Back to Journals » Cancer Management and Research » Volume 12
Diagnostic and Predictive Values of 18F-FDG PET/CT Metabolic Parameters in EGFR-Mutated Advanced Lung Adenocarcinoma
Authors Hong IK , Lee JM, Hwang IK, Paik SS , Kim C, Lee SH
Received 20 April 2020
Accepted for publication 16 July 2020
Published 28 July 2020 Volume 2020:12 Pages 6453—6465
DOI https://doi.org/10.2147/CMAR.S259055
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Yong Teng
Il Ki Hong,1 Jeong Mi Lee,2 In Kyoung Hwang,2 Seung Sook Paik,2 Chanwoo Kim,3 Seung Hyeun Lee4
1Department of Nuclear Medicine, Kyung Hee University Medical Center, Kyung Hee University School of Medicine, Seoul, South Korea; 2Department of Internal Medicine, Graduate School, Kyung Hee University, Seoul, South Korea; 3Department of Nuclear Medicine, Kyung Hee University Hospital at Gangdong, Kyung Hee University School of Medicine, Seoul, South Korea; 4Division of Pulmonary, Allergy, and Critical Care Medicine, Department of Internal Medicine, Kyung Hee University Medical Center, Kyung Hee University School of Medicine, Seoul, South Korea
Correspondence: Seung Hyeun Lee Email [email protected]
Purpose: The clinical implications of the metabolic parameters of 18F-fluorodeoxyglucose positron emission tomography-computed tomography (18F-FDG PET/CT) in epidermal growth factor receptor (EGFR)-mutated lung cancer are not fully understood. The aim of this study was to evaluate the diagnostic and prognostic utility of the parameters in EGFR-mutated lung cancer patients.
Patients and Methods: We retrospectively enrolled 134 patients with advanced lung adenocarcinoma (72 EGFR-negative and 62 EGFR-positive). We evaluated the correlation between EGFR mutational status and the maximum standardized uptake value (SUVmax), as well as the associations between treatment outcomes in EGFR-mutated patients and various metabolic parameters of primary tumors. For the best predictive parameters, we calculated the metabolic tumor volume (MTV) and total lesion glycolysis (TLG) using two SUV cutoffs: 1.5 (MTV1.5, TLG1.5) and 2.5 (MTV2.5, TLG2.5).
Results: Mean SUVmax was lower for EGFR-mutated tumors compared with EGFR wild-type (6.11 vs 10.41, p < 0.001) tumors. Low SUVmax was significantly associated with positive EGFR mutation (odds ratio = 1.74). Multivariate analysis for survival demonstrated that high MTV1.5, TLG1.5, MTV2.5, and TLG2.5 were independently associated with shorter progression-free survival (PFS) and overall survival (OS), and the highest hazard ratios were found in TLG1.5 (3.26 for PFS and 4.62 for OS).
Conclusion: SUVmax may be predictive for EGFR mutational status, and MTV and TLG of primary tumors may be promising prognostic parameters; 18F-FDG PET/CT has potential utility for the risk stratification of EGFR-mutated patients treated with targeted therapy.
Keywords: lung cancer, 18F-fluorodeoxyglucose positron emission tomography-computed tomography, epidermal growth factor receptor mutation, survival, metabolic parameters
Introduction
Lung cancer is the most common cause of cancer mortality globally. In 2013, the estimated global lung cancer incidence was 1.8 million, and the estimated number of fatal cases was 1.6 million.1 According to the Korean National Lung Cancer registry data, the crude incidence rate of lung cancer was 43.9 per 100,000, and the age-standardized mortality rate was 19.8 per 100,000 in 2012.2,3 Adenocarcinoma is the most frequent histologic subtype, and it accounts for 50% of all lung cancers.4 Although new treatment modalities, including molecular-targeted therapy and immune checkpoint inhibitors, have demonstrated remarkable survival benefits in patients with advanced lung adenocarcinoma,4,5 the prognosis is still poor, and more individualized therapeutic strategies should be established to improve clinical outcomes.
18F-fluorodeoxyglucose positron emission tomography-computed tomography (18F-FDG PET/CT) is a functional imaging modality that measures the glucose metabolism of tumors. Due to its advantages over conventional techniques such as CT scan, it is widely used in lung cancer management; its uses include staging at diagnosis, response evaluation after systemic treatment, re-staging after neoadjuvant treatment, and surveillance after curative resection or stereotactic radiosurgery.6,7 To extend its application in practice, numerous studies have evaluated the possible utility of the semi-quantitative metabolic parameters obtained from 18F-FDG PET/CT images. Standardized uptake value (SUV) is the most widely investigated across various human malignancies; some studies reported the association between maximal SUV (SUVmax) and poor survival of non-small-cell lung carcinoma (NSCLC) patients,8–10 while others showed no association.11–14 These conflicting data partially result from the limitation of SUVmax; it is vulnerable to statistical noise, as it is measured using a single voxel analysis.15 Thus, recent studies have focused on the volumetric parameters, including metabolic tumor volume (MTV) and total lesion glycolysis (TLG), which reflect metabolic tumor burden.
