Back to Journals » OncoTargets and Therapy » Volume 9
Dendrobium candidum inhibits MCF-7 cells proliferation by inducing cell cycle arrest at G2/M phase and regulating key biomarkers
Authors Sun J, Guo Y, Fu X, Wang Y, Liu Y, Huo B, Sheng J, Hu X
Received 29 July 2015
Accepted for publication 22 September 2015
Published 21 December 2015 Volume 2016:9 Pages 21—30
DOI https://doi.org/10.2147/OTT.S93305
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Faris Farassati
Jing Sun,1 Yidi Guo,1 Xueqi Fu,1–3 Yongsen Wang,1 Ye Liu,1 Bo Huo,1 Jun Sheng,4 Xin Hu1–3
1School of Life Sciences, 2Key Laboratory for Molecular Enzymology and Engineering of Ministry of Education, 3National Engineering Laboratory of AIDS Vaccine, School of Life Sciences, Jilin University, Changchun, 4Yunnan Research Centre for Advance Tea Processing, Yunnan Agricultural University, Kunming, People’s Republic of China
Background: Breast cancer is one of the most frequently occurring cancers in women. In recent years, Dendrobium candidum has played a part in antihyperthyroidism and anticancer drugs. This study aims to examine the antitumor effect of D. candidum on breast cancer.
Methods: Human breast cancer cell line MCF-7 and normal breast epithelial cell line MCF10A were used to observe the effects of D. candidum treatment on human breast cancer. 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay was employed to examine the cell proliferation of the MCF-7 and MCF10A cells. Western blot analysis and reverse transcription polymerase chain reaction were used to detect the key molecules and biomarkers in breast cancer pathology. Cell cycle was analyzed by using Becton Dickinson FACScan cytofluorometer.
Results: The results indicated that D. candidum significantly decreased cell viability at different concentrations compared to the control group (P<0.05). D. candidum-treated MCF-7 cells in the G2/M phase was significantly increased compared to the control group (P<0.05). The messenger RNA levels of estrogen receptor alpha, IGFBP2, IGFBP4, and GATA3 were significantly decreased, and the messenger RNA and protein levels of ELF5, p53, p21, p18, CDH1, CDH2, and p12 were significantly increased, compared to the control group (P<0.05). The protein levels of estrogen receptor alpha, PGR, GATA3, and Ki67 were significantly decreased and the protein levels of p53 and ELF5 were significantly increased compared to the control group (P<0.05). The general apoptosis biomarker, Bcl-2, was significantly decreased and the Bax was significantly increased compared to the control group (P<0.05). In contrast to that in MCF-7, D. candidum does not affect cell proliferation at any concentration and any time points in normal breast epithelial cells, MCF10A cells.
Conclusion: D. candidum could decrease the cell viability of MCF-7 cells by inducing cell cycle arrest at the G2/M phase and regulating the key biomarkers in breast cancer cells.
Keywords: breast cancer, D. candidum, proliferation, biomarker, inhibition
Introduction
Dendrobium candidum is one of the most medicine-valuable orchids, mainly distributes in Southeast and South Asia, such as the People’s Republic of China, Japan, etc.1,2 There are many functions for D. candidum, including 1) the dried stems of D. candidum can strengthen the stomach activity in traditional Chinese medicine;3 2) D. candidum also plays an important role in preventing cataract development, relieving throat inflammation, and fatigue; 3) could reduce peripheral vascular obstruction and improve immunity; and 4) in recent years, D. candidum has played a part in antihyperthyroidism and anticancer drugs.4 Therefore, as an important traditional Chinese medical herb, it has been with a higher clinical value and potential application.4
Breast cancer is one of the most frequently occurring cancer in women worldwide.5 Approximately 14.2% (in the People’s Republic of China) and 26.4% malignant tumor patients (in the USA) are diagnosed with breast cancer annually.6 Recently, scientists have explored many differential gene expressions to advance individualized treatment or to assist in the cancer prognosis,7 such as the estrogen receptor (ER), progesterone receptor,8 and human epidermal growth factor-2.9 Meanwhile, many drugs that target these molecules have been designed.
