Back to Archived Journals » Innovation and Entrepreneurship in Health » Volume 6
Developing a technology-based intervention for empowering cancer patients from low-income backgrounds: a lifecycle approach
Authors Varanasi AP , Helzlsouer KJ
Received 6 December 2017
Accepted for publication 26 February 2019
Published 15 May 2019 Volume 2019:6 Pages 1—7
DOI https://doi.org/10.2147/IEH.S133549
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Professor Rubin Pillay
Video abstract presented by Arti Patel Varanasi.
Views: 133
Arti Patel Varanasi,1 Kathy J Helzlsouer2
1Advancing Synergy, LLC, Baltimore, MD 21227, USA; 2Division of Cancer Control and Population Sciences, National Cancer Institute, Bethesda, MD 20892, USA
Abstract: Despite significant advances across the cancer continuum from prevention through survivorship, achieving uniform high-quality, patient-centered care for all remains a challenge. We have entered the era of precision medicine and multi-modal treatment where the promise of cure is great. Yet fragmented care and disparities in cancer outcomes persist, especially among those individuals from low income and under-resourced backgrounds. Thoughtful, mindful, and purposeful implementation of technology-based innovations have the potential to empower cancer patients and improve long-term outcomes and overall quality of life. These innovations have the potential to provide access to early detection and treatment and to extend and streamline cancer care coordination efforts by complementing and enhancing provider roles throughout the cancer continuum. Technology can be leveraged to mitigate limited resources and in turn improve long-term cancer outcomes for low-income cancer patients. While innovations in technology span a broad spectrum of advancement, we focus here on a technology-based intervention that serves the end goal of empowering patients throughout the cancer continuum as an example of what can be done to support cancer care and management through technology-based innovations.
Keywords: cancer, low income, technology, innovation, intervention, empower
Background
Significant progress has been achieved in treating and caring for cancer patients since the signing of the United States National Cancer Act in 1971.1 Novel treatment modalities coupled with earlier detection have improved the outcomes for cancer patients.2 The number of cancer survivors in the USA is projected to reach 20.3 million by 2026.3 As the population ages, the number of new cancer cases is estimated to increase from 1.6 to 2.3 million cases per year or by 45% in the 20-year period from 2010 to 2030.4 Globally, at a minimum, the number of new cancer cases is anticipated to reach 21.7 million by 2030.5 The increased incidence and prevalence of cancer married with soaring cancer-related needs and costs and a projected decrease in the number of cancer-specific health professionals, including oncologists, puts an increasing burden on the cancer care delivery system in the United States and abroad.1
Achieving patient-centered high-quality cancer care for all remains elusive. This is particularly pronounced in populations that are traditionally underserved and experience cancer health disparities in screening, diagnosis, treatment, and outcomes related to their socioeconomic status, racial/ethnic background, or geographic location.6–10 Technology-based innovations rooted in acceptability, utility, and sustainability offer promise to address disparities in care across these populations.
Technology-based innovations potentially can facilitate patient–provider interaction and empower patients and their caregivers to ultimately achieve and sustain high-quality, patient-centered cancer care. These innovations span the cancer continuum ranging from preventive approaches to digital health solutions to diagnostics to devices. For sustained empowerment and impact, these innovations must meet the needs of the concerned patients and be supported by their communities. In other words, as noted by Kuijpers et al, the innovation must “provide cancer survivors with the skills, knowledge, and motivation to positively influence their health status, which is commonly referred to as patient empowerment”,11 a key element in self-management and improved outcomes.12 This approach makes intuitive sense, but the realities of implementation and scalability are far more challenging. Thus, the first step in developing or considering an existing innovation for use in an under-resourced setting is determining the buy-in and uptake of the proposed innovation.
