Back to Journals » HIV/AIDS - Research and Palliative Care » Volume 12
Determinants of Virological Failure Among Patients on First-line Antiretroviral Therapy in Central Oromia, Ethiopia: A Case–Control Study
Authors Abdullahi IJ, Deybasso HA , Adlo AM
Received 12 September 2020
Accepted for publication 1 December 2020
Published 14 December 2020 Volume 2020:12 Pages 931—939
DOI https://doi.org/10.2147/HIV.S281672
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Bassel Sawaya
Ilili Jemal Abdullahi,1 Haji Aman Deybasso,2 Abebe Megerso Adlo2
1ICAP Ethiopia Central Office, Addis Ababa, Ethiopia; 2Department of Public Health, Adama Hospital Medical College, Adama, Oromia, Ethiopia
Correspondence: Abebe Megerso Adlo Tel +251 911 04 45 25
Email [email protected]
Background: Viral suppression among people living with HIV is the main goal of antiretroviral therapy (ART). Despite the treatment, more than four million people and over 55 thousand patients globally and in Ethiopia had virological failure respectively in 2017. The purpose of this study was to identify determinants of virological failure among patients on ART in central Oromia, Ethiopia.
Methods: A facility-based unmatched case–control study was conducted. Sample size was calculated using EpiInfo software, and 504 randomly selected patient’s records were analyzed. Case to control ratio was one to one. Separate sampling frames were prepared for cases and controls using medical registration numbers (MRN) from patients’ registrations data. Cases and controls were selected from respective sampling frames. Respective patient charts were traced using the MRN, and data were extracted from patient charts using data abstraction format. The data were entered using EpiInfo version seven and analyzed using SPSS version 20.0. Descriptive statistics and binary logistic regression were used for the analysis. Adjusted odds ratio with its 95%CI was used to present strength association. A P-value of < 0.05 was used to determine statistical significance of the association.
Results: Age < 15 years (AOR=3.72, 95%CI: 1.70– 8.12), baseline CD4 count < 100/mm3 (AOR=2.31, 95%CI: 1.05– 5.08), TDF-3TC-NVP regimen (AOR=2.81, 95%CI: 1.28– 6.18), nondisclosure of HIV status (AOR=6.78, 95%CI: 3.24– 14.15) and history of poor adherence (AOR=11.19, 95%CI: 5.65– 22.15) were factors independently associated with virological failure among the study participants.
Conclusion: Younger age, low baseline CD4 count, TDF/NVP containing regimen, poor adherence, and nondisclosure of HIV status were significant determinants of virological failure. Therefore, adherence counseling and paying due attention to patients having these determinants are required to achieve adequate viral load suppression.
Keywords: ART, ART case–control, Ethiopia, HIV, Oromia, virological failure
Plain Language Summary
HIV infection has been a global public health problem for over four decades. Antiretroviral treatment (ART) aims at controlling replication of HIV in the human body thereby preventing AIDS, the disease caused by this virus. This treatment is being given to millions of people infected by HIV. Despite the treatment, millions of people in the world and thousands of patients in Ethiopia having the virus are not responding to ART. The purpose of this study was to identify factors contributing to resistance of HIV to ART in central Oromia, Ethiopia.
To the contributing factors, we compared equal number of patients with controlled viral load (252) and those with uncontrolled viral load (252). Medical records of the patients from both groups were selected by lottery and data were extracted from the patient charts using data abstraction format. We analyzed the data using a scientifically acceptable statistical method. As a result we found several factors contributing to the resistance of the virus.
Accordingly, younger age, low CD4 count at the start of ART, nevirapine containing regimen, poor adherence, and nondisclosure of HIV status were some of the factors. Therefore, adherence counseling and paying due attention to patients having these determinants is required to achieve adequate viral load suppression.