Until now, many evolutionarily conserved key molecules have been discovered, which are involved in a variety of cellular processes including tumor suppression.10 These molecules could induce tumor suppression or inhibition via enhancing cell cycle arrest, repairing damaged DNA, and causing apoptosis by regulating key gene expression.11,12 Previous reports show that the therapeutic drugs interfere with DNA replication and in a further step can affect the development of tumor cells via the cell cycle. The G0, G1, and sub-G1 phase of cell cycle arrest could always induce the cell apoptosis and inhibit the proliferation of tumor cells.13
The present study evaluated the antitumor effect of D. candidum on breast cancer. The key molecules involved in cell proliferation of breast cancer cells, including ERα, PGR, GATA3, p53, Ki67, and ELF5, were examined to evaluate and analyze the antitumor effects of D. candidum on the MCF-7 breast cancer cell line.
Materials and methods
Materials and chemicals
D. candidum were generously donated by Prof Jun Sheng (Yunnan Research Centre for Advance Tea Processing, Yunnan Agricultural University, Kunming, People’s Republic of China). Polyvinylidene fluoride membranes were purchased from EMD Millipore (Billerica, MA, USA). The necessary apparatus for sodium dodecyl sulfate polyacrylamide gel electrophoresis and Western blot were bought from Bio-Rad.
Cells culture
The human breast cancer cell line MCF-7 and the human breast epithelial cell line MCF10A were generously donated by Prof Xin Hu (Jilin University, Changchun, People’s Republic of China) and cultured as described in a previous article.14 No ethics statement was required from the institutional review board for the use of these cell lines. Briefly, MCF-7 cells were cultured in Dulbecco’s Modified Eagle’s Medium (DMEM, Gibco, Carlsbad, CA, USA) supplemented with 10% fetal bovine serum, 100 IU/mL penicillin/streptomycin. MCF10A cells were cultured in Ham’s F12 medium and DMEM with 2.5 mM L-glutamine (DMEM:F12, Gibco), 5% horse serum (Gibco), 20 ng/mL epidermal growth factor (PeproTech, Rocky Hill, NJ, USA), 100 ng/mL cholera toxin (Sigma-Aldrich Co., St Louis, MO, USA), 10 μg/mL insulin (Sigma-Aldrich), 0.5 mg/mL hydrocortisone (Sigma-Aldrich), and 100 IU/mL penicillin/streptomycin. Cells were maintained at 37°C and 5% CO2 in a humidified incubator.
MTT assay
Cells were seeded in a 96-well plate to a final concentration of 5,000 cells/well and incubated in growth media with varying concentrations of D. candidum for 12, 24, 48, and 72 hours. Medium was removed and fresh medium was added to each well along with 10 mL of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) solution (5 mg/mL). After 4 hours of incubation at 37°C, the medium was drained and replaced by 150 mL of dimethyl sulfoxide. The plates were read at wavelength of 490 nm using a microplate reader (BioTek, Winooski, VT, USA). Six reduplicate wells were used for each treatment, and experiments were repeated three times.
Western blot analysis
Cells were treated with 2 mg/mL D. candidum for 48 and 72 hours. Western blot was performed as previously described.15 The cells were collected in ice-cold phosphate-buffered saline and lysed in ice-cold whole cell extraction buffer containing 25 mM β-glycerophosphate (pH 7.3), 5 mM ethylenediaminetetraacetic acid, 2 mM ethylene glycol tetraacetic acid, 5 mM β-mercaptoethanol, 1% Triton X-100, 0.1 M NaCl, and a protease inhibitor mixture (Roche Applied Science, Penzberg, Germany). The protein concentration cell lysates were determined by using the Bradford method and boiled in sodium dodecyl sulfate (SDS) sample buffer (50 mM Tris, pH 6.8; 100 mM dithiothreitol; 2% SDS; 0.1% bromophenol blue; and 10% glycerol). The proteins were separated on 8%–10% SDS polyacrylamide gel and electrotransferred to polyvinylidene fluoride membrane. After blocking with 3% bovine serum albumin-Tris-Hcl buffer saline Tween, primary antibodies (PGR, MILLIPORE 2437345; ELF5, Abcam ab136119; GATA3, Abcam ab11693; ERα, Santa Cruz Biotechnology sc-542; p53, Abcam ab26; Ki67, Abcam ab15580) were detected using horseradish peroxidase-conjugated antirabbit antibodies and visualized on Tanon-5200 Chemiluminescent Imaging System (Tanon Science & Technology, Shanghai, People’s Republic of China). β-Actin (ZSGB-BIO TA-09, Beijing, People’s Republic of China) was also tested to confirm equal loading.
