Back to Journals » International Journal of Nanomedicine » Volume 11
Current application of phytocompound-based nanocosmeceuticals for beauty and skin therapy
Received 20 January 2016
Accepted for publication 24 February 2016
Published 11 May 2016 Volume 2016:11 Pages 1987—2007
DOI https://doi.org/10.2147/IJN.S104701
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Thomas Webster
Palanivel Ganesan,1,2 Dong-Kug Choi1,2
1Department of Applied Life Science, Nanotechnology Research Center, 2Department of Biotechnology, College of Biomedical and Health Science, Konkuk University, Chungju, Republic of Korea
Abstract: Phytocompounds have been used in cosmeceuticals for decades and have shown potential for beauty applications, including sunscreen, moisturizing and antiaging, and skin-based therapy. The major concerns in the usage of phyto-based cosmeceuticals are lower penetration and high compound instability of various cosmetic products for sustained and enhanced compound delivery to the beauty-based skin therapy. To overcome these disadvantages, nanosized delivery technologies are currently in use for sustained and enhanced delivery of phyto-derived bioactive compounds in cosmeceutical sectors and products. Nanosizing of phytocompounds enhances the aseptic feel in various cosmeceutical products with sustained delivery and enhanced skin protecting activities. Solid lipid nanoparticles, transfersomes, ethosomes, nanostructured lipid carriers, fullerenes, and carbon nanotubes are some of the emerging nanotechnologies currently in use for their enhanced delivery of phytocompounds in skin care. Aloe vera, curcumin, resveratrol, quercetin, vitamins C and E, genistein, and green tea catechins were successfully nanosized using various delivery technologies and incorporated in various gels, lotions, and creams for skin, lip, and hair care for their sustained effects. However, certain delivery agents such as carbon nanotubes need to be studied for their roles in toxicity. This review broadly focuses on the usage of phytocompounds in various cosmeceutical products, nanodelivery technologies used in the delivery of phytocompounds to various cosmeceuticals, and various nanosized phytocompounds used in the development of novel nanocosmeceuticals to enhance skin-based therapy.
Keywords: nanodelivery technologies, skincare, nanophytocompounds, nanophytocosmeceuticals, nanotherapy
Introduction
Cosmeceutical research is a rapid growing area in personal care sectors, which extends from facial products to skin and body products.1–3 A forecasted report suggested that by 2018, the global market for cosmeceuticals will reach ~42 billion dollars and lead to a great demand in near future.4 Even though various synthetic compounds are currently available in the world market, human toxicity is increasing with cosmeceutical interference in certain treatments.5–8 Alternatively, plant-derived natural, bioactive compounds show enhanced beauty roles, along with health benefits against diseases.9–15 Plant-derived bioactive compounds have been used for centuries in cosmeceuticals for a wide variety of beauty treatments for the skin, face, lips, hair, and nail care with beneficial actions against photoaging, inflammation, hair loss, lip care, psoriasis, and ultraviolet (UV) toxicity.13–21 Further, UV radiation causes skin tanning and endothelial cell necrosis and suppresses immunological functions. Some natural phytobioactive compounds can prevent these deleterious effects with skin care.20,22–25
Phytobioactive compounds used in cosmeceuticals include catechins, gallic acids, epicatechins, curcumin, hydroxylbenzoic and cinnamic acids, quercetin, ascorbic acids, luteolin, alpha and beta carotene, complex polysaccharides, and fatty acids.26–31 These compounds, in addition to their cosmetic effects, enhance antibacterial, antifungal, anticarcinogenic, and anti-inflammatory biological actions.31–37 Even though various macrosized phytobioactive compounds are used in cosmeceutical formulations, their solubility and formulation type have limitations in enhancing the effect of phyto-based cosmeceuticals and therapy.38–41 The major limitations of phyto-based cosmeceutical therapy include less skin penetration, lower prolongevity, less final quality and lower whitening effects. These qualities depend on the solubility and size of the active phytocompounds.40,42 This leads us in search of novel, highly promising technologies for enhanced skin health efficiency of cosmeceutical products.
Nanotechnology solves most issues with higher protective and skin health-enhancing efficiency. Various nanotechnology methods are involved in active roles in beauty enhancement, such as nanoemulsions, liposomes, solid lipid nanoparticles, smaller sized nanoparticles, hydrogels, and dendrimers.4,43,44 Commercially, many synthetic compounds are used in nanocosmeceuticals that cause toxicity and skin health concerns. Natural phytocompounds are in a greater demand.45–47 Nanoscale phytocompounds used in cosmeceutical products are gradually increasing among products such as sun screens, antiaging, and UV protectants.47–56 Fewer phyto-based cosmeceutical nanoproducts available in the market are reviewed, namely, vitamin E in acne lotion, tea extracts in nanoshampoo, green tea extracts in the ageless skin care miracle kit, and vitamin E and aloe in UV protection moisture lotion. These phyto-based cosmeceutical nanoproducts contribute to the higher efficacy of the products mentioned for skin-based nanotherapy.57,58
Recent studies suggest that phyto-based nanocosmeceuticals will play a greater dual role in the near future for their enhanced protective, beauty, and health benefits.4,43,44,59 Therefore, the current review focuses on the different types of nanosized cosmeceuticals, current nanotechnology usage in the development of nanosized compounds or carriers for the delivery of phytobioactive compounds, and various nanosized phytobioactive compounds used in cosmeceutical therapy for their enhanced skin therapeutic applications.
Phytoconstituent-based nanocosmeceuticals for skin therapy
Nanoantiaging
Aging is the critical factor that causes skin collagen to lose its physical appearance in many ways such as oil production decrease, dry skin, texture loss, age spots, and loose skin. Aging causes skin thinning, and the end result is wrinkle formation.60,61 This is not limited to the aged population; younger individuals exposed to various harsh environmental conditions such as infrared and ultraviolet rays, chemical pollution, and other physical stresses can also be affected.62,63 Currently, various synthetic and natural antiaging creams using nanosize technology are available on the market with enhanced skin protection to moisturize, lift, and whiten skin.64–68 Recently, Lohani et al4 reviewed some nanoproducts produced by few companies for enhanced beauty, such as Hydra Zen Cream, Revitalift, and Skin Caviar Ampoules that contain phytocompounds, using various nanotechnologies. Nanosized phytobioactive compounds, such as curcumin and vegetable oils, show enhanced skin appearance by various antioxidative mechanisms. The aforementioned products prevent skin aging due to oxidative stress and premature aging.69–73 The combination of polyphenols with retinols enhances skin protection. Other than nanosizing, few delivery systems have been used to develop nanoantiaging creams and lotions for the enhanced activity of bioactive compounds to deeper layers of skin. Recently, a nanoemulsion system was constructed using Eysenhardtia platycarpa leaf flavanones with 70 nm nanoparticles that enhanced antiaging activity. Polymeric nanoparticles of E. platycarpa leaf flavanones with the particle sizes between 156 nm and 202 nm also showed the enhanced antiaging effects.74 In addition, coencapsulation of resveratrol and curcumin showed enhanced delivery of phytocompounds for disease treatment with their antioxidant activities and thereby delays aging effects.75,76
Nanomoisturizers
Skin dehydration can be prevented by moisturizers, which support skin beauty and enhance skin flexibility by maintaining moisture.77 A moisture-preventing barrier on the skin is the stratum corneum, which maintains the skin moisture. Dehydration results in the abundant loss of water from the skin.78 Moisturizer creates a thin film on the outer surface of the skin maintaining skin moisture and can be effectively achieved by various nanotechnological systems.79 Currently, nanoemulsions, nanoliposomes, and solid lipid nanocarriers are effectively used in moisturizer formulations with active phytoingredients and enhance the skin’s beauty.79,80 Further, skin hydration is effectively enhanced with prolonged activity achieved by the solid lipid nanoparticle systems with low viscosity and nongreasy effects.80 Currently, many phyto-based nanomoisturizing delivery systems are in starting phases of development. Among them, safranol was used in the development of solid lipid nanoparticles with enhanced moisturizing effects, along with UV protecting activity. In this system, the minimum nanoparticle diameter was 100 nm.80 Similarly, rice bran oil nanoemulsions were also developed with enhanced moisturizing effects to improve various skin diseases, including atopic dermatitis and psoriasis.69 Opuntia ficus-indica (L.) extract mill nanoemulsions were developed with varying particle size from 92 nm to 233 nm and had enhanced moisturizing effects.79 Currently, various vegetable oil nanoemulsions were studied for their potential use as a moisturizer product. In the future, vegetable oils will be a great alternative for enhanced moisturizing effects for dry skin-related diseases.69
Nanoskin cleanser
Skin cleanser plays an active role in maintaining skin health.81,82 A thin hydrolipid film covers the skin containing cellular debris from the decomposition products of corneocytes and secretions from eccrine, sebaceous, and apocrine glands. Hydrolipid films on the skin surface may attract undesirable microorganisms, which in turn accelerate the odor production with the metabolized products from the sweat glands.83 Skin cleaning directly promotes the removal of skin surface bacteria, which reduces odor production. Currently, skin cleaning products are available on the market from macro- to nanosize containing both synthetic and natural products.84,85 Research is being performed on several phyto-based nanocompounds for enhanced skin cleaning. Recently, phyto-based extracts used in cleansers showed enhanced skin cleansing activity by reducing pore size and skin oil content. However, they are only in the budding stage. The effectiveness of using phyto-based compounds in skin cleansing and related to skin disease such as acne86 is well studied. Similarly, nanosized liposomal lauric acid was constructed in the size range of 113 nm and showed enhanced antimicrobial activity against the acne.87 Another study, using niosomes by combination of lauric acid and curcumin, showed enhanced antimicrobial activity against acne-caused skin infections.88 These studies suggest that phyto-based nanotechnologies show enhanced effects on skin cleanser activity on skin disease. Further, phyto-based nanotechnologies will pave the way for the development of skin cleanser products with phytocompounds with enhanced skin cleaning activity.
Nanosunscreens
Sunscreens protect the skin from the harmful sun rays exposure.89 Commercially, sunscreens available are in the forms of creams and lotions that contain synthetic compounds, which act as a barrier when the harmful rays hit the skin. Sunscreens prevent deep UV ray penetration and inhibit irritation and other consequences. However, synthetic sunscreens have disadvantages, including formation of a chalky layer, greasiness, smelliness, decreased appeal, and toxicity.90 A few of the mentioned disadvantages are overcome by use of natural phyto-based active ingredients. Recently, Bulla et al91 studied the usage of phyto-based bioactive compounds derived from Schinus terebinthifolius Raddi in sunscreens and found that they enhance antioxidant and sun blocking activities. Similarly, bioactive-rich dried extracts of Brazilian Lippia species showed higher photoprotective activity.92 Even though phytobioactive compounds show increased sunscreen effects, their stability and skin protective effects were greatly enhanced by using nanotechnology delivery systems in the sunscreen cream and lotions.73,93,94 Recently, saffranol was constructed using solid lipid nanoparticles and had enhanced sun screening activity in the size range of 103–233 nm.80 Similarly, nanostructured lipid carriers were constructed using rice bran oil and raspberry oil and had enhanced sunscreen activities with higher antioxidant and UV protective activities.95 In another study, phytoflavanoid-encapsulated poly(lactic-co-glycolic) acid nanoparticles were constructed with the particle size of 90–100 nm that had enhanced sunscreen activity along with increased antioxidant activity. In another study, a pomegranate seed oil nanoemulsion was constructed with various nanoparticle sizes and showed increased skin protection against photodamage with enhanced antioxidant activities.96 The same research group also studied the effect of pomegranate seed oil nanoemulsions on human erythrocytes and found increased photosafety of lipid skin membranes.97 The earlier research suggests that the development of novel nanosized phytocompounds will enhance sunscreen products with fewer toxic effects.
Nanohair care
Hair care is the most promising cosmeceutical sector. People of all ages are concerned about their hair. Hair loss may occur at any age. Hair loss conditions are associated with scalp inflammation and growth disorders, which vary with the individual.98–101 This affects the social status of individuals due to changes in physical appearance of the increased hair loss by vellus follicles transformations.102 Several plant-derived phytobioactive compounds are extensively used in the hair care products.103–105 Recently, phytochemicals from rice and artichokes are used to enhance the hair growth, color, and appearance and protect hair from UV-induced degradation.104 Similarly, pomegranate hydroethanolic extract was used in the reduction of the hair follicle red fading effect and enhanced hair color retention.103 The mixture of phyto-based bioactive compounds also showed enhanced hair growth in the anagen phase. Along with hair growth, the phyto-based bioactive compound mixture protects from hair loss and breaking. Recently, a nanocosmeceutical-based approach of promoting hair growth was achieved using delivery technologies, such as niosomes and liposomes, to hair follicular cells.106–113 Curcumin-based liposomes were constructed and had 70% enhanced penetration of the curcumin to the hair growth, irrespective of liposomes type in the size range of 213–320 nm.114 Similarly, a curcumin-based cyclodextrin complex also showed enhanced follicle penetration and hair growth enhancement.115 In addition, the combination of polyarginine and oleic acid-modified nanoparticles in size range of 140–143 nm showed enhanced skin penetration of follicular or nonfollicular cell pathways.116 These studies confirm that higher penetration of bioactive molecules from the plants can be enhanced by nanotechnology to develop better hair growth, disease treatment, and beauty therapies.
Nanonail care
Nanonail care is a promising novel cosmetic and health care sector. Nanonail care particles are developed using synthetic particles and show increased protective effects against nail infections. This treatment increases nail glow and toughness in mammals.43,44 However, the use of synthetic nanoparticles has a decreased market impact compared to natural products. Therefore, the demand of phyto-based nail care products is gradually increasing. Natural, plant-based pigments mostly consist of phytobioactive compounds and have various therapeutic effects. Bioactive compounds from plants are rich in color such as carotene (orange color), chlorophyll (green), and curcumin (yellow), lycopene (reddish orange), anthocyanin (red/purple), and annatto (yellow/orange). These compounds were used in the traditional practices of mixing colored extracts with honey, ghee, or coconut oil for skin and nail care.117,118 These bioactive compounds have antibacterial, antifungal, antiviral, and antioxidant health enhancing effects. Even though these compounds are used in foods and cosmetics at the macro- or microlevels, the use of the natural pigments at the nanolevel is limited. Recently, asynthetic nanoemulsion was developed with enhanced antifungal activity against nail infections.119 Turmeric and henna are rich in phytobioactive compounds, and their role in the nail care is well known. However, the role of nanosized turmeric and henna bioactive compounds remains unstudied. Further, during nanosizing, the colors and their effects may enhance or change, which will give future researchers a broad range of the nanonail care cosmetics to study.
Nanolip care
Nanolip cosmetics are another growing nanosector. Currently, various synthetic and metal particles are used at the nanosize for effective lip protection, enhanced color. These products last longer and have a homogenous distribution.4 The use of nanometal particles enhances the even distribution of pigments over the lips to increase protection. Since lips are most vulnerable skin-type, caution is necessary when using synthetic and metallic nanoparticles. Generally, phytoconstituents are used in the lip care at the macro- and microlevels such as coconut oil, curcumin, and other bioactive compounds that have increased protective activity.120,121 Recently, plant betalains, a novel dietary-colored indole-derived pigments, were efficiently used in the lip care products and the extraction of those compounds and their encapsulation enhances storage stability122–124 and applications. Bixa orellana was used as a colorant for lip jelly and was found to have higher organoleptic lipstick properties. Similarly, propolis was also used in the development of a novel lipstick. However, the use of nanotechnology in phyto-based lipstick development is still in the budding stage.