Introduction
AIDS-related illnesses remain among the leading causes of death around the globe. Every day, 5000 people are infected and there were 1.8 million new HIV infections globally in 2017, which is far from the target set to end AIDS by 2030.1–3 In Ethiopia, 14,872 people died because of HIV/AIDS and there were 247,250 AIDS orphans in 2017.4 Oromia region accounts for 30% of deaths and 37% of AIDS orphans in the country.5,6 The worrisome fact is not only the risk of HIV transmission, but also the transmission of the resistant virus to ART naïve patients.7
In the fight against the HIV/AIDS pandemic, provision of the ART is to achieve optimal viral suppression; that is to less than 1000 copies per/mL of the plasma. Failure to achieve this target viral load (VL) is known as virological failure, which is a VL count of more than 1000 copies/mL in two consecutive results at least three months apart.3,4 When the VL is adequately suppressed, damage to the immune system will be minimized and risk of opportunistic infections that leads to morbidity and mortality will decrease.8,9 Fourfold reductions in the average number of HIV doubles survival time for the patient on ART.10 Risk of HIV transmission is very low with optimal viral suppression.4,9,11
Despite this evidence, more than four million people who were on ART, had unsuppressed VL and there were about one million deaths from HIV globally in 2017.12 The majority of patients on ART, but with unsuppressed VL, have carried drug resistance mutated viruses. Researchers reported more than 60% resistance to at least one antiretroviral (ARV) drug among patients on ART.13–16 Overall ART treatment failure was 15.9% while the virological failure was 6.3%.17 A study that determined resistance among HIV-infected children (less than two years) and with high VL reported 75% ARV resistance of which 52% existed prior to ART (transmitted resistance virus).18
Diversified factors were associated with ART failure in general and with the virological failure in particular.19,20 Factors that determine ART outcome may differ from one context to another. Therefore, understanding factors that determine the virological failure among patients on ART is important to inform HIV/AIDS prevention and control program stakeholders. Furthermore, as routine VL monitoring service expanded recently in Ethiopia and evidence on determinants of the virological failure are scarce in similar settings in general, and in the current study setting in particular, this study is important to contribute to filling the evidence gap.
Methods and Materials
Study Design, Area and Period
We employed an unmatched case–control study design in Oromia region, central Ethiopia, from December 2018 to January 2019. Oromia is one of the HIV/AIDS epidemic highly affected regions in the country. At the time of data collection, 409 health facilities provide ART services and more than 100,000 patients were on ART in the region.21 There were 10 high-load towns accounting for over 40% of clients receiving the treatment in the region. Among clients receiving ART in the 10 towns, 58% are receiving the treatment in four high-load towns namely, Adama, Bishoftu, Mojo, and Asela. Catchment areas of these four towns were specific study areas for the current study. Four hospitals and eight health centers are providing ART services and at the end of June 2018, 22,853 clients were receiving ART in these towns. Adama Regional Laboratory serves as a referral laboratory for all health facilities in the study area to determine VL for all clients receiving the treatment in the four towns.
Study Population and Eligibility Criteria
Source population for this study was all clients enrolled for ART in the four major towns (Adama, Bishoftu, Mojo, and Asela) during the study period, while study population was patients enrolled for the services and had documented viral loadVL result from October 1, 2017 to September 30, 2018 in the regional reference laboratory. Cases were those patients on ART for more than six months and with VL not reduced to the target (plasma viral load of >1000 copies/mL) on two consecutive tests with a three-month interval with adherence support.4 These include all patients who failed to attain the target including those who had inadequate VL suppression (reduced VL but still above the target), constant VL above the target and those with increased VL. The controls were those patients with adequately suppressed VL, that is to the plasma VL of ≤1000 copies/mL on routine viral load test done in one year prior to the data collection (October 1, 2017 to September 30, 2018).4
Sample Size Determination and Sampling Procedure
Sample size was calculated using EpiInfo StatCalc an unmatched case–control formulae.22 Assumptions such as 95%CI 80% power and one-to-one case-to-control ratio were used in the sample size determination. Four determinants of virological failure identified in other studies, such as younger age group, longer duration on ART, low CD4 count and not receiving single tablet daily regimen were used as determinant variables. These determinants were among those reported in studies in in the country and in other similar African countries.19,20,23–27 Under the assumptions, calculation using the variable “five year or more duration on ART” gave largest sample size; that was 504 and consequently, 252 cases and 252 controls were included in this study.
From the 12 ART sites in the study area, five health facilities namely, Adama and Bishoftu Hospitals, Geda, St Fransico, and Mojo health centers were randomly selected. All of these health facilities send collected samples for VL test to Adama Regional Laboratory, where data clerks record the unique ART number, name of referring health facility, test result, and many more variables. From that database, a list of routine viral load tests done from October 1, 2017 to September 30, 2018 for the selected ART sites was generated.