Quantitative reverse transcription polymerase chain reaction
Cells were treated with 2 mg/mL D. candidum for 48 hours. RNA isolation and quantitative reverse transcription polymerase chain reaction (qRT-PCR) assays were performed as described in previous articles.14,16 Total RNA from cells was extracted with TRIzol reagent (Invitrogen, Waltham, MA, USA) following the manufacturer’s instruction, and complementary DNA was synthesized using Moloney murine leukemia virus reverse transcriptase with random primers. Complementary DNA was generated with BioTeke super RT kit (BioTek) according to the manufacturer’s protocol. qRT-PCR was performed using SYBR Premix Ex Taq™ (TaKaRa, Dalian, People’s Republic of China). Primers are listed in Table 1.
  | Table 1 RT-PCR primer sequences (all sequences from 5′ to 3′) |
Cell cycle analysis
Cells were treated with graded concentrations of D. candidum (0–2 mg/mL) for 48 hours. Cell cycle distribution was evaluated using cell cycle detection kit (BestBio, People’s Republic of China) following the manufacturer’s instruction. Briefly, the cells were harvested, washed twice with phosphate-buffered saline, and fixed at 4°C for 1 hour with 70% ethanol, and then stained with a propidium iodide solution (containing RNase) at 4°C for 30 minutes. At least 20,000 cells were analyzed by Becton Dickinson FACScan cytofluorometer (Mansfield, MA, USA). Cell cycle distribution was calculated using ModFIT cell cycle analysis software (version 2.01.2; Becton Dickinson).
Statistical analysis
The quantitative analysis of the Western blot images was performed by using the computer-assisted software Image Total Tech (2.0 version). In brief, the images of Western blot were scanned with the Typhoon and digitalized, and saved in JPEG format. The values of the Western blot bands were calculated. Data were presented as mean ± SD of at least three independent experiments. Statistical analysis of data was performed by Student’s t-test for two-group comparison. P<0.05 represents the statistically significant difference.
Results
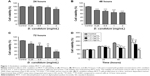
D. candidum decreases the cell viability of MCF-7 cells
In order to observe the antitumor proliferation effects of D. candidum, MTT assay was used to examine the cell viability of the MCF-7 cells. First, the median lethal dose value of D. candidum was calculated; the preliminary study showed that the median lethal dose of D. candidum is 2 mg/mL (data not shown). Second, the appropriate concentration of D. candidum I was investigated. The MTT assay results indicated that the treatment of D. candidum could significantly decrease the cell viability at different concentrations (0.25, 0.5, 1, and 2 mg/mL) compared to the control group at 24, 48, and 72 hours, respectively (Figure 1A–C, P<0.05). The cell viability was also decreased, followed by the increased concentration (range from 0.25 to 2 mg/mL) of D. candidum on different time points. However, when the concentration of D. candidum was higher than 2 mg/mL (4 and 8 mg/mL), the cell viability was increased slightly compared to the concentration of 2 mg/mL (data not shown). Therefore, in the following experiments, 2 mg/mL was used as the concentration of D. candidum.
Meanwhile, Figure 1D also showed that the cell viability significantly increased, followed by the increased treatment time of D. candidum, compared to the control group at 12 hours (from 24 to 72 hours) (Figure 1D, P<0.05). This result suggests that D. candidum illustrated the optimized antiproliferation effect at 48 hours treatment at all of the D. candidum concentrations.
D. candidum triggers MCF-7 cell cycle arrest at G2/M phase
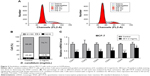
The outcomes of cell cycle assay showed that the percentage of the D. candidum-treated MCF-7 cells in the G2/M phase was significantly increased compared to the control group (Figure 2A and B, P<0.05). Also, the percentage of the MCF-7 cells in G2/G0 phase in D. candidum-treated group increased, and S phase decreased compared to the control group (Figure 2B).
Furthermore, the cell cycle arrest-related CDK family proteins transcription has also been examined by using the qRT-PCR assay. The results indicated that the treatment of D. candidum significantly decreased the transcription of CDK family members compared to the control group (Figure 2C, P<0.05).