Most of the studies that have evaluated the prognostic values of the volumetric parameters were conducted on early-stage, operable NSCLC. According to a meta-analysis, high SUVmax, MTV, and TLG were significantly associated with a high risk of recurrence and death in patients who received surgical resection.10 However, studies on the clinical utility of those parameters in advanced diseases are rare, and only 3 studies have been reported to date.12,16,17 One study reported that high whole-body MTV and TLG, rather than SUVmax, were associated with poor survival in stage IV NSCLC, suggesting that global metabolic tumor burden is associated with prognosis.16 In contrast, the two other studies simultaneously evaluated MTV and TLG from whole-body and primary tumors and reported that the MTV of a primary tumor was independently associated with progression-free survival (PFS)17 and overall survival (OS).12,17 The latter suggests the prognostic utility of the volumetric parameters measured in primary tumors, not in all the target lesions, even in advanced NSCLC.
EGFR mutations are major driver mutations occurring in lung adenocarcinomas.18 Approximately 15% of Caucasian and 30%–40% of Asian patients with lung adenocarcinoma have tumors with EGFR mutations.19 Although these patients have shown prolonged PFS when treated with EGFR-tyrosine kinase inhibitors (TKIs), the response rate is no more than 70%–80%, and resistance inevitably occurs after approximately 11 months of treatment in most patients.20–22 Thus, it is important to determine predictive and prognostic factors before TKI treatment to improve clinical outcomes.
To address this issue, we conducted this study to determine whether metabolic parameters of 18F-FDG PET/CT can discriminate between EGFR-mutated lung adenocarcinomas and EGFR wild-types and predict treatment response and survival in patients treated with first-line EGFR-TKIs.
Patients and Methods
Study Subjects and Data Collection
We retrospectively recruited patients who received systemic treatment for histologically confirmed, locally advanced, or metastatic lung adenocarcinoma at the Kyung Hee University Hospital, a tertiary referral hospital in South Korea, between March 2016 and November 2019. Patients who did not have available follow-up data, initial 18F-FDG PET/CT images, a history of other cancers, other driving genetic alterations including ROS1 and ALK fusions, and previous history of chemotherapy or radiotherapy were excluded. EGFR-negative patients were all treated with pemetrexed/platinum doublet with or without pemetrexed continuation maintenance chemotherapy, while EGFR-mutant patients were treated with EGFR-TKIs, including gefitinib, erlotinib, or afatinib as a first-line treatment.
All patients underwent staging workup, including chest computed tomography (CT), brain magnetic resonance imaging, and 18F-FDG PET/CT. TNM staging was performed according to the 8th edition of the International Association for the Study of Lung Cancer TNM staging system.23 Tumor response was examined by CT after every two cycles of systemic treatment and evaluated according to the Response Evaluation Criteria in Solid Tumors (RECIST) 1.1.24 For EGFR-TKIs, a single cycle comprised 4 weeks of treatment. We reviewed electronic medical records to obtain demographic information, past medical or social history, and clinical data of all participants. This study protocol was approved by the Clinical Research Ethics Committee of the Kyung Hee University Hospital (KHUH2020-03-088), and written informed consent was obtained from all patients who were alive, but it was waived for patients who had died. All research was carried out in compliance with the Declaration of Helsinki.
EGFR Mutation Testing
All the EGFR tests were performed using the tumor tissues. Genomic DNA was extracted from formalin-fixed, paraffin-embedded, 5-µm-thick tissue sections using the High Pure Template Preparation Kit (Roche Applied Science, Mannheim, Germany). The extracted DNA was stored at −20°C until analysis. EGFR Pyro Kit (QIAGEN Korea Ltd., Seoul, Korea) and PyroMark Q24 System (QIAGEN Korea Ltd., Seoul, Korea) were used to detect EGFR mutations by real-time polymerization chain reaction (PCR). The primer sets covered mutations or deletions spanning exons 18 to 21 of the genes encoding the tyrosine kinase domain of EGFR. The results were interpreted according to the manufacturer’s instructions.