Several frameworks, including the IDEAS (integrate, design, assess, and share) framework and the lifecycle approach, exist for modeling the implementation of technology-based solutions.13,14 Each of these frameworks emphasizes “assessment” as a step in the process. This lifecycle approach described here is modeled after the technology development lifecycle which is defined as a progression through a series of differing stages of development.14 The lifecycle starts with end-user input followed by design and development, then focus groups and testing, subsequently by modifications and updates, and finally implementation and future iterations based on additional end-user input (Figure 1). An evidence-based design is the result of this iterative process and input from the patient and provider end users through focus groups and testing. The nature of involving the patient in the lifecycle from the beginning inherently empowers them.12,15 The idea with the lifecycle approach is to enable experiences that inform the lifecycle and subsequently yield information on how care and resources are used and optimized. This information on care and resource optimization in turn provides a measure of patient empowerment. Additionally, wrapped around any framework should be a thoughtful, mindful, and purposeful mindset that places the cancer patient at the center. This patient-centric approach ensures that the perspective of the patient is paramount and under consideration at all times. Within this mindset, any innovation that is introduced will have greater chances of acceptability, utility, and sustainability.
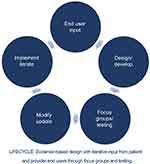 | Figure 1 Lifecycle approach to developing technology-based innovations. |
An ideal way to illustrate this approach is through a case study of empowering patients through technology-enhanced navigation using a lifecycle approach. The benefits of patient navigation in alleviating barriers to treatment initiation and adherence have been well documented.16–18 Patient navigation is an outgrowth of observations by surgical oncologist, Dr Harold Freeman in 1990. Trained as a surgeon, Dr Freeman noted that he was seeing far too many women with late-stage breast cancer in the operating room. He felt the cancers should have been caught earlier and sought to address barriers to cancer screening, diagnosis, treatment, and supportive care. Patient navigation was born at Harlem Hospital. The first cancer patient navigation program demonstrated an increase in the 5-year breast cancer survival rate at Harlem Hospital from 39% to 70%.17 Since 1990, patient navigation has become a part of the cancer care continuum with patient navigators helping patients and their families weave through the complexities of a cancer diagnosis and beyond.19–27 Numerous studies have highlighted the benefits of cancer care coordination, including patient navigation and survivorship care planning, in achieving high-quality cancer care.28–32 As an evidence-based approach to managing treatment and care through the cancer care continuum, cancer patient navigation offers a sustainable solution to addressing the increasing costs of cancer care and for empowering patients.33,34 Extending navigation services to all groups and settings remains a challenge. Technology-based innovations offer one approach to meeting the challenge.
Case study
Rationale
The case study described here is rooted in the question: “Can technology be leveraged using a lifecycle approach to empower cancer patients while optimizing care and resources?”. It challenges the reader to think about resources the health community can leverage to empower cancer patients and optimize care and resources through technology-based innovations.
The case study recounts the design and results of the Technology Enhanced Navigation℠ Trial (TEN℠ Trial).35,36 The randomized clinical trial stemmed from the motivation to find ways to better support low-income women going through breast cancer treatment. Progress in cancer treatment has resulted in improved survival but with an increasing complexity of treatment presenting more challenges for optimum cancer care delivery. Treatment delays, failure to take medication regularly, visit cancellations, missed appointments, and poor symptom management can prevent getting the right dose at the right time and affect cancer outcomes. These issues are more likely to happen among poor, underserved women, including racial and ethnic minorities, who often face limited psychosocial support, financial challenges, and physical and mental health comorbidities.20,35,36
Data from cancer patients and research studies have shown that the poor and underserved have poor cancer outcomes.6,37–42 Socioeconomic and racial disparities in receipt of appropriate adjuvant therapy for breast cancer are well documented.8,9 The completion of treatment regimens for breast cancer that often include a combination of radiation, chemotherapy, and hormonal therapy is particularly challenging for low-income patients who commonly present with comorbidities, endure financial constraints, have limited psychosocial support, and are care for in under-resourced communities.6,9,35 This situation leads to disparities related to race, age, geography, which are more than often tied to income level.37,42 Additionally, this creates both an information gap and information overload along with social isolation and an increasing reliance on self-care.6 The pillars of long-term survival post-diagnosis, including initiating and completing appropriate treatment, receiving supportive care addressing their psychosocial needs, and accessing information and resources to keep them informed from diagnosis through survivorship, are often limited in low-income cancer patients.6,35
