Back to Journals » International Journal of Nanomedicine » Volume 14
Defining the process parameters affecting the fabrication of rosuvastatin calcium nanoparticles by planetary ball mill
Authors Alshora D, Ibrahim M , Elzayat E , Almeanazel OT, Alanazi F
Received 1 March 2019
Accepted for publication 6 May 2019
Published 27 June 2019 Volume 2019:14 Pages 4625—4636
DOI https://doi.org/10.2147/IJN.S207301
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Thomas Webster
Doaa Alshora,1 Mohamed Ibrahim,1,2 Ehab Elzayat,1 Osaid T Almeanazel,1 Fars Alanazi1
1Kayyali Chair for Pharmaceutical Industries, Department of Pharmaceutics, College of Pharmacy, King Saud University, Riyadh, Saudi Arabia; 2Department of Pharmaceutics, College of Pharmacy, Al-Azhar University, Assiut, Egypt
Purpose: Rosuvastatin calcium (ROSCa) nanoparticles were fabricated by planetary ball mill to enhance ROSCa dissolution rate and bioavailability.
Methods: Milling time factors (milling cycle time and number as well as pause time) were explored. The effect of different milling ball size, speed, and solid-to-solvent ratio were also studied using Box–Behnken factorial design. The fabricated nanoparticles were evaluated in term of physicochemical properties and long-term stability.
Results: The obtained data revealed that the integrated formulation and process factors should be monitored to obtain desirable nanoparticle attributes in terms of particle size, zeta potential, dissolution rate, and bioavailability. The optimized ROSCa nanoparticles prepared by milling technique showed a significant enhancement in the dissolution rate by 1.3-fold and the plasma concentration increased by 2-fold (P<0.05). Moreover, stability study showed that the optimized formula of ROSCa nanoparticles exhibits higher stability in long-term stability conditions at 30°C with humidity of 60%.
Conclusion: Formulation of ROSCa as nanoparticles using milling technique showed a significant enhancement in both dissolution rate and plasma concentration as well as stability compared with untreated drug.
Keywords: rosuvastatin calcium, nanoparticles, planetary ball mill, Box–Behnken, in vitro dissolution, stability, pharmacokinetics
Introduction
During the last few decades, nanotechnology participated in medical and pharmaceutical fields. It has a significant use in pharmaceutics to improve drug solubility and thus enhancing its dissolution rate and bioavailability. Nanonization is carried out via two different techniques, either bottom-up or top-down.1 After the advancement and improvement occurred in nanoparticle machine area, top-down techniques gain a special interest in pharmaceutical research and industrial community.2 Milling is one of the effective top-down techniques. It can be operated in dry and wet methods, but wet milling showed a high capability in reducing the particle size to nanaosize than dry method.3
Planetary ball mill is a powerful machine used to reduce the particle size to nanosize depending on centrifugal accelerating force rather than gravitational accelerating force, which makes the process more efficient and accomplished in a short time.4 Several parameters related to the planetary ball mill can affect the resultant particles’ properties. These factors include i) milling time-related parameters (pause time, number of milling cycles and time of each cycle), ii) speed, iii) milling ball size, and iv) solid to solvent ratio. Nanosization by planetary ball mill significantly resulted in enhancing the dissolution rate and bioavailability of different drugs, like clarithromycin5 and naproxen.6
Rosuvastatin calcium (ROSCa), antihyperlipidemic drug, is a class II drug that characterized by poor water solubility, that lower its low dissolution rate, which in turn affects its. Different techniques were used to enhance and improve the dissolution rate and bioavailability of ROSCa, including liquisolid technique,7 solid dispersion,8 cyclodextrin complexation,9 self-nanoemulsifying technique.10 nanosponge structure,11 and hexagonal liquid crystalline nanodispersion technique.12 ROSCa nanosization by planetary ball mill is expected to enhance its dissolution rate and bioavailability by reducing the particle size of the drug.13
In this study, different process parameters that can affect the fabrication of ROSCa nanoparticles by planetary ball mill viz., milling time factors (pause time, number of cycles, and time of each cycle) were studied individually. Other parameters that affect the milling efficiency such as milling ball size, milling speed, and solid to solvent ratio were also studied and optimized as independent parameters using Box–Behnken design. The effect of these parameters on the nanoparticles attributer (response), namely particle size (nm), zeta potential (mV) and dissolution efficiency (%) will be studied.