Two sampling frames, one for cases and the second for controls were prepared using the unique ART number. For the cases, a list of all patients recorded as having virological failure was prepared separately. Then a list of all patients who achieved the target viral suppression was prepared. Both samples of cases and controls were selected by random sampling technique using SPSS software from respective sampling frames.28 Names of the health facilities and unique ART number of selected patients were used to obtain patient charts from each health facility to extract additional variables which were not available at the regional laboratory.
Study Variables
Virological failure, a persistent VL exceeding 1000 copies/mm3 based on two consecutive tests within a three-month interval after at least six months of ART, was the outcome variable of the study. Sociodemographic characteristics such as age, sex, educational level, religion, marital status, residence, and other clinical characteristics such as tuberculosis co-infection, WHO clinical stage at initiation, baseline CD4 cell count, first-line regimen given, and duration on ART, nutritional status, ART adherence and history of ART regimen change were the independent variables of the study.
Data Collection Procedure
Data extraction form was developed by the investigators after reviewing national comprehensive ART guidelines, available recording and reporting formats at the ART clinics and extensive literature review.4 Eight degree-level health professionals were recruited to extract data from patient folders. They have basic ART training and experience of working in the ART clinics. Lists of selected patient unique ART number and their respective health facility names were given to the data collectors. They extracted required data elements from patient charts using the tool under close supervision of the authors of this paper.
Data Quality Assurance
The data collection tool was pretested for its comprehensiveness before the actual data collection and necessary amendments were made. Experienced health professionals were used for the data collection and were trained on the tool before the commencement of the data collection. There were daily checking of the completed tool and debriefing sessions by supervisors to the data collectors to ensure quality of the data. Supervisors have also randomly selected patient charts and checked against the completed data extraction tool.
Data Processing and Analysis
Data were entered in to computer using EPIInfo version seven and analyzed using SPSS version 20.0.22,28 Descriptive statistics were computed to report the demographic and clinical characteristics of cases and controls. Bivariate analysis was performed to assess for a crude association between the independent and the outcome variables. Variables with a P-value of ≤0.25 at the bivariate analysis were selected for the final model.29 Multivariable binary logistic regression modeling was used to identify independent determinants of virological failure. Hosmer–Lemeshow goodness-of-fit test was used to assess model fitness (P-value was 0.243) implying that the model fits the data.29 Multicollinearity between the independent variables was also checked. Tolerance of ≤0.1 was considered to indicate the existence of multicollinearity and removed one of the collinear variables from the model.30 Finally, variables that have a strong association with the virological failure were identified based on the odds ratio with its 95%CI. A P-value ≤0.05 was used to declare statistical significance.
Ethical Consideration
This study was approved by Adama Hospital Medical College Institutional Review Board and it was conducted in accordance with the Declaration of Helsinki. Individual study participant’s consent was waived by the review board as the data source of medical records of patients. Permission letters to the selected health facilities were obtained from Oromia Regional Health Bureau. Permission was also secured from respective health facility leaders and all data were anonymously registered using codes without personal identifiers such as names of the patients. The data were kept confidential throughout the process of the study and individuals based analysis of data was made in the current study.
Results
Sociodemographic Characteristics
Five hundred and four participants, 252 cases and 252 controls were included in this study. The median ages were 29 years (IQR=18–35) and 34 years (IQR=28–40) for cases and the controls respectively. Nearly a quarter of the cases, 60 (23.8%) and 17 (6.7%) of the controls were younger than 15 years of age at enrolment to ART (Table 1).
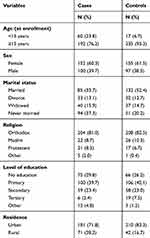 |
Table 1 Sociodemographic Characteristics of Patients Receiving ART in Central Oromia, Ethiopia, 2018 |
Clinical Characteristics
The baseline median CD4 counts of cases and controls were 175/mm3 (IQR=99–291) and 223/mm3 (IQR=135–343) respectively. Slightly over half 142 (56.3%) of the cases were presented to care with WHO disease stage of III or IV, while 116 (46.0%) of the controls were in this category at the time of enrollment. Proportion of the study participants with a history of TB among case and control were 24% and 23% respectively, while 23.4% of the controls and 33.7% of the cases had some form of malnutrition at baseline (Table 2).