Acquisition of PET/CT Images and Calculation of Metabolic Parameters
All patients underwent PET/CT scanning with Gemini TF16 PET/CT scanners (Philips Medical Systems, Cleveland, OH) before the initiation of systemic treatment. After fasting for at least 6 hours, 5–6 MBq/kg of 18F-FDG was administered intravenously, and scanning was performed after 60 minutes. Low-dose unenhanced CT scans were first obtained for attenuation correction, and then PET scans (1 min per bed) were obtained. The values of the metabolic parameters were assessed by two experienced nuclear medicine physicians (I.K.H and C.K) using MIRADA XD3 software (MIRADA Medical, Oxford, UK). Physicians dragged the cursor from the center of the primary tumor to a point near the edge of the lesion, and the software automatically outlined a three-dimensional volume of interest (VOI) on the tumor. MTV and TLG were calculated using two SUV cutoff values of 1.5 and 2.5 for this semi-automated contouring system to exclude physiologic FDG uptake of the non-tumor tissue. MTV was defined as the total tumor volume inside the boundaries of the VOI. TLG was calculated as the product of MTV and the mean SUV of the lesion.
Statistical Analyses
Baseline characteristics between groups were compared using Fisher’s exact test. The optimal cutoff for SUVmax for the discrimination of EGFR mutation was identified using receiver operating characteristic (ROC) curve analysis. The optimal cutoff values of SUVmax, MTV, and TLG for survival analysis were defined using the point with the lowest p-value on the Log rank test for progression-free survival (PFS) for all possible values of each parameter. PFS and OS were defined as the periods from the first day of treatment to disease progression/death and death from any cause, respectively. Data on patients without tumor recurrence or death were censored at the last follow-up. Associations between clinical parameters and EGFR mutational status or survival were evaluated by univariate analysis using the Log rank test, and subsequently, the multivariate Cox’s proportional hazard regression was conducted with adjustment for parameters with p-values < 0.3 from the univariate analysis. Survival curves were generated using the Kaplan–Meier method. P-values < 0.05 were considered significant. All analyses were performed using SPSS version 20.0 (IBM Corporation, Armonk, NY, USA) and MedCalc (MedCalc Software Ltd, Ostend, Belgium).
Results
Patient Characteristics
During the study period, 372 patients were newly diagnosed with NSCLC. Of them, 160 underwent first-line treatment with platinum-based doublet for those without EGFR mutation or EGFR-TKIs for those with EGFR mutation. Eleven patients who had only external 18F-FDG PET/CT images, 5 without unavailable survival data, 3 with history of other cancers, 4 with genetic alterations other than EGFR mutation, and 3 who received other cancer treatments were excluded. Finally, a total of 134 (72 EGFR-negative and 62 EGFR-positive) patients were eligible for analysis.
The clinical characteristics of the study population are summarized in Table 1. All subjects were Korean, and their median age was 69 years (range, 39–81 years). Forty-five (34%) patients were female. Sixty-seven (50%) patients were aged ≥ 65 years. Seventy-six (57%) patients were current or former smokers. One hundred and eight (80%) patients had an Eastern Cooperative Oncology Group (ECOG) performance status of 0 or 1. Nine (7%) patients had stage IIIB, and 125 (93%) patients had stage IV. Twenty-eight (20%) patients had metastases at > 3 organs. Thirty-nine (29%) and 22 (16%) patients had brain and liver metastasis, respectively. Thirty-five (57%) patients had exon 19 deletions, 25 (47%) patients had L858R point mutations, and 2 patients had uncommon mutations (G719X and S768I, respectively) among EGFR-mutated patients. Compared with EGFR-negative patients, the proportions of females (52% vs 18%, p = 0.046) and those who had never smoked (61% vs 28%, p = 0.017) were higher in the EGFR-mutated group.
 |
Table 1 Characteristics of Patients with Advanced Lung Adenocarcinomas |
Association Between SUVmax and EGFR Mutational Status
The SUVmax was significantly lower in the EGFR-mutated group (mean, 6.11 ± 2.77) than in the EGFR-negative group (mean, 10.41 ± 5.61, p < 0.001) (Figure 1A). ROC curve analysis was performed to determine the optimal cutoff for discriminating EGFR mutational status. At the cutoff of 9.6, the AUC was 0.760 (95% CI: 0.673–0.833, p < 0.001), with a sensitivity of 55.6% and a specificity of 89.5% (Figure 1B). The proportion of tumors with low SUVmax (< 9.6) was significantly higher in the EGFR-mutated group than in the EGFR wild-type group (84% vs 42%, p = 0.010) (Table 1).