D. candidum regulates the key molecules involving in the MCF-7 proliferation
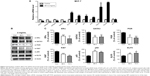
To clarify the specific mechanism of the antitumor proliferation effects of D. candidum, the key molecules involved in MCF-7 proliferation were examined. First, we examined the apoptosis- or proliferation-related molecules by using the qRT-PCR assay. The results showed that the messenger RNA (mRNA) levels of ERα, IGPBP2, IGFBP4, and GATA3 were significantly reduced, and the mRNA levels of ELF5, p53, p21, p18, CDH1, CDH2, and p12 were significantly increased compared to the control group (Figure 3A, P<0.05). From the mRNA detection results, we found that apoptosis biomarkers (ELF5, p21, CDH1, CDH2, and p12) were increased; therefore, we also examined the protein levels of these biomarkers. The result also confirmed the similar changes of the ELF5, p21, CDH1, CDH2, and p12 protein as the mRNA levels (Figure S1).
Furthermore, the general biomarkers for apoptosis, such as Bcl-2 and Bax, were was also detected. The results indicated that Bcl-2 was significantly decreased and Bax was significantly increased compared to the control group (Figure S2).
Second, the breast cancer biomarkers (ERα, PGR, and GATA3) and oncogenes (p53, Ki67, and ELF5) were also examined by using the Western blot assay. The results indicated that the protein levels of ERα, PGR, GATA3, and Ki67 were significantly decreased and the protein levels of p53 and ELF5 were significantly increased compared to the control group (Figure 3B and C, P<0.05).
D. candidum did not affect the normal breast epithelial MCF10A cell growth
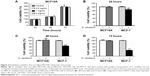
Does D. candidum affect the normal breast epithelial cell lines proliferation? MCF10A cells were used to evaluate the effects of D. candidum on normal breast cell lines. In contrast to the effects in cancer cells exposed to various concentrations, the MCF10A cells showed no differences in cell proliferation at the concentration of 0.5, 1, and 2 mg/mL (Figure 4A, P>0.05). Meanwhile, D. candidum (at the concentration of 2 mg/mL) also did not affect cells proliferation in MCF10A cells at 24 hours (Figure 4B), 48 hours (Figure 4C), and 72 hours (Figure 4D) compared to that in MCF-7 cells (P>0.05).
Discussion
Previous studies reported that D. candidum could be served as a kind of promising functional food or medicine, but little scientific findings about its therapeutic effects were explored.17,18 There are many important components in D. candidum, including benzenes and their derivatives (with high concentration), lignans, flavonoids, phenolic, and lactone; meanwhile, 18 kinds of new pigments were also discovered in D. candidum.19 Recently, many studies have reported that D. candidum has some potential therapeutic functions on various pathologic forms, including hyperglycemic, immunity, cancer, and inflammation.20 Bao et al21 found that D. candidum could inhibit the HepG2 liver cancer cell proliferation in vitro and preliminarily proved the anticancer effects of D. candidum. Although the studies have showed the therapeutic effects of D. candidum, the specific mechanisms of the anticancer effects have not been fully clarified.
The cell proliferation is the most important characteristic of the tumor cells and could be indicated by the cell viability. Therefore, in this study, we observed the cell viability in the control and D. candidum-treated cells, and explored the antiproliferation effects of D. candidum in vitro. The results indicated that the treatment of D. candidum could significantly decrease the cell viability at different concentrations (0.25, 0.5, 1, and 2 mg/mL) compared to the control group at 24, 48, and 72 hours, respectively (Figure 1A–C, P<0.05). Tokgun et al22 proved that the extracts of D. candidum have cytotoxic effects on human breast carcinoma cell line MCF-7 cells. Zhao et al17 found that D. candidum could inhibit the proliferation of the HCT-116 cells. Our result is consistent with these two studies.
In this study, we demonstrated that D. candidum inhibits the MCF-7 cell viability by enhancing the cell cycle arrest in the G2/M phase. Ben Sahra et al23 proved that the metformin could inhibit the proliferation of prostate cancer cells by blocking the cell cycle in the G2/M phase and decreasing the cell growth. Achari et al24 also reported that microRNA-34c affects the cell cycle mainly by inducing an arrest in the G2/M phase and frequently triggers the inhibitory effects of the breast cancer cell proliferation. Together with the above two studies, our study also showed that the induction of the G2/M cell cycle arrest in cancer cells could inhibit the cancer cells’ proliferation. Therefore, we explored the key molecules that regulate the processes of the breast cancer cell cycle arrest.
