Back to Journals » Clinical Ophthalmology » Volume 9
Delayed manifestation of bilateral scleral thinning after I-BRITE® procedure and review of literature for cosmetic eye-whitening procedures
Authors Moshirfar M, McCaughey M, Fenzl C, Santiago-Caban L, Kramer G, Mamalis N
Received 30 November 2014
Accepted for publication 15 January 2015
Published 4 March 2015 Volume 2015:9 Pages 445—451
DOI https://doi.org/10.2147/OPTH.S78390
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Majid Moshirfar,1 Michael V McCaughey,2 Carlton R Fenzl,3 Luis Santiago-Caban,4 Gregory D Kramer,3 Nick Mamalis3
1Department of Ophthalmology, Francis I Proctor Foundation, University of California San Francisco, San Francisco, CA, USA; 2University of New Mexico School of Medicine, Albuquerque, NM, USA; 3John A Moran Eye Center, University of Utah, Salt Lake City, UT, USA; 4Department of Ophthalmology, University of Puerto Rico School of Medicine, San Juan, PR, USA
Purpose: To report a case of delayed-onset bilateral scleral thinning and calcium deposition following a cosmetic ocular-whitening procedure (I-BRITE®).
Methods: A 33-year-old male patient with a history of right-sided ptosis repair and left-sided anterior uveitis had previously undergone bilateral I-BRITE treatment for chronic conjunctival hyperemia. Four years after the procedure, the patient was referred to our institution with bilateral scleral thinning and overlying calcific depositions. A literature review was performed through PubMed from 1980 through 2014 using the search terms ‘cosmetic’, ‘ocular’, ‘conjunctivectomy’, ‘regional conjunctivectomy’, ‘I-BRITE’, ‘eye-whitening’, ‘scleritis’, ‘necrotizing scleritis’, ‘anterior uveitis’, ‘mitomycin C’, ‘5-fluorouracil’, and ‘bevacizumab’, along with associated cross-referencing from relevant articles.
Results: Examination of the patient revealed bilateral necrotizing scleritis within the nasal region of both eyes. Calcified plaques were also present within the areas of scleromalacia, along with epithelial defects demonstrated with fluorescein staining. Although evidence of previous intraocular inflammation was apparent within the left eye, there were no active signs of inflammation evident within either eye on initial presentation. Complication rates reported in the literature include: scleral thinning (1.8%), calcific plaque formation (2.9%), fibrovascular proliferation (13%), diplopia (1.2%), elevation of intraocular pressure (4.2%), and recurrence of conjunctival hyperemia (2.1%).
Conclusion: Cosmetic ocular whitening procedures have an attendant high complication rate, and have been associated with several adverse postoperative complications, which have in turn generated several reservations regarding the veritable benefit of the procedure. Many postsurgical complications may demonstrate delayed apparition, varying from several months to several years after primary surgical intervention as in the case reported here.
Keywords: necrotizing scleritis, scleromalacia, cosmetic ocular whitening
Corrigendum for this paper has been published
Introduction
Cosmetic eye-whitening procedures for the treatment of chronic conjunctival hyperemia have recently become a topic of interest due to accumulating evidence of severe postoperative complications associated with their practice.1,2 A large proportion of patients have undergone the so-called regional conjunctivectomy procedure in South Korea, along with many patients in the US who have undergone a similar procedure known as I-BRITE® (Boxer Wachler Vision Institute, Beverly Hills, CA, USA). Each respective procedure entails resection involving significant amounts of bulbar conjunctiva with or without resection of Tenon’s capsule components within the nasal and temporal regions of the palpebral fissure, with concomitant intraoperative and postoperative mitomycin C (MMC) administration topically, subconjunctivally, or both.1 There are a multitude of worrisome postoperative complications that have been reported in association with these procedures, including, but not limited to: scleral thinning with or without calcified plaques, diplopia induced by fibrovascular growth, necrotizing scleritis, and elevated intraocular pressure (IOP).1,2 Herein, we report a case of bilateral scleral thinning with associated calcified plaque formation in a patient who had undergone a cosmetic eye-whitening procedure 4 years prior to presentation.