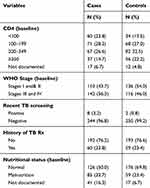 |
Table 2 Clinical Characteristics of Patients Receiving ART in Central Oromia, Ethiopia, 2018 |
Antiretroviral Treatment-Related Characteristics
The median number of months that the study participants were on ART was 88 months (IQR=64–121) for cases and 84 months (IQR=54–114) for the controls. The commonest regimen for controls and cases was TDF-3TC-EFV, 50.4% and 24.6% respectively. There were a higher proportion of participants among cases 145 (57.5%) with history of regimen shift or substitution, while only 70 (27.8%) control had a similar history (Table 3).
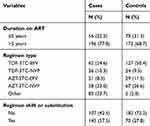 |
Table 3 Antiretroviral Medication-related Characteristics of Patients Receiving ART in Central Oromia, Ethiopia, 2018 |
Behavioral Characteristics
Regarding disclosure of HIV status of the study participants, the majority, 205 (81.3%) of the case and 238 (94.4%) of the controls had documented disclosure of their HIV status. This shows that disclosure among cases was more than 10% lower than that of the controls. Regarding the adherence, 91 (36.1%) of the cases and 15 (6.0%) controls had a history of poor adherence, that is adherence level of <85%. Congruent with the disclosure status, poor adherence was >30% higher among cases than the controls.
Factors Associated with Unsuppressed Viral Loads
In the binary logistic regression analysis where cases were coded to “1” and controls were coded to “0”, age, residence, WHO stage (baseline), baseline CD4 count, nutritional status, duration on ART, type of regimen, history of regimen shift/substitution, disclosure status and history of poor adherence came out to have a P-value ≤0.25 and became candidate variables for the final regression model. When adjusted for potential confounders in the final model, the multivariable logistic regression, age <15 years, baseline CD4 <100/mm3, TDF-3TC-NVP regimen, nondisclosure of HIV status and poor adherence came out to be independent determinants of unsuppressed VL or failure in adequate suppression of the VL.
Accordingly, odds of failure in the adequate suppression of VL was almost four times higher (AOR=3.71, 95%CI: 1.7–8.1) among children aged <15 years at enrollment compared to older patients. Starting ART after CD4 count decreased was associated with failure in adequate suppression of the VL; patients with baseline CD4 counts <100/mm3 had higher odds (AOR=2.31, 95%CI: 1.1–5.1) of the unsuppressed VL compared to those patients with baseline CD4 ≥350/mm3. In addition, patients who were taking TDF-3TC-NVP had nearly three times higher odds (AOR=2.81, 95%CI: 1.3–6.2) of having unsuppressed VL compared to those treated with TDF-3TC-EFV; and also, patients who did not disclose their HIV status to others had more than six times higher odds (AOR=6.77, 95%CI: 3.2–14.2) of unsuppressed VL compared to their counterpart. Furthermore, adherence status is another variable with a significant association with unsuppressed VL; patients who had a history of poor adherence had higher odds (AOR=11.19, 95%CI: 5.7–22.2) of having unsuppressed VL compared to those who had fair-to-good adherence status (Table 4).
 |
Table 4 Determinants of Virological Failure Among Patients on ART in Central Oromia, Ethiopia, 2018 |
Discussion
Our finding revealed that children (age <15 years) had higher odds of unsuppressed VL compared to those ≥15 years. This result is consistent with studies done in Uganda and South Africa.26,27 Our practical experience in Ethiopia shows that 78% of children younger than 10 years are being treated with NVP-containing regimens, and the majority of the children included in this study were also receiving NVP-based regimen, which is nonoptimal and although not preferred as first-line ARV regimen.31 It is recommended to use LPV/r-based regimen for younger children since it is superior to NVP-based regimens in long-term virological suppression.8,9,32 There is also a high level of drug resistance (most of this is non-nucleotide reverse transcriptase inhibitor resistance) reported among children, which will affect the virological suppression.7 Moreover, the formulation of LPV/r currently available in Ethiopia is the liquid one, which has a bitter taste, and can affect the adherence of the children to treatment. The other possible explanation for higher odds of unsuppressed VL among children could be a gap with adjusting the ARV dose as children grow fast and continuously. If the dose is not adjusted when weight increases, the concentration of the drug will be suboptimal and unable to control viral replication. In addition, children who attend school might not follow the medication schedule on weekdays.
