Back to Journals » Risk Management and Healthcare Policy » Volume 13
Critically Ill Patients with Coronavirus Disease 2019 in a Designated ICU: Clinical Features and Predictors for Mortality
Authors Wang ZH, Shu C, Ran X, Xie CH, Zhang L
Received 16 May 2020
Accepted for publication 7 July 2020
Published 20 July 2020 Volume 2020:13 Pages 833—845
DOI https://doi.org/10.2147/RMHP.S263095
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Professor Marco Carotenuto
Zhao-Hua Wang,1,* Chang Shu,2,* Xiao Ran,1 Cui-Hong Xie,1 Lei Zhang2
1Department of Critical Care Medicine, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430030, People’s Republic of China; 2Department of Surgery, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430030, People’s Republic of China
*These authors contributed equally to this work
Correspondence: Lei Zhang
Department of Surgery, Tongji Hospital, 1095 Jiefang Avenue, Wuhan 430030, People’s Republic of China
Tel +86-27-83665312
Email [email protected]
Background: Coronavirus disease 2019 (COVID-19) is a worldwide pandemic outbreak with a high mortality. Prognostic factors of critically ill patients with COVID-19 have not been fully elucidated yet.
Methods: In the present study, 59 patients with COVID-19 from the intensive care unit of the Caidian Branch of Tongji Hospital were enrolled. Epidemiological, demographic, clinical, laboratory, radiological, treatment data, and clinical outcomes were collected. Prognostic factors were statistically defined.
Results: Of the 59 patients studied (67.4± 11.3 years), 38 patients were male, 51 had underlying diseases, and 41 patients died during admission. Compared with the survivors, the deceased patients were of older age, had more smoking history, severer fatigue, and diarrhea, a higher incidence of multiple organ injuries, more deteriorative lymphopenia and thrombocytopenia, remarkably impaired cellular immune response, and strengthened cytokine release. Age higher than 70 (OR=2.76, 95% CI=1.45– 5.23), arrhythmia (OR=4.76, 95% CI=1.59– 14.25), and a Sequential Organ Failure Assessment (SOFA) score above 4 (OR=5.16, 95% CI=1.29– 20.55) were identified as risk factors for mortality of patients.
Conclusion: Critically ill COVID-19 patients aged higher than 70, arrhythmia, or a SOFA score above 4 have a high risk of mortality, and need prior medical intervention.
Keywords: severe acute respiratory coronavirus 2, pneumonia, critically ill, mortality, prognostic factor
Introduction
Since December 2019, an outbreak of coronavirus disease 2019 (COVID-19) caused by severe acute respiratory coronavirus 2 (SARS-CoV-2) has been declared a global worldwide pandemic.1
Up to now, no specific therapeutic agents or vaccines for COVID-19 are available. Several therapies, such as remdesivir and favipiravir, are under investigation, but the antiviral efficacy of these drugs is not yet known. The high ability of infection and limited treatment strategies caused one of the major characters of COVID-19, that is a rapid progression from sole pulmonary infection to multiple organ dysfunction (shock, acute respiratory distress syndrome [ARDS], cardiac injury, and acute kidney injury [AKI]), which manifest as extraordinary high mortality in such critically ill COVID-19 patients.
Several previous descriptive cohort studies have reported epidemiological, demographic, and clinical characteristics of COVID-19 patients, as well as severe cases.2,3 Though advanced age, dyspnea, anorexia, and underlying comorbidities are more common in critically ill patients,2,4 the risk factors contributing to the mortality of critically ill COVID-19 have not been fully identified.
In the present study, we present details of 59 critically ill patients with SARS-CoV-2 infection, admitted to the intensive care unit (ICU) of Caidian Branch of Tongji Hospital, and then identified prognostic factors for mortality of these critically ill patients. Those data may facilitate the screening of the most dangerous critically ill COVID-19 patients and set the priority of medical intervention to them.