As noted earlier, navigation programs offer a potential solution to overcome disparities in treatment, improve receipt of treatment and thus improve cancer outcomes, but few studies have rigorously explored the impact of technology-enhanced patient navigation models on long-term treatment adherence.17,19,35,36,43–45 Thus, the goal of the TEN℠ Trial was to determine if “virtual” interaction with a navigator, who was a trained health professional, versus information alone would improve adherence, symptom management, and quality of life among low-income breast cancer patients undergoing adjuvant breast cancer treatment.35,36
Application design
The design of the TEN℠ application was based on evidence and need as vocalized by breast cancer care providers and breast cancer patients. The statement “TEN℠ is part of my medical team” summarizes the sentiments expressed by focus group participants convened to evaluate the feasibility and acceptability of the TEN℠ application.35,36 The focus group was comprised of low-income breast cancer patients with varying computer expertise. A common theme raised by the group was the isolation they experienced during treatment and the advantage of being connected to information that was filtered and tailored for them. Thus, there was overwhelming support for the face-to-face interaction with the navigator through video chat.35,36 Most significantly, the women viewed the TEN℠ application as a “medical tool” that should be part of their medical team and as their friend.35,36
The TEN℠ application was designed as a readily accessible, cost-efficient, web-based knowledge and communication portal designed to support women in navigating through the complexities of cancer treatment and to get the right treatment and information at the right time. The TEN℠ application offers a novel and innovative approach to provide personalized support to individuals by including opportunities for a virtual chat between the patient and the navigator, tailored information, and most importantly, limits feelings of isolation which in turn empowers the patient. Features of the TEN℠ application include a simple, secure login, and easy-to-use interface with topics identified by icons.35,36 The topical areas connected users to specific, vetted Internet links and team-produced videos and documents. Intervention participants had access to video chat and messaging capabilities.35,36 The application included areas to include information on staging, recommended treatment, and provider contact information.35,36 Lastly, the questionnaire module supported questionnaire administration and tracking for both health/quality of life assessments and usability.35,36 As part of the lifecycle approach, end-user (ie, patients and providers) input was obtained prior to designing the application user interface and after the end user-informed user interface was designed.
Study design
The virtual navigation program supported by the TEN℠ application was evaluated in a simple non-crossover randomized clinical trial comparing the technology-enhanced navigator program to Internet access alone.35,36 Eligible patients were low income and newly diagnosed with stage 0–III breast cancer, for whom adjuvant therapy with chemotherapy, hormone therapy, and/or radiation therapy were part of the recommended treatment plan.
Recruitment and enrollment
Recruitment was facilitated through breast cancer treatment insurance-type programs managed by the Maryland Department of Health and Mental Hygiene and through a network of oncology care providers across the state of Maryland. To eliminate any bias related to technology access, all study participants were provided with a netbook computer and Internet access for the durations of the study. Participants were enrolled from across the state of Maryland where adequate wireless broadband service was available. All patients met low-income level criteria as defined by the Housing and Urban Development Guidelines at the time of the study.46 Informed consent and randomization to the navigator arm (intervention) or the Internet-access only arm (comparison) occurred during the enrollment visit, a majority of which took place in the study participant’s home. The patient empowerment process began during the enrollment visit. Study participants were given the computer along with a Quick Start Guide and a longer User Guide with detailed instructions on connecting to the Internet and using the netbook computer and the navigation application. They also received hands-on training on how to use the device and web-based application, including completion of the online questionnaires. Many of the patients had little to no computer experience and they were able to master the tool during the 2-hr enrollment and training session conducted by the study coordinator.
Implementation
The trial was designed to compare access to a navigator plus information vetted and tailored for breast cancer patient to providing access to information alone. A week after enrollment, the study coordinator contacted the participants to assess any problems they may have with the computer. Within 2 weeks of enrollment, participants on the navigator intervention arm were assigned and contacted by a nurse or social worker navigator. Contact was either by telephone of videoconferencing using the computer. Ultimately, the novelty of videoconferencing disappeared and a majority of the interactions were via telephone.35,36 The navigators attempted to contact study participants approximately every 2 weeks or more often as needed (Helzlsouer 2016a, Helzlsouer 2016b). Assessments included patient-reported outcomes based on validated questionnaires for functionality, quality of life, self-efficacy, and usability of the application. Status of treatment completion, hormone initiation, unscheduled outpatient visits, emergency room visits, and hospitalizations were obtained from medical records key study outcomes included treatment adherence, fatigue, quality of life, satisfaction with care, and technology feasibility and usability.
