Back to Archived Journals » Research and Reviews in Parkinsonism » Volume 7
Descriptive symptom terminology used by Parkinson’s patients and caregivers
Authors Mursaleen LR, Stamford JA ![]() , Butterfield T, Edwards G, Kustow P
, Butterfield T, Edwards G, Kustow P ![]() , Kustow PA, Griffith S, Dudgeon GMK
, Kustow PA, Griffith S, Dudgeon GMK ![]() , Dudgeon MG
, Dudgeon MG ![]()
Received 28 April 2017
Accepted for publication 28 August 2017
Published 1 November 2017 Volume 2017:7 Pages 71—78
DOI https://doi.org/10.2147/JPRLS.S140697
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Peter Hedera
Leah R Mursaleen,1,2 Jon A Stamford,1,2 Tim Butterfield,2 Gaynor Edwards,2,3 Penny Kustow,2 Paul A Kustow,2 Simon Griffith,2 Gilly M K Dudgeon,2 Mike G Dudgeon2
1The Cure Parkinson’s Trust, London UK, 2Parkinson’s Movement, London, UK, 3Spotlight YOPD, Rye, UK
Background: The ability to accurately describe symptoms is a critical facet of patient–physician interaction and represents both a conduit and a barrier to diagnosis and treatment.
Objective: The objective of this study was to discuss the range and breadth of symptom description to provide insights into symptom complexity, patient interest and understanding, as well as possible communication barriers between the patient and the physician.
Patients and methods: Using a synthesis of information from an online survey of 407 people with Parkinson’s and a focus group of 7 people with Parkinson’s and 3 care partners, we examined the descriptors used by patients and carers to describe a range of motor and nonmotor symptoms in Parkinson’s disease.
Results: We found that patient descriptors were more extensive generally for nonmotor than motor symptoms, and that the terminology used to describe neuropsychiatric symptoms was particularly detailed and extensive.
Conclusion: Since many nonmotor symptoms are not visible and require interrogation by physicians and articulation by patients, these are areas of particular vulnerability in the patient–physician communication loop.
Keywords: Parkinson’s disease, symptoms, communication, cognition, symptom language, symptom terminology, symptom descriptors, neuropsychiatric terms
Introduction
Parkinson’s disease (PD) is clinically diagnosed by the presence of bradykinesia, rigidity, tremor and postural instability.1 These motor symptoms, thus, largely account for its categorization as a movement disorder.
Motor symptoms are only one facet of PD. More recently, the importance of nonmotor symptoms in the symptomatology of PD has been acknowledged. Nonmotor symptoms include (but are not limited to) apathy, depression, anxiety, somnolence, gastrointestinal disturbances, paresthesias, insomnia, constipation, urinary frequency and urgency.2 This change of emphasis is reflected in our current thinking of PD as a complex neuropsychopathologic condition. In many cases, the nonmotor symptoms of PD reflect not only the pathology of PD itself, but also the adverse event profile of the drugs used to treat the condition.3 Irrespective of the source, these components define the PD patient experience.
Although these are not the only factors explaining perceived quality of life,4 there has been much interest over the last several years on the relevance of motor and nonmotor symptoms as determinants of quality of life in people with PD,4–11 with nonmotor symptoms having collectively a greater impact than motor symptoms on the decline of health-related quality of life.5,8–10 Common nonmotor concerns among patients include sleep disturbance,4,7,9 fatigue4,5,11 and neuropsychiatric symptoms such as depression, anxiety and cognition.4–7,9,11 Nonetheless, there remains a lack of clarity on the use of individual nonmotor descriptors.
Since the motor symptoms formed the basis of the diagnostic criteria, they are relatively well understood by physician and patient alike. However, the variety and complexity of many nonmotor symptoms and their sometimes distant relationship to PD, which remains primarily labeled as a movement disorder, probably forms a communication barrier between patient and physician. In this report, we look at the range of terms used spontaneously by patients to describe the motor and nonmotor symptoms and relate this to the complexity of each.
Participants and methods
The results presented here represent a synthesis of information from an online survey of 407 people with PD and a small focus group (see below). The survey data used were part of a larger survey of 492 people with PD, collected between 13 July and 9 September 2015, parts of which have been reported elsewhere.11 The focus group took place on the 12 August 2016 at The Cure Parkinson’s Trust, a UK-based research charity. Completion of the survey and/or participation in the focus group was deemed to be consent from the participants.
The focus group consisted of 10 people, 7 with Parkinson’s and 3 care partners. There were six male and four female participants. The participants with PD, aged between 44 and 77 years, had been diagnosed for 4–21 years and were chosen in order to represent a wide range of experience with the condition. All participants were residents in the southeast of the UK.