Materials and methods
Materials
Rosuvastatin calcium (ROSCa) was purchased from Beijing Mesochem Technology CO. Ltd. (Beijing, China). Polyvinylpyrrolidone (PVP K-30) MWt. 40,000 was obtained from Loba Chemie (India). Methanol was obtained from Fisher Chemical, UK. Hydrochloric acid was obtained from Panreac, Barcelona, Spain. All other ingredients were of analytical grade and used as received.
Methods
Preparation of ROSCa nanoparticles
ROSCa nanoparticles were prepared in two steps. ROSCa powder was first subjected to dry milling using planetary ball mill (Pulverisette 7 Premium, Fritsch Co. Germany), in order to decrease the particles size to less than 20 µm. since the received raw material was analyzed for particle size and it was found 618 µm. The dry milled ROSCa powder was then subjected to wet milling using aqueous solution containing 10%w/w PVP K30 as stabilizer.14 The suspension of ROSCa and PVP solution was then nanosized by planetary ball mill using zirconium balls (0.1 mm) at 800 rpm. Thereafter, the obtained nanosuspension was then dried using freeze-drier (Alpha 1–4 LD Plus, Martin Christ Gefriertrocknugs anlagen GmbH, Osterode am Harz, Germany) pending further investigations.
Impact of milling time
In this part, the effect of milling time factors (pause time, number of cycle, and time of each cycle) on the particle size, zeta potential, and DE% of ROSCa was estimated. According to our preliminary study (data not shown), it was found that the milling process was inefficient when the number of milling cycles exceeded 3 cycles. Therefore, the milling time was kept constant at 3 cycles each of 10 mins, and the milling time factors were changed to study their impact on nanoparticles attributes.
Pause time means the stopping or cooling time between cycles during the whole milling process. In this study, the pause time was adjusted into 3 different intervals 5, 10, and 15 mins, and its effect on ROSCa nanoparticles was determined by particle size and dissolution rate.
The effects of the number of milling cycles were studied on the nanoparticles attributer. One, two, and three milling cycles were examined.
Milling times of 4, 7, and 10 mins were examined for their impact on ROSCa nanoparticles.
Experimental design
After investigating the effect of milling time, milling cycle, and pause time on the properties of ROSCa nanoparticles, the effects of other independent parameters were evaluated using factorial design.
Experiment was designed by applying Box–Behnken design using statistical program (statgraphics Centurion program Version 17.2.02.). Three independent factors, milling speed (A), milling ball size (B), and drug:solvent ratio (C) were studied for their effects on four responses (dependent parameters) including particle size, zeta potential, and dissolution efficiency after 120 mins, DE 120 as shown in Table 1). ROSCa nanoparticles were prepared as described previously.
 |
Table 1 Variables in Box–Behnken design |
Particle size analysis
The particle size of all formulation was measured for both nanosuspension (before drying) and freeze-dried nanoparticles using Malvern zetasizer (Nano S, Worcestershire, UK). Small amount of the nanoparticles (in case of after drying) were reconstituted in 10 mL of deionized water and vortexed. The particle size was measured immediately.
Zeta potential measurements
The zeta potential for nanosuspension and freeze-dried nanoparticles was measured using Malvern zetasizer (Nano S, Worcestershire, UK) Small amount of the nanoparticles (in case of after drying) were reconstituted in 10 mL of deionized water and vortexed. The particle size was measured immediately.
X-ray powder diffraction (XRPD)
The X-ray diffraction analysis was done for the best obtained formula and compared to the raw drug in order to study its crystallinity. The X-ray diffraction patterns of the powder samples were obtained using RIGAKU diffractometer (Japan) which was equipped with curved graphite crystal monochromator, automatic divergence slit, and automatic controller PW/1710. The target used was CuKα radiation operating at 40 kV and 40 mA (λkα =1.5418 Å). The diffraction patterns were achieved using continuous scan mode with 2Ɵ° ranging from 4° to 60°.
Transmission electron microscopy (TEM)
In order to characterize the external and internal shape of nanoparticles, transmission electron microscope was used (FEI Tecnai G2 20 TWIN, USA) that was operated at 200 kV. The point-to-point resolution of this microscope was less than 0.25 nm and line to line resolution <0.10 nm.