Analysis of Predictive Factors of EGFR Mutational Status
In the univariate analysis, the female gender, no history of smoking, and low SUVmax were significantly associated with positive EGFR mutation (all p < 0.05). In the multivariate analysis, the female gender (HR = 2.65, 95% CI: 1.13–5.19), no history of smoking (HR = 1.28, 95% CI: 1.05–4.12), and low SUVmax (HR = 3.14, 95% CI: 1.23–5.57) were independently associated with EGFR positivity (Table 2).
 |
Table 2 Analysis Results for the Variables Associated with EGFR Mutational Status |
Survival Analysis for the Patients Receiving EGFR-TKIs as a First-Line Treatment
The optimal cutoff values for the survival analysis, as determined by the Log rank test, were 3.54 for SUVmax, 14.08 for MTV1.5, 69.86 for TLG1.5, 4.28 for MTV2.5, and 12.64 for TLG2.5 (Figure S1). Survival analysis results on clinical and metabolic parameters are summarized in Table 3. The median PFS for all study subjects was 15.3 months (range, 2.2–30.4 months). The univariate analysis showed metastases in 3 or more organs, gefitinib/erlotinib use, and high SUVmax showed a trend of association with shorter PFS. In addition, smoking, high MTV, and TLG, regardless of SUV cutoffs, were significantly associated with shorter PFS (all p < 0.05). Multivariate analysis showed that smoking (HR = 2.08, 95% CI: 1.02–4.26), MTV1.5 (HR = 3.12, 95% CI: 1.35–6.67), TLG1.5 (HR = 3.26, 95% CI: 1.45–7.32), MTV2.5 (HR = 2.16, 95% CI: 1.05–4.44), and TLG2.5 (HR = 2.19, 95% CI: 1.10–4.68) were independently associated with shorter PFS. Kaplan–Meier survival curves showed that patients with high values of metabolic parameters were likely to have poor survival in terms of PFS (Figure 2).
 |
Table 3 Survival Analyses Results According to Clinicopathologic and Metabolic Parameters in EGFR-Mutated Patients |
 |
Figure 2 Kaplan–Meier survival curves for progression-free survival (PFS). (A) SUVmax, (B) MTV1.5, (C) TLG1.5, (D) MTV2.5, (E) TLG2.5. P-values were determined using the Log rank test. |
The median OS for all study subjects was 28.5 months (range, 2.2–56.0 months). Univariate analysis showed that male sex, poor performance status, and low SUVmax showed a trend of association with OS. Metastases in 3 or more organs and high MTV and TLG, regardless of SUV cutoffs, were significantly associated with shorter OS (all p < 0.05). Multivariate analysis showed that metastases in 3 or more organs (HR = 2.11, 95% CI: 1.04–3.61), MTV1.5 (HR = 4.18, 95% CI: 1.28–10.85), TLG1.5 (HR = 4.62, 95% CI: 1.26–12.11), MTV2.5 (HR = 2.04, 95% CI: 1.08–6.25), and TLG2.5 (HR = 1.87, 95% CI: 1.02–6.33) were independently associated with shorter OS. Kaplan–Meier survival curves showed that patients with high values of metabolic parameters were likely to have poor survival in terms of OS (Figure 3).
 |
Figure 3 Kaplan–Meier survival curves for overall survival (OS). (A) SUVmax, (B) MTV1.5, (C) TLG1.5, (D) MTV2.5, (E) TLG2.5. P-values were determined using the Log rank test. |
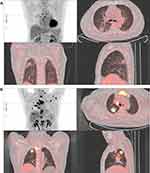
Representative cases of EGFR-mutant patients with different PFS and OS according to different MTV and TLG of the primary tumors are presented in Figure 4.
Discussion
The present data demonstrate that SUVmax is significantly lower in EGFR-mutated lung adenocarcinoma than in the EGFR wild-type, and low SUVmax (< 9.6) may be predictive of EGFR mutation. In addition, volumetric parameters such as high MTV and TLG measured in the primary tumor were associated with shorter PFS and OS in EGFR-mutant patients treated with first-line EGFR-TKIs, even after adjusting for the effects of clinical parameters. To the best of our knowledge, this is the first report identifying that 18F-FDG PET/CT parameters of primary tumors may have predictive and prognostic values in advanced EGFR-mutated lung adenocarcinoma.