Results
From a total of 150 interested individuals screened, 102 were enrolled and 98 (49 on each study arm) included in the final analysis.35,36 The presence of comorbidities at baseline was assessed and found that almost half of the women in both study arms at four or more reported diseases.35,36 Further, about half of those had either anxiety or depression. This speaks to the challenges of addressing cancer treatment in a complex environment where women are dealing with multiple disease burdens at the same time. All of the study participants on the intervention (navigator) arm completed recommended chemotherapy and initiated recommended hormone therapy, whereas four study participants on the control arm did not complete one or more recommended adjuvant treatments.35,36 A high level of use and agreement on ease-of-use and confidence in using the portal was observed.35,36 The usability assessment of the web-based application obtained 3 months after enrollment was similar between the two groups with approximately 75% reporting that they used the program frequently and that it was easy to use.35,36 Notably, none of the study participants reported having access to navigator services beyond initial diagnosis and surgery.35,36 This reality mimics what often occurs in cancer care and clearly illustrates the need for a technology-based approach to providing the needed support and access to services during cancer treatment and beyond.
In summary, adherence to treatment was better with the addition of a navigator compared to control. End-user involvement and iterative development supported the high degree of usability. More significant is that computer competence was achievable after brief training (~2 hrs) even among those with no prior computer experience. The use of the application and overall confidence in the ability to access reliable information was high thus assisting into patient empowerment.35,36
Lifecycle: experiences that inform
With the TEN℠ case study in mind, it is apparent that the lifecycle is made up of experiences. These experiences inform and extend and feed into a larger lifecycle. Further, the outcomes from the lifecycle determine what happens next. In the lifecycle approach, we look to 1) sustain impact, 2) continue the lifecycle, and 3) add relevant components based on the experience of the lifecycle. Collectively, the lifecycle experiences inform and empower. The lifecycle approach inherently ties into the optimization of care and resources, which involves the following five steps: 1) identifying the issue; 2) assessing the situation; 3) taking action; 4) evaluating the impact; and 5) sustaining the solution (Figure 2). Further, this approach involves the human and the technology working together hand-in-hand. Building further on care and resource optimization is being able to incorporate findings and new information into the lifecycle. For example, through our TEN℠ lifecycle, we found challenges to sustainability to include: connectivity costs, compliance with questionnaires among the control group who had no personal interaction with a navigator transition to survivorship care, independent navigation versus direct line of care, hours of operation, mobile population, intermittent phone service, length and multiplicity of treatments, and transition to survivorship care. Despite these challenges, the decreasing costs of electronic devices, such as tablets, phone, and portable computers, and improved accessibility to connectivity open a new avenue for patient empowerment with the lifecycle approach providing a framework for optimizing care and resources by leveraging human capital and technology.47 Ultimately, technology-based innovations can be used to empower low-income cancer patients and optimize care and resources using a lifecycle approach that actively engages patients and values their input as part of the process.48
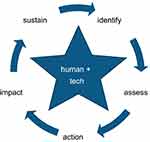 | Figure 2 Care and resource optimization. |
Conclusions
Together the case study and lifecycle approach described here provide an example of a framework for developing, evaluating, and implementing technology-based innovations for low-income cancer patients across the cancer continuum. There are an increasing number of evidence-based approaches to improving the cancer experience from prevention through survivorship. Davis and Oakley-Girvan (2017) outline key elements of patient empowerment and user-centered application design, development, and testing to ensure that applications will meet the needs of cancer survivors.48 Although the innovation described in the case study is a digital health-based solution, innovations can be solutions ranging from a process to a device that ultimately result in a paradigm shift from the status quo to a new approach to addressing the growing cancer burden, especially in under-resourced settings. Innovations with low cost are needed to address disparities among low-income and low-resourced communities. The ultimate test of implementation and sustainability is having the correct alignment of resources and buy-in. For example, with the advent of the human papillomavirus vaccine and novel approaches to screening and treatment, cervical cancer incidence and prevalence has the potential to be drastically reduced.49–51 White Tsu notes that “We already know what causes cervical cancer, how to prevent it, and how to treat it, even in resource-constrained settings”, and yet the solution is “in large part, sustained political commitment”.50 The challenge is to think and look outside of the proverbial box and begin from the patient perspective considering their lifestyles and available resources to identify innovations with high impact and relatively low cost. These innovations fill gaps in care and ultimately serve to empower the cancer patient beyond their initial diagnosis and positively impact their long-term outcomes.