Drug content of ROSCa nanoparticles
In a volumetric flask of 10 mL, 10 mg of nanoparticles was dissolved in 10 mL methanol; then, 1 mL was taken in 10 mL volumetric flask and completed to the volume with 0.1 N HCl. The absorbance was measured at λ 244 nm against a blank made of stabilizer only following the same procedure. The drug content experiment was carried out in triplicate.
In vitro dissolution study
The in vitro dissolution profile of ROSCa nanoparticles was studied using a USP-II dissolution apparatus (Pharma Test, DT 70, Germany). The dissolution experiment was carried out in 500 mL of 0.1 N HCl as a dissolution medium at 37°C and 50 rpm. Aliquots of 5 mL sample were withdrawn at pre-determined time intervals (5, 10, 15, 30, 60, 90, and 120 mins) using poroplast-kerze filter. The absorbance was measured spectrophotometrically at 244 nm against a suitable blank. The dissolution efficiency after 120 mins was calculated according to Khan Equation:15
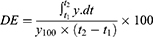
where, y is the percentage of drug dissolved, DE is the area under the dissolution curve between time points t1 and t2 expressed as a percentage of the curve at maximum dissolution, y100, over the same time period.
Pharmacokinetic studies
Animal
Rabbits (weight 4 kg) were obtained from the College of Pharmacy experimental animal care center (King Saud University, Riyadh, Saudi Arabia). The animals fasted for 24 hrs prior to the administration of the drug, the drug was administered orally through oral gavage in order to assure the dose uniformity. The study was approved by the Ethical Committee (Protocol IRB no. 80–23) by Experimental Animal Care Unit. The experimental protocol was in agreement with the Guide of the National Institution of Health (NIH) for the Care and Use of Laboratory Animals.
Drug administration
Two groups of animals were administered the calculated animal dose equivalent to 10 mg. Both raw ROSCa and optimized nanoparticle formulation were suspended in 0.5% Na-CMC prior administration and administered orally by gastric tube. Aliquots of blood sample were collected from ear vein in heparinized tube at different time intervals for at least 24 hrs. The blood sample was centrifuged at 6,000 for 20 mins to separate the plasma. The samples were analyzed using developed UPLC-MS/MS method.14 Different pharmacokinetics parameters such as Cmax, Tmax, AUC, rate of absorption, and rate of elimination were calculated using a non-compartmental model analysis.
Analysis of ROSCa in rabbit plasma
Sample preparation
The plasma samples were processed by protein precipitation method. Simply, 0.2 mL of plasma sample was combined with 50 µL internal standard Prednisolone (100 µg/mL) and 0.75 mL methanol, and then vortexed for 1 min. After centrifugation at 15,000 rpm (15,000× g) for 10 mins, 750 µL of the supernatant liquid was transferred to a sample vial and 5 µL was injected into the LC/MS/MS for quantitative analysis.
Chromatographic conditions
In this study, a validated UPLC-MS/MS (UPLC: Waters Acquity, Milford, MA, USA) was employed to determine the concentration of rosuvastatin in rabbit plasma [12]. The chromatographic conditions involved the use of a BEH C18 column (50 mm×2.1 mm, 1.7 µm) with a mobile phase composed of acetonitrile and 0.1% formic acid (35:65, volume: volume) at a flow rate of 0.25 mL/min using prednisolone as the internal standard. The eluted compounds were detected by tandem mass spectrometry using TQ detector (Waters Corp., Milford, MA) equipped with an electrospray ionization (ESI) source operating in positive ionization mode. The quantification was performed with multiple reactions monitoring (MRM) mode. Selection of ionization pairs (m/z) was shown as follows: rosuvastatin: 482.097→258.072 (cone voltage 60 V, collision energy 34 V), Prednisolone: 403.172→385.224 (cone voltage 42 V, collision energy 13 V).
Stability studies
Stability studies were done for untreated drug powder and nanoparticles of the optimized formula according to ICH guidelines.16 The effect of long-term storage conditions at 30±2 Co, 60%±5% RH on formulations was determined by characterizing the particle size and the dissolution rate for 12 months.
Statistical analysis
One-way analysis of variance (ANOVA) was used for the comparison. A value of P<0.05 was denoted as a significant difference throughout the current study. (Statgraphics Centurion program Version 17.2.02.).