Phytoconstituent-based nanodelivery systems in cosmeceuticals for beauty and health care
Limitations of synthetic bioactive ingredients used in cosmeceuticals increase the demand of nanosized phyto-based bioactive ingredients that can be applied by nanodelivery systems.64,69,80,93 Nanodelivery systems enhance the solubility of the active phytobioactive compounds and penetrate through the skin. Penetration enhancement of active phyto-based ingredients was achieved through suitable nanodelivery techniques that led to increased cosmeceutical effectiveness.50,51,96,110,125,126 In addition, phyto-based bioactive ingredients solve most skin diseases, and thereby, nanotechnology enhances the therapeutic value of dual role cosmeceuticals. Further, nanoparticle formulation increases retention of the product on the outer skin layer. Various nanodelivery techniques used in the formulation of cosmeceuticals are of solid lipid nanoparticles, nanostructured lipid carriers, liposomes, phytosomes, nanoemulsions, nanoniosomes, nanocapsules, and nanolipospheres.4,57,70 Solid lipid nanoparticle-loaded resveratrol showed enhanced permeation in the porcine skin.127 Similarly, colloidal silica emulsion loaded with quercetin also showed enhanced human skin permeation owing to the nanosizing of the compounds.128 Even though various nanotechnology systems enhanced deeper penetration of phytocompounds that are currently available in cosmeceuticals, technology usage depends upon the type of bioactive ingredients, such as hydrophilic or hydrophobic, gel or emulsion, size, degradability, and toxicity. In this section, we discuss the type of nanotechnology delivery techniques used for the development of phytobioactive compound-based nanocosmeceuticals.
Solid lipid nanoparticles
Solid lipid nanoparticles are among the nanotechnologies extensively used in both pharmaceuticals and cosmeceuticals.45,48,49 Commercially, many cosmeceutical companies produce cosmeceuticals using this technology to enhance beauty and therapy. This nanoparticle system developed from microemulsion systems using solid lipids such as stearic acid, cetyl, and palmitic acid along with plant-derived phytocompounds such as flavonoids, phenolic acids, and other active compounds. Solid lipid nanoparticles have advantages in stability, higher production, and long-term storage compared to other conventional and nanodelivery techniques.44,50,80,110 Solid lipid nanoparticles also boost the cosmeceutical efficiency by enhancing properties that includes hydration, lower uptake of the systemic circulations, therapeutic values, prolonged, and sustained release in the skin and hair. Recently, sesamol-loaded solid lipid nanoparticle was constructed with the particle size of 127 nm for usage in skin cream, which increased the apoptotic nature of the skin.129 Vitamin A-loaded solid lipid nanoparticles were constructed with the particle size of 200 nm and increased skin moisture retention and skin occlusive effects in porcine skin.130,131 Similarly, curcumin, another bioactive compound, was constructed using solid lipid nanoparticles with the particle size of 210 nm. This packaging of curcumin showed enhanced activity in hydrogels prepared for inflammation treatment of porcine skin.132 However, some solid lipid nanoparticles showed a higher particle size with the addition of quercetin, an average of 574 nm. This size can be reduced with the addition of silica, decreasing the average size to 483 nm causing enhanced skin penetration of quercetin with higher photostability and skin protection.128,133 In another study, caffeine-loaded solid lipid nanoparticles were constructed, with a particle size of 182 nm and enhanced skin permeation of the bioactive compound with higher skin protection.134 Similarly, resveratrol-loaded solid lipid nanoparticles with a particle size of 180 nm showed higher skin permeation with enhanced protection.135,136 Lutein-loaded solid lipid nanoparticles with the various nanosizes showed increased skin protection from the UV light and antioxidant activity retention, thereby prolonging skin health.137 From the earlier research studies, solid lipid nanoparticles made with plant-based bioactive compounds suggest that cosmeceutical efficiency and health benefits are increased. Solid lipid nanoparticles may promote this sector with further development of phyto-based solid lipid nanoparticle formulations with multiple applications in therapy-based nanocosmeceuticals.
Nanostructured lipid carriers
Nanostructured lipid carriers are another important delivery technology used in cosmeceuticals to deliver natural and synthetic bioactive ingredients with increased efficiency.138–140 This technology has more advantages than other nanotechnologies in bulk production, increased stability of the carriers, and is different from solid lipid nanocarriers with the use of liquid lipids such as oleic acid. In addition, nanostructured lipid carrier has different structures such as imperfect type with different lipid structures, formless type with lipid preventing crystallization, and multiple type with the lipid oil containing bioactive compounds thereby limit the expulsion of bioactive compounds, which is a major drawback in solid lipid nanoparticles. Several phytobioactive compounds are highly used in the preparation of nanostructured lipid carriers with high efficiency in the topical applications, thereby protecting the skin and enhancing beauty. Okonogi and Riangjanapatee141 recently developed a nanostructured lipid carrier for topical delivery of lycopene with particle size ranging from 150 nm to 160 nm. Similarly, a lutein-nanostructured lipid carrier with particle size of 166–350 nm was constructed and tested for UV protection in the pig ear skin studies. These studies confirm that the lutein nanoparticle formation and their skin protective effects vary with nanocarrier type. Solid lipid nanocarriers showed higher lutein photostability followed by nanostructured lipid carrier and nanoemulsion. Moreover, the stability of all three types of nanocarriers showed better photoprotection of the skin.137
In another study, a quercetin-loaded nanostructured lipid carrier was constructed with the particle size of 282 nm and showed enhanced topical delivery of bioactive compounds with higher antioxidant activity. This quercetin product enhances the beauty of the skin from preventing oxidative stress.138 Similarly, nanostructured lipid carrier was constructed using phenylethyl resorcinol with a particle size of 218 nm and showed enhanced photostability and water stability, which may be applied as a whitening agent in the near future.142,143 Resveratrol was also constructed using nanostructured lipid carrier systems in the size range of 110 nm and showed enhanced skin protection with higher antioxidant activity, than solid lipid nanoparticle-loaded resveratrol.144 Further E-resveratrol greatly protects skin through the lipid-based nanoparticle system, under UV radiation.145 In certain studies, some bioactive compounds, such as squalene, promote drug absorption to the hair follicles, for example, a squalene-enriched nanostructured lipid carrier with a particle size of 208–265 nm is used to treat hair follicles of individuals suffering from alopecia areata.146 From these recent research studies, we confirm that nanostructured lipid carrier systems will open a gateway for phytobioactive compounds to enhance beauty and personal care.
Nanosingle or multiemulsions
Single or multiple nanoemulsions are currently used in various cosmetic products for their specific application using sizes between 10 nm and 300 nm with enhanced activity.69,147,148 The major advantages of using nanoemulsions in cosmetics are their unique size, which can enhance the bioactive activity of both hydrophilic and lipophilic types. However, this method has limitations in cosmeceutical production due to instability of emulsions during storage.149–158 Further, the emulsion core compounds direct whether a single or multiple nanoemulsions can be formed based on their number of core compounds in the nanosized emulsions.159,160 Multiple emulsions have advantages over single emulsions, due to their wider applications in cosmeceuticals, which can further reduce cosmeceutical cost. Several plant-derived bioactive compounds are used in nanoemulsions, such as flavonoids and polyphenols. Their bioactive role enhances cosmeceutical value with lower toxicity.161–164 Recently, quercetin-rich nanoemulsions, in the size range of 200–300 nm, were prepared from the ethanolic extract of Achyrocline satureioides and showed increased skin retention of bioactive compounds with enhanced antioxidant activities in porcine skin studies.164 In another study, genistein, a soy bioactive compound, coloaded with tocomin, acts as multiple nanoemulsions with the particle size of 173–208 nm and showed enhanced skin protection from UV damage. This treatment will be useful in the sunscreen lotion production.165 Similarly, retinyl palmitate was used in the production of nanoemulsions in the size range of <275 nm and showed enhanced skin penetration with increased inner skin protection.166 Similarly curcumin nanoemulsions were developed using a self-nanoemulsifying method with the mean particle size of 85 nm and showed enhanced transdermal availability with high permeability and without degradation.71 This might be useful in the skin care sectors for developing novel skin cream emulsions. In another study, lutein was successfully encapsulated into nanoemulsions in the size range of 150–350 nm and increased the release of the active compound compared to other lipid carriers. It showed comparatively better protection than corn oil suspended lutein.137 Nanoemulsions are highly used in skin-based beauty therapy. Achyrocline extract nanoemulsions show enhanced protection against herpes virus infections.161 Even though these phytocompound-rich single or multiple nanoemulsions showed better protective effects in various cosmeceuticals compared to commercial macroformulations, the stability of nanoemulsions is a big issue in the production and commercialization of these delivery technologies in the cosmeceutical sectors.
Lipid-based nanocapsules
Lipid-based nanocapsules show promising effects in biomedical applications along with cosmeceuticals.59,167–169 These delivery systems have enhanced effects in the delivery of phytobioactive compounds in oral therapy, which boosts the cosmeceutical sectors for the development of cosmeceuticals with enhanced activity by topical application. However, synthetic compounds are extensively developed using this technique,170 and phytocompounds used for cosmeceuticals are limited. Recently, resveratrol and curcumin were constructed using lipid core nanocapsules at 201 nm with an increased retention of polyphenols in the skin and increased skin protection.59 This confirms that coencapsulation enhances the bioavailability compounds in the skin and promotes skin protection against physiological environments. This technology will be potentially useful in the near future of the cosmeceuticals sectors. Similarly, an individual phytocompound of resveratrol was constructed using lipid-derived nanocapsules at 207 nm that enhanced skin protection with higher bioavailability of those bioactive compounds under UV radiation.145 Chitosan hydrogels combine nanocapsules with the active compounds of capsaicinoids and enhance skin protection by the control release of those compounds.171–173 The same research group studied the effects of capsaicinoids in nanocapsules and found greater skin care with sustained release.172 These studies confirm that phyto-derived bioactive compounds can be used for the construction of nanocapsules to apply in cosmeceutical sectors and beauty therapies.
Nanophytosomes
Phytosomes are another novel technology, particularly for plant polyphenolics, which enhance bioavailability of phytobioactive compounds to the skin5 and in other therapeutic applications174–177 for skin beauty and health care. The greater advantage of these techniques involves chemical bonding between the polyphenolics and the lipid bilayer and thereby enhances the physical stability of bioactive compounds, which prolong the compound’s effectiveness.178,179 Phytosomes are extensively used in cosmeceutical sectors for their beauty and therapeutic applications with the phyto-derived crude extracts or with the individual bioactive compounds.179,180 Recently, extracts from the Citrus auranticum and Glycyrrhiza glabra were used for the construction of phytosomes and for the skin aging studies. The researchers confirmed that phytosomes enhance the bioavailability of those compounds in cream and thereby protect skin from aging.181 Further, nanosizing of those phytosomes increases the bioavailability of those compounds for beauty and health care applications.182,183 Gold nanoparticle-bound phytosomes containing phytoextracts rich in quercetin were constructed and had enhanced quercetin efficiency at a particle size of 100 nm.184 Similarly, rutin-based phytosomes were developed with enhanced compound bioavailability and antioxidant activity.185
Nanoliposomes
Liposomes are effective in carrying phytobioactive compounds in cosmeceuticals. Products containing liposomes increase skin protection and beauty qualities.43,186–189 Generally, nanoliposomes are spherical, have a uni- or multilamellar structures, and are a few nanometers in size. Liposomes have high efficiency in phyto-based compounds delivery to the dermal layer. Nanoliposomes are used in cosmeceutical products such as antiaging cream, sun block cream, hair cream, and skin moisturizer, which contain synthetic as well as phytobioactive compounds.43,187–189 The major advantages of nanoliposomes are their unique size, they can encapsulate hydrophilic and hydrophobic bioactive compounds, UV absorbing lipids can protect the skin and the bioactive compounds without further degradation.43,187 Figure 1 shows the possible nanoliposome mechanism in reducing skin wrinkles. Among nanoliposomes that contain phytobioactive compounds, curcumin was studied with enhanced stability and prolonged activity of transdermal application.190 Recently, curcumin-loaded 80 nm nanoliposomes were constructed and showed skin protection.191 The same research group focused on 110 nm nanoliposome development with long circulating nanoliposomes for skin care and disease treatment.192 The combination of curcumin with hyaluronic acid enhances the bioavailability of curcumin in the skin and body, which leads to many novel nanoapproaches for the skin-based nanotherapautics and beauty.193 Even though several phytocompound-encapsulated liposome technologies are used in cosmeceuticals and therapy, the nanosize of liposomes is still in the budding stages with the active phyto-derived compounds. Phytocompound-encapsulated liposomes will be an innovative technology in the near future for application in therapy-based beauty.
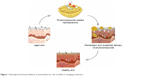  | Figure 1 Nanoliposome-based delivery of antioxidants for skin wrinkles in antiaging treatment. |
Nanoniosomes
Niosomes are extensively used in cosmeceutical sectors since it has multiple advantages over liposomes such as higher stability, enhanced skin penetration, low toxicity, and higher protection of the bioactive compounds.194–196 Niosome sizes vary from nanometer to micrometer, the lower the size greater the efficiency. Nanoniosomes are smaller, unilamellar structures in the size range of 10–100 nm.197–200 Niosomes are extensively used to deliver antioxidants such as ascorbic acid, resveratrol, and ellagic acid through skin and are used orally to treat diseases.196,201–207 Even though commercially many cosmeceutical creams and lotions contain niosomes with synthetic compounds, phyto-derived bioactive compound usage is a new sector in the development of nanoniosomes. Recently, Tavano et al202 produced a slightly larger niosome in the size range of 471–565 nm containing phyto-derived antioxidants such as resveratrol, alpha-tocopherol, and curcumin. This product showed enhanced antioxidant activity to the skin with increased skin permeation activity for cosmeceutical applications. This study confirmed that phyto-derived compounds can be effectively used in noisome formulation for cosmeceutical applications without change in bioactivity and enhanced skin protection. However, the size is a big factor, and reduction to nanosize with alteration in production methods may enhance their activity with a broad range of applications in cosmeceutical sectors. Recently, individual phytocompounds, such as curcumin, were developed using nanoniosome methods in the size range of 91 nm and showed enhanced skin protective activity.208–215 From the earlier studies, phytocompound-containing nanoniosomes may be useful for both cosmeceuticals and pharmaceutical sectors.
Nanoethosomes, glycerosomes, and hyalurosomes
Ethosome, glycerosomes, and hyalurosomes are modified liposomes, named based on their compounds used in the phospholipids, such as ethanol, glycerol, or sodium hyaluronate. They are modified for the enhanced delivery of the active ingredients to the skin and exclusively used in cosmeceutical sectors for their therapy and beauty treatments.216,217 Further, nanosizing of vesicles with incorporation of phytocompounds showed enhanced protective and beauty activity.218–221 Recently, quercetin glycerosomes were developed in the size range of 80–110 nm with a unilamellar structure and showed enhanced skin protective activity.222 This may be used in the future for the production of antioxidant skin cream. Ethosomes nanovesicles in the size range of 128 nm were prepared with polyphenolic extracts of Fraxinus angustifolia leaf and bark and showed enhanced wound healing and skin protection activity.223 Similar to the ethosomes, nanosized hyalurosomes of liquorice extract were prepared in the size range of 100 nm and enhanced skin beauty.224 The same research group also developed curcumin-based nanohyalurosomes with a minimal particle size of 112 nm and showed enhanced skin beauty-based therapy.193 From these studies, extracts can be used to develop nanovesicles, which could be efficiently used in the cosmeceutical sector, and it will give a broader scope of research and development for cosmeceutical products with multiple efficiencies.
Fullerene
Fullerene is yet another novel technology made of carbon atoms of 1 nm size, consisting of even numbers known as C60, which is used in the cosmeceutical sectors to carry phytocompounds. Fullerene itself acts as an antioxidant and prevents premature skin aging.225–229 Fullerene delivers vitamins to effectively enhance the skin. Recently, some researchers developed a fullerene nanocapsule with ascorbic acid and vitamin E. It showed enhanced skin protective activity against premature aging by its antioxidant activities.230,231 Further, skin protection was also enhanced by the fullerenes with some modifications.232–234 Fullerene-based photodynamic therapy was used for skin-based disease treatment with the bioactive compounds, such as curcumin.235,236 However, further study on the application of various phyto-derived bioactive compounds with fullerenes in cosmeceutical-based skin therapy has yet to be carried out. This will give way for the development of the new cosmeceutical products with better protective activity by the carrier and bioactive compounds.