Although mutational analysis using tumor tissue or body fluid is the standard method for determining EGFR mutational status, it is sometimes limited by issues such as invasiveness of the tissue acquisition procedures and inaccessibility or insufficiency of sample tissues. Thus, metabolic parameters obtained from the 18F-FDG PET/CT data have been evaluated for non-invasive predictions of EGFR mutation.25–28 SUVmax, representing the most active metabolic location of the tumor, is the most studied, and several studies have shown that low SUVmax is associated with EGFR mutation.26,28,29 Previous studies have demonstrated that glucose transporter 1 (GLUT1), which serves important functions in glucose transport, may be regulated by reactive oxygen species (ROS).30,31 Chen et al recently observed that ROS activity was lower in EGFR-mutant lung cancer cells than in wild-type cells, and identified the role of NADPH oxidase 4 (NOX4)/ROS/GLUT1 axis in glucose metabolism; downregulated NOX4 expression led to a decrease in ROS activity resulting in decreased GLUT1 expression. The decreased FDG uptake in EGFR-mutant cells can partially be explained by this mechanism.29 However, the data on the value of SUVmax in predicting EGFR mutation are still conflicting, as some studies have failed to prove associations.25,27,32,33 One of the reasons for the conflicting results among studies is the differences in the study design and patient characteristics across the studies. All the aforementioned studies enrolled EGFR-mutated patients of all clinical stages. Some studies enrolled all NSCLC histologic subtypes,25,28 while others did not.26,27,33 EGFR mutation is dominant in adenocarcinomas, and tumors harboring this mutation become heterogeneous as they progress due to the clonal evolution.34,35 Thus, their metabolic characteristics could vary with the clinical stages. In addition, the detection of EGFR mutation is more important in advanced than in early-stage lung cancer because the presence or absence of the mutation determines treatment strategy; whether TKIs should be used for frontline treatment or not. Thus, we ensured that our population was more homogenous, consisting of advanced EGFR-mutated adenocarcinomas, and we successfully demonstrated that SUVmax, even in advanced stages, can predict EGFR mutational status at the cutoff of 9.6. Our data are consistent with those from previous studies,28,36 and they suggest that PET/CT may be a helpful non-invasive modality for predicting EGFR mutation. Our results should be validated and the optimal cutoff should be determined in further large-scale investigations. In addition, the clinical significance of other metabolic parameters as predictors for EGFR mutations need to be evaluated, as a recent study has demonstrated that MTV could discriminate the EGFR-positive patients from those without the mutation.37
In contrast to SUV, which reflects the high metabolic lesion of the tumor, MTV and TLG incorporate metabolic burden and disease extent, and they may have better predictive values than SUV. Several studies have demonstrated that volumetric parameters have prognostic values in advanced NSCLC treated with chemotherapy.12,16,17 However, the clinical significance of those parameters in EGFR-mutated patients receiving TKIs may differ in wild-type patients treated with chemotherapy since the mechanism by which TKIs induce cancer cell death is different from that of cytotoxic agents. In addition, tumors with EGFR mutations are metabolically indolent, as shown in previous studies and our present study. To date, only 3 studies have investigated the predictive value of the various metabolic parameters in advanced EGFR-mutated NSCLC.11,38,39 We have summarized their results in Table 4. One study reported that high SUVmax (> 12) was independently associated with PFS.38 Other studies reported that high whole-body TLG was significantly associated with poor survival in patients treated with EGFR-TKIs.11,39 However, the latter two studies evaluated MTV and TLG from whole-body lesions.
 |
Table 4 Summary of Published Data and the Present Study on the Association Between 18F-FDG PET/CT Metabolic Parameters and Clinical Outcome of Advanced EGFR-Mutated Lung Adenocarcinoma |
Although it may be ideal to calculate whole-body volumetric information to ascertain the exact metabolic tumor burden, it is time-consuming, especially in patients with extensive metastasis; thus, it may be impractical despite its proven significance. We aimed at finding simpler, image-based markers that could be readily used in clinical practice, and hypothesized that volumetric parameters of the primary tumor have prognostic values. To determine the best parameters, we used two SUV cutoffs to calculate MTV and TLG. The present data showed that high MTV and TLG, regardless of the SUV cutoffs, were independently associated with poor PFS and OS. The HRs of MTV1.5 (3.12 for PFS and 4.18 for OS) and TLG1.5 (3.26 for PFS and 4.62 for OS) were higher than those of MTV2.5 (2.16 for PFS and 2.04 for OS) and TLG2.5 (2.19 for PFS and 1.89 for OS), suggesting that the predictive performance of volumetric parameters may be better at the cutoff SUV of 1.5 than 2.5. The exact reason for the better performance of the volumetric parameters with low SUV cutoff is not clear; however, we postulate that the low cutoff permits an MTV that is reflective of a more exact metabolic burden of primary tumors compared with a high cutoff because EGFR-mutant tumors are metabolically indolent.26,28,29 Our results are supported by a previous report demonstrating that MTV and TLG of the primary tumor, rather than SUVmax, were associated with clinical outcomes in NSCLC patients treated with chemotherapy.12
Although SUV has been extensively studied and was proven to be related with prognosis in NSCLC patients across all stages, it has several limitations as an optimal metabolic parameter; it depends on various patient-related (body size, blood glucose levels) and technical (reconstruction method, partial volume effect) factors,40 and does not account for tumor volume, which can be related to the T descriptor in the staging system that is associated with prognosis. Especially, SUVmax is the value of a single voxel in the region of interest that is subject to statistical variability due to noise in a reconstructed PET/CT image.15 The clinical usefulness of volumetric parameters and optimal SUV cutoffs in calculating metabolic tumor burden should be validated in large-scale studies.
