Back to Journals » Clinical Ophthalmology » Volume 16
Defining the Clinical Role of Swept-Source Anterior Segment Optical Coherence Tomography in Eyes Undergoing Strabismus Surgery
Authors Pujari A , Patil V, Chauhan N, Thangavel R, Phuljhele S, Saxena R
Received 14 March 2022
Accepted for publication 26 April 2022
Published 5 May 2022 Volume 2022:16 Pages 1449—1465
DOI https://doi.org/10.2147/OPTH.S365622
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Amar Pujari, Vinay Patil, Nidhi Chauhan, Rajeswari Thangavel, Swati Phuljhele, Rohit Saxena
Dr. Rajendra Prasad Centre for Ophthalmic Sciences, All India Institute of Medical Sciences, New Delhi, India
Correspondence: Amar Pujari, Dr. Rajendra Prasad Centre for Ophthalmic Sciences, All India Institute of Medical Sciences, Room No. 212, RPC-1, AIIMS, New Delhi, India, Email [email protected]
Purpose: To describe the clinical role of swept-source anterior segment optical coherence tomography in extraocular muscle imaging in various strabismic conditions.
Study Design: Prospective observational study.
Participants: A total of 1104 extraocular muscles of 150 subjects were imaged. Normal eyes (640 muscles), eyes undergoing re-operation (240 muscles), paralytic strabismic eyes (16 muscles), non-paralytic strabismic eyes (20 muscles), post-traumatic strabismic eyes (64 muscles), eyes with thyroid disease (64 muscles), and other atypical cases (60 muscles) were imaged. In all cases, up to four recti muscles were evaluated in a given eye depending on the need.
Results: The mean age was 38.15 ± 16.25 years. Overall, muscle insertion was identified in over 90% of the subjects. Apart from normal muscles (n = 860, 93.68%), the location was identified in previously operated eyes (n = 54, 90%), in traumatic muscle slippage (n = 2, 11.11%), in ocular coat ectasia (n = 11, 91.66%), in thyroid muscle hypertrophy, in operated scleral fixated intraocular lens with strabismus, and other challenging cases. In all circumstances, clinically useful information was obtained from the anterior 4– 14 millimetres of the rectus muscle, which in turn helped in active surgical planning and overall management.
Conclusion: The SS-ASOCT with its wider and deeper scanning abilities was able to locate the extraocular muscle insertion in a variety of strabismic conditions. Therefore, this tool can be used in the preoperative periods for optimal surgical planning and management.
Keywords: swept source anterior segment imaging, extraocular muscle imaging, imaging in strabismus, ASOCT imaging of extraocular muscles
Introduction
Strabismus is a common condition in ophthalmology clinics.1 It primarily manifests as visual axis misalignment due to various causes, therefore, its correction is essential to achieve binocular single-vision, as well as for cosmetic reasons. Routinely, while planning a surgery, imaging is not necessary, however, in certain scenarios where muscle-specific information is required then imaging is inevitable. In this regard, the role of magnetic resonance imaging (MRI), computed tomography (CT) and ultrasound biomicroscopy (UBM) are being explored for more than two decades.2 The MRI and CT provide details with respect to muscle belly and its surroundings, nevertheless, they also provide dynamic changes along muscles whenever required. However, they rarely provide any information with respect to muscle location on the sclera or its distances from the angle/limbus, moreover, they are still expensive (per setting), and they need a significant amount of time for imaging and assessment. The third imaging modality, that is, UBM on the other hand does provide some information with respect to muscle location over sclera, but again, it has its own set of limitations including being a contact procedure and others (technical difficulties).3 Therefore, in strabismus subspecialty we need a non-contact, portable and more widely available cheaper imaging tool, which can show the local muscle details.
In recent times, a non-contact imaging tool seems to fulfill many of these criteria. The tool is anterior segment optical coherence tomography (ASOCT).4 It is an optical instrument, which works on different wavelengths (from 840 to 1140nm), and tries to provide cross-sectional details of the ocular coat and the muscle. Its inbuilt caliper further eases our efforts, where it tries to provide a definite scale for measurements from any defined landmark (such as anterior chamber angle/limbus).4
Previously, using swept-source anterior segment optical coherence tomography (SS-ASOCT) the identification of extraocular muscles including the medial rectus, lateral rectus, inferior rectus, and the superior rectus has been highlighted. By positioning the eye in abduction, adduction, sursum-duction and deorsum-duction gazes, all four recti can be imaged.5–7 Nevertheless, identification of muscle in operated eyes, and comparison of its abilities with UBM has also been highlighted.8 However, the ASOCT seems to possess much more potential than those described till now; hence, this article tries to fill this lacunae by actively exploring its ability in a variety of unusual case scenarios Moreover, it also provides detailed insights into common fallacies during imaging and active measures or recommendations to overcome the same.