Results and discussion
Impact of milling time factors
Pause time during the milling procedure, as a process factor, was not discussed briefly in the milling procedures. Figure 1 shows the effect of milling time factors (pause time, number of milling cycles and time of each milling cycle) on the particle size and DE% of ROSCa nanoparticles. Generally, ROSCa particle size was remarkably reduced to less than 2500 nm by milling in comparison to the untreated material (618 µm). The data also revealed that the size of the milled drug particles increased with increasing the pause time during the milling cycles. The particle size increased from 461.8±16.68 nm to 1513.56±55.2 with increasing the pause time from 5 to 15 mins. Dynamically, during the milling process, the grinding balls are subjected to high rotational speed, and the difference between the speed of the ball and the grinding jar lead to interaction between the impact and fraction forces, which in turn releases a high energy that used in particle size reduction.17 While during the pause time, the non-movement condition was pronounced and the ball will then settle down with the aid of gravity force and, by prolonging pause time, the opportunity for the particle to re-aggregate and form a large particle is high. Therefore, by increasing the pause time from 5 to 15 mins, the non-movement period increased and thus, the tendency to for the particles to form large particle size prevails.
The in-vitro dissolution study showed that there was an inverse correlation between the DE% and the pause time. For example, the calculated DE% showed marked and significant reduction from 56.15% to 38.01% (P<0.05) with increasing the pause time from 5 to 15 mins (Figure 1). Prolonging pause time allows the small particles obtained from the milling procedure to re-aggregate again pending the next milling cycle, resulting in enlargement of the particles at the end of milling. Therefore, a reduction in the dissolution rate due to the enlargement in the particle size occurred with longest pause time.3
The present study shows that the pause time affected the particle size as well as the dissolution rate significantly and it must be kept as short as possible. Pause time of 5 mins was selected as the optimum pause time condition that resulted in a smaller particle size with higher dissolution rate.
In order to study the effect of number of milling cycles, both pause time and time of each cycle were kept constant at 5 and 10 mins, respectively. The results showed that by increasing the number of cycles from 1 to 3 cycles, a significant reduction in the particle size was observed. Increasing the number of milling cycles (increasing milling time) might allow more chance to reduce the particle size with aid of beads collisions. The results showed also that increasing milling cycle resulted in a significant enhancement of the drug dissolution rate positive effect on dissolution rate from 69.9±3.7% to 78.8±4.9% by increasing the milling cycles from 2 to 3 cycles. These results are conformed with theory that stated that decreasing the particles size will enhance the dissolution rate.3 In addition, increasing the number of cycles gives a reasonable time for efficient particle size reduction, which might result in increasing the particles’ surface area that, in turn, enhanced the dissolution rate.3 The same results were obtained by Liu et al,18 who studied the effect of the number of cycles on the particle size of itraconazole and indomethacin during milling procedure. They found that the particle sizes of both drugs significantly decreased with increasing the number of cycles from 2 to 10 cycles.18
Another factor that might influence the milling process is the time of milling cycle. The effects of different cycle times (4, 7, and 10 mins) on particle size and dissolution rate of ROSCa nanoparticles were studied. Similar to the number of cycles, increasing the cycle time from 4 to 10 mins markedly and significantly decrease the particle size from 3088±171.03 nm to 448.3±31.60 nm, respectively. Consequently, ROSCa dissolution rate was increased significantly from 69.04±3.3% after 4 mins to 78.7±4.9% after 10 mins. This result could be attributed to the relation between the milling time and the collision occurred between the milling ball. So, by increasing the milling time, a high collision will occur, and this gives a chance for large particle to be milled into smaller size.19 Bartos and coworker20 succeeded to decrease meloxicam particle size from 35 µm to 126 nm by increasing the milling time to 50 mins.
The present study showed that milling time factors played a crucial role in particle size reduction. The pause time should be kept as minimum as possible while milling time including (number of cycles and time of each cycle) should be at optimum limit. Therefore, milling of ROSCa for 3 cycles with 10 mins of each and a pause time of minutes was considered as optimum milling conditions that produced small particle size with highest dissolution rate. These milling conditions will be adopted during studying other formulation and process factors.