Carbon nanotubes
Carbon nanotubes are a novel technology currently used in the cosmeceutical sectors particularly for the skin care. Similar to fullerene, carbon nanotubes alone act as an antioxidant at 100 nm in size.237,238 In addition to their uses in cosmeceutical sectors, they are effective in delivery systems for phytocompounds in biomedical applications.226,239,240 Recently, curcumin was conjugated to a single wall carbon nanotube for the effective delivery.241 The role of hyaluronic acids in the delivery of drugs using carbon nanotubes was reviewed recently,242 and further clinical trials are needed to assess their biosafety. However, a study on the application of carbon nanotubes with phytocompounds in skin care and cosmeceuticals is yet to be done.
Phytoconstituents in nanosize cosmeceuticals and skin therapy
Nanosize phytoconstituents are gaining popularity among the active ingredients in the cosmeceuticals, as they enhance beauty and have therapeutic roles in diseases such as antiaging, UV protection, and prevention of skin-related diseases.190,243–246 Increased skin protective activity of bioactive compounds is related to polyphenol content along with their reduced size. Compounds vary from macro to nano and with skin penetration using different delivery techniques.193,247–251 Recently, resveratrol encapsulated in the solid lipid nanoparticle showed enhanced skin uptake with increased protection and antioxidant activity.144 Solid lipid nanoparticles were compared nanostructured lipid carrier systems and found similar efficiencies with increased retention in the skin. Nanodelivery technologies also enhance the compound bioavailability not only to the skin but also to the brain to treat disorders such as Parkinson’s disease.252 Further modification of bioactive compounds along with the nanodelivery technologies can enhance skin bioavailability to increase protection.145 Similarly, quercetin encapsulated into a solid lipid nanoparticle system showed increased skin retention of quercetin with greater antioxidant activity.138,253,254 Genistein-loaded nanoemulsions were incorporated into hydrogels in the size range of 250 nm and enhanced delivery of the bioactive compound to the skin for a beauty-based therapy. In addition, coencapsulation of resveratrol and curcumin with lipid core nanocapsules increased delivery of the resveratrol to the skin.59 Further, several other phytobioactive compounds were efficiently encapsulated into various nanodelivery systems to enhance beauty and therapy applications. Specific compounds, their skin protecting activity, and beauty applications are discussed in the following sections. Some of the nanophytobioactive compounds and its role in beauty and skin therapy are listed in Table 1.
  | Table 1 Nanosized phytocompounds used in cosmeceuticals |
Nano Aloe vera
Aloe vera consists of several active constituents such as proteins, minerals, carbohydrates, and vitamins from the inner portions of the leaf, which are widely used in the skin care and the medicine.255–259 Aloe gel greatly enhances moisturizing activity and antiaging by enhancing the synthesis of collagen and elastin fibers. In addition, it also helps in the wound healing.257–263 The higher moisture, bioactive compound, and mineral content of aloe gel enhance the moisturizing effects and skin protection. Therefore, aloe is often used in cosmetic products such as creams, lotions, and gels.189,264,265 However, for their enhanced bioactivity of the aloe compounds, nanodelivery techniques are recently used in the cosmeceutical sections. Further, the use of nanosize A. vera in the preparation of cosmeceutical products is now in early stages. Few nanotechniques enhanced skin care activities. A. vera gel extract liposomes were constructed, with the particle size of 200 nm, that enhanced skin collagen synthesis and growth of skin cell lines, which confirm that it may be used to enhance skin care products.189 Similarly, some researchers patented nanoemulsion technology varying the nanosize containing A. vera extract. The resulting product caused skin rejuvenation and had antiwrinkle activity. In addition, coencapsulation of A. vera with curcumin enhances phytocompound delivery to the skin and protects antioxidant activity.266 From the earlier studies, nanosizing bioactive compounds from A. vera enhances beauty applications. Further development of various nanodelivery techniques is a good research opportunity.
Nanocurcumin
Curcumin is the active compound in turmeric, which is native to India and other Asian countries. Turmeric is widely used in cooking, medicine, beauty, and health products.267–273 Turmeric as a crude extract or tuber powder normally used for skin care among South Indian women in their daily routine owing to its multibeneficial activities such as antiaging, moisture retention, antioxidant activity, and natural beauty products with yellow pigments.274–281 Although turmeric has multiple beneficial activities, its bright yellow color and instability under certain environmental conditions make researchers look for alternative delivery technologies for skin care and beauty applications. Nanodelivery techniques such as solid lipid nanocarriers, nanoliposomes, nanoniosomes, and nanoemulsions solve most of their difficulties with turmeric and enhance the bioavailability of its polyphenolic compounds in cosmeceutical product.4,43,44 Curcumin nanotransfersomes, nanoethosomes, and nanoliposomes were prepared and studied for the skin hydration and UV protection activity. Three nanodelivery technologies enhanced the delivery of curcumin to the skin with greater hydration and UV protective in cream containing the nanoformulations. Greater efficiency among the nanodelivery techniques was observed in the order of nanotransfersomes having the most efficiency, nanoethosomes and nanoliposomes having the least. They also varied in size from 167 nm to 262 nm.219 In another study, curcumin nanoethosomes were prepared with particle sizes of 216 nm and enhanced antiwrinkle properties. Further, skin permeation of the curcumin-loaded nanoliposome with particle sizes of 82–92 nm showed higher skin protection.191 Similarly, nanohyaluronosomes with particle sizes of 112–220 nm enhanced curcumin bioavailability to the skin and will potentially be used for beauty-based therapies in near future.193 Similarly, curcumin-loaded niosomes were prepared with enhanced bioavailability in the skin.282 In addition to the individual compounds, coadministration of curcumin with resveratrol in the lipid core nanocapsules increased delivery of the phytocompounds to the skin with better antioxidant potential and skin care.59 Likewise, codelivery of curcumin with resveratrol in the nanosized niosomal formulations enhanced skin beauty with increased antioxidant activity.202
Nanovitamin E
Vitamin E is heat stable fat soluble compound most commonly used in cosmeceuticals for skin protection properties such as antiwrinkle, enhanced skin moisturizing, and prevention of skin disease.283 Since most cosmeceutical products contain the precursor of active tocopherol and its cleavage products in the skin is minimal, which leads to a decrease in skin protection. Even though vitamin E is naturally present in human skin, most of it is not active due to prolonged solar irradiation. Nanodelivery techniques solve issues with sufficient delivery along with prolonged and sustained release of vitamin E associated with enhanced beauty. Vitamin E nanoemulsion was prepared using nanoemulsion techniques and enhanced bioavailability of the vitamin compared to microsized emulsions.284 Vitamin E was also prepared using solid lipid nanoparticle delivery systems with size equivalent to 292 nm and enhanced skin protection activity. Further, solid lipid nanoparticle vitamin E preparation enhanced vitamin E delivery for cosmeceutical-based skin therapy.285–290 Teo et al291 reported that vitamin E-enriched palm oil nanoemulsions with particle sizes of 94 nm enhanced product stability and may be useful for their novel nanosized cosmeceuticals. In near future, vitamin E will be delivered with suitable nanodelivery technologies in therapy-based cosmeceuticals for enhanced activity, stability, and beauty care.
Nanovitamin C
Vitamin C is a vitamin abundant in plants and has multiple roles in the beauty and health care, involving collagen synthesis.292,293 Vitamin C readily undergoes degradation when exposed to harsh environments and UV radiation. A continuous supply of vitamin C compounds enhances the skin’s beauty. Nanodelivery techniques help sustain vitamin C supplementation without changes in its biological activity after applying to the skin. Recently, several nanotechnological approaches have been published using vitamin C for skin health and beauty, such as solid lipid nanoparticles and nanostructured lipid carriers.294 Vitamin C-encapsulated solid lipid nanoparticle was prepared with a particle size of 200–225 nm. Solid lipid nanoparticle enhanced the carrier of the vitamin against apoptotic effects.295 Similarly, nanoliposomes were constructed using vitamin C that enhanced skin permeation activities with different particle sizes, ranging from 66 nm to 130 nm.296 In addition, vitamin C nanoliposomes had enhanced stability and higher antioxidant activity compared to ordinary liposomes after 60 days of storage.297 In another study, a vitamin C and gold-loaded nanofiber mask were prepared for the effective delivery of vitamins to the face. The liposomal preparation delivery of vitamins through mask enhanced facial beauty.298 Therefore, vitamin C can be delivered to the skin using nanodelivery technologies for enhanced skin beauty-based therapies.
Nanoresveratrol
Resveratrol is another polyphenolic compound with multiple beneficial roles in quenching free radicals and protecting skin against harmful environmental conditions.299 Polyphenolic compounds inhibit oxidative mechanisms and UV-induced radiation. They are widely used in functional foods for skin care and health.56,300–304 In comparison to other antioxidants, resveratrol shows higher efficiency of skin protection against UV-B radiation.305,306 However, resveratrol stability and beauty effects are limited due to compound degradation under harsh environmental conditions. To overcome this, nanodelivery techniques such as solid lipid nanoparticles, nanoemulsions, and transfersomes showed enhanced resveratrol stability and delivery to the skin.136,307,308 Recently, microemulsions were constructed using resveratrol with particle sizes of 66–68 nm that enhanced resveratrol bioavailability to the skin to protect against UV radiation.309 Recently, three different nanodelivery techniques, such as solid lipid nanocarrier, nanostructured lipid carrier, and nanoemulsions, were studied for resveratrol delivery efficiency to the skin. Among them, the solid lipid nanocarrier preparation has the most enhanced resveratrol delivery to the skin.310 Analyzing resveratrol nanodelivery studies, we conclude resveratrol may be highly protective through nanodelivery techniques and may be used in the preparation of natural nanocosmeceutical products to enhance beauty.
Nanoquercetin
Quercetin is another phytoactive flavonoid compound having efficiency in multiple aspects such as skin care and beauty through antioxidant activities; thereby, it enhances skin beauty and personal care.311–314 In comparison to other flavonoids, quercetin showed higher skin protecting efficiency and enhanced beauty due to its abundance of OH groups. This leads many researchers to analyze the multiple efficiencies of quercetin in cosmetics. However, the major drawback of quercetin applications is its stability and penetrating efficiency, which is solved by nanodelivery techniques. Solid lipid nanoparticle and nanostructured lipid carrier nanodelivery systems were compared for the effective delivery of the quercetin to the skin. The nanostructured lipid carrier showed higher quercetin retention in the skin membrane with a particle size of ~282 nm.138 The same research group also suggests that quercetin in the solid lipid nanoparticle preparation has increased skin protection against UV radiation of human skin.253 Nanoglycerosomes were also prepared with particle sizes between 80 nm and 110 nm that enhanced skin delivery of quercetin along with its photoprotective effects against oxidative damage.222 Similarly, quercetin-rich nanoemulsions and A. satureioides nanoemulsions were prepared with nanoparticles ranging from 200 nm to 300 nm. This preparation had higher skin retention of the phytocompounds with increased skin protecting activity.164 In another study, quercetin-loaded nanocapsules were constructed with a particle size of 228 nm for sustained delivery of quercetin to the skin to increase antioxidant activity.315 From these studies, we conclude that quercetin may be used in the preparation of various nanosized cosmeceuticals products with increased stability, prolonged beautifying effects, and without harmful side effects.
Nanogreen tea
Natural green tea extract is rich source of bioactive compounds, such as catechin, epigallectocatechin, epicatechin, and epigallecto-3-catechin, which are powerful antioxidants widely used in skin care and beauty treatments.316–320 The individual potential of these compounds also varies in usage for development of cosmeceutical products. Green tea components have multiple skin care roles such as antiaging and prevention of UV-induced photoaging.316,320–323 The bioactive compounds readily undergo functional group loss during the exposure to environmental conditions and decrease the stability of the compounds, which limits the application of these compounds in cosmeceutical products. Nanodelivery techniques enhance the bioavailability of these compounds in cosmeceutical products, thereby enhancing their usage in the novel development of green tea-based cosmeceuticals. Recently, Montenegro324 listed group of phytocompounds usage in commercial products. Among them, green tea catechins were used in antiaging cream preparation with fullerene as the delivery agent. Fang et al325 also prepared green tea catechin liposomes with nanoparticle sizes of 130–268 nm that enhanced antioxidant activity in the skin. In another study, green tea liposomes, with a reduced particle size of ~138 nm, enhanced green tea catechins by ethanol incorporation into the liposomes for enhanced transdermal delivery.326 Similarly, green tea polyphenol nanoliposomes were developed using milk fat globule membranes and soy lipids to enhance antioxidant activity in the cell model.327 These studies suggest that green tea polyphenols can be sufficiently nanodelivered to various skin care products for their sustained beautifying effects.
Nanogenistein
Genistein is one of the major isoflavones found in many cereal plants and has skin protecting activities, including antioxidant, collagen synthesis, UV protection, and antiaging benefits.328–333 Even though genistein has skin protecting activity and enhances beauty, its sustained effect is still limited due to lower stability in cosmeceutical products. In order to enhance stability and prolong activity, nanosized delivery technologies have greater impact in cosmeceutical sectors. Genistein-loaded nanoemulsions were prepared with 250 nm sized nanoparticles and enhanced delivery of isoflavones to the skin with higher skin protecting activity.334 In another study, nanosized liposome-encapsulated genistein in nanoparticles the size of 84 nm was tested on rat skin, with or without hair.335 In addition to the nanosized liposomes, the hairs on the skin also affect the delivery of the genistein compound to the skin.
Nanolycopene
Lycopene is another natural phytobioactive compound with multiple applications in skin care, including antiaging and antioxidant activity.336–345 Owing to its lipophilic nature and decreased delivery to the skin, lycopene is of limited usage in skin care products. In addition to its lipophilic nature, lycopene readily undergoes degradation during light and air exposure, which further limits its usage in cosmeceuticals. However, nanosizing lycopene solves most of these problems and enhances its bioavailability.141,346,347 Recently, lycopene ethosomes and transfersomes were constructed in the nanosize of 153 nm and 124 nm, respectively, which enhance the bioavailability lycopene to the skin with potential antioxidant activity. Further, it enhances the penetration of lycopene to the inner cell and nucleus, which could be useful in skin protection and care.348 Research focusing on nanodelivery in cosmeceutical products containing lycopene is still limited.
Nanotoxicity
The major concern of using nanocosmeceuticals is toxicity owing to the small size, allowing the particles to cross the membrane easily and interact with other proteins and cells. This may lead to the other effects by their subsequent metabolic products.349 Before applying the products, extensive clinical screening must be done, even though many reports suggest that polymers in cosmeceuticals have no deleterious effects. Further, natural products have no side effects. Extensive use of these compounds may also lead to side effects. Further clinical support is needed before nanotechnology proceeds to commercialization and product development.
Conclusion
The greater demand of the nanosized phyto-based bioactive compound usage in the development of nanocosmeceuticals for skincare-based therapy such as moisturizing, sun block, anti-aging, whitening effects along with skin health leads many researchers in search of novel nanocosemecutical-based therapy. Nanosized cosmeceuticals with phyto-based bioactive compounds retain higher amounts of bioactive compounds in the skin, are more stable, and enhance skin appeal with prolong time. Nanosized compounds range in size. Increasing the nanosize limits the penetration ability of bioactive compounds to the skin, which limits beautifying and therapeutic activities. The solubility types and the cosmeceutical application of phyto-based bioactive compounds determine the delivery technology for nanocosmeceutical development. Further research of their mechanisms is necessary to target site release from the delivery agents. This further boosts nanosized phytobioactive compound-based cosmeceutical research and skin health.
Acknowledgment
This work was supported by the Konkuk University in 2015.
Disclosure
The authors report no conflicts of interest in this work.