Methods
A chart review of a single patient who received a cosmetic eye-whitening procedure at an outside institution was performed. Medical records detailing preoperative, intraoperative, and postoperative findings were obtained from outside providers. Information including, but not limited to, preoperative signs and symptoms, intraoperative procedure, patient demographics, visual acuity, medication regimen, complications, and postoperative complication–related therapy were obtained through the record review.
A thorough examination was performed, including quantification of best corrected and uncorrected visual acuities, measurement of IOP, and slit-lamp and dilated fundus examinations, and slit-lamp photography. Histopathologic analysis was also performed.
A literature review was performed through PubMed from 1980 through 2014 using the search terms ‘cosmetic’, ‘ocular’, ‘conjunctivectomy’, ‘regional conjunctivectomy’, ‘I-BRITE’, ‘eye-whitening’, ‘scleritis’, ‘necrotizing scleritis’, ‘anterior uveitis’, ‘mitomycin C’, ‘5-fluorouracil’, and ‘bevacizumab’, along with associated cross-referencing from relevant articles. As a result of various inter-investigational discrepancies associated with reported complications, aggregation of patients (rather than total eye quantity) was used for quantitative analysis.
Case report
A 33-year-old male with a previous history of chronic conjunctival hyperemia and past surgical history of right-sided congenital ptosis repair at age 5 years and bilateral cosmetic eye-whitening procedure was referred to our institution for evaluation and management of bilateral scleral thinning. During the patient’s previously performed cosmetic procedure, subsequent to conjunctival excision, unilateral cauterization of the left eye was required to achieve hemostasis. Intraoperative 0.02% MMC was also administered topically to the nasal conjunctivae of each eye for a duration of 1 minute. This was then followed by injection of 5-fluorouracil and bevacizumab into the remaining conjunctiva. Postoperative topical 0.02% MMC was additionally prescribed four times per day for 7 days.
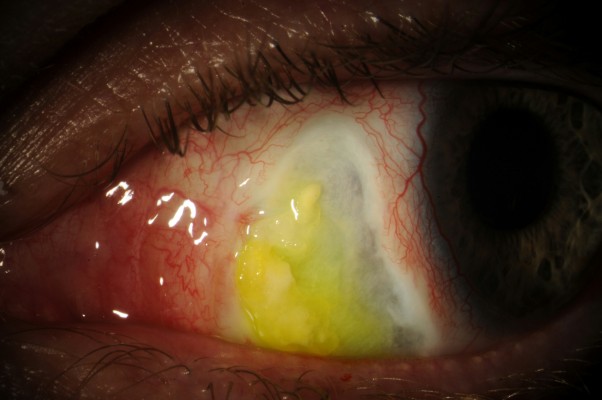
Three weeks prior to presentation at our institution, the patient was treated with topical moxifloxacin for an episode of conjunctivitis within the left eye. During a follow-up visit, the patient developed anterior uveitis for which topical steroids were prescribed; despite this treatment, the inflammation did not resolve. At this point, the patient was then referred to our quaternary care facilities for further evaluation. On initial presentation, the patient did not have any associated pain or other ocular symptoms, and demonstrated uncorrected visual acuity of 20/30 in the right eye and 20/70 in the left eye; tonometric evaluation was not performed. Slit-lamp examination revealed bilateral nasal scleral thinning and corresponding regional scleral avascularity, which was more prominent in the left eye. Associated overlying calcific plaque formation and prominent accompanying epithelial irregularities visualized with fluorescein staining were also present (Figures 1 and 2). Bilateral pronounced enlargement of nasal and temporal limbal vessels was observed, along with unilateral accumulation of pigment on the anterior lenticular capsule and detached synechiae within the left eye. Despite this finding, there were no signs of active anterior uveitis detected on examination. Although there was high suspicion that these findings were attributable to the patient’s previously performed eye-whitening procedure, a full immunologic work-up was initiated (Antinuclear Antibodies (ANA), Perinuclear Anti-Neutrophil Cytoplasmic Antibodies (P-ANCA), Cytoplasmic Anti-Neutrophil Cytoplasmic Antibodies (C-ANCA), Angiotensin-Converting Enzyme (ACE), Rheumatoid Factor (RF)) to exclude any potential immunologic etiologies. The patient was prescribed 100 mg doxycycline orally and 1,000 mg ascorbic acid orally to be taken twice per day, and 3.5 g erythromycin ointment to apply nightly, along with aggressive administration of lubricating ointment, preservative-free artificial tears, and autologous serum tears. The moxifloxacin was stopped.