Acknowledgments
The case study described above is the result of the work of the Technology-Enhanced Navigation (TEN) Clinical Trial Study Team, comprising the Prevention and Research Center with Dr Kathy Helzlsouer as the study Principal Investigator, Advancing Synergy, the Maryland Department of Health and Mental Hygiene, funding for the clinical trial from Susan G. Komen for the Cure, and referring providers across the state of Maryland.
Disclosure
The authors report no conflicts of interest in this work.
References
1.
2. Cronin KA, Lake AJ, Scott S, et al. Annual report to the nation on the status of cancer, part I: national cancer statistics. Cancer. 2018;124(13):2785–2800. doi:10.1002/cncr.31551. Epub 2018 May 22.
3. Miller KD, Siegel RL, Lin CC, et al. Cancer treatment and survivorship statistics, 2016. CA Cancer J Clin. 2016;66(4):271–289. doi:10.3322/caac.21349
4. Smith BD, Smith GL, Hurria A, Hortobagyi GN, Buchholz TA. Future of cancer incidence in the united states: burdens upon an aging, changing nation. J Clin Oncol. 2009;27(17):2758–2765. doi:10.1200/JCO.2008.20.8983
5.
6. Bettencourt BA, Schlegel RJ, Talley AE, Molix LA. The breast cancer experience of rural women: a literature review. Psychooncology. 2007;16(10):875–887. doi:10.1002/pon.1235
7. Budman DR. Dose and schedule as determinants of outcomes in chemotherapy for breast cancer. Semin Oncol. 2004;31(6 Suppl 15):3–9. doi:10.1053/j.seminoncol.2004.11.021
8. Wildiers H, Reiser M. Relative dose intensity of chemotherapy and its impact on outcomes in patients with early breast cancer or aggressive lymphoma. Crit Rev Oncol Hematol. 2011;77(3):221–240. doi:10.1016/j.critrevonc.2010.02.002
9. Du Xianglin L, Gor BJ. Racial disparities and trends in radiation therapy after breast-conserving surgery for early-stage breast cancer in women, 1992 to 2002. Ethn Dis. 2007;17(1):122–128.
10. Haggstrom DA, Quale C, Smith-Bindman R. Differences in the quality of breast cancer care among vulnerable populations. Cancer. 2005;104(11):2347–2358. doi:10.1002/cncr.21443
11. Kuijpers W, Groen WG, Loos R, et al. An interactive portal to empower cancer survivors: a qualitative study on user expectations. Support Care Cancer. 2015;23(9):2535–2542. doi:10.1007/s00520-015-2605-0
12. Aujoulat I, d’Hoore W, Deccache A. Patient empowerment in theory and practice: polysemy or cacophony? Patient Educ Couns. 2007;66(1):13–20. pii:S0738-3991(06)00324-7. doi:10.1016/j.pec.2006.09.008.
13. Mummah SA, Robinson TN, King AC, Gardner CD, Sutton S. IDEAS (integrate, design, assess, and share): a framework and toolkit of strategies for the development of more effective digital interventions to change health behavior. J Med Internet Res. 2016;18(12):e317. pii:v18i12e317. doi:10.2196/jmir.5927.
14. Borycki EM, Househ M, Kushniruk AW, Kuziemsky C. Use of qualitative methods across the software development lifecycle in health informatics. Stud Health Technol Inform. 2011;164:293–297.