Methods
To explore the clinical role of SS-ASOCT (CASIA-2, Tomey Corp, Nagoya, Japan) in various extraocular muscle disorders the index study was undertaken. The study was approved by the institution [All India Institute of Medical Sciences, New Delhi, India. IEC-83/07.02.2020] ethical committee for extraocular muscle imaging. Over a period of 20 months, the study assessed over 150 subjects (more than 18 years) with extraocular muscle disorders who underwent strabismus surgery for various reasons. Before imaging, from each subject, a written informed consent was obtained, and during study period, the study strictly adhered to the tenants of the declaration of Helsinki.
Patients undergoing extraocular muscle surgery for their ocular misalignment were primarily included (between 18 and 80 years). After general information and consent, the subjects were made to sit in front of the machine and were asked to position their chin along the chin rest. The ASOCT scanning was set into pre-operative cataract mode and imaging was initiated. To screen the medial rectus muscle, the eye was positioned in an abducted position, similarly, to screen the lateral rectus, superior rectus, and the inferior rectus muscles, the eye was positioned in adduction, depression, and elevated positions respectively. Scanning was initiated by identifying the anterior chamber angle on OCT screen. The horizontal (while imaging horizontal recti) and the vertical (while imaging vertical recti) scan lines were positioned between the center of the pupil and the anterior sclera, and then using the joystick, the sclera was screened till the fornices. In the end, the angle was seen on one side of the screen and the muscle on the other, leading to a perfectly positioned muscle in the centre of the scan.
The scan was captured when the muscle-specific hypointense layer was visualized from angle to insertion, the distance was then measured by placing a straight scale calliper between the two points. In case of difficulties due to extraocular movement restrictions, deep seated eyes, and patients not willing to consent, such eyes/subjects were excluded from the study.
Over a period of 20 months, a total of 80 normal subjects, 30 patients undergoing re-operation, 4 patients with paralytic strabismus, 10 patients with non-paralytic strabismus, 8 patients with post-traumatic strabismus, 8 patients with thyroid eye disease, 3 patients with ocular coat ectasia and strabismus, 5 patients with scleral fixed intraocular lens patients and strabismus, and 2 patients with suspected congenital muscle anomaly were imaged.
All scans were performed by the first author (AP) and intraoperative correlation was noted by three different authors. The subjects demographic details, ASOCT details, intraoperative features were entered in an excel sheet and the statistical analysis was performed using Stata software 12.1.
Results
Demography
The mean age of 150 subjects was 38.15±16.25 years. Amongst these, 81 were male and 69 were female. Bilateral imaging was performed in almost all the cases. Amongst the various indications, 918 muscles were normal, 60 muscles were in operated eyes, 4 muscles were in paralytic eyes, 20 muscles were in non-paralytic eyes, 18 muscles were in post-traumatic squints, 64 muscles were in eyes with thyroid eye disease, and 20 muscles in other conditions.
Muscle Identification: From Easy to Difficult
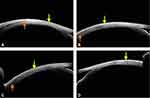
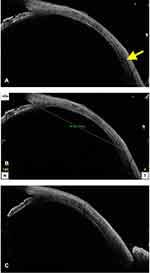
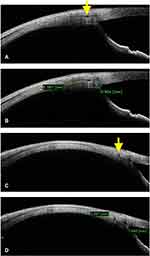
On ASOCT, a certain amount of clarity was necessary to visualize and to label the given rectus muscle. Upon initiation of longitudinal scanning for horizontal recti, the clearly visible hypointense layer within a defined distance from the angle was considered indicative of an extra ocular muscle. It was the anterior most portion or the insertional portion of the given rectus muscle. It usually began as a narrow hypointense layer at 50% depth and progressed posteriorly as a well-defined layer with increasing thickness (Figure 1). After a few millimeters, this hypointense layer assumed a much darker and thicker configuration due to its transition from tendon to belly portion; however, the exact point at which such transition happened was not clear in many cases.