The other determinant of having unsuppressed VL was baseline CD4 <100/mm3. This is similar to study in South Africa that showed an association between CD4 cell count of less than 100/mm3 at ART start with unsuppressed VL.26 Similar result was also reported in a study done in Vietnam that showed an association between unsuppressed VL with lower CD4 cell count.25 A study from western Kenya also revealed higher odds of treatment failure among patients with low baseline CD4.24
Researchers identified higher odds of unsuppressed VL among children who were treated with TDF-3TC-NVP regimens compared to those on TDF-3TC-EFV. This could be due to the palatability of the preparation, since TDF-3TC-EFV formulation is a fixed-dose combination of a single pill taken once daily. A study from Vietnam showed an association between the fixed-dose (single tablet daily) regimen with suppressed VL.25 Other studies also showed that, from non-nucleoside reverse transcriptase inhibitor, efavirenz use was associated with better virological outcome compared to nevirapine.33,34
Patients with poor adherence had more than 10 times higher odds of unsuppressed VL compared to those with good adherence. This is also consistent with the results reported by other research. Studies from Uganda, Kenya, Cameron and Haiti have consistently shown increased odds of high VL among those with poor adherence.14,27,35,36 High viral load among those with poor adherence occurs for the obvious reason, that optimal drug concentration is needed to control viral replication. It is reported that adherence at or above 95% is required for durable viral suppression.8,9
The other important predictor of high viral load identified in our study was disclosure status. Those who did not disclose their status to others had higher odds of unsuppressed VL. This is mainly due to difficulty in adherence to treatment and follow-up among those who did not disclose their status. Those who disclosed reported more responses of support, encouragement, and lower depression, which can have an indirect positive effect on treatment outcome.37,38 Whereas, a study done in the UK showed no effect of disclosure on the outcome of the treatment. This discrepancy could be due to differences in setup (with relative ease of good adherence to ART without the need to disclose in the UK) and variation in the characteristic of the population studied in the two settings.39 For children, disclosure of HIV status to the children themselves has a health benefit, including increased adherence to treatment and higher odds of retention among disclosed.40,41 This could be another reason for the difference in odds of viral suppression among those to whom the status was disclosed and to whom it was not.
In this study, sex of the study participants did not come out as independent determinant of virological failure; and this is consistent with a previous study.25 In contrast to some previous studies, variables such as history Tb co-infection, baseline nutritional status, duration on first-line ART were not significantly associated with virological failure in our study.19,23,27,42 This difference could signify difference of the determinants from one context to another.
Limitations of the Study
In this study, we were able to identify several determinants of virological failure; yet we acknowledge some limitations of the study at the same time. One of the limitations was the fact that we used existing routinely recorded facility data. As a result, we failed to capture some potentially important variables either due to missing records or the variables may not be required for patient care purposes. Therefore, we were not able to include some of the variables that we planned to analyze. Furthermore, although the study setting was an area with a high patient load, generalization of the results to more peripheral settings of the region should be done with precaution. Nevertheless, given good data collection procedure, design of the study and modeling, the result of this study remains important for scholars and program implementers.
Conclusion
Younger age (<15 years), low baseline CD4 count (<100/mm3), nevirapine-based regimen, nondisclosure of HIV status and history of poor adherence were independent determinants of virological failure among patients on ART in the current study setting. Therefore, all HIV/AIDS program stakeholders should pay due attention to patients with these identified determinates. Furthermore, we recommend researchers to conduct more comprehensive research that can address the limitations of this study.
Data Sharing Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Acknowledgment
We acknowledge the study participants, data collectors, ART providers and facility managers of the included health facilities for their respective contributions without which this study would not have been realized.
Author Contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Joint United Nations Programme on HIV/AIDS (UNAIDS). GLOBAL AIDS UPDATE 2017, Ending AIDS, Progress Towards the 90–90–90 Targets. 2017.
2. UNAIDS. Joint United Nations Programme on HIV/AIDS UNAIDS. UNAIDS; 2017.
3. World Health Organization. Guidelines for Managing Advanced HIV Disease and Rapid Initiation of Antiretroviral Therapy. 2017.
4. Ministry of Health, Ethiopia. National Consolidated Guidelines for Comprehensive Hiv Prevention, Care and Treatment. 2018.