References
Li DH, Wu ZM, Martini N, Wen JY. Advanced carrier systems in cosmetics and cosmeceuticals: a review. J Cosmet Sci. 2011;62(6):549–563. | ||
Taib SHM, Abd Gani SS, Ab Rahman MZ, Basri M, Ismail A, Shamsudin R. Formulation and process optimizations of nano-cosmeceuticals containing purified swiftlet nest. RSC Adv. 2015;5(53):42322–42328. | ||
Boonme P, Junyaprasert VB, Suksawad N, Songkro S. Microemulsions and nanoemulsions: novel vehicles for whitening cosmeceuticals. J Biomed Nanotechnol. 2009;5(4):373–383. | ||
Lohani A, Verma A, Joshi H, Yadav N, Karki N. Nanotechnology-based cosmeceuticals. ISRN Dermatol. 2014;2014:14. [843687]. | ||
Chanchal D, Swarnlata S. Novel approaches in herbal cosmetics. J Cosmet Dermatol. 2008;7(2):89–95. | ||
Efird JT, Holly EA, Cordier S, et al. Beauty product-related exposures and childhood brain tumors in seven countries: results from the SEARCH International Brain Tumor Study. J Neurooncol. 2005;72(2):133–147. | ||
Markowitz K. Pretty painful: why does tooth bleaching hurt? Med Hypotheses. 2010;74(5):835–840. | ||
Washam C. Beastly beauty products: exposure to inorganic mercury in skin-lightening creams. Environ Health Perspect. 2011;119(2):A80–A81. | ||
Alviano DS, Alviano CS. Plant extracts: search for new alternatives to treat microbial diseases. Curr Pharm Biotechnol. 2009;10(1):106–121. | ||
Durmic Z, Blache D. Bioactive plants and plant products: effects on animal function, health and welfare. Anim Feed Sci Technol. 2012;176(1–4):150–162. | ||
González-Molina E, Domínguez-Perles R, Moreno DA, García-Viguera C. Natural bioactive compounds of Citrus limon for food and health. J Pharmaceut Biomed. 2010;51(2):327–345. | ||
Vasanthi HR, ShriShriMal N, Das DK. Phytochemicals from plants to combat cardiovascular disease. Curr Med Chem. 2012;19(14):2242–2251. | ||
Gismondi A, Canuti L, Grispo M, Canini A. Biochemical composition and antioxidant properties of Lavandula angustifolia miller essential oil are shielded by propolis against UV radiations. Photochem Photobiol. 2014;90(3):702–708. | ||
Mustapa AN, Martin A, Mato RB, Cocero MJ. Extraction of phytocompounds from the medicinal plant Clinacanthus nutans Lindau by microwave-assisted extraction and supercritical carbon dioxide extraction. Ind Crop Prod. 2015;74:83–94. | ||
Yamasaki K. Bioactive saponins in Vietnamese ginseng, Panax vietnamensis. Pharm Biol. 2000;38:16–24. | ||
Afaq F, Zaid MA, Khan N, Dreher M, Mukhtar H. Protective effect of pomegranate-derived products on UVB-mediated damage in human reconstituted skin. Exp Dermatol. 2009;18(6):553–561. | ||
Kohl E, Steinbauer J, Landthaler M, Szeimies RM. Skin ageing. J Eur Acad Dermatol Venereol. 2011;25(8):873–884. | ||
Maruki-Uchida H, Kurita I, Sugiyama K, Sai M, Maeda K, Ito T. The protective effects of piceatannol from passion fruit (Passiflora edulis) seeds in UVB-irradiated keratinocytes. Biol Pharm Bull. 2013;36(5):845–849. | ||
Park M, Han J, Lee CS, Soo BH, Lim KM, Ha H. Carnosic acid, a phenolic diterpene from rosemary, prevents UV-induced expression of matrix metalloproteinases in human skin fibroblasts and keratinocytes. Exp Dermatol. 2013;22(5):336–341. | ||
Wu Y, Matsui MS, Chen JZ, et al. Antioxidants add protection to a broad-spectrum sunscreen. Clin Exp Dermatol. 2011;36(2):178–187. | ||
Yaar M, Gilchrest BA. Photoageing: mechanism, prevention and therapy. Br J Dermatol. 2007;157(5):874–887. | ||
Hwang BM, Noh EM, Kim JS, et al. Curcumin inhibits UVB-induced matrix metalloproteinase-1/3 expression by suppressing the MAPK-p38/JNK pathways in human dermal fibroblasts. Exp Dermatol. 2013;22(5):371–374. | ||
Bae JY, Choi JS, Kang SW, Lee YJ, Park J, Kang YH. Dietary compound ellagic acid alleviates skin wrinkle and inflammation induced by UV-B irradiation. Exp Dermatol. 2010;19(8):e182–e190. | ||
Fiorentino DF, Chen RO, Stewart DB, Brown KK, Sundram UN. The direct cellular target of topically applied pimecrolimus may not be infiltrating lymphocytes. Br J Dermatol. 2011;164(5):996–1003. | ||
Li N, Deng L, Xiang L, Liang Y. Photoprotective effect of tea and its extracts against ultraviolet radiation-induced skin disorders. Trop J Pharm Res. 2014;13(3):475–483. | ||
Black HS. The role of nutritional lipids and antioxidants in UV-induced skin cancer. Front Biosci. 2015;7:30–39. | ||
Bogdan Allemann I, Baumann L. Antioxidants used in skin care formulations. Skin Therapy Lett. 2008;13(7):5–9. | ||
Grether-Beck S, Marini A, Jaenicke T, Krutmann J. Effective photoprotection of human skin against infrared A radiation by topically applied antioxidants: results from a vehicle controlled, double-blind, randomized study. Photochem Photobiol. 2015;91(1):248–250. | ||
Jung S, Darvin ME, Chung HS, et al. Antioxidants in Asian-Korean and caucasian skin: the influence of nutrition and stress. Skin Pharmacol Physiol. 2014;27(6):293–302. | ||
Masaki H. Role of antioxidants in the skin: anti-aging effects. J Dermatol Sci. 2010;58(2):85–90. | ||
Pandel R, Poljšak B, Godic A, Dahmane R. Skin photoaging and the role of antioxidants in its prevention. ISRN Dermatol. 2013;2013:930164. | ||
Godic A, Poljsak B, Adamic M, Dahmane R. The role of antioxidants in skin cancer prevention and treatment. Oxid Med Cell Longev. 2014;2014;6:860479. | ||
Graf J. Antioxidants and skin care: the essentials. Plast Reconstr Surg. 2010;125(1):378–383. | ||
Nguyen G, Torres A. Systemic antioxidants and skin health. J Drugs Dermatol. 2012;11(9):e1–e4. | ||
Schempp CM, Meinke MC, Lademann J, Ferrari Y, Brecht T, Gehring W. Topical antioxidants protect the skin from chemical-induced irritation in the repetitive washing test: a placebo-controlled, double-blind study. Contact Dermatitis. 2012;67(4):234–237. | ||
Udompataikul M, Sripiroj P, Palungwachira P. An oral nutraceutical containing antioxidants, minerals and glycosaminoglycans improves skin roughness and fine wrinkles. Int J Cosmet Sci. 2009;31(6):427–435. | ||
Zablotska LB, Chen Y, Graziano JH, et al. Protective effects of B vitamins and antioxidants on the risk of arsenic-related skin lesions in Bangladesh. Environ Health Perspect. 2008;116(8):1056–1062. | ||
Chermahini SH, Majid FAA, Sarmidi MR. Cosmeceutical value of herbal extracts as natural ingredients and novel technologies in anti-aging. J Med Plants Res. 2011;5(14):3074–3077. | ||
Fisk WA, Lev-Tov HA, Clark AK, Sivamani RK. Phytochemical and botanical therapies for rosacea: a systematic review. Phytother Res. 2015;29(10):1439–1451. | ||
Krausz A, Gunn H, Friedman A. The basic science of natural ingredients. J Drugs Dermatol. 2014;13(8):937–943. | ||
Yildiz H, Abuaf OK. Use of cosmeceuticals during pregnancy and lactation period. Turkderm Arch Turk D. 2013;47(4):194–199. | ||
Kidd PM. Bioavailability and activity of phytosome complexes from botanical polyphenols: the silymarin, curcumin, green tea, and grape seed extracts. Altern Med Rev. 2009;14(3):226–246. | ||
Golubovic-Liakopoulos N, Simon SR, Shah B. Nanotechnology use with cosmeceuticals. Semin Cutan Med Surg. 2011;30(3):176–180. | ||
Meghea A. Pharmaceuticals and cosmeceuticals based on soft nanotechnology techniques with antioxidative, immunostimulative and other therapeutic activities. Recent Pat Nanotechnol. 2008;2(2):137–145. | ||
Ascenso A, Ribeiro HM, Marques HC, Simoes S. Topical delivery of antioxidants. Curr Drug Deliv. 2011;8(6):640–660. | ||
Felippi CC, Oliveira D, Ströher A, et al. Safety and efficacy of antioxidants-loaded nanoparticles for an anti-aging application. J Biomed Nanotechnol. 2012;8(2):316–321. | ||
Sapino S, Carlotti ME, Cavalli R, et al. Photochemical and antioxidant properties of gamma-oryzanol in beta-cyclodextrin-based nanosponges. J Incl Phenom Macro. 2013;75(1–2):69–76. | ||
Bossio O, Gomez-Mascaraque LG, Fernandez-Gutierrez M, Vazquez-Lasa B, Roman JS. Amphiphilic polysaccharide nanocarriers with antioxidant properties. J Bioact Compat Pol. 2014;29(6):589–606. | ||
Farboud ES, Nasrollahi SA, Tabbakhi Z. Novel formulation and evaluation of a Q10-loaded solid lipid nanoparticle cream: in vitro and in vivo studies. Int J Nanomed. 2011;6:611–617. | ||
Kaur IP, Kapila M, Agrawal R. Role of novel delivery systems in developing topical antioxidants as therapeutics to combat photoageing. Ageing Res Rev. 2007;6(4):271–288. | ||
Manosroi A, Chutoprapat R, Sato Y, et al. Antioxidant activities and skin hydration effects of rice bran bioactive compounds entrapped in niosomes. J Nanosci Nanotechnol. 2011;11(3):2269–2277. | ||
Morabito K, Shapley NC, Steeley KG, Tripathi A. Review of sunscreen and the emergence of non-conventional absorbers and their applications in ultraviolet protection. Int J Cosmet Sci. 2011;33(5):385–390. | ||
Niculae G, Lacatusu I, Bors A, Stan R. Photostability enhancement by encapsulation of alpha-tocopherol into lipid-based nanoparticles loaded with a UV filter. Cr Chim. 2014;17(10):1028–1033. | ||
Castangia I, Manca ML, Caddeo C, et al. Faceted phospholipid vesicles tailored for the delivery of Santolina insularis essential oil to the skin. Colloid Surface B Colloids Surf B Biointerfaces. 2015;132:185–193. | ||
Manca ML, Castangia I, Caddeo C, et al. Improvement of quercetin protective effect against oxidative stress skin damages by incorporation in nanovesicles. Colloid Surface B Colloids Surf B Biointerfaces. 2014;123:566–574. | ||
Pando D, Caddeo C, Manconi M, Fadda AM, Pazos C. Nanodesign of olein vesicles for the topical delivery of the antioxidant resveratrol. J Pharm Pharmacol. 2013;65(8):1158–1167. | ||
Katz LM, Dewan K, Bronaugh RL. Nanotechnology in cosmetics. Food Chem Toxicol. 2015;85:127–137. | ||
Kaur IP, Kakkar V, Deol PK, Yadav M, Singh M, Sharma I. Issues and concerns in nanotech product development and its commercialization. J Control Release. 2014;193:51–62. | ||
Friedrich RB, Kann B, Coradini K, Offerhaus HL, Beck RC, Windbergs M. Skin penetration behavior of lipid-core nanocapsules for simultaneous delivery of resveratrol and curcumin. Eur J Pharm Sci. 2015;78:204–213. | ||
Sachs DL, Rittié L, Chubb HA, Orringer J, Fisher G, Voorhees JJ. Hypo-collagenesis in photoaged skin predicts response to anti-aging cosmeceuticals. J Cosmet Dermatol. 2013;12(2):108–115. | ||
Sherber NS. Topicals in skin rejuvenation: prescription topicals. Facial Plast Surg. 2014;30(1):12–15. | ||
Fu PP, Xia Q, Sun X, Yu H. Phototoxicity and environmental transformation of polycyclic aromatic hydrocarbons (PAHs)-light-induced reactive oxygen species, lipid peroxidation, and DNA damage. J Environ Sci Health C Environ Carcinog Ecotoxicol Rev. 2012;30(1):1–41. | ||
Yu H. Environmental carcinogenic polycyclic aromatic hydrocarbons: photochemistry and phototoxicity. J Environ Sci Health C Environ Carcinog Ecotoxicol Rev. 2002;20(2):149–183. | ||
Ahmad I, Akhter S, Ahmad MZ, et al. Collagen loaded nano-sized surfactant based dispersion for topical application: formulation development, characterization and safety study. Pharm Dev Technol. 2014;19(4):460–467. | ||
Chen HF, Wang ZY, Chen JJ, Fan XP, Qian GD. Preparation and aging behaviour of nano SiO2 modified PEA coating by in-situ polymerization. Rare Metal Mat Eng. 2004;33:9–12. | ||
Raza K, Singh B, Singla N, Negi P, Singal P, Katare OP. Nano-lipoidal carriers of isotretinoin with anti-aging potential: formulation, characterization and biochemical evaluation. J Drug Target. 2013;21(5):435–442. | ||
Ro J, Kim Y, Kim H, et al. Pectin micro- and nano-capsules of retinyl palmitate as cosmeceutical carriers for stabilized skin transport. Korean J Physiol Pha. 2015;19(1):59–64. | ||
Zhang T, Jin JH, Yang SL, Li G, Jiang JM. Preparation and characterization of poly(p-phenylene benzobisoxazole) (PBO) fiber with anti-ultraviolet aging. Acta Chim Sinica. 2010;68(2):199–204. | ||
Bernardi DS, Pereira TA, Maciel NR, et al. Formation and stability of oil-in-water nanoemulsions containing rice bran oil: in vitro and in vivo assessments. J Nanobiotechnology. 2011;9:44. | ||
Naz Z, Ahmad FJ. Curcumin-loaded colloidal carrier system: formulation optimization, mechanistic insight, ex vivo and in vivo evaluation. Int J Nanomed. 2015;10:4293–4307. | ||
Rachmawati H, Budiputra DK, Mauludin R. Curcumin nanoemulsion for transdermal application: formulation and evaluation. Drug Dev Ind Pharm. 2015;41(4):560–566. | ||
Yucel C, Quagliariello V, Iaffaioli RV, Ferrari G, Donsi F. Submicron complex lipid carriers for curcumin delivery to intestinal epithelial cells: effect of different emulsifiers on bioaccessibility and cell uptake. Int J Pharmaceut. 2015;494(1):357–369. | ||
Badea G, Lacatusu I, Badea N, Ott C, Meghea A. Use of various vegetable oils in designing photoprotective nanostructured formulations for UV protection and antioxidant activity. Ind Crop Prod. 2015;67:18–24. | ||
Domínguez-Villegas V, Clares-Naveros B, García-López ML, Calpena-Campmany AC, Bustos-Zagal P, Garduno-Ramirez ML. Development and characterization of two nano-structured systems for topical application of flavanones isolated from Eysenhardtia platycarpa. Colloid Surface B. 2014;116:183–192. | ||
Coradini K, Lima FO, Oliveira CM, et al. Co-encapsulation of resveratrol and curcumin in lipid-core nanocapsules improves their in vitro antioxidant effects. Eur J Pharm Biopharm. 2014;88(1):178–185. | ||
Friedrich RB, Mann B, Coradini K, Offerhaus HL, Beck RCR, Windbergs M. Skin penetration behavior of lipid-core nanocapsules for simultaneous delivery of resveratrol and curcumin. Eur J Pharm Sci. 2015;78:204–213. | ||