Combined with this longitudinal scanning, the addition of cross-sectional scanning further eased our understandings on muscle anatomy. This cross-sectional scanning was nothing but a simultaneously acquired scan during the previous imaging, but it was at 90 degrees to it. In general, the longitudinal scan provided the lengthwise details and the cross-sectional scan the widthwise details. Hence, the combined scanning was more productive than the isolated scan.
Amongst four recti, the identification of the medial rectus anterior portion was most challenging (technically) than the rest. On the contrary, identification of lateral rectus >superior rectus >inferior rectus was quite easy. This was not just related to exposure alone; in fact, the best exposure was achieved for the medial and the lateral rectus muscles. Here, the presence of thicker and quite clearly visible initial hypointense layer (tendon) eased the identification of anterior most insertion in three of the recti as compared to medial rectus muscle. With respect to exposure, the lid retraction was not at all necessary for medial or the lateral rectus muscles, but it was mandatory for the superior and the inferior recti. But while doing so, the inferior forniceal conjunctival folds were frequent interrupters for inferior rectus, thus the farther posterior visualization was difficult in many vertical recti cases.
To identify medial rectus insertion, firstly, a full amount of abduction was necessary, and it was possible in almost all the cases (unless restrictions were present), however presence of conjunctival fold or plica semilunaris often hindered the deeper visualization (mostly in operated cases, due to projection artefacts). In many naïve cases, the insertion was so thin that it was very difficult to mark the exact insertion; thus, it sometimes led to false labelling just posterior to actual insertion. Therefore, amongst the four recti, the identification of medial rectus insertion was technically more challenging as compared to others, whereas, exposure wise vertical recti were more challenging.
Out of 918 (100%) normal muscles, easy tendon visualization was possible in 860 (93.68%) muscles, whereas in remaining 58 (6.32%) muscles, a varying amount of difficult was encountered. Which included, presence of very thin tendon in 32 (3.48%) muscles (mostly the medial rectus) and deep-seated eyes with limited view in 26 (2.83%) muscles (mostly the vertical recti).
Length and Width Visualization
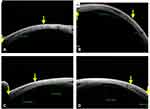
On ASOCT, with good duction movement and exposure, we were able to visualize a significant portion of any given recti. Along the medial rectus muscle, we were able to visualize up to 9 millimeters of its anterior length, for lateral rectus up to 14 millimeters, for inferior rectus up to 6 millimeters and for superior rectus again up to 5 millimeters of anterior lengths were visualized. (Figure 2) This possibility is important, because while using ASOCT alone we were able to visualize up to 15–30% of muscle length, which is the universally operated portion during all squint surgeries. Moreover, the cross-sectional scanning further aided the full width imaging of the muscle. Hence, the combined (longitudinal and the cross sectional) imaging augmented the understandings with respect to muscle presence and its orientation.
Tendon and Belly Identification
As mentioned above, the horizontal scanning of the horizontal recti and the vertical scanning for the vertical recti allowed antero-posterior visualization of the muscle. The initial portions of the lateral rectus, inferior rectus, and the superior rectus were well-defined, whereas such appearances were subtle while imaging the medial rectus muscle (Figures 1 and 2).
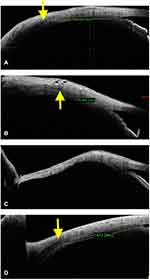
In general, the tendon appeared as a less thick hypointense layer at insertion, which then progressed posteriorly with much thicker and darker configurations. This was quite clear along all recti, but it was often more drastic along medial rectus muscle. Furthermore, in some cases, a reversal was noted, that is, a much thicker lateral rectus tendon was noted than its belly. (Figures 1 and 2) (Figures 3A and 4A, B) Hence, on ASOCT, a possible judgment regarding the location of tendon and belly can be made, but in some cases, it is better to be skeptical than over-assuming the location.