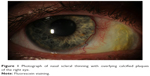  | Figure 1 Photograph of nasal scleral thinning with overlying calcified plaques of the right eye. |
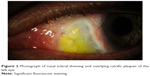  | Figure 2 Photograph of nasal scleral thinning and overlying calcific plaques of the left eye. |
Following the patient’s initial visit, immunologic panel results did not demonstrate atypicality. The left-sided calcific plaques were removed at the slit lamp and transported to ocular pathology for further analysis (Figure 3). Histopathologic analysis along with hematoxylin and eosin (H&E) as well as alizarin red stains confirmed the presence of calcium amongst regular scleral collagen lamellae. Considerable subjective relief of ocular irritation occurred after plaque removal, as a result of conjunctival re-epithelialization. This was performed 3 weeks after presentation. Serum tears were continued, and all epithelial defects resolved with continual aggressive utilization of preservative-free artificial tears and ointment over a 6-week period. During the patient’s last visit, the bilateral scleral thinning remained unchanged. This patient will continue to be followed closely for the foreseeable future and may eventually require amniotic membrane transplantation and/or tectonic graft placement in the event of regression.
Discussion
Methodology associated with each respective cosmetic eye-whitening procedure (regional conjunctivectomy and I-BRITE) involves dissection and excision of the greater portion of conjunctiva/Tenon’s capsule segments along with associated vasculature within the palpebral fissure. Varying combinations of 5-fluorouracil (specific to I-BRITE), bevacizumab, and 0.02% MMC either topically or subconjunctivally are administered intraoperatively with the stated objective of preventing excessive scar formation and recurrent vascular neogenesis. Postoperative administration of 0.02% MMC is then prescribed to be used four times daily for a period of 2–7 days contingent on preexisting thickness of the resected conjunctival tissue.1 Bevacizumab is used routinely during I-BRITE, and had been recently implemented during performance of regional conjunctivectomy, with 350 out of 1,713 patients having received the drug in a recent study.2
Cosmetic ocular-whitening procedures have been associated with complication rates as high as 82.9%.2 Postoperative complications demonstrate a high degree of variability with respect to subtype, severity, latency interval, and management technique (Table 1). Collective incidence of complications previously reported in the literature include: scleral thinning (1.8%), calcific plaque formation (2.9%), fibrovascular proliferation (13%), diplopia (1.2%), elevation of IOP (4.2%), and recurrence of conjunctival hyperemia (2.1%) (Table 2). Elevation of IOP has been attributed to prolonged use of postoperative topical corticosteroids, specifically fluorometholone acetate.1 Other reported complications include lymphangiectasis (0.1%), extraocular muscle fiber exposure (0.04%), dry eye (0.6%), chronic conjunctival epithelial defects (1%), decreased vision (1.8%), granuloma formation, and corneal ulceration.1–3 In 2011, the Korean Ministry of Health and Welfare issued an order for discontinuation of the regional conjunctivectomy procedure,2 and the American Society of Cataract and Refractive Surgery issued a clinical alert in March 2014 recommending use of alternative procedures for treatment of conjunctival hyperemia.3
To date, reported cases of necrotizing scleritis and other complications resulting from ocular-whitening procedures have taken place shortly following the procedure, up to an observed period of nearly 4 years.1–8 Within larger case series reports, average onset of diplopia and granuloma following initial cosmetic surgical intervention have been observed to occur with relative rapidity; ensuing after 1.6 and 2 months, respectively.1,4 Kwon et al documented a case series of patients with scleral thinning and calcification, some of whom showed similarities to the patient currently being discussed, that were first treated for complications anywhere from 12–36 months after surgery.7 However, the date of their initial presentation was less well-described. All generally associated postoperative complications have been reported to occur predominantly within the first 4–12 months following surgery.1,2 Interestingly, our patient presented 4 years after his performed procedure, denoting to our knowledge, one of the most protracted documented latency intervals associated with this procedure.