This study has several limitations. First, it is a retrospective study and selection bias is inevitable. Second, it was performed in a single institution and the sample size was small. To compensate for the small sample size and to collect relevant data, we analyzed various metabolic parameters at the various SUV cutoffs simultaneously, and two nuclear medicine specialists assessed the PET/CT images. Third, patients with other genetic alterations such as ROS1 and ALK fusion were not assessed because of the rarity of the alterations among lung adenocarcinoma patients (1%-5%). Finally, we focused on the baseline metabolic parameters rather than follow-up data after treatment. As suggested in a study on NSCLC patients treated with concurrent chemoradiation, the change in metabolic parameters from the baseline to the subsequent study may also have valuable clinical implications.41
Conclusion
In conclusion, SUVmax may be useful in predicting EGFR mutational status, while MTV and TLG, measured in the primary tumor, may be useful for predicting treatment response and survival in lung adenocarcinomas treated with TKIs. Our results highlight a practical, image-based method for the discrimination of EGFR-mutated tumors from wild-types and risk stratification before TKI use. If large-scale prospective studies confirm our results, PET/CT metabolic parameters, either alone or combined with other clinical parameters, can be used to select patients with TKI resistance and modify treatment strategy or prompt more rigorous follow-up. Our data also suggest the possible application of the parameters in other clinical settings, such as immunotherapy, which may facilitate the clinical utilization of PET/CT as a non-invasive predictive and prognostic modality in lung cancer management.
Acknowledgments
This research was supported by a grant from the Basic Research Program through the National Research Foundation funded by the Ministry of Science and ICT (2019R1F1A1041812) of the Republic of Korea.
Disclosure
The authors declare that they have no competing interests.
References
1. Fitzmaurice C, Dicker D, et al. The global burden of cancer 2013. JAMA Oncol. 2015;1(4):505–527. doi:10.1001/jamaoncol.2015.0735
2. Park JY, Jang SH. Epidemiology of lung cancer in Korea: recent trends. Tuberc Respir Dis (Seoul). 2016;79(2):58–69. doi:10.4046/trd.2016.79.2.58
3. Park CK, Kim SJ. Trends and updated statistics of lung cancer in Korea. Tuberc Respir Dis (Seoul). 2019;82(2):175–177. doi:10.4046/trd.2019.0015
4. Kim HC, Jung CY, Cho DG, et al. Clinical characteristics and prognostic factors of lung cancer in Korea: a pilot study of data from the korean nationwide lung cancer registry. Tuberc Respir Dis (Seoul). 2019;82(2):118–125. doi:10.4046/trd.2017.0128
5. Kim HC, Choi CM. Current status of immunotherapy for lung cancer and future perspectives. Tuberc Respir Dis (Seoul). 2020;83(1):14–19. doi:10.4046/trd.2019.0039
6. Hicks RJ, Kalff V, MacManus MP, et al. The utility of (18)F-FDG PET for suspected recurrent non-small cell lung cancer after potentially curative therapy: impact on management and prognostic stratification. J Nucl Med. 2001;42(11):1605–1613.