Utility in Re-Operations
In normal eyes, the ocular coat (including conjunctiva, sclera, and the uveal tissue) usually showed a regular contour. That is, it will be a perfect arc of ¼ of a circle with muscle at its 50% depth (Figures 1 and 2). Whereas in operated eyes these features are lost, and hence, the coats assume a thinner undulating configuration with variable surface scar (more in recessions). Under such circumstances, again the muscle identification process is challenging. In a recessed muscle, the tendon is implanted at a new site, and in a resected muscle, the belly is implanted at the old tendinous site. Hence, while imaging in operated eyes, a different set of understandings are must (this can be obtained from first authors our previous work, ref,13).
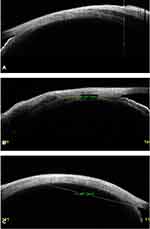
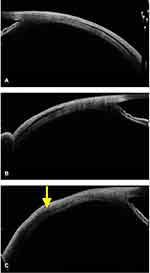
In a recessed muscle, the original muscle stump usually appears as a thick outward bump with thin undulating ocular coat thereafter (more along lateral rectus than medial rectus). (Figures 3B and 4C) Depending upon the amount of recession, the tendon can either be visualized within a notable distance (small recession 5–7 mm) or it may not be visualized till the fornices (more than 7 mm recession). In some cases with small recessions, the anterior tendon has a slightly more bulk as compared a naïve tendons, which can be attributed to muscle folding/piling at new insertion site.
In a resected eye, the muscle is noted at the original insertion site with a thick overall configuration. Again, this is due to the replacement of tendon by the muscle belly after its resection (Figures 3B, 4C, 5A-C and 6A).
Based on these features, we were able to predict the nature of surgery in around 90% (n=54) of the muscles (n=60), and based on which, we planned re-operation in the contralateral eye in presence of under-corrections (provided the primary eye is optimally operated) or the same eye in presence of over-corrections. However, in some 6 (10%) muscles, the characterization was quite difficult due to overlying scar (n=5), or due to restricted movements (n=1), in these cases, a detailed exploration was planned to achieve good correction.
Utility in Post-Traumatic Strabismus
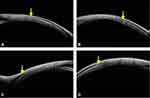
In strabismic eyes with a previous history of trauma, the routine MRI and CT were only be able to unveil the muscle belly and the surrounding bony changes, however, the muscle location on sclera was not possible. In these cases, the ASOCT was able to decode the location to a noticeable extent. In 8 subjects with history of trauma, a total of 18 (100%) muscles were imaged [(n=9 injured (50%), and (n=9) (50%) normal muscles for comparison]. In presence of belly injuries (n=7 muscle, 38.88%) the insertion appeared intact with only limited surface changes, whereas, in traumatic dis-insertions (n=2, 11.11%), the tendon was absent at the insertion with bare ocular coat (Figure 3C and 6B, 6C). This ASOCT knowledge was useful in surgical exploration/planning in those cases, and it was also useful in positive patient counselling.
The traumatic cases in here were of longer duration (6 months to 4 years), and had history of penetrating ocular injuries with variable diplopia. The cases were managed as per our hospital protocols, and the ASOCT was able to provide key information with respect to injured muscle.
Utility in Thyroid Eye Disease
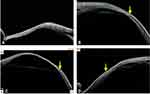
In thyroid eye disease, the cross-sectional images showed the changes along muscle tendon as well as belly. Till now, the B scan ultrasound, CT, and MRI have been used to characterize the muscle size, however, the ASOCT in presence of axially protruded globe was specially useful in visualizing the anterior portion of the muscle. The proptosis exposed a large scleral area; hence, the scanning was easy and quick in these eyes (provided restrictions were less). The thickened muscle belly and its tendon were clearly visualized, nevertheless, the thickness measurements were also attempted to monitor the patients who are on treatment (however, this needs some more time and observation to perfectly justify it). In our observation, we attempted 64 (100%) muscle imaging in 8 subjects, out which only 56 (87.5%) muscles clearly visualized (in rest, a thorough assessment was limited due to variable movement restrictions) (Figure 7).