The basis of discussion was formed by the results of question 16 of the 2015 Rallying to the Challenge: Outcomes and Measures survey, a free-text question which asked people with PD to list the five symptoms that most affect their quality of life.11 This question, answered by 407 people with PD, generated symptom descriptors which mapped to the following categories as described by Mathur et al:11
- Autonomic
- Bradykinesia
- Dyskinesia
- Fatigue
- Fine motor
- Gastrointestinal
- Neuropsychiatric
- Postural instability and gait dysfunction
- Rigidity
- Sensory
- Sleep
- Speech
- Tremor
These categorized symptom descriptors were then circulated to attendees at the focus group. Each category was discussed and attendees were invited to add any other terms they considered appropriate to describe symptoms within each category. All suggestions were accepted.
Results
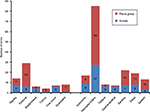
The survey question, answered by 407 people with PD, generated 90 different symptom terms which mapped to 13 categories11 (Tables S1 and S2). Twenty-five different motor symptom descriptors identified by the survey respondents were mapped to six categories (postural instability and gait dysfunction, rigidity, bradykinesia, tremor, dyskinesia, fine motor). Categories such as tremor and dyskinesia represented only single descriptors in each case, that is, the survey respondents only used the words “tremor” and “dyskinesia” to describe these symptoms. Conversely, the category of rigidity comprised seven terms, but mostly “rigidity” (48%) and “stiffness” (32%), as shown in Figure 1 and Table S1.
For the nonmotor symptoms described by the survey respondents, seven categories had 66 descriptors in total, with between 3 and 27 in each category (Figure 1; Table S2). Speech was represented by only three descriptors, most common of which was “speech difficulties” (85%). In contrast, neuropsychiatric symptoms covered 27 different descriptors. “Memory issues” (24%), “anxiety” (19%) and “depression” (15%) were the most prevalent (Table S2).
Figure 1 also shows analogous data for the focus group. The focus group participants added 43 descriptors to the six motor symptom categories, with between 1 and 23 distinct descriptors for each category (Table S1). Only one additional descriptor (picking up change) was added to the fine motor category, whereas 23 new descriptors were assigned to the postural instability and gait dysfunction category, ranging from lay terms such as “tendency to run” to the more clinical such as “festination” (Table S1).
An additional 108 symptom descriptors were added to the seven nonmotor categories, with between 1 and 58 in each case (Figure 1; Table S2). A single descriptor (slow digestion) was mapped to the gastrointestinal symptoms category. By comparison, consideration of neuropsychiatric symptoms by the focus group elicited a further 58 symptom descriptors ranging from single words such as “anger” and “tearfulness” to more detailed terms, for example, “translating thoughts into words” and “inability to learn new things” (Table S2).
Discussion
Motor symptoms provide only a partial picture of PD. Over the last decade, increased focus on the broader symptomatology of PD has led to the recognition of many nonmotor symptoms as important facets of this complex condition. While many of the motor symptoms are readily understood and described, the same cannot be said of the nonmotor symptoms. In some cases, it is unclear even whether these relate to the underlying pathology or the treatment of the condition. These present significant challenges in symptom categorization.
We took as our starting point the published categorization of symptoms by Mathur et al.11 This categorization was conducted by a physician with PD and attempted to synthesize patient descriptions of clinical symptoms. This amounted to 13 categories to cover all patient descriptions. Of the six motor categories, four match the clinical criteria used to diagnose PD.1 The two additional categories (fine motor and dyskinesia), especially in the case of dyskinesia, could be seen as pharmacologically induced rather than explicit symptomatology, but are nonetheless included on the basis that they form part of the patient experience of PD.
Nonmotor symptomatology was broadly subdivided into seven categories encompassing much of the breadth of patient experience which is not always routinely tapped by clinical investigation in the consulting room. The results appear to reflect two findings – firstly, the larger number of terms used to describe nonmotor symptoms and secondly, the strikingly detailed dissection of neuropsychiatric language used by patients and caregivers (Figure 1; Table S2).
The apparently greater attention to nonmotor over motor symptoms is consistent with the current understanding of their pivotal importance in patients’ quality of life.2,5,8–13 The particular focus on the cognitive terminological landscape is interesting. This finding suggests that within the neuropsychiatric landscape, cognitive symptoms appear to be a particular area of concern.
Another possibility is that this may reflect the inadequacy of existing descriptive language. One might assume, for instance, that the better understood symptoms require fewer descriptors. For example, “tremor” is commonplace and clearly well defined. In the survey, all respondents (n=112) used the word tremor, with the focus group adding only a further three terms. Conversely, issues with balance, posture and gait, mapped as “postural instability and gait dysfunction”, attracted more discussion in the focus group. This is especially the case for the “neuropsychiatric” category, which prompted much discussion and description (Figure 1).