Patients and Methods
Patients
From February 2, 2020 to March 1, 2020, a total of 59 critically ill COVID-19 patients from the intensive care unit of Caidian Branch of Tongji Hospital were enrolled in this single-center and retrospective study. Caidian Branch of Tongji Hospital (also named Wuhan Sino-German Friendship Hospital) is designated as the hospital to diagnose and treat the COVID-19 patients in Wuhan City, China.
Laboratory confirmation of SARS-CoV-2 infection was performed on throat swab samples by using real-time PCR assay, as previously described.3,4 According to the WHO interim guidance and Diagnostic and Treatment Program of COVID-19 (Version 7.0) published by the National Health Commission of the People’s Republic of China, all patients were diagnosed with severe pneumonia induced by SARS-CoV-2 infection who required mechanical ventilation, had inspiratory oxygen fraction (FiO₂) ≥60%, or had the shock or organ failure.4,6 The clinical outcomes were monitored up to March 8, 2020, the final date of follow-up. Informed Oral Consent was obtained from the patients, and written informed consent was waived due to the rapid emergence of the disease. This oral consent from the patients was acceptable and approved by the Institutional Review Board of Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology. This study was conducted after Institutional Review Board approval from the Hospital.
Data Collection
The data of epidemiological, clinical, laboratory, and radiological characteristics and treatment and outcomes were collected with data collection forms from electronic medical records. Initial investigations included a complete blood count, coagulation profile, and serum biochemical test (including renal and liver function, cardiac biomarkers). The serum levels of C-reactive protein (CRP), procalcitonin (PCT), ferritin, and cytokines of all patients were measured by clinical laboratory. Peripheral blood samples were collected from some patients and examined for lymphocyte subsets by flow cytometry. The immunoglobulin (Ig) M of influenza A and B, respiratory syncytial virus, adenovirus, parainfluenza virus, Legionella pneumophila, mycoplasma pneumoniae, and chlamydia pneumoniae were detected by chemiluminescence immunoassay. And immune-related factors (IgA, IgG, IgM, C3, and C4) were also measured. The Glasgow Coma Scale, Sequential Organ Failure Assessment (SOFA), and Acute Physiology and Chronic Health Evaluation II (APACHE II) scores were determined on the day of ICU admission. The date of disease onset was defined as the day when the first symptom was noticed. The intervals of the time of onset of symptoms to confirmation of disease, to hospital admission, to ICU admission, to death were calculated.
The complications were also recorded. ARDS and shock were defined according to the above-mentioned guidances of WHO and Chinese government for COVID-19. AKI was defined based on the serum level of creatinine (Cr).7 Cardiac injury was confirmed if hypersensitive cardiac troponin I was above the 99th percentile upper reference limit or new abnormalities were shown in electrocardiography.3 The other complications included liver dysfunction, hyperglycemia, gastrointestinal hemorrhage, pneumothorax, urinary tract infection, bacteremia, and hospital-acquired pneumonia. The treatments were analyzed, such as high-flow nasal cannula, mechanical ventilation (MV), extracorporeal membrane oxygenation (ECMO), renal replacement therapy, vasoconstrictive agents, common antiviral agents (oseltamivir and arbidol and/or ribavirin), lopinavir/ritonavir, glucocorticoids, antibiotics, traditional Chinese medicines (Lianhua Qingwen granules and Xuebijing), immunoglobulin, interferon (IFN) α-2b, and the convalescent plasma.
Statistical Analysis
Statistical analyses were performed with SAS statistical software (SAS Institute Inc., release 9.3, USA) and R version 3.6.2. The continuous variables were expressed as the mean±standard deviation (SD) or median (IQR), while categorical variables were expressed as frequency (proportions). Student’s t-test and Wilcoxon rank-sum test were used for the comparison of quantitative data between survivors and non-survivors as appropriate; the Chi-square test and Fisher exact method were used for the comparison of qualitative data between survivors and non-survivors as appropriate. Crude hazard ratios (HR) and 95% confidence intervals (CIs) were calculated by using Cox survival analysis. The survival curves were compared by Log rank test. We conducted stratified analysis according to the interval from the onset of symptoms to hospital admission or age. Two-tailed P<0.05 was considered statistically significant.