Utility in Ocular Coat Disorders
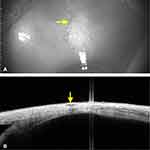
The other important indication was imaging in ocular coat ectatic disorder or staphylomatous eyes. These cases had a primary retinal problem and later developed sensory exotropia, who wished to get corrected cosmetically, however, previously we denied surgery due to the critical location of ectasia and subsequent risk of uveal tissue prolapse. But with the ASOCT in our armamentarium, we evaluated 3 subjects with staphyloma and tried to image 12 (100%) muscles. Out of these, 11 muscles (91.66%) were closely related to the ectatic site but not within it, hence, they were planned for surgery with a successful outcome through fornix-based approach. In one case, the muscle was absent or it had been lost. Therefore, the ASOCT was able to delineate the extent of ectasia, the location of muscle insertion, and the distance between the two quite accurately. In Figure 8A-D, it can be noted that the muscle insertions lie well away from the ectatic margins, thus they were handled with fewer chances of uveal tissue prolapse. In all these cases, the stretched ocular coat had pushed the muscles more posteriorly than actively pulling them. This ASOCT guided safe evaluation and the reaffirmation of negotiable ocular coat status helped in positive surgical planning and execution in all cases.
Utility in Surgical Planning Under Rare Circumstances
The other rare scenarios where the ASOCT guided knowledge was necessary were, 1) the eyes with sensory exotropia having scleral fixated intra ocular lens (SFIOL) in situ, and 2) the eyes with suspected congenital rectus muscle anomalies.
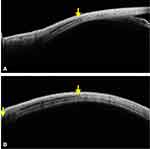
Previously, while operating on an eye with SFIOL in situ, a serious haptic damage was encountered, hence from that time onwards we undertook an ASOCT guided evaluation in all eyes. In a total of 6 subjects who have undergone SFIOL, a corrective strabismus surgery was planned. A total of 16 muscles (and 25 contralateral muscles) in and around the previous scars were evaluated (12 horizontal recti due to the proximity of haptic and 4 inferior recti due to extending scar). The location of the haptic, its proximity with the muscle and its depth were assessed (Figures 9 and 10). In all cases (12 muscles or 100%), the relation between haptic and the muscle were thoroughly studied. Based on this a successful surgery was executed with no untoward complication in any of the case.
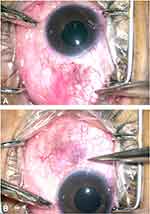 |
Figure 9 In a subject with operated SFIOL, a significant amount of conjunctival scarring can be seen along the nasal (A) and the temporal (B) bulbar surfaces. |
In one subject with suspected inferior rectus hypoplasia/aplasia, the ASOCT was able to identify the muscle features at its insertion. Including inferior rectus muscle, the horizontal recti were also imaged in the same eye, and all four recti in the contralateral eye to co-relate the anatomy and to confirm the positions. The muscle features were normal at insertion (Figure 6D), and subsequently, the MRI revealed reduced muscle bulk. The normal insertional features were confirmed during intraoperative exploration. Hence, it was a likely case of primary inferior rectus palsy rather than the hypoplasia/aplasia.
Discussion
The role of ASOCT in identifying extra ocular muscles is being explored since nearly a decade. Its ability to take measurements in normal eyes and its correlation with intraoperative distances has been highlighted on many occasions.9–12 However, a better justification for ASOCT use in strabismus can be made if it’s role in the active diagnosis and/or management is defined. In this direction previously, we have described the role of SS-ASOCT in identifying the nature of extraocular muscle surgery in cases with lost surgical details.13 Where we noted that the ASOCT did provide novel information with respect to the nature of surgery, possible amount of surgery, and the options for subsequent planning. Moreover, the procedure was quick and non-contact with wider imaging facilities, therefore, as of now it seems to be a good additive tool to the traditional ultrasound biomicroscope/CT/MRI.
Apart from re-surgeries, the ASOCT can also be used in other cases as highlighted in the results section. Over a period of 20 months, we imaged a variety of cases where the localization of extra ocular muscle was essential. Which included ocular coat ectasia, post traumatic squints, thyroid disease associated muscle hypertrophy, operated cases of SFIOL, congenital muscle anomalies and others. In all these cases, ASOCT was able to provide necessary insights which were earlier thought to be not possible. In many conditions, even in presence of routine CT and MRI, the ASOCT was able to provide the crucial information, which other imaging modalities failed to reveal, and most importantly based such information a feasible surgical approach was possible. Thus, ASOCT seems to be useful in educating the patients, as well as in minimizing the “explorative surgical approaches”.