Experimental design
In this part, the effects of milling speed (A), milling ball size (B), and solid to solvent ratio (C) on the quality attributes of ROSCa nanoparticles were evaluated. In order to study the interaction between those parameters, Box–Behnken design was applied. The desirability in this part was to formulate ROSCa nanoparticles that have: small particle size, high zeta potential, and high dissolution rates. Therefore, different formulations were designed using Box–Behnken factorial design using the statistical analysis as shown in Table 2. The effect of three independent factors including speed (A), ball size (B), solid to solvent ratio (C), their quadratic (AA, BB, CC), and interactive effects on the dependent parameters (responses) including particles size, zeta potential, and dissolution efficiency (120 mins) were studied as shown in Table 2.
 |
Table 2 Composition of different ROSCa nanoparticles formulations based on Box–Behnken factorial design |
Effect on particle size
ANOVA analysis (Table 3) showed the significance of all possible effects of independent factors on ROSCa nanoparticles size. It was found that the effect of milling ball size on nanoparticle size was more noticeable, but insignificant (P=0.21) in comparison to the milling speed and solid to solvent ratio. ANOVA analysis showed that none of the independent factors had a significant individual effect on the particle size (>0.05), but the interaction of milling speed and milling ball size exhibited the most pronounced effect on ROSCa nanoparticles size (P=0.06). In case of F5 and F1, for example, the particle size of F5 (500 rpm, 0.5 mm, 0.625) was 875.4±2.76 nm and it enlarged to 2817.66±140.01 nm in F1 (800 rpm, 1 mm, 0.625). Moreover, the response surface plot for particle size (Figure 2A) showed that increasing the milling ball size from 0.5 to 1 mm and speed from 400 to 800 rpm caused enlarging in the particle size. The relation between the particle size and speed could be clarified the energy produced during rotation. So, at low speed (200–400 rpm) the particle size decreased due to the higher energy inside the milling bowel that led to impaction and breakage of the particles. Further increment in the speed may result in enlargement in the particle size due to agglomeration which mainly occurred due to the breakage of repulsive force prepared by the stabilizer.18 Increasing the solid content did not significantly affect nanoparticle size. However, Ghosh and co-workers found that increasing the solid content from 2% to 5% significantly decreased the particle size.21
 |
Table 3 Analysis of variance for the effect of independent parameters on particle size, zeta potential and DE% with respect to speed, ball size, and solid solvent ratio |
 |
Figure 2 Response surface plot for the effect of independent factors speed and ball size on (A) particle size, (B) zeta potential, and (C) DE%. Abbreviation: DE, dissolution efficiency. |
Effect on zeta potential
The target of nanoparticle formulation designed for enhanced drug dissolution is to increase the negative value of zeta potential which indicates the stability of the dispersion system. ANOVA table (Table 3) showed that the individual effect of solid to solvent ratio and quadratic effect of speed have positive effects on zeta potential and maximized its value (P<0.05). In contrast, the results showed that the milling ball size significantly decreased zeta potential value (P=0.01), while the reduction of zeta potential due to the effect of solid:solvent ratio was found more significant (P=0.003). In addition, the significant effects were observed in case of the quadratic effects of both milling speed (P=0.01) and milling ball size (P=0.009), and the interaction between them also exhibited significant effect (P=0.06) on the zeta potential value. According to the results, the independent factors that exerted a significant effect on zeta potential can be ranked as follows, depending on both sum of squares and P-value, solid to solvent ratio (C) > quadratic effect of ball size (BB) > quadratic effect of speed (AA) > ball size (B).
The 3D response surface plot for zeta potential (Figure 2B) showed that increasing the speed from 200 to 500 rpm led to a decrease in the negative value of zeta potential, while further increment to 800 rpm led to raising zeta potential negative value but in an insignificant manner (P>0.05). Moreover, the milling ball size affected the zeta potential in an opposite way. The zeta potential value increased while using small ball size (0.1–0.5 mm) and it significantly decreased with larger ball size of 1 mm. For example, F1 and F12 nanoparticle formulations both were milled at 800 rpm using solid to solvent ratio of 0.625 but with different ball sizes. It was found that the negative value of zeta potential decreased (−28.5±6.5 to −22.9±3.9 mV) with increasing the ball size from 0.1 to 1 mm in case of F1.