7. Sheikhbahaei S, Mena E, Yanamadala A, et al. The value of FDG PET/CT in treatment response assessment, follow-up, and surveillance of lung cancer. AJR Am J Roentgenol. 2017;208(2):420–433. doi:10.2214/AJR.16.16532
8. Sasaki R, Komaki R, Macapinlac H, et al. [18F]fluorodeoxyglucose uptake by positron emission tomography predicts outcome of non-small-cell lung cancer. J Clin Oncol. 2005;23(6):1136–1143. doi:10.1200/JCO.2005.06.129
9. Hoang JK, Hoagland LF, Coleman RE, Coan AD, Herndon JE
10. Liu J, Dong M, Sun X, Li W, Xing L, Yu J. Prognostic value of 18F-FDG PET/CT in surgical non-small cell lung cancer: a meta-analysis. PLoS One. 2016;11(1):e0146195. doi:10.1371/journal.pone.0146195
11. Wang D, Zhang M, Gao X, Yu L. Prognostic value of baseline 18F-FDG PET/CT functional parameters in patients with advanced lung adenocarcinoma stratified by EGFR mutation status. PLoS One. 2016;11(6):e0158307. doi:10.1371/journal.pone.0158307
12. Sharma A, Mohan A, Bhalla AS, et al. Role of various metabolic parameters derived from baseline 18F-FDG PET/CT as prognostic markers in non-small cell lung cancer patients undergoing platinum-based chemotherapy. Clin Nucl Med. 2018;43(1):e8–e17. doi:10.1097/RLU.0000000000001886
13. Chung HW, Lee KY, Kim HJ, Kim WS, So Y. FDG PET/CT metabolic tumor volume and total lesion glycolysis predict prognosis in patients with advanced lung adenocarcinoma. J Cancer Res Clin Oncol. 2014;140(1):89–98. doi:10.1007/s00432-013-1545-7
14. Liao S, Penney BC, Wroblewski K, et al. Prognostic value of metabolic tumor burden on 18F-FDG PET in nonsurgical patients with non-small cell lung cancer. Eur J Nucl Med Mol Imaging. 2012;39(1):27–38. doi:10.1007/s00259-011-1934-6
15. de Langen AJ, Vincent A, Velasquez LM, et al. Repeatability of 18F-FDG uptake measurements in tumors: a metaanalysis. J Nucl Med. 2012;53(5):701–708. doi:10.2967/jnumed.111.095299
16. Liao S, Penney BC, Zhang H, Suzuki K, Pu Y. Prognostic value of the quantitative metabolic volumetric measurement on 18F-FDG PET/CT in Stage IV nonsurgical small-cell lung cancer. Acad Radiol. 2012;19(1):69–77. doi:10.1016/j.acra.2011.08.020
17. Yoo SW, Kim J, Chong A, et al. Metabolic tumor volume measured by F-18 FDG PET/CT can further stratify the prognosis of patients with Stage IV non-small cell lung cancer. Nucl Med Mol Imaging. 2012;46(4):286–293. doi:10.1007/s13139-012-0165-5
18. Jett JR, Carr LL. Targeted therapy for non-small cell lung cancer. Am J Respir Crit Care Med. 2013;188(8):907–912. doi:10.1164/rccm.201301-0189PP
19. Shi Y, Au JS, Thongprasert S, et al. A prospective, molecular epidemiology study of EGFR mutations in Asian patients with advanced non-small-cell lung cancer of adenocarcinoma histology (PIONEER). J Thorac Oncol. 2014;9(2):154–162. doi:10.1097/JTO.0000000000000033
20. Mok TS, Wu YL, Thongprasert S, et al. Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma. N Engl J Med. 2009;361(10):947–957. doi:10.1056/NEJMoa0810699
21. Rosell R, Carcereny E, Gervais R, et al. Erlotinib versus standard chemotherapy as first-line treatment for European patients with advanced EGFR mutation-positive non-small-cell lung cancer (EURTAC): a multicentre, open-label, randomised Phase 3 trial. Lancet Oncol. 2012;13(3):239–246. doi:10.1016/S1470-2045(11)70393-X
22. Sequist LV, Yang JC, Yamamoto N, et al. Phase III study of afatinib or cisplatin plus pemetrexed in patients with metastatic lung adenocarcinoma with EGFR mutations. J Clin Oncol. 2013;31(27):3327–3334. doi:10.1200/JCO.2012.44.2806
23. Goldstraw P, Chansky K, Crowley J, et al. The IASLC lung cancer staging project: proposals for revision of the TNM Stage Groupings in the Forthcoming (Eighth) Edition of the TNM classification for lung cancer. J Thorac Oncol. 2016;11(1):39–51. doi:10.1016/j.jtho.2015.09.009
24. Eisenhauer EA, Therasse P, Bogaerts J, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45(2):228–247. doi:10.1016/j.ejca.2008.10.026
