.1186/s12885-016-2471-2
27. Baird AM, Leonard J, Naicker KM, Kilmartin L, O’Byrne KJ, Gray SG. IL-23 is pro-proliferative, epigenetically regulated and modulated by chemotherapy in non-small cell lung cancer. Lung Cancer. 2013;79(1):83–90. doi:10.1016/j.lungcan.2012.10.003
28. Li J, Zhang L, Zhang J, et al. Interleukin 23 regulates proliferation of lung cancer cells in a concentration-dependent way in association with the interleukin-23 receptor. Carcinogenesis. 2013;34(3):658–666. doi:10.1093/carcin/bgs384
29. Bujang MA. A step-by-step process on sample size determination for medical research. Malays J Med Sci. 2021;28(2):15–27.
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