5. The Ethiopian Public Health Institute. HIV Related Estimates and Projections for Ethiopia–2017. Addis Ababa, Ethiopia; 2017.
6. The Ethiopian Public Health Institute. HIV Related Estimates andProjections for Ethiopia. 2015.
7. World Health Organization. Guidelines on the Public Health Response to Pretreatment HIV Drug Resistance. 2017.
8. Ministry of Health, Federal Democratic Republic of Ethiopia. National Guidelines for Comprehensive Hiv Prevention, Care and Treatment. 2018.
9. World Health Organization. Consolidated Guidelines on the Use of Antiretroviral Drugs for Treating and Preventing Hiv Infection: Recommendations for a Public Health Approach. 2016.
10. Arnaout RA, Lloyd AL, O’Brien TR, et al. A simple relationship between viral load and survival time in HIV-1 infection. Proc Natl Acad Sci. 1999;96(20):11549–11553. doi:10.1073/pnas.96.20.11549
11. UNAIDS. Global AIDS Updates. 2017.
12. UNAIDS. Global AIDS Update 2018, Miles To Go, Closing Gaps Breaking Barriers, Righting Injustices. 2018.
13. Gill MM, Hoffman HJ, Bobrow EA, et al. Detectable viral load in late pregnancy among women in the Rwanda option B+ PMTCT program: enrollment results from the Kabeho study. PLoS One. 2016;11(12):e0168671. doi:10.1371/journal.pone.0168671
14. Zoufaly A, Jochum J, Hammerl R, et al. Virological failure after 1 year of first-line ART is not associated with HIV minority drug resistance in rural Cameroon. J Antimicrob Chemother. 2015;70(3):922–925. doi:10.1093/jac/dku470
15. Etta EM, Mavhandu L, Manhaeve C, et al. High level of HIV-1 drug resistance mutations in patients with unsuppressed viral loads in rural northern South Africa. AIDS Res Ther. 2017;14(1). doi:10.1186/s12981-017-0161-z.
16. Townsend D, Troya J, Maida I. et al. First HAART in HIV-infected patients with high viral load: value of HIV RNA levels at 12 weeks to predict virologic outcome. J Int Assoc Physicians AIDS Care. 2009;8:314–317.
17. Endalamaw A, Mekonen M, Geremew D, Ambaw F, Tesera H, Habtewold TD. Still poor HAART adherence has great impact on HIV/AIDS treatment failure in Ethiopia. biorxiv. 2018. doi:10.1101/440743.
18. Amani-Bosse C, Dahourou DL, Malateste K, et al. Virological response and resistances over 12 months among HIV-infected children less than two years receiving first-line lopinavir/ritonavir-based antiretroviral therapy in Cote d’Ivoire and Burkina Faso: the MONOD ANRS 12206 cohort. J Int AIDS Soc. 2017;20(1):21362. doi:10.7448/IAS.20.01.21362
19. Mulisa D, Tesfa M, Mullu Kassa G, Tolossa T. Determinants of first line antiretroviral therapy treatment failure among adult patients on ART at central Ethiopia: un-matched case control study. BMC Infect Dis. 2019;19(1):1024. doi:10.1186/s12879-019-4651-6
20. Zoufaly A, Jochum J, Hammerl R, et al. Determinants of HIV-1 drug resistance in treatment-naïve patients and its clinical implications in an antiretroviral treatment program in Cameroon. J Int AIDS Soc. 2014;17:19615. doi:10.7448/IAS.17.4.19615
21. Oromia Regional Health Bureau. Performance Review for HIV Epidemic Control, 2016–2017 Finfinne/Addis Ababa, Ethiopia. 2017.