The current exercise re-enforces that the ASOCT is quite useful in many strabismic conditions, and hence the justification to use this tool whenever required (more so if a particular set of patient load is high). Nevertheless, in absence of this tool with a strabismus surgeon, a collaboration with the anterior segment surgeon can be sought, just to use whenever necessary.
Till now no large-scale or longitudinal study has highlighted the role of ASOCT in strabismic conditions as the index study does, hence through our experiences and errors, we would like to emphasize the common fallacies, which we faced during imaging. Moreover, we would also like to propose few recommendations to maximize the OCT benefits in extraocular muscle imaging.
Common Errors and Fallacies in Muscle Identification
While imaging, a word of caution on following pitfalls is must.
- Postoperatively, the muscle imaging needs to be cautiously judged till 4 weeks. Because the muscle space often erroneously appears as large hypointense layer, which settles over a period with the resolution of edema and teno-conjunctival flattening.
- The large caliber conjunctival vessels can erroneously be judged as muscle areas. This can be avoided by carefully observing the muscle-specific characteristics (Figure 11).
- In presence of dense conjunctival folds or scars (more so along the medial rectus), the muscle-specific identities can be misleading. Hence, here only a presumption can be made, and more definite conclusions are better avoided.
- The identification of thin tendon/belly can be difficult sometimes, hence, in these cases, a co-relational scan between the longitudinal and the horizontal scans can be considered for better clarity.
- Naïve surgeons can falsely label the subconjunctival dark areas as muscle. Which can be avoided by examining them more clearly by noticing the focal and dis-continuous nature of the artifacts.
Recommendations for Muscle Imaging
Full Muscle Length and Width Scanning
As we know, the horizontal recti have vertical insertions, and the vertical recti have horizontal insertions. Therefore, while identifying any rectus insertion, a thorough end-to-end scanning is necessary. This can be achieved by selecting the 360-degree (vertical as well as horizontal) scanning mode under pre-operative cataract protocol. Under this, the horizontal scan line provides a lengthwise details, and the vertical scan line provides the cross-sectional details (with respect to horizontal recti, vice-versa for vertical recti) (Figures 1 and 2). Furthermore, a raster scan under bleb mode can also be used to assess the cross-sectional details.
Correlational Judgement Between Longitudinal and the Vertical Scans
The next task is to derive maximum information from the above two scans combined. Before freezing the images, the joystick can be moved vertically as well as horizontally to visualize the muscle in real time. This basically provides a cumulative information which can otherwise be missed or under-visualized on a single scan or a frozen scan. In addition, this also accurately dictates the depth of the muscle and thus avoids the misinterpretation due to accessary subconjunctival spaces (Figure 12).
Identification of Muscle Depth
The third step is the visualization of ocular coat in its entirety. Its thickness usually spans more than 10 millimeters (from conjunctival surface to inner scleral/choroidal surface), and the muscle-specific hypointense layer often lies at the midway or around half of its depth. This is important, because, as we move from limbus to fornices, the overall ocular coat thickness increases, hence, what we identify as muscle can be very superficial at the insertion and will be deep posteriorly. Moreover, the identified muscle must be traceable anteriorly as well as posteriorly as a single continuous layer (Figures 1 and 2).
Measurement from a Fixed Anatomical Landmark
Once the muscle is identified and confirmed, then its location needs to be measured from a fixed anatomical landmark. The measurement must be replicable by all and should be readily visible. In this regard, the angle seems to be more reliable with visually convincing fixed features. It is easily identifiable and most importantly it hardly has any differences with the outer limbus readings (Figures 1-6 and 8).
Therefore, pre-op distances can be measured from the angle>limbus and intraoperatively the same can be correlated with the outer limbus.
Controlled Interpretation Using Contralateral Eye
Once the identification, confirmation and the measurements are done, then it needs to be verified with the contralateral eye or the best control eye. The appearances, measurements, and other tissue characters can be compared here in the operated eyes and post-traumatic eyes, so that the original location can be confirmed, and subsequent surgery can be planned.
Limitations
The limitations of our study were, 1) the whole observations were made by only few surgeons (mainly first author), and many were subjective. However, in presence of frankly visible muscle details the disagreements were less. 2) The appearances of the medial rectus tendon were quite unusual, which could be attributed to excessive abduction stretch (of the relaxed medial rectus); however, it is less likely, because such features were absent while scanning the lateral rectus or the rest. 3) The limbal and the angle measurements may differ, but the differences seem to be less than 1 mm. In fact, in some cases, the muscle presence and its rough estimations were more important than the perfect readings. 4) As the current technology is only limited to cross-sectional imaging, the actual enface mappings14 were genuinely lacking in our study.