Cognition and the broader academic field of neuropsychiatry are areas replete with circumlocution and neologism. It is no surprise that the vocabulary used by people with PD is of a more circuitous nature and generally descriptive rather than didactic. Whereas “tremor” appears to be universally accepted and understood, neuropsychiatry encompasses a cornucopia of different symptoms and descriptors (Table S2). It is perhaps of interest in this context that the Movement Disorder Society - Unified Parkinson’s Disease Rating Scale has five questions on neuropsychiatric aspects of PD out of a total of 50 questions.14 The data from this study appear to suggest a mismatch between clinical and patient description and/or understanding.
Of course, any categorization has its limitations and some symptom descriptors might fit legitimately into more than one category. For example, olfactory hallucinations could be seen as sensory or neuropsychiatric and drooling could be seen as autonomic or gastrointestinal. It is striking, for instance, that the Non-Motor Symptom assessment scale for Parkinson’s Disease questionnaire focuses strongly on sleep, gastrointestinal function, autonomic and cognitive aspects of PD,15 but does not explicitly address fatigue or speech, areas highlighted by this study.
Irrespective of the restrictions of categorization, the results, nonetheless, highlight the greater concentration of patient minds on nonmotor symptomatology. Since many of these symptoms are not visible and require interrogation by physicians and articulation by patients, these are areas of particular vulnerability in the communication loop.
Acknowledgments
The authors thank Helen Matthews and Tom Isaacs for their support and contribution in designing, organizing and facilitating the focus group. The authors confirm that no financial or material support was received for this study.
Disclosure
The authors report no conflicts of interest in this work.
References
Jankovic J. Parkinson’s disease: clinical features and diagnosis. J Neurol Neurosurg Psychiatry. 2008;79(4):368–376. | ||
Chaudhuri KR, Tolosa E, Schapira AH, Poewe W. Non-Motor Symptoms of Parkinson’s Disease. 2nd ed. Oxford: Oxford University Press; 2014. | ||
MacMahon D, McIntosh S. Managing the major non-motor symptoms of Parkinson’s disease. Geriatr Med. 2007;43–49. | ||
Qin Z, Zhang L, Sun F; Chinese Parkinson Study Group, et al. Health related quality of life in early Parkinson’s disease: impact of motor and non-motor symptoms, results from Chinese levodopa exposed cohort. Parkinsonism Relat Disord. 2009;15(10):767–771. | ||
Rahman S, Griffin HJ, Quinn NP, Jahanshahi M. Quality of life in Parkinson’s disease: the relative importance of the symptoms. Mov Disord. 2008;23(10):1428–1434. | ||
Gomez-Esteban JC, Zarranz JJ, Lezcano E, et al. Influence of motor symptoms upon the quality of life of patients with Parkinson’s disease. Eur Neurol. 2007;57(3):161–165. | ||
Duncan GW, Khoo TK, YarnallAJ, et al. Health-related quality of life in early Parkinson’s disease: the impact of nonmotor symptoms. Mov Disord. 2014;29(2):195–202. | ||
Santos-García D, de la Fuente-Fernández R. Impact of non-motor symptoms on health-related and perceived quality of life in Parkinson’s disease. J Neurol Sci. 2013;332(1):136–140. | ||
Antonini A, Barone P, Marconi R, et al. The progression of non-motor symptoms in Parkinson’s disease and their contribution to motor disability and quality of life. J Neurol. 2012;259(12):2621–2631. | ||
Martinez-Martin P, Rodriguez-Blazquez C, Kurtis MM, Chaudhuri KR; NMSS Validation Group. The impact of non-motor symptoms on health-related quality of life of patients with Parkinson’s disease. Mov Disord. 2011;26(3):399–406. | ||
Mathur S, Mursaleen L, Stamford J, DeWitte S, Robledo I, Isaacs T. Challenges of improving patient-centered care in Parkinson’s disease. J Parkinsons Dis. 2017;7(1):163–174. | ||
Shearer J, Green C, Counsell CE, Zajicek JP. The impact of motor and non motor symptoms on health state values in newly diagnosed idiopathic Parkinson’s disease. J Neurol. 2012;259(3):462–468. | ||
Stamford J. Non-motor symptoms of Parkinson’s in the spotlight. Br J Neurosci Nurs. 2012;8(5):266. | ||
Goetz CG, Tilley BC, Shaftman SR, et al. Movement disorder society-sponsored revision of the unified Parkinson’s disease rating scale (MDS-UPDRS): Scale presentation and clinimetric testing results. Mov Disord. 2008;23(15):2129–2170 | ||
Chaudhuri KR, Martinez-Martin P, Schapira AH, et al. International multicenter pilot study of the first comprehensive self-completed nonmotor symptoms questionnaire for Parkinson’s disease: the NMSQuest Study. Mov Disord. 2006;21(7):916–923. |
Supplementary materials
  | Table S1 Motor symptoms |
  | Table S2 Nonmotor symptoms Abbreviations: REM, rapid eye movement sleep behavior disorder; OCD, obsessive compulsive disorder. |
 © 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2017 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms
and incorporate the Creative Commons Attribution
- Non Commercial (unported, 3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.