Effect on dissolution efficiency (DE)
The ANOVA table (Table 3) showed that only the milling ball size exerted insignificant effect on DE (P=0.05), while milling speed and solid to solvent ratio exhibited insignificant effects (P<0.05). In contrast, the quadratic effect of both ball size and solid to solvent ratio significantly decreased the DE value (P-value was 0.06 and 0.02, respectively). The interaction effect between ball size and solid:solvent ratio also affected the DE significantly (P=0.03). Increasing the ball size from 0.1 to 1 mm and solid to solvent ratio from 0.25 to 1, resulted in slowing the dissolution rate (decreased DE), as the case of formulations F1 and F12 that were prepared using same milling speed (800 rpm) and solid:solvent ratio (0.625), Figure 2C. F1 nanoparticle formula was manufactured using the largest milling ball size (1 mm), and the resulting particle size was large (2817.66 nm); therefore, the drug exhibited a reduction in DE to 60.5%. In contrast, smallest ball size was used in preparing F12 nanoparticle formula, which resulted in smaller particle size (1018.80 nm), and in turn, the DE increased (71.34%). In Figure 2C, DE was enhanced from 64.1% to 71.9% with increasing the ball size from 0.1 to 0.5 mm and the solid to solvent ratio from 0.25 to 0.625, respectively. Further enlargement in the ball size and increment solid to solvent ratio led to marked reduction in the DE to reach 56.6% as seen in F6.
Optimization of the milling process and formulation condition for ROSCa nanoparticles
The multiple response optimization was used to optimize ROSCa nanoparticles with the following desirability: smaller particle size, maximum zeta potential, and maximum DE. Table 4 illustrates the optimum milling conditions for the production of the optimized ROSCa nanoparticles formula stabilized by 10% PVP. The data showed that the optimized ROSCa nanoparticle formula could be prepared by planetary ball milling using a milling speed of 446 rpm, milling ball size of 0.1 mm, and solid to solvent ratio of 0.25 (Table 4). This optimized formula was manufactured according to the previously mentioned milling conditions and characterized for particle size, zeta potential, and DE in comparison to the predicted values obtained from the statistical program (Table 4). The observed values of the optimized ROSCa nanoparticle formula (zeta potential, particle size, and DE%) were found highly correlated with their predicted values.
 |
Table 4 The predicted and observed values for optimized ROSCa nanoparticles attributes |
Characterization of ROSCa optimized nanoparticle formula
Transmission electron microscopy (TEM)
TEM analysis was carried out to allow studying the internal structure and morphology of ROSCa nanoparticles. The images obtained from TEM showed that the nanoparticles have a spherical shape with a particle size of less than 400 nm as seen in Figure 3A. Figure 3B shows the internal structure of the particles in which the nanoparticles contain a core (drug) coated with a layer of the stabilizing polymer.
 |
Figure 3 TEM images for ROSCa nanoparticles optimized formula: (A) intact particles; (B) internal section.Abbreviation: ROSCa, rosuvastatin calcium. |
X-ray powder diffraction (XRPD)
To determine the crystalline nature of the raw ROSCa and nanoparticles of the optimized formula, XRPD studies were performed. The XRPD pattern of untreated ROSCa showed two characteristic peaks at 2θ degree of 31.8 and 45.5 degrees. These sharp peaks indicate certain crystallinity of untreated ROSCa. The XRPD spectrum of ROSCa nanoparticles optimized formula showed complete disappearance of ROSCa characteristic diffraction peaks, and this could be due to the ability of the higher concentration of PVP (10%) to transform ROSCa to amorphous form. The addition of PVP as stabilizer plays an important role in not only changing the crystalline form to amorphous one but also inhibiting the rate crystal growth22,23
In vitro and in vivo evaluation
The in vitro dissolution study showed that the dissolution rate of the optimized ROSCa nanoparticle formula was significantly enhanced compared with raw ROSCa. The drug exhibited 79% dissolution from the optimized nanoparticle formula, while the untreated raw drug showed only 61% after 120 mins (Figure 4A).
 |
Figure 4 In vitro dissolution rate (A) and plasma concentration–time curve (B) of ROSCa optimized nanoparticle formula compared with the untreated drug.Abbreviation: ROSCa, rosuvastatin calcium. |
The solubility of untreated drug was carried out in previous work,24 and it was 0.3 mg/mL. In addition, the solubility of the drug in nanoparticles compared to the untreated drug was enhanced, and this might contribute to the enhanced drug dissolution rate in case of nanoparticles.
By performing the in vivo study, the results showed that milling technique was significantly efficient not only in enhancing the dissolution rate but also in improving the bioavailability. Plasma concentration–time curve showed that the plasma concentration was increased significantly by 2-fold for ROSCa nanoparticles compared with untreated drug. (Figure 4B).