We examined the mRNA levels of the tumor growth-associated biomarkers, including ERα, IGPBP2, IGFBP4, GATA3, and the mRNA levels of the apoptosis-associated biomarkers, including ELF5, p53, p21, p18, CDH1, CDH2, and p12. The results showed that all of the tumor growth biomarkers (ERα, IGPBP2, IGFBP4, and GATA3) were significantly decreased or inhibited when treated with D. candidum. Also, all of the apoptosis biomarkers (ELF5, p53, p21, p18, CDH1, CDH2, and p12) were significantly increased. We also detected the protein levels and found that the breast cancer markers (ERα, PGR, and GATA3) were significantly decreased. While the oncogene Ki67 was significantly decreased, the oncogenes p53 and ELF5 were significantly increased. Grawenda et al25 reported that p53 mutation could represent the prognosis and harbor potential utility as a clinical tool. Si et al26 found that the expression of GATA3 is concurrently downregulated during breast cancer progression. Vernet-Tomas et al27 proved that the expression of p53 in breast cancer patients could predict a tumor with low probability of non-sentinel nodes infiltration. Fernandes et al28 also reported that PGR, ESR1, human epidermal growth factor-2, and Ki67 could act as the prognostic biomarker in different molecular subtypes of breast cancer. Comparing the above studies,25–28 we systematically analyzed the key molecules or biomarker, which could provide the targeting pathway for the therapy in clinical.
In addition, the normal breast epithelial cell line MCF10A was also used as a control to examine the cancer specificity of D. candidum. Treatment with D. candidum at any concentration and any time points caused no inhibitory effect on cell proliferation, suggesting that D. candidum selectivity inhibits MCF-7 breast cancer cell proliferation.
In conclusion, D. candidum could selectivity inhibit the breast cancer cell (MCF-7) proliferation by inducing cell cycle arrest at the G2/M phase and regulating the key biomarkers in breast cancer cells. Meanwhile, D. candidum does not affect the normal breast epithelial cells (MCF10A) growth.
Acknowledgments
Jun Sheng and Xin Hu contributed equally to this work.
This work was supported by grants obtained from the China Postdoctoral Science Foundation (Grant numbers 2012T50290 and 20100481059).
Disclosure
The authors report no conflicts of interest in this work.
References
Zhao P, Wu F, Feng FS, Wang WJ. Protocorm-like body (PLB) formation and plant regeneration from the callus culture of Dendrobium candidum Wall ex Lindl. In vitro Cell Dev Biol-Plant. 2008;44:178–185. | ||
Jones WE, Kuehnle AR, Arumuganathan K. Nuclear DNA content of 26 orchid (Orchidaceae) genera with emphasis on Dendrobium. Ann Bot. 1998;82:189–194. | ||
Shao H, Zhang LQ, Li JM, Wei RC. Advances in research of Dendrobium officinale. Chin Tradit Herbal Drugs. 2004;35:109–112. | ||
Shao BM, Xu W, Dai H, Tu P, Li Z, Gao XM. A study on the immune receptors for polysaccharides from the roots of Astragalus membranaceus, a Chinese medicinal herb. Biochem Biophys Res Commun. 2004;320:1103–1111. | ||
Alghamdi IG, Hussain II, Alghamdi MS, El-Sheemy MA. The power of the age standardized incidence rate to discover the gene link between cancer diseases: development of a new epidemiological method to save money, time and effort for genetic scientists. Onco Target Ther. 2015;8:677–687. | ||
Wang YC, Wei LJ, Liu JT, Li SX, Wang QS. Comparison of cancer incidence between China and the USA. Cancer Biol Med. 2012;9(2):128–132. | ||
Pathmanathan N, Balleine RL. Ki67 and proliferation in breast cancer. J Clin Pathol. 2013;66:512–516. | ||
Mohsin SK, Weiss H, Havighurst T, et al. Progesterone receptor by immunohistochemistry and clinical outcome in breast cancer: a validation study. Mod Pathol. 2004;17:1545–1554. | ||