22. CDC. Epi Info TM 7 User Guide. Vol. 645. 2016.
23. Bayu B, Tariku A, Bulti A, et al. Determinants of virological failure among patients on highly active antiretroviral therapy in University of Gondar referral hospital, Northwest Ethiopia: a case–control study. HIV AIDS (Auckl). 2017;9:153–159. doi:10.2147/HIV.S139516
24. Kwobah M, Mwangi CW, Koech AK, Simiyu JN, M. Siika A. Factors associated with first-line antiretroviral therapy failure amongst HIV-infected african patients: a case-control study. World J AIDS. 2012;02(04):271–278. doi:10.4236/wja.2012.24036
25. Rangarajan S, Colby DJ, Giang LT, et al. Factors associated with HIV viral load suppression on antiretroviral therapy in Vietnam. J Virus Eradication. 2016;2(2):8. doi:10.1016/S2055-6640(20)30466-0
26. Joseph Davey D, Abrahams Z, Feinberg M, et al. Factors associated with recent unsuppressed viral load in HIV-1-infected patients in care on first-line antiretroviral therapy in South Africa. Int J STD AIDS. 2018;29(6):603–610. doi:10.1177/0956462417748859
27. Bulage L, Ssewanyana I, Nankabirwa V, et al. Factors associated with virological non-suppression among HIV-positive patients on antiretroviral therapy in uganda, august 2014–july 2015. BMC Infect Dis. 2017;17(1):326. doi:10.1186/s12879-017-2428-3
28. IBM. IBM SPSS Statistics 20 Brief Guide. Vol. 170. 2011.
29. Zhang Z. Model building strategy for logistic regression: purposeful selection. Ann Transl Med. 2016;4:7.
30. Senaviratna NAMR, Cooray A. Diagnosing multicollinearity of logistic regression model. Asian J Probab Stat. 2019;1–9. doi:10.9734/ajpas/2019/v5i230132.
31. Annual performance report. HIV/AIDS Case Team of Federal Minstry of Health, Ethiopia. 2018.
32. Barlow-Mosha L, Angelidou K, Lindsey J, et al. Nevirapine- versus lopinavir/ritonavir-based antiretroviral therapy in hiv-infected infants and young children: long-term follow-up of the IMPAACT p1060 randomized trial. Clin Infect Dis. 2016;63(8):1113–1121. doi:10.1093/cid/ciw488
33. Crawford K, Wakabi S, Magala F, Kibuuka H, Liu M, Hamm TE. Evaluation of treatment outcomes for patients on first-line regimens in US President’s emergency plan for AIDS relief (PEPFAR) clinics in Uganda: predictors of virological and immunological response from RV288 analyses: predictors of response to first-line ART from RV288 analysis. HIV Med. 2015;16:95–104.
34. Tanuma J, Matsumoto S, Haneuse S, et al. Long-term viral suppression and immune recovery during first-line antiretroviral therapy: a study of an HIV-infected adult cohort in Hanoi, Vietnam. J Int AIDS Soc. 2017;20(4):e25030. doi:10.1002/jia2.25030
35. Jean Louis F, Buteau J, François K, et al. Virologic outcome among patients receiving antiretroviral therapy at five hospitals in Haiti. PLoS One. 2018;13(1):e0192077. doi:10.1371/journal.pone.0192077
36. Cherutich P, Kim AA, Kellogg TA, et al. Detectable HIV viral load in Kenya: data from a population-based survey. PLoS One. 2016;11(5):e0154318. doi:10.1371/journal.pone.0154318
37. Medley A, Garcia-Moreno C, McGill S, Maman S. Rates, barriers and outcomes of HIV serostatus disclosure among women in developing countries: implications for prevention of mother-to-child transmission programmes. Bull World Health Organ. 2004;9.
38. Patel R, Ratner J, Gore-Felton C, et al. HIV disclosure patterns, predictors, and psychosocial correlates among HIV positive women in Zimbabwe. AIDS Care. 2011;24(3):358–368. doi:10.1080/09540121.2011.608786
39. Daskalopoulou M, Lampe FC, Sherr L, For the ASTRA Study Group. Non-disclosure of HIV status and associations with psychological factors, ART non-adherence, and viral load non-suppression among people living with HIV in the UK. AIDS Behav. 2017;21(1):184–195. doi:10.1007/s10461-016-1541-4
40. Krauss B. Guideline on HIV Disclosure Counselling for Children Up to 12 Years of Age. 2011.
41. Arrivé E, Dicko F, Amghar H, et al. HIV status disclosure and retention in care in HIV-infected adolescents on antiretroviral therapy (ART) in West Africa. PLoS One. 2012;7(3):e33690. doi:10.1371/journal.pone.0033690
42. Ahmed M, Merga H, Jarso H. Predictors of virological treatment failure among adult HIV patients on first-line antiretroviral therapy in Woldia and Dessie hospitals, Northeast Ethiopia: a case-control study. BMC Infect Dis. 2019;19(1):305. doi:10.1186/s12879-019-3924-4