Shim JH, Park JH, Lee JH, Lee DY, Lee JH, Yang JM. Moisturizers are effective in the treatment of xerosis irrespectively from their particular formulation: results from a prospective, randomized, double-blind controlled trial. J Eur Acad Dermatol Venereol. 2016;30(2):276–281. | ||
Berardesca E, Barbareschi M, Veraldi S, Pimpinelli N. Evaluation of efficacy of a skin lipid mixture in patients with irritant contact dermatitis, allergic contact dermatitis or atopic dermatitis: a multicenter study. Contact Dermatitis. 2001;45(5):280–285. | ||
Ribeiro RC, Barreto SM, Ostrosky EA, da Rocha-Filho PA, Veríssimo LM, Ferrari M. Production and characterization of cosmetic nanoemulsions containing Opuntia ficus-indica (L.) mill extract as moisturizing agent. Molecules. 2015;20(2):2492–2509. | ||
Khameneh B, Halimi V, Jaafari MR, Golmohammadzadeh S. Safranal-loaded solid lipid nanoparticles: evaluation of sunscreen and moisturizing potential for topical applications. Iran J Basic Med Sci. 2015;18(1):58–63. | ||
Lee BM, An S, Kim SY, et al. Topical application of a cleanser containing extracts of folium, and var. reduces skin oil content and pore size in human skin. Biomed Rep. 2015;3(3):343–346. | ||
Werschler WP, Trookman NS, Rizer RL, Ho ET, Mehta R. Enhanced efficacy of a facial hydrating serum in subjects with normal or self-perceived dry skin. J Clin Aesthet Dermatol. 2011;4(2):51–55. | ||
Troccaz M, Gaïa N, Beccucci S, et al. Mapping axillary microbiota responsible for body odours using a culture-independent approach. Microbiome. 2015;3(1):3. | ||
Coret CD, Suero MB, Tierney NK. Tolerance of natural baby skin-care products on healthy, full-term infants and toddlers. Clin Cosmet Investig Dermatol. 2014;7:51–58. | ||
Mitani K, Takano F, Kawabata T, et al. Suppression of melanin synthesis by the phenolic constituents of sappanwood (Caesalpinia sappan). Planta Med. 2013;79(1):37–44. | ||
Yang D, Pornpattananangkul D, Nakatsuji T, et al. The antimicrobial activity of liposomal lauric acids against Propionibacterium acnes. Biomaterials. 2009;30(30):6035–6040. | ||
Pornpattananangkul D, Fu V, Thamphiwatana S, et al. In vivo treatment of Propionibacterium acnes infection with liposomal lauric acids. Adv Healthc Mater. 2013;2(10):1322–1328. | ||
Liu CH, Huang HY. In vitro anti-propionibacterium activity by curcumin containing vesicle system. Chem Pharm Bull. 2013;61(4):419–425. | ||
Ho R, Teai T, Meybeck A, Raharivelomanana P. UV-protective effects of phytoecdysteroids from Microsorum grossum extracts on human dermal fibroblasts. Nat Prod Commun. 2015;10(1):33–36. | ||
Pollack AZ, Buck Louis GM, Chen Z, et al. Bisphenol A, benzophenone-type ultraviolet filters, and phthalates in relation to uterine leiomyoma. Environ Res. 2015;137:101–107. | ||
Bulla MK, Hernandes L, Baesso ML, et al. Evaluation of photoprotective potential and percutaneous penetration by photoacoustic spectroscopy of the Schinus terebinthifolius raddi extract. Photochem Photobiol. 2015;91(3):558–566. | ||
Polonini HC, Brandao MAF, Raposo NRB. A natural broad-spectrum sunscreen formulated from the dried extract of Brazilian Lippia sericea as a single UV filter. RSC Adv. 2014;4(107):62566–62575. | ||
Bennet D, Kang SC, Gang J, Kim S. Photoprotective effects of apple peel nanoparticles. Int J Nanomedicine. 2014;9:93–108. | ||
Shetty PK, Venuvanka V, Jagani HV, et al. Development and evaluation of sunscreen creams containing morin-encapsulated nanoparticles for enhanced UV radiation protection and antioxidant activity. Int J Nanomedicine. 2015;10:6477–6491. | ||
Niculae G, Lacatusu I, Badea N, Stan R, Vasile BS, Meghea A. Rice bran and raspberry seed oil-based nanocarriers with self-antioxidative properties as safe photoprotective formulations. Photochem Photobiol Sci. 2014;13(4):703–716. | ||
Baccarin T, Mitjans M, Ramos D, Lemos-Senna E, Vinardell MP. Photoprotection by Punica granatum seed oil nanoemulsion entrapping polyphenol-rich ethyl acetate fraction against UVB-induced DNA damage in human keratinocyte (HaCaT) cell line. J Photochem Photobiol B. 2015;153:127–136. | ||
Baccarin T, Mitjans M, Lemos-Senna E, Vinardell MP. Protection against oxidative damage in human erythrocytes and preliminary photosafety assessment of Punica granatum seed oil nanoemulsions entrapping polyphenol-rich ethyl acetate fraction. Toxicol In Vitro. 2015;30(1 Pt B):421–428. | ||
Goodier M, Hordinsky M. Normal and aging hair biology and structure ‘aging and hair’. Curr Probl Dermatol. 2015;47:1–9. | ||
Perera E, Yip L, Sinclair R. Alopecia areata. Curr Probl Dermatol. 2015;47:67–75. | ||
Tobin DJ. Age-related hair pigment loss. Curr Probl Dermatol. 2015;47:128–138. | ||
Trüeb RM. Effect of ultraviolet radiation, smoking and nutrition on hair. Curr Probl Dermatol. 2015;47:107–120. | ||
Oh JW, Kloepper J, Langan EA, et al. A guide to studying human hair follicle cycling in vivo. J Invest Dermatol. Epub 2015 Sep 9. | ||
Dario MF, Pahl R, de Castro JR, et al. Efficacy of Punica granatum L. hydroalcoholic extract on properties of dyed hair exposed to UVA radiation. J Photochem Photobiol B. 2013;120:142–147. | ||
Fernandez E, Martinez-Teipel B, Armengol R, Barba C, Coderch L. Efficacy of antioxidants in human hair. J Photochem Photobiol B. 2012;117:146–156. | ||
Le Floc’h C, Cheniti A, Connétable S, Piccardi N, Vincenzi C, Tosti A. Effect of a nutritional supplement on hair loss in women. J Cosmet Dermatol. 2015;14(1):76–82. | ||
Blume-Peytavi U, Vogt A. Human hair follicle: reservoir function and selective targeting. Brit J Dermatol. 2011;165(Suppl 2):13–17. | ||
Bolzinger MA, Briancon S, Pelletier J, Chevalier Y. Penetration of drugs through skin, a complex rate-controlling membrane. Curr Opin Colloid Interface Sci. 2012;17(3):156–165. | ||
Główka E, Wosicka-Frackowiak H, Hyla K, et al. Polymeric nanoparticles-embedded organogel for roxithromycin delivery to hair follicles. Eur J Pharm Biopharm. 2014;88(1):75–84. | ||
Mura S, Pirot F, Manconi M, Falson F, Fadda AM. Liposomes and niosomes as potential carriers for dermal delivery of minoxidil. J Drug Target. 2007;15(2):101–108. | ||
Papakostas D, Rancan F, Sterry W, Blume-Peytavi U, Vogt A. Nanoparticles in dermatology. Arch Dermatol Res. 2011;303(8):533–550. | ||
Rancan F, Amselgruber S, Hadama S, et al. Particle-based transcutaneous administration of HIV-1 p24 protein to human skin explants and targeting of epidermal antigen presenting cells. J Control Release. 2014;176:115–122. | ||
Shamma RN, Aburahma MH. Follicular delivery of spironolactone via nanostructured lipid carriers for management of alopecia. Int J Nanomed. 2014;9:5449–5460. | ||
Vogt A, Blume-Peytavi U. Selective hair therapy: bringing science the fiction. Exp Dermatol. 2014;23(2):83–86. | ||
Jung S, Otberg N, Thiede G, et al. Innovative liposomes as a transfollicular drug delivery system: penetration into porcine hair follicles. J Invest Dermatol. 2006;126(8):1728–1732. | ||
Konrádsdóttir F, Ogmundsdóttir H, Sigurdsson V, Loftsson T. Drug targeting to the hair follicles: a cyclodextrin-based drug delivery. AAPS PharmSciTech. 2009;10(1):266–269. | ||
Desai PR, Shah PP, Hayden P, Singh M. Investigation of follicular and non-follicular pathways for polyarginine and oleic acid-modified nanoparticles. Pharm Res. 2013;30(4):1037–1049. | ||
Saikia AP, Ryakala VK, Sharma P, Goswami P, Bora U. Ethnobotany of medicinal plants used by Assamese people for various skin ailments and cosmetics. J Ethnopharmacol. 2006;106(2):149–157. | ||
Abbasi AM, Khan MA, Ahmad M, Zafar M, Jahan S, Sultana S. Ethnopharmacological application of medicinal plants to cure skin diseases and in folk cosmetics among the tribal communities of North-West Frontier Province, Pakistan. J Ethnopharmacol. 2010;128(2):322–335. | ||
Pannu J, McCarthy A, Martin A, et al. NB-002, a novel nanoemulsion with broad antifungal activity against dermatophytes, other filamentous fungi, and Candida albicans. Antimicrob Agents Chemother. 2009;53(8):3273–3279. | ||
Dweck AC. Natural ingredients for colouring and styling. Int J Cosmet Sci. 2002;24(5):287–302. | ||
Kamairudin N, Gani SS, Masoumi HR, Hashim P. Optimization of natural lipstick formulation based on pitaya (Hylocereus polyrhizus) seed oil using D-optimal mixture experimental design. Molecules. 2014;19(10):16672–16683. | ||
Al-Alwani MA, Mohamad AB, Kadhum AA, Ludin NA. Effect of solvents on the extraction of natural pigments and adsorption onto TiO2 for dye-sensitized solar cell applications. Spectrochim Acta A Mol Biomol Spectrosc. 2015;138:130–137. | ||
Khan MI, Giridhar P. Plant betalains: chemistry and biochemistry. Phytochemistry. 2015;117:267–295. | ||
Ravichandran K, Palaniraj R, Saw NM, et al. Effects of different encapsulation agents and drying process on stability of betalains extract. J Food Sci Technol. 2014;51(9):2216–2221. | ||
Pirayesh Islamian J, Mehrali H. Lycopene as a carotenoid provides radioprotectant and antioxidant effects by quenching radiation-induced free radical singlet oxygen: an overview. Cell J. 2015;16(4):386–391. | ||
Wu Y, Li YH, Gao XH, Chen HD. The application of nanoemulsion in dermatology: an overview. J Drug Target. 2013;21(4):321–327. | ||
Carlotti ME, Sapino S, Ugazio E, Gallarate M, Morel S. Resveratrol in solid lipid nanoparticles. J Disper Sci Technol. 2012;33(4):465–471. | ||
Scalia S, Franceschinis E, Bertelli D, Iannuccelli V. Comparative evaluation of the effect of permeation enhancers, lipid nanoparticles and colloidal silica on in vivo human skin penetration of quercetin. Skin Pharmacol Physiol. 2013;26(2):57–67. | ||
Geetha T, Kapila M, Prakash O, Deol PK, Kakkar V, Kaur IP. Sesamol-loaded solid lipid nanoparticles for treatment of skin cancer. J Drug Target. 2015;23(2):159–169. | ||
Jenning V, Gysler A, Schafer-Korting M, Gohla SH. Vitamin A loaded solid lipid nanoparticles for topical use: occlusive properties and drug targeting to the upper skin. Eur J Pharm Biopharm. 2000;49(3):211–218. | ||
Jenning V, Schäfer-Korting M, Gohla S. Vitamin A-loaded solid lipid nanoparticles for topical use: drug release properties. J Control Release. 2000;66(2–3):115–126. | ||
Zamarioli CM, Martinsh RM, Carvalho EC, Freitas LAP. Nanoparticles containing curcuminoids (Curcuma longa): development of topical delivery formulation. Rev Bras Farmacogn. 2015;25(1):53–60. | ||
Scalia S, Mezzena M. Photostabilization effect of quercetin on the UV filter combination, butyl methoxydibenzoylmethane-octyl methoxycinnamate. Photochem Photobiol. 2010;86(2):273–278. | ||
Puglia C, Offerta A, Tirendi GG, et al. Design of solid lipid nanoparticles for caffeine topical administration. Drug Deliv. 2016;23(1):36–40. | ||
Caddeo C, Teskac K, Sinico C, Kristl J. Effect of resveratrol incorporated in liposomes on proliferation and UV-B protection of cells. Int J Pharm. 2008;363(1–2):183–191. | ||
Teskac K, Kristl J. The evidence for solid lipid nanoparticles mediated cell uptake of resveratrol. Int J Pharm. 2010;390(1):61–69. | ||
Mitri K, Shegokar R, Gohla S, Anselmi C, Müller RH. Lipid nanocarriers for dermal delivery of lutein: preparation, characterization, stability and performance. Int J Pharm. 2011;414(1–2):267–275. | ||
Bose S, Michniak-Kohn B. Preparation and characterization of lipid based nanosystems for topical delivery of quercetin. Eur J Pharm Sci. 2013;48(3):442–452. | ||
Doktorovova S, Souto EB. Nanostructured lipid carrier-based hydrogel formulations for drug delivery: a comprehensive review. Expert Opin Drug Deliv. 2009;6(2):165–176. | ||
Jain A, Garg NK, Jain A, et al. A synergistic approach of adapalene-loaded nanostructured lipid carriers, and vitamin C co-administration for treating acne. Drug Dev Ind Pharm. Epub 2015 Nov 17:1–9. | ||
Okonogi S, Riangjanapatee P. Physicochemical characterization of lycopene-loaded nanostructured lipid carrier formulations for topical administration. Int J Pharm. 2015;478(2):726–735. | ||
Fan HF, Liu GQ, Huang YQ, Li Y, Xia Q. Development of a nanostructured lipid carrier formulation for increasing photo-stability and water solubility of Phenylethyl Resorcinol. Appl Surf Sci. 2014;288:193–200. | ||
Fan HF, Zhou HF, Ma CL, Huang YQ, Li Y, Xia Q. A novel method for the improved skin whitening effect based on nanostructured lipid carrier. Mol Cryst Liq Cryst. 2014;593(1):232–242. | ||
Gokce EH, Korkmaz E, Dellera E, Sandri G, Bonferoni MC, Ozer O. Resveratrol-loaded solid lipid nanoparticles versus nanostructured lipid carriers: evaluation of antioxidant potential for dermal applications. Int J Nanomedicine. 2012;7:1841–1850. | ||
Detoni CB, Souto GD, da Silva AL, Pohlmann AR, Guterres SS. Photostability and skin penetration of different E-resveratrol-loaded supramolecular structures. Photochem Photobiol. 2012;88(4):913–921. | ||
Lin YK, Al-Suwayeh SA, Leu YL, Shen FM, Fang JY. Squalene-containing nanostructured lipid carriers promote percutaneous absorption and hair follicle targeting of diphencyprone for treating alopecia areata. Pharm Res. 2013;30(2):435–446. | ||
Nemitz MC, Moraes RC, Koester LS, Bassani VL, von Poser GL, Teixeira HF. Bioactive soy isoflavones: extraction and purification procedures, potential dermal use and nanotechnology-based delivery systems. Phytochem Rev. 2015;14(5):849–869. | ||
Schwarz JC, Baisaeng N, Hoppel M, Löw M, Keck CM, Valenta C. Ultra-small NLC for improved dermal delivery of coenyzme Q10. Int J Pharmaceut. 2013;447(1–2):213–217. | ||