15. Ludman EJ, McCorkle R, Bowles EA, et al. Do depressed newly diagnosed cancer patients differentially benefit from nurse navigation? Gen Hosp Psychiatry. 2015;37(3):236–239. doi:10.1016/j.genhosppsych.2015.02.008
16. Freeman HP, Muth BJ, Kerner JF. Expanding access to cancer screening and clinical follow-up among the medically underserved. Cancer Pract. 1995;3(1):19–30.
17. Freeman HP. The history, principles, and future of patient navigation: commentary. Semin Oncol Nurs. 2013;29(2):72–75. doi:10.1016/j.soncn.2013.02.002
18. Nguyen TU, Kagawa-Singer M. Overcoming barriers to cancer care through health navigation programs. Semin Oncol Nurs. 2008;24(4):270–278. doi:10.1016/j.soncn.2008.08.007
19. Freund KM, Battaglia TA, Calhoun E, et al. National cancer institute patient navigation research program: methods, protocol, and measures. Cancer. 2008;113(12):3391–3399. doi:10.1002/cncr.23960
20. Ell K, Vourlekis B, Xie B, et al. Cancer treatment adherence among low-income women with breast or gynecologic cancer: a randomized controlled trial of patient navigation. Cancer. 2009;115(19):4606–4615. doi:10.1002/cncr.24500
21. Esparza A. Patient navigation and the American Cancer Society. Semin Oncol Nurs. 2013;29(2):91–96. doi:10.1016/j.soncn.2013.02.004
22. Ferrante JM, Chen PH, Kim S. The effect of patient navigation on time to diagnosis, anxiety, and satisfaction in urban minority women with abnormal mammograms: a randomized controlled trial. J Urban Health. 2008;85(1):114–124. doi:10.1007/s11524-007-9228-9
23. Fillion L, de Serres M, Cook S, Goupil RL, Bairati I, Doll R. Professional patient navigation in head and neck cancer. Semin Oncol Nurs. 2009;25(3):212–221. doi:10.1016/j.soncn.2009.05.004
24. Gabram SG, Lund MJ, Gardner J, et al. Effects of an outreach and internal navigation program on breast cancer diagnosis in an urban cancer center with a large african-american population. Cancer. 2008;113(3):602–607. doi:10.1002/cncr.23568
25. Haylock PJ, Cantril C. Patient navigation in cancer care. Introduction. Semin Oncol Nurs. 2013;29(2):71. doi:10.1016/j.soncn.2013.02.001
26. Hopkins J, Mumber MP. Patient navigation through the cancer care continuum: an overview. J Oncol Pract. 2009;5(4):150–152. doi:10.1200/JOP.0943501
27. Krebs LU, Burhansstipanov L, Watanabe-Galloway S, Pingatore NL, Petereit DG, Isham D. Navigation as an intervention to eliminate disparities in American Indian communities. Semin Oncol Nurs. 2013;29(2):118–127. doi:10.1016/j.soncn.2013.02.007
28. Basu M, Linebarger J, Gabram SG, Patterson SG, Amin M, Ward KC. The effect of nurse navigation on timeliness of breast cancer care at an academic comprehensive cancer center. Cancer. 2013;119(14):2524–2531. doi:10.1002/cncr.28024
29. Cantril C, Haylock PJ. Patient navigation in the oncology care setting. Semin Oncol Nurs. 2013;29(2):76–90. doi:10.1016/j.soncn.2013.02.003
30. Raich PC, Whitley EM, Thorland W, Valverde P, Fairclough D;
31. Whitley EM, Raich PC, Dudley DJ, et al. Relation of comorbidities and patient navigation with the time to diagnostic resolution after abnormal cancer screening. Cancer. 2017;123(2):312–318. doi:10.1002/cncr.30316
32. Freund KM, Battaglia TA, Calhoun E, et al. Impact of patient navigation on timely cancer care: the patient navigation research program. J Natl Cancer Inst. 2014;106(6):dju115. doi:10.1093/jnci/dju115
33. Pratt-Chapman M, Willis A. Community cancer center administration and support for navigation services. Semin Oncol Nurs. 2013;29(2):141–148. doi:10.1016/j.soncn.2013.02.009
34. Ramsey S, Whitley E, Mears VW, et al. Evaluating the cost-effectiveness of cancer patient navigation programs: conceptual and practical issues. Cancer. 2009;115(23):5394–5403. doi:10.1002/cncr.24603
35. Helzlsouer K, Appling S, Gallichio L, et al. A pilot study of a virtual navigation program to improve adherence among low-income breast cancer patients. J Oncol Navig Surviv. 2016;7(7):20–29.