Pharmacokinetics parameters calculated from plasma concentration–time curve of ROSCa are shown in Table 5. The maximum plasma concentration significantly increased by 2-fold after the administration of ROSCa nanoparticles optimized formula (P<0.05). The area under the curve (AUC0-t) also increased significantly by 1.5 fold (P<0.05).
 |
Table 5 The plasma pharmacokinetic parameters after administration of optimized nanoparticle formula and pure drug (ROSCa) in a dose equivalent to 10 mg to rabbits (n=3) |
Stability study
The effect of long-term storage condition of temperature 30°C and humidity of 60% on the particle size of ROSCa nanoparticles optimized formula is illustrated in Figure 5. The results showed a significant particle size enlargement after 3 months in the nanoparticles formulation compared with freshly prepared nanoparticles. The particle size increased from 611.06±60.01 nm to 1768.18±389.55 nm. After that, the enlargement in the particle size is considered as insignificant (Figure 5).
The effect of temperature and humidity on the dissolution rate of ROSCa nanoparticles optimized formula was also studied (Figure 5). The results of storage of raw ROSCa showed that the dissolution rate decreased by 50% after 3 months, while no significant decrease in the dissolution rate was observed thereafter. In case of the optimized nanoparticle formula, the dissolution rate of ROSCa from nanoparticles decreased only by 10% after 3 months and by less than 20% after 6 months after which no significant change was observed. Therefore, ROSCa nanoparticles showed some kind of higher stability over the untreated one. Moreover, the drug content analysis showed no loss in the drug content compared to the initial one. The decrease in the drug dissolution rate in case of the optimized nanoparticle formula could be contributed to the enlargement in the particle size.
Conclusion
It is very important to study the effect of different process parameters on the particle size and dissolution rate. The results showed that for best milling pause time should be kept as minimum as possible, while both number of milling cycles and time of each cycle showed maximized, but for certain limit. Box–Behnken factorial design was used, and it showed up the effect of formulation and process parameters on the attributes of ROSCa nanoparticles prepared by planetary ball milling. The optimized ROSCa nanoparticle formula showed enhancement in ROSCa dissolution rate and plasma concentration with smaller particle size, in addition to as higher stability over 6 months in compassion to the untreated ROSCa.
Acknowledgment
This project was financially supported by King Saud University, Vice Deanship of Research Chairs, Kayyali Chair for Pharmaceutical Industry, through grant number DO-2017.
Author contributions
All authors contributed towards data analysis, drafting and critically revising the paper, gave final approval of the version to be published, and agreed to be accountable for all aspects of the work.
Disclosure
The authors report no potential conflicts of interest in relation to this work.
References
1. Junghanns JAH, Müller RH. Nanocrystal technology, drug delivery and clinical applications. Int J Nanomed. 2008;3:295–309. PMCID: PMC2626933.
2. Kumar CSSR. Nanotechnology tools in pharmaceutical R&D. Materialstoday. 2009;12:24–30. Electron microscopy special issue. ISSN:1369 7021 © Elsevier Ltd. doi:10.1016/S1369-7021(10)70142-5
3. Alshora D, Ibrahim MA, Alanazi FK. Nanotechnology from particle size reduction to enhancing aqueous solubility. In: Grumezescu AM, editor. Surface Chemistry of Nanobiomaterials. Application of Nanobiomaterials. Vol. 3. Cambridge, USA: Elsevier; 2016:163–191.
4. Fritsch: Planetary ball mill. Retrieved from: http://www.fritsch-international.com/sample-preparation/milling/planetary-mills/details/product/pulverisette-7-premium-line/.
5. Shahbaziniaza M, Foroutana SM, Bolourchiana N. Dissolution rate enhancement of clarithromycin using ternary ground mixtures: nanocrystal formation. Iran J Pharm Res. 2013;12(4):587–598.
6. Patnaik S, Kurdekar AD, Chunduri LAA, Prathibha C, Sai Vamsi Krishna V, Venkataramaniah K. Naproxen-soluplus® nano formulations for enhanced oral bioavailability. Int J Pharm. 2017;7:99–112.