Calligaris S, Comuzzo P, Bot F, et al. Nanoemulsions as delivery systems of hydrophobic silybin from silymarin extract: effect of oil type on silybin solubility, in vitro bioaccessibility and stability. LWT Food Sci Technol. 2015;63(1):77–84. | ||
Cerqueira-Coutinho C, Santos-Oliveira R, dos Santos E, Mansur CR. Development of a photoprotective and antioxidant nanoemulsion containing chitosan as an agent for improving skin retention. Eng Life Sci. 2015;15(6):593–604. | ||
Goncalves VSS, Rodriguez-Rojo S, De Paz E, Mato C, Martin A, Cocero MJ. Production of water soluble quercetin formulations by pressurized ethyl acetate-in-water emulsion technique using natural origin surfactants. Food Hydrocolloid. 2015;51:295–304. | ||
Ha TVA, Kim S, Choi Y, et al. Antioxidant activity and bioaccessibility of size-different nanoemulsions for lycopene-enriched tomato extract. Food Chem. 2015;178:115–121. | ||
Hategekimana J, Chamba MVM, Shoemaker CF, Majeed H, Zhong F. Vitamin E nanoemulsions by emulsion phase inversion: effect of environmental stress and long-term storage on stability and degradation in different carrier oil types. Colloid Surf A. 2015;483:70–80. | ||
Hategekirnana J, Masamba KG, Ma JG, Zhong F. Encapsulation of vitamin E: effect of physicochemical properties of wall material on retention and stability. Carbohydr Polym. 2015;124:172–179. | ||
Lu LY, Liu Y, Zhang ZF, et al. Pomegranate seed oil exerts synergistic effects with trans-resveratrol in a self-nanoemulsifying drug delivery system. Biol Pharm Bull. 2015;38(10):1658–1662. | ||
Sigward E, Corvis Y, Doan BT, et al. Preparation and evaluation of multiple nanoemulsions containing gadolinium (III) chelate as a potential magnetic resonance imaging (MRI) contrast agent. Pharm Res. 2015;32(9):2983–2994. | ||
Walker RM, Decker EA, McClements DJ. Physical and oxidative stability of fish oil nanoemulsions produced by spontaneous emulsification: effect of surfactant concentration and particle size. J Food Eng. 2015;164:10–20. | ||
Zhang J, Bing L, Reineccius GA. Formation, optical property and stability of orange oil nanoemulsions stabilized by Quallija saponins. Lwt Food Sci Technol. 2015;64(2):1063–1070. | ||
McClements DJ. Advances in fabrication of emulsions with enhanced functionality using structural design principles. Curr Opin Colloid Interface Sci. 2012;17(5):235–245. | ||
Schwarz JC, Klang V, Karall S, Mahrhauser D, Resch GP, Valenta C. Optimisation of multiple W/O/W nanoemulsions for dermal delivery of aciclovir. Int J Pharm. 2012;435(1):69–75. | ||
Bidone J, Argenta DF, Kratz J, et al. Antiherpes activity and skin/mucosa distribution of flavonoids from achyrocline satureioides extract incorporated into topical nanoemulsions. Biomed Res Int. 2015;2015:238010. | ||
Fasolo D, Bassani VL, Teixeira HF. Development of topical nanoemulsions containing quercetin and 3-O-methylquercetin. Pharmazie. 2009;64(11):726–730. | ||
Fasolo D, Schwingel L, Holzschuh M, Bassani V, Teixeira H. Validation of an isocratic LC method for determination of quercetin and methylquercetin in topical nanoemulsions. J Pharm Biomed Anal. 2007;44(5):1174–1177. | ||
Zorzi GK, Caregnato F, Moreira JC, Teixeira HF, Carvalho EL. Antioxidant effect of nanoemulsions containing extract of achyrocline satureioides (Lam) D.C.-Asteraceae. AAPS PharmSciTech. Epub 2015 Sep 11. | ||
Brownlow B, Nagaraj VJ, Nayel A, Joshi M, Elbayoumi T. Development and in vitro evaluation of vitamin E-enriched nanoemulsion vehicles loaded with genistein for chemoprevention against UVB-induced skin damage. J Pharm Sci. 2015;104(10):3510–3523. | ||
Clares B, Calpena AC, Parra A, et al. Nanoemulsions (NEs), liposomes (LPs) and solid lipid nanoparticles (SLNs) for retinyl palmitate: effect on skin permeation. Int J Pharm. 2014;473(1–2):591–598. | ||
Calderilla-Fajardo SB, Cázares-Delgadillo J, Villalobos-Garcia R, Quintanar-Guerrero D, Ganem-Quintanar A, Robles R. Influence of sucrose esters on the in vivo percutaneous penetration of octyl methoxycinnamate formulated in nanocapsules, nanoemulsion, and emulsion. Drug Dev Ind Pharm. 2006;32(1):107–113. | ||
Olvera-Martínez BI, Cázares-Delgadillo J, Calderilla-Fajardo SB, Villalobos-Garcia R, Ganem-Quintanar A, Quintanar-Guerrero D. Preparation of polymeric nanocapsules containing octyl methoxycinnamate by the emulsification-diffusion technique: penetration across the stratum corneum. J Pharm Sci. 2005;94(7):1552–1559. | ||
Weiss-Angeli V, Poletto FS, de Marco SL, et al. Sustained antioxidant activity of quercetin-loaded lipid-core nanocapsules. J Nanosci Nanotechnol. 2012;12(3):2874–2880. | ||
Guterres SS, Alves MP, Pohlmann AR. Polymeric nanoparticles, nanospheres and nanocapsules, for cutaneous applications. Drug Target Insights. 2007;2:147–157. | ||
Contri RV, Katzer T, Ourique AF, et al. Combined effect of polymeric nanocapsules and chitosan hydrogel on the increase of capsaicinoids adhesion to the skin surface. J Biomed Nanotechnol. 2014;10(5):820–830. | ||
Contri RV, Kaiser M, Poletto FS, Pohlmann AR, Guterres SS. Simultaneous control of capsaicinoids release from polymeric nanocapsules. J Nanosci Nanotechnol. 2011;11(3):2398–2406. | ||
Contri RV, Soares RM, Pohlmann AR, Guterres SS. Structural analysis of chitosan hydrogels containing polymeric nanocapsules. Mater Sci Eng C Mater Biol Appl. 2014;42:234–242. | ||
Ajazuddin AS, Saraf S. Applications of novel drug delivery system for herbal formulations. Fitoterapia. 2010;81(7):680–689. | ||
Allam AN, Komeil IA, Abdallah OY. Curcumin phytosomal softgel formulation: development, optimization and physicochemical characterization. Acta Pharm. 2015;65(3):285–297. | ||
El-Gazayerly ON, Makhlouf AI, Soelm AM, Mohmoud MA. Antioxidant and hepatoprotective effects of silymarin phytosomes compared to milk thistle extract in CCl4 induced hepatotoxicity in rats. J Microencapsul. 2014;31(1):23–30. | ||
Hou Z, Li Y, Huang Y, et al. Phytosomes loaded with mitomycin C-soybean phosphatidylcholine complex developed for drug delivery. Mol Pharm. 2013;10(1):90–101. | ||
Pandita A, Sharma P. Pharmacosomes: an emerging novel vesicular drug delivery system for poorly soluble synthetic and herbal drugs. ISRN Pharm. 2013;2013:348186. | ||
Shakeri A, Sahebkar A. Phytosome: a fatty solution for efficient formulation of phytopharmaceuticals. Recent Pat Drug Deliv Formul. 2015;10(1):7–10. | ||
Bombardelli E. Phytosome: new cosmetic delivery system. Boll Chim Farm. 1991;130(11):431–438. | ||
Damle M, Mallya R. Development and evaluation of a novel delivery system containing phytophospholipid complex for skin aging. AAPS PharmSciTech. Epub 2015 Aug 20. | ||
Gunasekaran T, Haile T, Nigusse T, Dhanaraju MD. Nanotechnology: an effective tool for enhancing bioavailability and bioactivity of phytomedicine. Asian Pac J Trop Biomed. 2014;4(Suppl 1):S1–S7. | ||
Saraf S, Gupta A, Alexander A, Khan J, Jangde M, Saraf S. Advancements and avenues in nanophytomedicines for better pharmacological responses. J Nanosci Nanotechnol. 2015;15(6):4070–4079. | ||
Demir B, Barlas FB, Guler E, et al. Gold nanoparticle loaded phytosomal systems: synthesis, characterization and in vitro investigations. RSC Adv. 2014;4(65):34687–34695. | ||
Singh D, Rawat MS, Semalty A, Semalty M. Rutin-phospholipid complex: an innovative technique in novel drug delivery system-NDDS. Curr Drug Deliv. 2012;9(3):305–314. | ||
de Pera M, Coderch L, Fonollosa J, de la Maza A, Parra JL. Effect of internal wool lipid liposomes on skin repair. Skin Pharmacol Appl Skin Physiol. 2000;13(3–4):188–195. | ||
Kaur IP, Agrawal R. Nanotechnology: a new paradigm in cosmeceuticals. Recent Pat Drug Deliv Formul. 2007;1(2):171–182. | ||
Raza K, Shareef MA, Singal P, Sharma G, Negi P, Katare OP. Lipid-based capsaicin-loaded nano-colloidal biocompatible topical carriers with enhanced analgesic potential and decreased dermal irritation. J Liposome Res. 2014;24(4):290–296. | ||
Takahashi M, Kitamoto D, Asikin Y, Takara K, Wada K. Liposomes encapsulating aloe vera leaf gel extract significantly enhance proliferation and collagen synthesis in human skin cell lines. J Oleo Sci. 2009;58(12):643–650. | ||
Patra S, Roy E, Madhuri R, Sharma PK. The next generation cell-penetrating peptide and carbon dot conjugated nano-liposome for transdermal delivery of curcumin. Biomater Sci. 2015;4(3):418–429. | ||
Chen Y, Wu Q, Zhang Z, Yuan L, Liu X, Zhou L. Preparation of curcumin-loaded liposomes and evaluation of their skin permeation and pharmacodynamics. Molecules. 2012;17(5):5972–5987. | ||
You J, Dai DB, He WJ, et al. [Preparation of curcumin-loaded long-circulating liposomes and its pharmacokinetics in rats]. Zhongguo zhongyao zazhi/China J Chin Materia Medica. 2014;39(7):1238–1242. [Chinese]. | ||
Manca ML, Castangia I, Zaru M, et al. Development of curcumin loaded sodium hyaluronate immobilized vesicles (hyalurosomes) and their potential on skin inflammation and wound restoring. Biomaterials. 2015;71:100–109. | ||
Moghassemi S, Hadjizadeh A. Nano-niosomes as nanoscale drug delivery systems: an illustrated review. J Control Release. 2014;185:22–36. | ||
Moghassemi S, Parnian E, Hakamivala A, et al. Uptake and transport of insulin across intestinal membrane model using trimethyl chitosan coated insulin niosomes. Mater Sci Eng C Mater Biol Appl. 2015;46:333–340. | ||
Pando D, Matos M, Gutiérrez G, Pazos C. Formulation of resveratrol entrapped niosomes for topical use. Colloid Surf B. 2015;128:398–404. | ||
Coviello T, Trotta AM, Marianecci C, et al. Gel-embedded niosomes: preparation, characterization and release studies of a new system for topical drug delivery. Colloids Surf B Biointerfaces. 2015;125:291–299. | ||
Khazaeli P, Pardakhty A, Shoorabi H. Caffeine-loaded niosomes: characterization and in vitro release studies. Drug Deliv. 2007;14(7):447–452. | ||
Pardakhty A, Shakibaie M, Daneshvar H, Khamesipour A, Mohammadi-Khorsand T, Forootanfar H. Preparation and evaluation of niosomes containing autoclaved Leishmania major: a preliminary study. J Microencapsul. 2012;29(3):219–224. | ||
Varshosaz J, Taymouri S, Pardakhty A, Asadi-Shekaari M, Babaee A. Niosomes of ascorbic acid and alpha-tocopherol in the cerebral ischemia-reperfusion model in male rats. Biomed Res Int. 2014;2014:816103. | ||
Muzzalupo R, Tavano L, Lai F, Picci N. Niosomes containing hydroxyl additives as percutaneous penetration enhancers: effect on the transdermal delivery of sulfadiazine sodium salt. Colloids Surf B Biointerfaces. 2014;123:207–212. | ||
Tavano L, Muzzalupo R, Picci N, de Cindio B. Co-encapsulation of lipophilic antioxidants into niosomal carriers: percutaneous permeation studies for cosmeceutical applications. Colloids Surf B Biointerfaces. 2014;114:144–149. | ||
Tavano L, Pinazo A, Abo-Riya M, et al. Cationic vesicles based on biocompatible diacyl glycerol-arginine surfactants: physicochemical properties, antimicrobial activity, encapsulation efficiency and drug release. Colloids Surf B Biointerfaces. 2014;120:160–167. | ||
Tavano L, Aiello R, Ioele G, Picci N, Muzzalupo R. Niosomes from glucuronic acid-based surfactant as new carriers for cancer therapy: preparation, characterization and biological properties. Colloids Surf B Biointerfaces. 2014;118:7–13. | ||
Tavano L, Alfano P, Muzzalupo R, de Cindio B. Niosomes vs microemulsions: new carriers for topical delivery of capsaicin. Colloids Surf B Biointerfaces. 2011;87(2):333–339. | ||
Tavano L, Gentile L, Oliviero Rossi C, Muzzalupo R. Novel gel-niosomes formulations as multicomponent systems for transdermal drug delivery. Colloids Surf B Biointerfaces. 2013;110:281–288. | ||
Tavano L, Muzzalupo R, Mauro L, Pellegrino M, Andò S, Picci N. Transferrin-conjugated pluronic niosomes as a new drug delivery system for anticancer therapy. Langmuir. 2013;29(41):12638–12646. | ||
Ascenso A, Raposo S, Batista C, et al. Development, characterization, and skin delivery studies of related ultradeformable vesicles: transfersomes, ethosomes, and transethosomes. Int J Nanomed. 2015;10:5837–5851. | ||
Bseiso EA, Nasr M, Sammour O, Abd El Gawad NA. Recent advances in topical formulation carriers of antifungal agents. Indian J Dermatol Ve. 2015;81(5):457–463. | ||
Choi JH, Cho SH, Yun JJ, Yu YB, Cho CW. Ethosomes and transfersomes for topical delivery of ginsenoside Rh1 from red ginseng: characterization and in vitro evaluation. J Nanosci Nanotechno. 2015;15(8):5660–5662. | ||
Guo F, Wang J, Ma M, Tan FP, Li N. Skin targeted lipid vesicles as novel nano-carrier of ketoconazole: characterization, in vitro and in vivo evaluation. J Mater Sci Mater M. 2015;26(4):175. | ||
Khan NR, Harun MS, Nawaz A, Harjoh N, Wong TW. Nanocarriers and their actions to improve skin permeability and transdermal drug delivery. Curr Pharm Des. 2015;21(20):2848–2866. | ||
Nasr M, Abdel-Hamid S, Alyoussef AA. A highlight on lipid based nanocarriers for transcutaneous immunization. Curr Pharm Biotechnol. 2015;16(4):371–379. | ||
Rahman M, Akhter S, Ahmad J, Ahmad MZ, Beg S, Ahmad FJ. Nanomedicine-based drug targeting for psoriasis: potentials and emerging trends in nanoscale pharmacotherapy. Expert Opin Drug Deliv. 2015;12(4):635–652. | ||
Singh D, Pradhan M, Nag M, Singh MR. Vesicular system: versatile carrier for transdermal delivery of bioactives. Artif Cell Nanomed B. 2015;43(4):282–290. | ||
Shukla P, Gupta G, Singodia D, et al. Emerging trend in nano-engineered polyelectrolyte-based surrogate carriers for delivery of bioactives. Expert Opin Drug Deliv. 2010;7(9):993–1011. | ||