In addition to creating a predisposition for the development of necrotizing scleritis/infectious scleritis,9,10 MMC has been known to cause scleral ulcerations/calcifications, corneal edema, limbal stem cell deficiency, and iritis, with a highly variable postoperative onset of symptoms, ranging from days to years.11–22 MMC is an alkylating agent that covalently crosslinks DNA, and leads to the inhibition of DNA, RNA, and protein synthesis.23 It follows that regions of the ocular surface that have been exposed to MMC will therefore experience protracted wound healing along with impaired revascularization, particularly with intraoperative addition of bevacizumab (a recombinant humanized monoclonal antibody prohibiting interaction of vascular endothelial growth factor-A with its associated receptors). Areas of avascularity in regions exposed to intraoperative and postoperative MMC during pterygium excision have been observed to persist for longer than 7 years postoperatively.24 In addition, a decreased density of goblet cells within affected conjunctival regions has also been documented.21 By virtue of the normal physiologic relative avascularity of the sclera, it is solely dependent on the adjacent blood supply provided by the choroidal and episcleral vessels.25 Disruption of this crucial vascular network can result in necrosis of scleral tissue accompanied by a prospective possibility for several catastrophic consequences. Development of ischemic sclerae as a postoperative complication of cosmetic eye-whitening procedures is thought to constitute the underlying etiology for the resultant ocular morbidities (conjunctival epithelial defects, scleral thinning/plaque formation, fibrovascular adhesions) that have been documented in postoperative patients.4 Indeed, regions of avascularity typify associated pathological alterations that were observed within our patient, as well as other previously recognized cases of scleromalacia.5,6 Intraoperative cauterization with resultant injury to episcleral vessels will undoubtedly result in a certain degree of vascular compromise within the immediate subjected area. It has been logically inferred that performance of intraoperative cauterization will likely result in increased probability of vascular impotence, resulting in scleral ischemia.1,6 Notably, our patient received intraoperative unilateral cauterization within the left eye, which correlates with the increased severity of scleral thinning observed. On account of the collective antiproliferative augmentative effects resulting from combined use, use of intraoperative 5-fluorouracil (an antimetabolite that disrupts function of RNA and DNA), bevacizumab, and MMC should be avoided, especially after instances of required intraoperative cauterization. Avoidance of postoperative MMC is also recommended due to resultant prolonged wound healing associated with its use.
After definitive exclusion of autoimmune and infectious etiologies, an appropriate therapeutic approach can be instituted. Therapeutic options for noninfectious, nonimmunologic causes of necrotizing scleritis vary depending upon severity and progression. Conservative treatment may be instituted at the clinician’s discretion for a discrete period of time to monitor for signs of regression or progression. We initially recommend calcific plaque removal for facilitation of re-epithelialization. This may aid in significant improvement of symptoms, sparing the patient from undergoing unnecessary surgical intervention. Application of ethylenediaminetetraacetic acid (EDTA) may also be an option for treatment of calcific plaques. However, the authors have no experience with it. Implementation of autologous serum tears may also be considered, especially for cases demonstrating recalcitrance of conjunctival epithelial defect resolution. In more severe cases, additional treatment options available for utilization include placement of amniotic membrane grafts, autologous conjunctival flaps, or simultaneous placement of both.7,8,26–29 Other forms of tissue media available for scleral surface repair include sclera, cornea, pericardium, fascia lata, dermis, and cartilage.29–37
After initiation of treatment, close patient follow-up should be continued for a prolonged period of time to monitor for progression or recurrence of symptoms. This specific population of postoperative patients remains at significant risk for potentially catastrophic postoperative complications several years following initial surgical intervention. It is of particular importance that a thorough surgical history is obtained from every patient in order to anticipate and properly manage any of several ensuing sequelae that may later occur.