25. Liu A, Han A, Zhu H, et al. The role of metabolic tumor volume (MTV) measured by [18F] FDG PET/CT in predicting EGFR gene mutation status in non-small cell lung cancer. Oncotarget. 2017;8(20):33736–33744. doi:10.18632/oncotarget.16806
26. Minamimoto R, Jamali M, Gevaert O, et al. Prediction of EGFR and KRAS mutation in non-small cell lung cancer using quantitative (18)F FDG-PET/CT metrics. Oncotarget. 2017;8(32):52792–52801. doi:10.18632/oncotarget.17782
27. Caicedo C, Garcia-Velloso MJ, Lozano MD, et al. Role of [(1)(8)F]FDG PET in prediction of KRAS and EGFR mutation status in patients with advanced non-small-cell lung cancer. Eur J Nucl Med Mol Imaging. 2014;41(11):2058–2065. doi:10.1007/s00259-014-2833-4
28. Cho A, Hur J, Moon YW, et al. Correlation between EGFR gene mutation, cytologic tumor markers, 18F-FDG uptake in non-small cell lung cancer. BMC Cancer. 2016;16:224. doi:10.1186/s12885-016-2251-z
29. Chen L, Zhou Y, Tang X, et al. EGFR mutation decreases FDG uptake in nonsmall cell lung cancer via the NOX4/ROS/GLUT1 axis. Int J Oncol. 2019;54(1):370–380. doi:10.3892/ijo.2018.4626
30. Liemburg-Apers DC, Willems PH, Koopman WJ, Grefte S. Interactions between mitochondrial reactive oxygen species and cellular glucose metabolism. Arch Toxicol. 2015;89(8):1209–1226. doi:10.1007/s00204-015-1520-y
31. Fiorentini D, Prata C, Maraldi T, et al. Contribution of reactive oxygen species to the regulation of Glut1 in two hemopoietic cell lines differing in cytokine sensitivity. Free Radic Biol Med. 2004;37(9):1402–1411. doi:10.1016/j.freeradbiomed.2004.07.022
32. Putora PM, Fruh M, Muller J. FDG-PET SUV-max values do not correlate with epidermal growth factor receptor mutation status in lung adenocarcinoma. Respirology. 2013;18(4):734–735. doi:10.1111/resp.12083
33. Zhu L, Yin G, Chen W, et al. Correlation between EGFR mutation status and F(18) -fluorodeoxyglucose positron emission tomography-computed tomography image features in lung adenocarcinoma. Thorac Cancer. 2019;10(4):659–664. doi:10.1111/1759-7714.12981
34. Chang YS, Choi CM, Lee JC. Mechanisms of epidermal growth factor receptor tyrosine kinase inhibitor resistance and strategies to overcome resistance in lung adenocarcinoma. Tuberc Respir Dis (Seoul). 2016;79(4):248–256. doi:10.4046/trd.2016.79.4.248
35. Kohsaka S, Petronczki M, Solca F, Maemondo M. Tumor clonality and resistance mechanisms in EGFR mutation-positive non-small-cell lung cancer: implications for therapeutic sequencing. Future Oncol. 2019;15(6):637–652. doi:10.2217/fon-2018-0736
36. Guan J, Xiao NJ, Chen M, et al. 18F-FDG uptake for prediction EGFR mutation status in non-small cell lung cancer. Medicine (Baltimore). 2016;95(30):e4421. doi:10.1097/MD.0000000000004421
37. Yang B, Wang QG, Lu M, et al. Correlations study between (18)F-FDG PET/CT metabolic parameters predicting epidermal growth factor receptor mutation status and prognosis in lung adenocarcinoma. Front Oncol. 2019;9:589. doi:10.3389/fonc.2019.00589
38. Ng KS, King Sun C, Boom Ting K, Ting Kun AY. Prognostic factors of EGFR-mutated metastatic adenocarcinoma of lung. Eur J Radiol. 2020;123:108780. doi:10.1016/j.ejrad.2019.108780
39. Keam B, Lee SJ, Kim TM, et al. Total lesion glycolysis in positron emission tomography can predict gefitinib outcomes in non-small-cell lung cancer with activating EGFR mutation. J Thorac Oncol. 2015;10(8):1189–1194. doi:10.1097/JTO.0000000000000569
40. Bottino MC, Thomas V, Janowski GM. A novel spatially designed and functionally graded electrospun membrane for periodontal regeneration. Acta Biomater. 2011;7(1):216–224. doi:10.1016/j.actbio.2010.08.019
41. Huang W, Fan M, Liu B, et al. Value of metabolic tumor volume on repeated 18F-FDG PET/CT for early prediction of survival in locally advanced non-small cell lung cancer treated with concurrent chemoradiotherapy. J Nucl Med. 2014;55(10):1584–1590. doi:10.2967/jnumed.114.142919
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.