Verma P, Pathak K. Therapeutic and cosmeceutical potential of ethosomes: an overview. J Adv Pharm Technol Res. 2010;1(3):274–282. | ||
Hua S. Lipid-based nano-delivery systems for skin delivery of drugs and bioactives. Front Pharmacol. 2015;6:219. | ||
Kaur CD, Saraf S. Topical vesicular formulations of Curcuma longa extract on recuperating the ultraviolet radiation-damaged skin. J Cosmet Dermatol. 2011;10(4):260–265. | ||
Lopes CM, Coelho PB, Oliveira R. Novel delivery systems for anti-allergic agents: allergic disease and innovative treatments. Curr Drug Deliv. 2015;12(4):382–396. | ||
Vanić Z, Holæter AM, Skalko-Basnet N. (Phospho) lipid-based nanosystems for skin administration. Curr Pharm Des. 2015;21(29):4174–4192. | ||
Manca ML, Castangia I, Caddeo C, et al. Improvement of quercetin protective effect against oxidative stress skin damages by incorporation in nanovesicles. Colloids Surf B Biointerfaces. 2014;123:566–574. | ||
Moulaoui K, Caddeo C, Manca ML, et al. Identification and nanoentrapment of polyphenolic phytocomplex from Fraxinus angustifolia: in vitro and in vivo wound healing potential. Eur J Med Chem. 2015;89:179–188. | ||
Castangia I, Caddeo C, Manca ML, et al. Delivery of liquorice extract by liposomes and hyalurosomes to protect the skin against oxidative stress injuries. Carbohydr Polym. 2015;134:657–663. | ||
Dellinger AL, Cunin P, Lee D, et al. Inhibition of inflammatory arthritis using fullerene nanomaterials. PLoS One. 2015;10(4):e0126290. | ||
Dombi GW, Purohit K, Martin LM, Yang SC. Collagen gel formation in the presence of a carbon nanobrush. J Mater Sci Mater Med. 2015;26(1):5356. | ||
Ngan CL, Basri M, Tripathy M, Abedi Karjiban R, Abdul-Malek E. Skin intervention of fullerene-integrated nanoemulsion in structural and collagen regeneration against skin aging. Eur J Pharm Sci. 2015;70:22–28. | ||
Souto GD, Pohlmann AR, Guterres SS. Ultraviolet A irradiation increases the permeation of fullerenes into human and porcine skin from C(6)(0)-poly(vinylpyrrolidone) aggregate dispersions. Skin Pharmacol Physiol. 2015;28(1):22–30. | ||
Tim M. Strategies to optimize photosensitizers for photodynamic inactivation of bacteria. J Photochem Photobiol B Biol. 2015;150:2–10. | ||
Ito S, Itoga K, Yamato M, Akamatsu H, Okano T. The co-application effects of fullerene and ascorbic acid on UV-B irradiated mouse skin. Toxicology. 2010;267(1–3):27–38. | ||
Ito Y, Warner JH, Brown R, et al. Controlling intermolecular spin interactions of La@C(82) in empty fullerene matrices. Phys Chem Chem Phys. 2010;12(7):1618–1623. | ||
Inui S, Aoshima H, Ito M, Kobuko K, Itami S. Inhibition of sebum production and Propionibacterium acnes lipase activity by fullerenol, a novel polyhydroxylated fullerene: potential as a therapeutic reagent for acne. J Cosmet Sci. 2012;63(4):259–265. | ||
Inui S, Aoshima H, Nishiyama A, Itami S. Improvement of acne vulgaris by topical fullerene application: unique impact on skin care. Nanomedicine. 2011;7(2):238–241. | ||
Inui S, Mori A, Ito M, Hyodo S, Itami S. Reduction of conspicuous facial pores by topical fullerene: possible role in the suppression of PGE2 production in the skin. J Nanobiotechnology. 2014;12:6. | ||
Yin R, Hamblin MR. Antimicrobial photosensitizers: drug discovery under the spotlight. Curr Med Chem. 2015;22(18):2159–2185. | ||
Zhang Z, Chen X, Rao W, Long F, Yan L, Yin Y. Preparation of novel curcumin-imprinted polymers based on magnetic multi-walled carbon nanotubes for the rapid extraction of curcumin from ginger powder and kiwi fruit root. J Sep Sci. 2015;38(1):108–114. | ||
Gupta S, Bansal R, Gupta S, Jindal N, Jindal A. Nanocarriers and nanoparticles for skin care and dermatological treatments. Indian Dermatol Online J. 2013;4(4):267–272. | ||
Ilbasmis-Tamerc S, Degim T. A feasible way to use carbon nanotubes to deliver drug molecules: transdermal application. Expert Opin Drug Deliv. 2012;9(8):991–999. | ||
Demming A. Nanotechnology under the skin. Nanotechnology. 2011;22(26):260201. | ||
Sahoo NG, Bao H, Pan Y, et al. Functionalized carbon nanomaterials as nanocarriers for loading and delivery of a poorly water-soluble anticancer drug: a comparative study. Chem Commun. 2011;47(18):5235–5237. | ||
Li H, Zhang N, Hao Y, et al. Formulation of curcumin delivery with functionalized single-walled carbon nanotubes: characteristics and anticancer effects in vitro. Drug Deliv. 2014;21(5):379–387. | ||
Tripodo G, Trapani A, Torre ML, Giammona G, Trapani G, Mandracchia D. Hyaluronic acid and its derivatives in drug delivery and imaging: recent advances and challenges. Eur J Pharm Biopharm. 2015;97(Pt B):400–416. | ||
Al-Rohaimi AH. Comparative anti-inflammatory potential of crystalline and amorphous nano curcumin in topical drug delivery. J Oleo Sci. 2015;64(1):27–40. | ||
Chaudhary H, Kohli K, Kumar V. Nano-transfersomes as a novel carrier for transdermal delivery. Int J Pharm. 2013;454(1):367–380. | ||
Chaudhary H, Kohli K, Kumar V. A novel nano-carrier transdermal gel against inflammation. Int J Pharm. 2014;465(1–2):175–186. | ||
Suwannateep N, Wanichwecharungruang S, Fluhr J, Patzelt A, Lademann J, Meinke MC. Comparison of two encapsulated curcumin particular systems contained in different formulations with regard to in vitro skin penetration. Skin Res Technol. 2013;19(1):1–9. | ||
Bonechi C, Martini S, Ciani L, et al. Using liposomes as carriers for polyphenolic compounds: the case of trans-resveratrol. PLoS One. 2012;7(8):e41438. | ||
Montanari J, Vera M, Mensi E, Morilla M, Romero E. Nanoberries for topical delivery of antioxidants. J Cosmet Sci. 2013;64(6):469–481. | ||
Sun Y, Du L, Liu Y, et al. Transdermal delivery of the in situ hydrogels of curcumin and its inclusion complexes of hydroxypropyl-beta-cyclodextrin for melanoma treatment. Int J Pharm. 2014;469(1):31–39. | ||
Thitilertdecha P, Guy RH, Rowan MG. Characterisation of polyphenolic compounds in Clerodendrum petasites S. Moore and their potential for topical delivery through the skin. J Ethnopharmacol. 2014;154(2):400–407. | ||
Zillich OV, Schweiggert-Weisz U, Eisner P, Kerscher M. Polyphenols as active ingredients for cosmetic products. Int J Cosmet Sci. 2015;37(5):455–464. | ||
Ganesan P, Ko HM, Kim IS, Choi DK. Recent trends in the development of nanophytobioactive compounds and delivery systems for their possible role in reducing oxidative stress in Parkinson’s disease models. Int J Nanomedicine. 2015;10:6757–6772. | ||
Bose S, Du YC, Takhistov P, Michniak-Kohn B. Formulation optimization and topical delivery of quercetin from solid lipid based nanosystems. Int J Pharm. 2013;441(1–2):56–66. | ||
Han SB, Kwon SS, Jeong YM, Yu ER, Park SN. Physical characterization and in vitro skin permeation of solid lipid nanoparticles for transdermal delivery of quercetin. Int J Cosmet Sci. 2014;36(6):588–597. | ||
Ali SW, Purwar R, Joshi M, Rajendran S. Antibacterial properties of aloe vera gel-finished cotton fabric. Cellulose. 2014;21(3):2063–2072. | ||
Balan BJ, Niemcewicz M, Kocik J, Jung L, Skopińska-Różewska E, Skopiński P. Oral administration of aloe vera gel, anti-microbial and anti-inflammatory herbal remedy, stimulates cell-mediated immunity and antibody production in a mouse model. Cent Eur J Immunol. 2014;39(2):125–130. | ||
Cui Y, Wang HY, Yao WR, Li YC, Xia XH, Qian H. Hepatoprotective effect of aloe vera polysaccharides against aflatoxin B-1-induced hepatotoxicity in rats. Fresen Environ Bull. 2014;23(7A):1635–1642. | ||
Gentilini R, Bozzini S, Munarin F, Petrini P, Visai L, Tanzi MC. Pectins from aloe vera: extraction and production of gels for regenerative medicine. J Appl Polym Sci. 2014;131(2):39760. | ||
Kang MC, Kim SY, Min YT, et al. In vitro and in vivo antioxidant activities of polysaccharide purified from aloe vera (Aloe barbadensis) gel. Carbohyd Polym. 2014;99:365–371. | ||
Fox LT, du Plessis J, Gerber M, van Zyl S, Boneschans B, Hamman JH. In vivo skin hydration and anti-erythema effects of aloe vera, aloe ferox and aloe marlothii gel materials after single and multiple applications. Pharmacogn Mag. 2014;10(Suppl 2):S392–S403. | ||
Hajheydari Z, Saeedi M, Morteza-Semnani K, Soltani A. Effect of Aloe vera topical gel combined with tretinoin in treatment of mild and moderate acne vulgaris: a randomized, double-blind, prospective trial. J Dermatol Treat. 2014;25(2):123–129. | ||
Imaga N, Osime E, Ezeji S, Okafor U. Effect of aloe vera gel and selenium on cisplatin-induced oxidative stress in Wister albino rats. FASEB J. 2014;28(Supp 1 LB155). | ||
Tabandeh MR, Oryan A, Mohammadalipour A. Polysaccharides of Aloe vera induce MMP-3 and TIMP-2 gene expression during the skin wound repair of rat. Int J Biol Macromol. 2014;65:424–430. | ||
Olsen DL, Raub W Jr, Bradley C, et al. The effect of aloe vera gel/mild soap versus mild soap alone in preventing skin reactions in patients undergoing radiation therapy. Oncol Nurs Forum. 2001;28(3):543–547. | ||
Tanaka M, Misawa E, Yamauchi K, Abe F, Ishizaki C. Effects of plant sterols derived from Aloe vera gel on human dermal fibroblasts in vitro and on skin condition in Japanese women. Clin Cosmet Investig Dermatol. 2015;8:95–104. | ||
Kitture R, Ghosh S, More PA, et al. Curcumin-loaded, self-assembled aloevera template for superior antioxidant activity and trans-membrane drug release. J Nanosci Nanotechno. 2015;15(6):4039–4045. | ||
Agrawal R, Sandhu SK, Sharma I, Kaur IP. Development and evaluation of curcumin-loaded elastic vesicles as an effective topical anti-inflammatory formulation. AAPS PharmSciTech. 2015;16(2):364–374. | ||
Balasubramanian S, Roselin P, Singh KK, Zachariah J, Saxena SN. Post harvest processing and benefits of black pepper, coriander, cinnamon, fenugreek and turmeric spices. Crit Rev Food Sci Nutr. Epub 2015 Mar 6. | ||
Deb L, Laishram S, Khumukcham N, et al. Past, present and perspectives of Manipur traditional medicine: a major health care system available for rural population in the North-East India. J Ethnopharmacol. 2015;169:387–400. | ||
Kocaadam B, Şanlier N. Curcumin, an active component of turmeric (Curcuma longa), and its effects on health. Crit Rev Food Sci Nutr. Epub 2015 Nov 3. | ||
Kumar A, Chetia H, Sharma S, Kabiraj D, Talukdar NC, Bora U. Curcumin resource database. Database. 2015;2015:bav070. | ||
Nimgulkar C, Ghosh S, Sankar AB, et al. Combination of spices and herbal extract restores macrophage foam cell migration and abrogates the athero-inflammatory signalling cascade of atherogenesis. Vascul Pharmacol. 2015;72:53–63. | ||
Srivastava A, Agarwal R, Chaturvedi TP, Chandra A, Singh OP. Clinical evaluation of the role of tulsi and turmeric in the management of oral submucous fibrosis: a pilot, prospective observational study. J Ayurveda Integr Med. 2015;6(1):45–49. | ||
Agarwal R, Gupta SK, Srivastava S, Agarwal P, Agrawal SS. Therapeutic potential of Curcuma longa, the golden spice of India, in drug discovery for ophthalmic diseases. Expert Opin Drug Discov. 2009;4(2):147–158. | ||
Goel A, Aggarwal BB. Curcumin, the golden spice from Indian saffron, is a chemosensitizer and radiosensitizer for tumors and chemoprotector and radioprotector for normal organs. Nutr Cancer. 2010;62(7):919–930. | ||
Gupta SC, Kismali G, Aggarwal BB. Curcumin, a component of turmeric: from farm to pharmacy. Biofactors. 2013;39(1):2–13. | ||
Gupta SC, Patchva S, Aggarwal BB. Therapeutic roles of curcumin: lessons learned from clinical trials. AAPS J. 2013;15(1):195–218. | ||
Gupta SC, Patchva S, Koh W, Aggarwal BB. Discovery of curcumin, a component of golden spice, and its miraculous biological activities. Clin Exp Pharmacol Physiol. 2012;39(3):283–299. | ||
Prasad S, Aggarwal BB. Turmeric, the golden spice: from traditional medicine to modern medicine. In: Benzie IFF, Wachtel-Galor S, editors. Herbal Medicine: Biomolecular and Clinical Aspects. 2nd ed. Boca Raton, FL: Taylor & Francis; 2011. | ||
Prasad S, Gupta SC, Tyagi AK, Aggarwal BB. Curcumin, a component of golden spice: from bedside to bench and back. Biotechnol Adv. 2014;32(6):1053–1064. | ||
Prasad S, Tyagi AK, Aggarwal BB. Recent developments in delivery, bioavailability, absorption and metabolism of curcumin: the golden pigment from golden spice. Cancer Res Treat. 2014;46(1):2–18. | ||
Rungphanichkul N, Nimmannit U, Muangsiri W, Rojsitthisak P. Preparation of curcuminoid niosomes for enhancement of skin permeation. Pharmazie. 2011;66(8):570–575. | ||
Korać RR, Khambholja KM. Potential of herbs in skin protection from ultraviolet radiation. Pharmacogn Rev. 2011;5(10):164–173. | ||
Kotyla T, Kuo F, Moolchandani V, Wilson T, Nicolosi R. Increased bioavailability of a transdermal application of a nano-sized emulsion preparation. Int J Pharm. 2008;347(1–2):144–148. | ||
Dingler A, Blum RP, Niehus H, Muller RH, Gohla S. Solid lipid nanoparticles (SLN/Lipopearls) – a pharmaceutical and cosmetic carrier for the application of vitamin E in dermal products. J Microencapsul. 1999;16(6):751–767. | ||
Nesseem D. Formulation of sunscreens with enhancement sun protection factor response based on solid lipid nanoparticles. Int J Cosmet Sci. 2011;33(1):70–79. | ||
Pardeike J, Hommoss A, Müller RH. Lipid nanoparticles (SLN, NLC) in cosmetic and pharmaceutical dermal products. Int J Pharm. 2009;366(1–2):170–184. | ||
Shidhaye SS, Vaidya R, Sutar S, Patwardhan A, Kadam VJ. Solid lipid nanoparticles and nanostructured lipid carriers – innovative generations of solid lipid carriers. Curr Drug Deliv. 2008;5(4):324–331. | ||