Conclusion
Cosmetic ocular whitening procedures such as regional conjunctivectomy and I-BRITE have exhibited exceedingly high rates of complications. Varying combinations of topical or subconjunctival MMC, 5-fluorouracil, and/or bevacizumab can cause regional avascularity, which may lead to scleral thinning and calcific plaque deposition, in addition to several other concerning ocular findings. Despite this, some variation of these procedures likely does have a place in ocular surgery. More work needs to be done in order to determine which combinations of the abovementioned medications achieve an acceptable safety profile. Physicians are encouraged to follow these patients closely, particularly during the first few postoperative months, to monitor for abnormalities that may develop. Correspondingly, sustained physician–patient correspondence is imperative, as complications may occur 4 or more years after surgery.
Disclosure
The authors report no conflicts of interest in this work.
References
Kim BH. Regional conjunctivectomy with postoperative mitomycin C to treat chronic hyperemic conjunctiva. Cornea. 2012;31(3):236–244. | ||
Lee S, Go J, Rhiu S, et al. Cosmetic regional conjunctivectomy with postoperative mitomycin C application with or without bevacizumab injection. Am J Ophthalmol. 2013;156(3):616–622.e3. | ||
ASCRS Cornea Clinical Committee. Clinical alert: eye-whitening procedure: regional conjunctivectomy with mitomycin-C application [press release]. Fairfax, VA: American Society of Cataract and Refractive Surgery. Available from: http://www.ascrs.org/node/1352. Accessed January 22, 2015. | ||
Rhiu S, Shim J, Kim EK, et al. Complications of cosmetic wide conjunctivectomy combined with postsurgical mitomycin C application. Cornea. 2012;31(3):245–252. | ||
Leung TG, Dunn JP, Akpek EL, Thorne JE. Necrotizing scleritis as a complication of cosmetic eye whitening procedure. J Ophthalmic Inflamm Infect. 2013;3(1):39. | ||
Shin HY, Kim MS, Chung SK. The development of scleromalacia after regional conjunctivectomy with the postoperative application of mitomycin C as an adjuvant therapy. Korean J Ophthalmol. 2013;27(3):208–210. | ||
Kwon HJ, Nam SM, Lee SY, Ahn JM, Seo KY. Conjunctival flap surgery for calcified scleromalacia after cosmetic conjunctivectomy. Cornea. 2013;32(6):821–825. | ||
Vo RC, Stafeeva K, Aldave AJ, et al. Complications related to a cosmetic eye-whitening procedure. Am J Ophthalmol. 2014;158(5):967–973. | ||
Kim BH. Surgical treatment of necrotic scleral calcification using combined conjunctival autografting and an amniotic membrane inlay filling technique. Eye (Lond). 2011;25(11):1484–1490. | ||
Okhravi N, Odufuwa B, McCluskey P, Lightman S. Scleritis. Surv Ophthalmol. 2005;50(4):351–363. | ||
Rubinfeld RS, Pfister RR, Stein RM, et al. Serious complications of topical mitomycin-C after pterygium surgery. Ophthalmology. 1992;99(11):1647–1654. | ||
Dunn JP, Seamone CD, Ostler HB, Nickel BL, Beallo A. Development of scleral ulceration and calcification after pterygium excision and mitomycin therapy. Am J Ophthalmol. 1991;112(3):343–344. | ||
Kaufman SC, Jacobs DS, Lee WB, Deng SX, Rosenblatt MI, Shtein RM. Options and adjuvants in surgery for pterygium: a report by the American Academy of Ophthalmology. Ophthalmology. 2013;120(1):201–208. | ||
Tsai YY, Lin JM, Shy JD. Acute scleral thinning after pterygium excision with intraoperative mitomycin C: a case report of scleral dellen after bare sclera technique and review of the literature. Cornea. 2002;21(2):227–229. | ||
Alsagoff Z, Tan DT, Chee SP. Necrotising scleritis after bare sclera excision of pterygium. Br J Ophthalmol. 2000;84(9):1050–1052. | ||