36. Helzlsouer K, Appling S, Scarvalone S, et al. Development and evaluation of a technology-enhanced interdisciplinary navigation program for low-income breast cancer patients. J Oncol Navig Surviv. 2016;7(4):10–18.
37. Bhargava A, Du XL. Racial and socioeconomic disparities in adjuvant chemotherapy for older women with lymph node-positive, operable breast cancer. Cancer. 2009;115(13):2999–3008. doi:10.1002/cncr.24363
38. Joslyn SA. Hormone receptors in breast cancer: racial differences in distribution and survival. Breast Cancer Res Treat. 2002;73(1):45–59. doi:10.1023/A:1015220420400
39. Katz ML, Young GS, Reiter PL, et al. Barriers reported among patients with breast and cervical abnormalities in the patient navigation research program: impact on timely care. Womens Health Issues. 2014;24(1):155. doi:10.1016/j.whi.2013.10.010
40. Nguyen-Pham S, Leung J, McLaughlin D. Disparities in breast cancer stage at diagnosis in urban and rural adult women: a systematic review and meta-analysis. Ann Epidemiol. 2014;24(3):228–235. doi:10.1016/j.annepidem.2013.12.002
41. Joslyn SA. Racial differences in treatment and survival from early-stage breast carcinoma. Cancer. 2002b;95(8):1759–1766. doi:10.1002/(ISSN)1097-0142
42. Griggs JJ, Culakova E, Sorbero ME, et al. Social and racial differences in selection of breast cancer adjuvant chemotherapy regimens. J Clin Oncol. 2007;25(18):2522–2527. doi:10.1200/JCO.2006.10.2749
43. Gustafson DH, McTavish FM, Stengle W, et al. Use and impact of eHealth system by low-income women with breast cancer. J Health Commun. 2005a;10(Suppl 1):195–218. doi:10.1080/10810730500263257
44. Gustafson DH, McTavish FM, Stengle W, et al. Reducing the digital divide for low-income women with breast cancer: a feasibility study of a population-based intervention. J Health Commun. 2005b;10(Suppl 1):173–193. doi:10.1080/10810730500263281
45. Freeman HP. Poverty, culture, and social injustice: determinants of cancer disparities. CA Cancer J Clin. 2004;54(2):72–77.
46.
47. Purnell JQ, Griffith J, Eddens KS, Kreuter MW. Mobile technology, cancer prevention, and health status among diverse, low-income adults. Am J Health Promot. 2014;28(6):397–402. doi:10.4278/ajhp.120816-ARB-396
48. Davis SW, Oakley-Girvan I. Achieving value in mobile health applications for cancer survivors. J Cancer Surviv. 2017;11(4):498–504. doi:10.1007/s11764-017-0608-1
49. White HL, Meglioli A, Chowdhury R, Nuccio O. Integrating cervical cancer screening and preventive treatment with family planning and HIV-related services. Int J Gynaecol Obstet. 2017;138(Suppl 1):41–46. doi:10.1002/ijgo.12194
50. Tsu VD. Cervical cancer prevention for all women: why now? Int J Gynaecol Obstet. 2017;138(Suppl 1):1–3. doi:10.1002/ijgo.12188
51. Fenton AT, Elliott MN, Schwebel DC, et al. Unequal interactions: examining the role of patient-centered care in reducing inequitable diffusion of a medical innovation, the human papillomavirus (HPV) vaccine. Soc Sci Med. .Soc Sci Med. 2018;200:238–248. doi: 10.1016/j.socscimed.2017.09.030.
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