7. Kamble PR, Shaikh KS, Chaudhari PD. Application of liquisolid technology for enhancing solubility and dissolution of rosuvastatin. Adv Pharm Bull. 2014;4:197–204. doi:10.5681/apb.2014.029
8. Ramu A, Vidyadhara S, Devanna N, Anusha C, Keerthi J. Formulation and evaluation of rosuvastatin fast dissolving tablets. Asian J Chem. 2013;25:5340–5346. doi:10.14233/ajchem
9. Akbari BV, Valaki BP, Maradiya VH, Akbari AK, Vidyasagar G. Enhancement of solubilityand dissolution rate of rosuvastatin calcium by complexation with β-cyclodextrin. Int J Pharm Biol Arch. 2011;2:511–520.
10. Amrutkar C, Salunkhe K, Chaudhari S. Study on self-nano emulsifying drug delivery system of poorly water- soluble drug rosuvastatin calcium. World J Pharm Res. 2014;3:2137–2151.
11. Gabr MM, Mortada SM, Sallam MA. Carboxylate cross-linked cyclodextrin: a nanoporous scaffold for enhancement of rosuvastatin oral bioavailability. Eur J Pharm Sci. 2018;111:1–12. doi:10.1016/j.ejps.2017.09.026
12. Gabr MM, Mortada SM, Sallam MA. Hexagonal liquid crystalline nanodispersions proven puperiority for enhanced oral delivery of rosuvastatin: in vitro characterization and in vivo pharmacokinetic study. J Pharm Sci. 2017;106:3103–3112. doi:10.1016/j.xphs.2017.04.060
13. Stengera F, Mendeb S, Schwedesc J, Peukertd W. Media milling in stirred media mills. Chem Eng Sci. 2005;60:4557–4565. doi:10.1016/j.ces.2005.02.057
14. Alshora DH, Ibrahim MA, Elzayat E, Almeanazel OT, Alanazi F. Rosuvastatin calcium nanoparticles: improving bioavailability by formulation and stabilization codesign. PLoS One. 2018;13(7):1–17. doi:10.1371/journal.pone.0200218
15. Ngwuluka NC, Lawal K, Olorunfemi PO, Ochekpe NA. Post-market in vitro bioequivalence study of six brands of ciprofloxacin tablets/caplets in Jos, Nigeria. Sci Res Essay. 2009;4(4):298–305.
16. Harmonisation ICH. Guidance for Industry Q1A (R2) Stability Testing of New Drug Substances and Products. Silver Spring: US Department of Health and Human Services, Food and Drug Administration; 2003.
17. Canakci A, Erdemir F, Varol T, Patir A. Determining the effect of process parameters on particle size in mechanical milling using the Taguchi method: measurement and analysis. Measurement. 2013;46:3532–3540. doi:10.1016/j.measurement.2013.06.035
18. Liu P, Rong X, Laru J, et al. Nanosuspensions of poorly soluble drugs: preparation and development by wet milling. Int J Pharm. 2011;411:215–222. doi:10.1016/j.ijpharm.2011.03.050
19. Knieke C, Sommer M, Peukert W. Identifying the apparent and true grinding limit. Powder Technol. 2009;195:25–30. doi:10.1016/j.powtec.2009.05.007
20. Bartos C, Szabó-Révész P, Bartos C, Katona G, Jójárt-Laczkovich O, Ambrus R. The effect of an optimized wet milling technology on the crystallinity, morphology and dissolution properties of micro- and nanonized meloxicam. Molecules. 2016;21(507):1–11. doi:10.3390/molecules21040507
21. Ghosh I, Schenck D, Bose S, Ruegger C. Optimization of formulation and process parameters for the production of nanosuspension by wet media milling technique: effect of vitamin E TPGS and nanocrystal particle size on oral absorption. Eur J Pharm Sci. 2012;47:718–772. doi:10.1016/j.ejps.2012.08.011
22. Nijlen TV, Brennan K, Mooter GV, Blaton N, Kinget R, Augustijns P. Improvement of the dissolution rate of artemisinin by means of supercritical fluid technology and solid dispersions. Int J Pharm. 2003;254:173–181. doi:10.1016/S0378-5173(03)00009-7
23. Patel DD. Kinetics and mechanisms of crystal growth inhibition of indomethacin by model precipitation inhibitors. [doctoral thesis]. Lexington: University of Kentucky; 2015.
24. Haq N, Shakeel F, Alanazi FK, Alshora DH, Ibrahim MA. Development and validation of a ‘green’ RP-HPLC method for the analysis of rosuvastatin: a step towards making liquid chromatography environmentally benign. Green Proc Synth. 2018;7:160–169. doi:10.1515/gps-2017-0023
 © 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2019 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.