Safianik B, Ben-Zion I, Garzozi HJ. Serious corneoscleral complications after pterygium excision with mitomycin C. Br J Ophthalmol. 2002;86(3):357–358. | ||
Dougherty PJ, Hardten DR, Lindstrom RL. Corneoscleral melt after pterygium surgery using a single intraoperative application of mitomycin-C. Cornea. 1996;15(5):537–540. | ||
Hayasaka S, Iwasa Y, Nagaki Y, Kadoi C, Matsumoto M, Hayasaka Y. Late complications after pterygium excision with high dose mitomycin C instillation. Br J Ophthalmol. 2000;84(9):1081–1082. | ||
Wan Norliza WM, Raihan IS, Azwa JA, Ibrahim M. Scleral melting 16 years after pterygium excision with topical mitomycin C adjuvant therapy. Cont Lens Anterior Eye. 2006;29(4):165–167. | ||
Saifuddin S, el Zawawi A. Scleral changes due to mitomycin C after pterygium excision: a report of two cases. Indian J Ophthalmol. 1995;43(2):75–76. | ||
Solomon A, Kaiserman I, Raiskup FD, Landau D, Frucht-Pery J. Long-term effects of mitomycin C in pterygium surgery on scleral thickness and the conjunctival epithelium. Ophthalmology. 2004;111(8):1522–1527. | ||
Lichtinger A, Pe’er J, Frucht-Pery J, Solomon A. Limbal stem cell deficiency after topical mitomycin C therapy for primary acquired melanosis with atypia. Ophthalmology. 2010;117(3):431–437. | ||
Glaubiger D, Ramu A. Antitumor antibiotics. In: Chabner BA, editor. Pharmacologic Principles of Cancer Treatment. Philadelphia, PA: WB Saunders; 1982:407–410. | ||
Raiskup F, Solomon A, Landau D, Ilsar M, Frucht-Pery J. Mitomycin C for pterygium: long term evaluation. Br J Ophthalmol. 2004;88(11):1425–1428. | ||
Watson PG, Young RD. Scleral structure, organisation and disease. A review. Exp Eye Res. 2004;78(3):609–623. | ||
Ma DH, Wang SF, Su WY, Tsai RJ. Amniotic membrane graft for the management of scleral melting and corneal perforation in recalcitrant infectious scleral and corneoscleral ulcers. Cornea. 2002;21(3):275–283. | ||
Hanada K, Shimazaki J, Shimmura S, Tsubota K. Multilayered amniotic membrane transplantation for severe ulceration of the cornea and sclera. Am J Ophthalmol. 2001;131(3):324–331. | ||
Karalezli A, Kucukerdonmez C, Borazan M, Akova Y. Successful treatment of necrotizing scleritis after conjunctival autografting for pterygium with amniotic membrane transplantation. Orbit. 2010;29(2):88–90. | ||
Ti SE, Tan DT. Tectonic corneal lamellar grafting for severe scleral melting after pterygium surgery. Ophthalmology. 2003;110(6):1126–1136. | ||
Mauriello JA Jr, Pokorny K. Use of split-thickness dermal grafts to repair corneal and scleral defects – a study of ten patients. Br J Ophthalmol. 1993;77(6):327–331. | ||
Chechelnitsky M, Mannis MJ, Chu TG. Scleromalacia after retinal detachment surgery. Am J Ophthalmol. 1995;119(6):803–804. | ||
Raviv T, Greenfield DS, Liebmann JM, Sidoti PA, Ishikawa H, Ritch R. Pericardial patch grafts in glaucoma implant surgery. J Glaucoma. 1998;7(1):27–32. | ||
Schein OD. The use of processed pericardial tissue in anterior ocular segment reconstruction. Am J Ophthalmol. 1998;125(4):549–552. | ||
Melamed S, Ashkenazi I, Belcher DC 3rd, Blumenthal M. Donor scleral graft patching for persistent filtration bleb leak. Ophthalmic Surg. 1991;22(3):164–165. | ||
Kosmin AS, Wishart PK. A full-thickness scleral graft for the surgical management of a late filtration bleb leak. Ophthalmic Surg Lasers. 1997;28(6):461–468. | ||
Mistlberger A, Biowski R, Grabner G. Repair of a late-onset filtering bleb leak using a corneal graft shaped with an excimer laser. Ophthalmic Surg Lasers. 2001;32(5):428–431. | ||
Soong HK, Meyer RF, Wolter JR. Fistula excision and peripheral grafts in the treatment of persistent limbal wound leaks. Ophthalmology. 1988;95(1):31–36. |
 © 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2015 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.