Hurrell T, Outhoff K. Human epidermal growth factor receptor 2-positive breast cancer, which cytotoxic agent best complements trastuzumab’s efficacy in vitro? Onco Targets Ther. 2013;6:693–701. | ||
Chiacchiera F, Simone C. Inhibition of p38 alpha unveils an AMPK-FoxO3A axis linking autophagy to cancer-specific metabolism. Autophagy. 2009;5:1030–1033. | ||
Brunet A, Bonni A, Zigmond MJ, et al. Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell. 1999;96(6):857–868. | ||
Dijkers PF, Medemadagger RH, Lammers JJ, Koenderman L, Coffer PJ. Expression of the pro-apoptotic Bcl-2 family member Bim is regulated by the forkhead transcription factor FKHR-L1. Curr Biol. 2000;10(19):1201–1204. | ||
Malumbres M, Barbacid M. Cell cycle, CDKs and cancer: a changing paradigm. Nat Rev Cancer. 2009;9:153–166. | ||
Lin T, Ponn A, Hu X, Law BK, Lu J. Requirement of the histone demethylase LSD1 in Snai1-mediated transcriptional repression during epithelial-mesenchymal transition. Oncogene. 2010;29:4896–4904. | ||
Guo YD, Fu X, Jin Y, et al. Histone demethylase LSD1-mediated repression of GATA-2 is critical for erythroid differentiation. Drug Des Devel Ther. 2015;9:3153–3162. | ||
van Bragt MP, Hu X, Xie Y, Li Z. RUNX1, a transcription factor mutated in breast cancer, controls the fate of ER-positive mammary luminal cells. Elife. 2014;3:e03881. | ||
Zhao X, Sun P, Qian Y, Suo H. D. candidum has in vitro anticancer effects in HCT-116 cancer cells and exerts in vivo anti-metastatic effects in mice. Nutr Res Pract. 2014;8:487–493. | ||
Wang Q, Sun P, Li G, Zhu K, Wang C, Zhao X. Inhibitory effects of Dendrobium candidum Wall ex Lindl. on azoxymethane- and dextran sulfate sodium-induced colon carcinogenesis in C57BL/6 mice. Oncol Lett. 2013;7:493–498. | ||
Li Y, Wang C, Wang F, et al. Chemical constituents of Dendrobium candidum. Zhongguo Zhong Yao Za Zhi. 2010;35:1715–1719. | ||
Li J, Li S, Huang D, Zhao X, Cai G. Advances in the of resources, constituents and pharmacological effect of Dendrobium officinale. Ke Ji Dao Bao. 2011;29:74–79. | ||
Bao LJ, Wang JH, Luo JP. Inhibitory effects of water extracts from four species of Dendrobiums on HelaS3 cells and HepG2 cell. J Anhui Agric Sci. 2008;36:15968–15970. | ||
Tokgun O, Akca H, Mammadov R, Aykurt C, Deniz G. Convolvulus galaticus, Crocus antalyensis, and Lilium candidum extracts show their antitumor activity through induction of p53-mediated apoptosis on human breast cancer cell line MCF-7 cells. J Med Food. 2012;15:1000–1005. | ||
Ben Sahra I, Laurent K, Loubat A, et al. The antidiabetic drug metformin exerts an antitumoral effect in vitro and in vivo through a decrease of cyclin D1 level. Oncogene. 2008;27:3576–3586. | ||
Achari C, Winslow S, Ceder Y, Larsson C. Expression of miR-34c induces G2/M cell cycle arrest in breast cancer cells. BMC Cancer. 2014;14:538. | ||
Grawenda AM, Moller EK, Lam S, et al. Interaction between p53 mutation and a somatic HDMX biomarker better defines metastatic potential in breast cancer. Cancer Res. 2015;75:698–708. | ||
Si W, Huang W, Zheng Y, et al. Dysfunction of the reciprocal feedback loop between GATA3 and ZEB2 induced nucleated repression programs contributes to breast cancer metastasis. Cancer Cell. 2015;27:822–836. | ||
Vernet-Tomas M, Banos N, Sabadell D, et al. p53 expression in breast cancer predicts tumor with low probability of non-sentinel nodes infiltration. J Obstet Gynaecol Res. 2015;41:1115–1121. | ||
Fernandes A, Bianchi G, Feltri AP, Perez M, Correnti M. Presence of human papillomavirus in breast cancer and its association with prognostic factors. Ecancermedicalscience. 2015;9:548. |
Supplementary materials
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.