Song C, Liu S. A new healthy sunscreen system for human: solid lipid nanoparticles as carrier for 3,4,5-trimethoxybenzoylchitin and the improvement by adding Vitamin E. Int J Biol Macromol. 2005;36(1–2):116–119. | ||
Wissing SA, Muller RH. Solid lipid nanoparticles (SLN) – a novel carrier for UV blockers. Pharmazie. 2001;56(10):783–786. | ||
Teo BS, Basri M, Zakaria MR, Salleh AB, Rahman RN, Rahman MB. A potential tocopherol acetate loaded palm oil esters-in-water nanoemulsions for nanocosmeceuticals. J Nanobiotechnology. 2010;8:4. | ||
Crisan D, Roman I, Crisan M, Scharffetter-Kochanek K, Badea R. The role of vitamin C in pushing back the boundaries of skin aging: an ultrasonographic approach. Clin Cosmet Investig Dermatol. 2015;8:463–470. | ||
Peh P, Lim NS, Blocki A, et al. Simultaneous delivery of highly diverse bioactive compounds from blend electrospun fibers for skin wound healing. Bioconjug Chem. 2015;26(7):1348–1358. | ||
Uner M, Wissing SA, Yener G, Muller RH. Skin moisturizing effect and skin penetration of ascorbyl palmitate entrapped in solid lipid nanoparticles (SLN) and nanostructured lipid carriers (NLC) incorporated into hydrogel. Pharmazie. 2005;60(10):751–755. | ||
Guney G, Kutlu HM, Genç L. Preparation and characterization of ascorbic acid loaded solid lipid nanoparticles and investigation of their apoptotic effects. Colloids Surf B Biointerfaces. 2014;121:270–280. | ||
Zhou W, Liu W, Zou L, et al. Storage stability and skin permeation of vitamin C liposomes improved by pectin coating. Colloids Surf B Biointerfaces. 2014;117:330–337. | ||
Yang S, Liu C, Liu W, et al. Preparation and characterization of nanoliposomes entrapping medium-chain fatty acids and vitamin C by lyophilization. Int J Mol Sci. 2013;14(10):19763–19773. | ||
Fathi-Azarbayjani A, Qun L, Chan YW, Chan SY. Novel vitamin and gold-loaded nanofiber facial mask for topical delivery. AAPS PharmSciTech. 2010;11(3):1164–1170. | ||
Yutani R, Teraoka R, Kitagawa S. Microemulsion using polyoxyethylene sorbitan trioleate and its usage for skin delivery of resveratrol to protect skin against UV-induced damage. Chem Pharm Bull. 2015;63(9):741–745. | ||
Ido Y, Duranton A, Lan F, Weikel KA, Breton L, Ruderman NB. Resveratrol prevents oxidative stress-induced senescence and proliferative dysfunction by activating the AMPK-FOXO3 cascade in cultured primary human keratinocytes. PLoS One. 2015;10(2):e0115341. | ||
Soeur J, Eilstein J, Lereaux G, Jones C, Marrot L. Skin resistance to oxidative stress induced by resveratrol: from Nrf2 activation to GSH biosynthesis. Free Radic Biol Med. 2015;78:213–223. | ||
Watanabe K, Shibuya S, Ozawa Y, Izuo N, Shimizu T. Resveratrol derivative-rich melinjo seed extract attenuates skin atrophy in Sod1-deficient mice. Oxid Med Cell Longev. 2015;2015:391075. | ||
Wittenauer J, Mackle S, Sußmann D, Schweiggert-Weisz U, Carle R. Inhibitory effects of polyphenols from grape pomace extract on collagenase and elastase activity. Fitoterapia. 2015;101:179–187. | ||
Pando D, Beltran M, Gerone I, Matos M, Pazos C. Resveratrol entrapped niosomes as yoghurt additive. Food Chem. 2015;170:281–287. | ||
Afaq F, Katiyar SK. Polyphenols: skin photoprotection and inhibition of photocarcinogenesis. Mini Rev Med Chem. 2011;11(14):1200–1215. | ||
Afaq F, Mukhtar H. Botanical antioxidants in the prevention of photocarcinogenesis and photoaging. Exp Dermatol. 2006;15(9):678–684. | ||
Nemen D, Lemos-Senna E. Preparation and characterization of resveratrol-loaded lipid-based nanocarriers for cutaneous administration. Quim Nova. 2011;34(3):408–413. | ||
Scognamiglio I, De Stefano D, Campani V, et al. Nanocarriers for topical administration of resveratrol: a comparative study. Int J Pharm. 2013;440(2):179–187. | ||
Juskaite V, Ramanauskiene K, Briedis V. Design and formulation of optimized microemulsions for dermal delivery of resveratrol. Evid Based Complement Alternat Med. 2015;2015:540916. | ||
Sun R, Zhao G, Ni S, Xia Q. Lipid based nanocarriers with different lipid compositions for topical delivery of resveratrol: comparative analysis of characteristics and performance. J Drug Deliv Sci Tec. 2014;24(6):591–600. | ||
Ali H, Dixit S. Quercetin attenuates the development of 7, 12-dimethyl benz (a) anthracene (DMBA) and croton oil-induced skin cancer in mice. J Biomed Res. 2015;29(2):139–144. | ||
Burak C, Brull V, Langguth P, et al. Higher plasma quercetin levels following oral administration of an onion skin extract compared with pure quercetin dihydrate in humans. Eur J Nutr. 2015;18(4):600–615. | ||
Maini S, Fahlman BM, Krol ES. Flavonols protect against UV radiation-induced thymine dimer formation in an artificial skin mimic. J Pharmacy Pharm Sci. 2015;18(4):600–615. | ||
Park EJ, Kim JY, Jeong MS, et al. Effect of topical application of quercetin-3-O-(2″-gallate)-alpha-l-rhamnopyranoside on atopic dermatitis in NC/Nga mice. J Dermatol Sci. 2015;77(3):166–172. | ||
Sahu S, Saraf S, Kaur CD, Saraf S. Biocompatible nanoparticles for sustained topical delivery of anticancer phytoconstituent quercetin. Pak J Biol Sci. 2013;16(13):601–609. | ||
Ahmad N, Mukhtar H. Cutaneous photochemoprotection by green tea: a brief review. Skin Pharmacol Appl Skin Physiol. 2001;14(2):69–76. | ||
Baumann L. Botanical ingredients in cosmeceuticals. J Drugs Dermatol. 2007;6(11):1084–1088. | ||
Fowler JF Jr, Woolery-Lloyd H, Waldorf H, Saini R. Innovations in natural ingredients and their use in skin care. J Drugs Dermatol. 2010;9(6 Suppl):S72–S81; quiz s82-73. | ||
Ikeda S, Kanoya Y, Nagata S. Effects of a foot bath containing green tea polyphenols on interdigital tinea pedis. Foot. 2013;23(2–3):58–62. | ||
Katiyar SK, Elmets CA. Green tea polyphenolic antioxidants and skin photoprotection (Review). Int J Oncol. 2001;18(6):1307–1313. | ||
Farris P. Idebenone, green tea, and coffeeberry extract: new and innovative antioxidants. Dermatol Ther. 2007;20(5):322–329. | ||
Huang CC, Fang JY, Wu WB, Chiang HS, Wei YJ, Hung CF. Protective effects of (-)-epicatechin-3-gallate on UVA-induced damage in HaCaT keratinocytes. Arch Dermatol Res. 2005;296(10):473–481. | ||
Xia J, Song X, Bi Z, Chu W, Wan Y. UV-induced NF-kappaB activation and expression of IL-6 is attenuated by (-)-epigallocatechin-3-gallate in cultured human keratinocytes in vitro. Int J Mol Med. 2005;16(5):943–950. | ||
Montenegro L. Nanocarriers for skin delivery of cosmetic antioxidants. J Pharm Pharm Res. 2014;2(4):73–92. | ||
Fang JY, Lee WR, Shen SC, Huang YL. Effect of liposome encapsulation of tea catechins on their accumulation in basal cell carcinomas. J Dermatol Sci. 2006;42(2):101–109. | ||
Fang JY, Hwang TL, Huang YL, Fang CL. Enhancement of the transdermal delivery of catechins by liposomes incorporating anionic surfactants and ethanol. Int J Pharm. 2006;310(1–2):131–138. | ||
Gülseren I, Corredig M. Storage stability and physical characteristics of tea-polyphenol-bearing nanoliposomes prepared with milk fat globule membrane phospholipids. J Agric Food Chem. 2013;61(13):3242–3251. | ||
Georgetti SR, Casagrande R, Vicentini FT, Baracat MM, Verri WA Jr, Fonseca MJ. Protective effect of fermented soybean dried extracts against TPA-induced oxidative stress in hairless mice skin. Biomed Res Int. 2013;2013:340626. | ||
Iovine B, Garofalo M, Orefice M, et al. Isoflavones in aglycone solution enhance ultraviolet B-induced DNA damage repair efficiency. Clin Exp Dermatol. 2014;39(3):391–394. | ||
Polito F, Marini H, Bitto A, et al. Genistein aglycone, a soy-derived isoflavone, improves skin changes induced by ovariectomy in rats. Br J Pharmacol. 2012;165(4):994–1005. | ||
Terra VA, Souza-Neto FP, Frade MA, et al. Genistein prevents ultraviolet B radiation-induced nitrosative skin injury and promotes cell proliferation. J Photochem Photobiol B Biol. 2015;144:20–27. | ||
Uzarska M, Czajkowski R, Schwartz RA, Bajek A, Zegarska B, Drewa T. Chemoprevention of skin melanoma: facts and myths. Melanoma Res. 2013;23(6):426–433. | ||
Waqas MK, Akhtar N, Mustafa R, Jamshaid M, Khan HM, Murtaza G. Dermatological and cosmeceutical benefits of Glycine max (soybean) and its active components. Acta Pol Pharm. 2015;72(1):3–11. | ||
de Vargas BA, Bidone J, Oliveira LK, Koester LS, Bassani VL, Teixeira HF. Development of topical hydrogels containing genistein-loaded nanoemulsions. J Biomed Nanotechnol. 2012;8(2):330–336. | ||
Kang KH, Kang MJ, Lee J, Choi YW. Influence of liposome type and skin model on skin permeation and accumulation properties of genistein. J Disper Sci Technol. 2010;31(8):1061–1066. | ||
Anunciato TP, da Rocha Filho PA. Carotenoids and polyphenols in nutricosmetics, nutraceuticals, and cosmeceuticals. J Cosmet Dermatol. 2012;11(1):51–54. | ||
Evans JA, Johnson EJ. The role of phytonutrients in skin health. Nutrients. 2010;2(8):903–928. | ||
Hu W, Dai D, Li W. Anti-aging effect of Blakeslea trispora powder on adult mice. Biotechnol Lett. 2013;35(8):1309–1315. | ||
Jenkins G, Wainwright LJ, Holland R, Barrett KE, Casey J. Wrinkle reduction in post-menopausal women consuming a novel oral supplement: a double-blind placebo-controlled randomized study. Int J Cosmet Sci. 2014;36(1):22–31. | ||
Lopes LB, VanDeWall H, Li HT, et al. Topical delivery of lycopene using microemulsions: enhanced skin penetration and tissue antioxidant activity. J Pharm Sci. 2010;99(3):1346–1357. | ||
Meinke MC, Darvin ME, Vollert H, Lademann J. Bioavailability of natural carotenoids in human skin compared to blood. Eur J Pharm Biopharm. 2010;76(2):269–274. | ||
Rizwan M, Rodriguez-Blanco I, Harbottle A, Birch-Machin MA, Watson RE, Rhodes LE. Tomato paste rich in lycopene protects against cutaneous photodamage in humans in vivo: a randomized controlled trial. Br J Dermatol. 2011;164(1):154–162. | ||
Sokoloski L, Borges M, Bagatin E. Lycopene not in pill, nor in natura has photoprotective systemic effect. Arch Dermatol Res. 2015;307(6):545–549. | ||
Stahl W, Heinrich U, Aust O, Tronnier H, Sies H. Lycopene-rich products and dietary photoprotection. Photochem Photobiol Sci. 2006;5(2):238–242. | ||
Stahl W, Sies H. Photoprotection by dietary carotenoids: concept, mechanisms, evidence and future development. Mol Nutr Food Res. 2012;56(2):287–295. | ||
Riangjanapatee P, Muller RH, Keck CM, Okonogi S. Development of lycopene-loaded nanostructured lipid carriers: effect of rice oil and cholesterol. Pharmazie. 2013;68(9):723–731. | ||
Riangjanapatee P, Okonogi S. Effect of surfactant on lycopene-loaded nanostructured lipid carriers. Drug Discov Ther. 2012;6(3):163–168. | ||
Ascenso A, Pinho S, Eleutério C, et al. Lycopene from tomatoes: vesicular nanocarrier formulations for dermal delivery. J Agric Food Chem. 2013;61(30):7284–7293. | ||
Raj S, Jose S, Sumod US, Sabitha M. Nanotechnology in cosmetics: opportunities and challenges. J Pharm Bioallied Sci. 2012;4(3):186–193. | ||
Pereira F, Baptista R, Ladeiras D, et al. Production and characterization of nanoparticles containing methanol extracts of Portuguese Lavenders. Measurement. 2015;74:170–177. | ||
Lacatusu I, Badea N, Murariu A, Nichita C, Bojin D, Meghea A. Antioxidant capacity of lipid nanoparticles loaded with rosemary extract. Mol Cryst Liq Cryst. 2010;523:260–272. | ||
Kumar N, Tharatha S, Chaiyasut C. Development and validation of simple isocratic high performance liquid chromatography-ultraviolet (HPLC-UV) method for determination of safflower yellow in Carthamus tinctorius (L.)-loaded nanostructured lipid carriers (NLC). Afr J Pharm Pharmacol. 2011;5(20):2335–2341. | ||
Chen-yu G, Chun-fen Y, Qi-lu L, et al. Development of a quercetin-loaded nanostructured lipid carrier formulation for topical delivery. Int J Pharm. 2012;430(1–2):292–298. | ||
Shen CY, Dai L, Shen BD, et al. Nanostructured lipid carrier based topical gel of ganoderma triterpenoids for frostbite treatment. Chin J Nat Med. 2015;13(6):454–460. | ||
Hwang SL, Kim JC. In vivo hair growth promotion effects of cosmetic preparations containing hinokitiol-loaded poly(epsilon-caprolacton) nanocapsules. J Microencapsul. 2008;25(5):351–356. | ||
Yang X, Lee HY, Kim JC. In vitro skin permeation of hinokitiol loaded in vesicles composed of behenyltrimethylammonium chloride and stearic acid. Drug Dev Ind Pharm. 2010;36(5):556–562. | ||
Suwannateep N, Wanichwecharungruang S, Haag SF, et al. Encapsulated curcumin results in prolonged curcumin activity in vitro and radical scavenging activity ex vivo on skin after UVB-irradiation. Eur J Pharm Biopharm. 2012;82(3):485–490. | ||
Abla MJ, Banga AK. Formulation of tocopherol nanocarriers and in vitro delivery into human skin. Int J Cosmet Sci. 2014;36(3): 239–246. | ||
Lu WC, Chiang BH, Huang DW, Li PH. Skin permeation of D-limonene-based nanoemulsions as a transdermal carrier prepared by ultrasonic emulsification. Ultrason Sonochem. 2014;21(2):826–832. | ||
Butnariu MV, Giuchici CV. The use of some nanoemulsions based on aqueous propolis and lycopene extract in the skin’s protective mechanisms against UVA radiation. J Nanobiotechnology. 2011;9:3. |
 © 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2016 The Author(s). This work is published and licensed by Dove Medical Press Limited. The
full terms of this license are available at https://www.dovepress.com/terms.php
and incorporate the Creative Commons Attribution
- Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted
without any further permission from Dove Medical Press Limited, provided the work is properly
attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