Conclusions
To conclude, the SS-ASOCT with its advanced scanning abilities can be used to locate the extra ocular muscles locally. In normal eyes, in operated eyes with or without surgical details, in post-traumatic eyes, in thyroid eye disease, in ocular coat disease, and in other conditions where muscle details were essential they were imaged using ASOCT, to get the reasonable information with respect to its presence or absence and its measurements. Thus, in future, this cross-sectional imaging tool can reliably be used as an interrogative tool or a surgical guide while planning surgery in routine as well as complex case scenarios.
Acknowledgment
Accepted for poster presentation at ARVO 2022.
Disclosure
The authors report no conflicting interests in this work.
References
1. Hashemi H, Pakzad R, Heydarian S, et al. Global and regional prevalence of strabismus: a comprehensive systematic review and meta-analysis. Strabismus. 2019;27(2):54–65.
2. Patel JR, Gunton KB. The role of imaging in strabismus. Curr Opin Ophthalmol. 2017;28(5):465–469.
3. Dai S, Kraft SP, Smith DR, Buncic JR. Ultrasound biomicroscopy in strabismus reoperations. J AAPOS. 2006;10(3):202–205.
4. Pujari A, Agarwal D, Sharma N. Clinical role of swept source optical coherence tomography in anterior segment diseases: a review. Semin Ophthalmol. 2021;1:1–8. doi:10.1080/14.08820538.2021.1897854
5. Hou LJ, Wu XM, Zhuo R, Zhang F, Lyu F. [The application of anterior segment optical coherence tomography in residual strabismus surgery]. Zhonghua Yan Ke Za Zhi. 2016;52(8):568–573. Chinese.
6. Ngo CS, Smith D, Kraft SP. The accuracy of anterior segment optical coherence tomography (AS-OCT) in localizing extraocular rectus muscles insertions. J AAPOS. 2015;19(3):233–236.
7. Pihlblad MS, Reynolds JD. Anterior Segment Optical Coherence Tomography of Previously Operated Extraocular Muscles. Am Orthoptic J. 2017;67:61–66.
8. Kaur S, Sukhija J, Korla S, Sachdeva K, Chaurasia S, Raj S. Comparison of the swept-source anterior segment optical coherence tomography and wide-field ultrasound biomicroscopy for imaging previously operated horizontal extraocular muscles.. J AAPOS. 2021;25(4):
9. Saffren BD, Yassin SH, Thau A, Nelson LB, Schnall B, Gunton KB. Anterior Segment Optical Coherence Tomography in Locating the Insertion of Horizontal Extraocular Muscles After Strabismus Surgery. J Pediatr Ophthalmol Strabismus. 2021;58(1):62–65.
10. Pihlblad MS, Erenler F, Sharma A, Manchandia A, Reynolds JD. Anterior Segment Optical Coherence Tomography of the Horizontal and Vertical Extraocular Muscles With Measurement of the Insertion to Limbus Distance. J Pediatr Ophthalmol Strabismus. 2016;53(3):141–145.
11. Rossetto JD, Cavuoto KM, Allemann N, McKeown CA, Capó H. Accuracy of Optical Coherence Tomography Measurements of Rectus Muscle Insertions in Adult Patients Undergoing Strabismus Surgery. Am J Ophthalmol. 2017;176:236–243.
12. Pihlblad MS, Troia A, Tibrewal S, Shah PR, Pre- I. Post-Operative Evaluation of Extraocular Muscle Insertions Using Optical Coherence Tomography: a Comparison of Four Devices. J Clin Med. 2019;8(10):1732.
13. Pujari A, Modaboyina S, Das D, et al. Clinical Role of Swept Source Anterior Segment Optical Coherence Tomography in Strabismus Re-Operation with Lost Surgical Details. Clin Ophthalmol. 2021;15:3581–3591.
14. Shibata K, Fujiwara A, Hamasaki I, et al. Shape analysis of rectus extraocular muscles with age and axial length using anterior segment optical coherence tomography. PLoS One. 2020;15(12):e0243382.
 © 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2022 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.