Results
Presenting Characteristics
There were 59 patients admitted to the ICU from February 2, 2020 to March 1, 2020. All patients were residents of Wuhan, China. The mean age was 67.4±11.3 years, and 26 (44.1%) were older than 70 years. Most patients were male, with a male/female (M/F) ratio of 1.81. Forty-seven (79.7%) patients had underlying diseases, including hypertension (31, 52.5%), chronic cardiac disease (13, 22.0%), and diabetes (15, 25.4%). Compared with the survivors, non-survivors had more smoking history (22.0% vs 0.0%, P<0.05). Baseline characteristics in the two groups are shown in Table 1.
 |
Table 1 Demographics and Baseline Characteristics of Critically Ill COVID-19 Pneumonia Patients |
The most common symptoms were fever (79.7%), cough (78.0%), dyspnea (55.9%), and chest tightness (52.5%). Less common symptoms were chilling, disorder of consciousness, hemoptysis, cyanosis, headache, myalgia, fatigue, anorexia, nausea/vomiting, and diarrhea. The median interval from onset of symptoms to confirmation of pneumonia was 4 days (IQR=1–7). The median duration from onset of symptoms to hospital admission was 12.0 days (IQR=8–17). The median duration from onset of symptoms to ICU admission was 16.0 days (IQR=13–23; Figure 1). Ground glass and consolidation areas were found in all patients’ bilateral lungs. The detailed information is listed in Table 2.
 |
Table 2 Symptoms, Comorbidities, and Treatments of Critically Ill COVID-19 Pneumonia Patients |
Vital Signs and Laboratory Parameters in Critically Ill Patients with COVID-19
The patients had normal mean arterial pressure and systolic blood pressure. On the day of ICU admission, APACHE II and SOFA scores were 15.3±6.4 and 7.2±8.5, respectively. The median of partial pressure of oxygen to fraction of inspired oxygen ratio was 163.1±142.7 mm Hg.
There are numerous laboratory abnormalities in critically ill patients. The patients had an elevated count of white blood cells (WBC) and neutrophil, lymphopenia, anemia, hypoalbuminema, high serum concentration of D-dimer, and lactate dehydrogenase (Figure 2). The patients also had prolonged prothrombin time (PT), higher levels of fibrinogen degradation product (FDP), alanine aminotransferase (ALT), aspartate aminotransferase (AST), creatinine, urea nitrogen, and blood glucose.
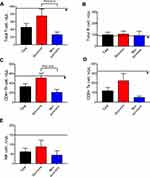
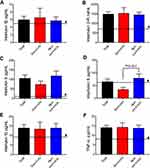
The serum levels of CRP, PCT, erythrocyto sedimentation rate (ESR), and ferritin were increased in critically ill patients with COVID-19 pneumonia. We measured the concentrations of IgA, IgG, IgM, C3, and C4, which were at normal levels in most of the patients (data not shown). The data of flow cytometric analysis of lymphocyte subsets showed that the concentrations of CD3+CD19-T cells (consisting of CD4+T and CD8+T cells), and natural killer (NK) cells were all reduced in the critically ill, and the differences of total T cells and CD4+T cells between survivors and non-survivors were statistically significant (P<0.05; Figure 3). Initial plasma interleukin (IL) 1β, IL2-R, IL6, IL8, IL10, and tumor necrosis factor (TNF)-α concentrations were higher in both survivors and non-survivors than in healthy adults (Figure 4). To confirm whether the patients had co-infection, we detected the common etiological factors of pneumonia. The data manifested that influenza A (9, 15.3%) was the predominant pathogen recovered, followed by adenovirus (1, 1.7%) and Mycoplasma pneumoniae (1, 1.7%). The data is shown in Table 3.
 |
Table 3 Vital Signs and Laboratory Parameters of the Patients |
Treatments, Organ Dysfunction, and Outcomes
In the series of critically ill patients, 45 (76.3%) were treated with high flow nasal cannula, 43 (72.9%) with non-invasive MV, 36 (61.0%) with invasive MV, two (3.4%) with extracorporeal membrane oxygenation (ECMO), 14 (23.7%) with renal replacement therapy, and 38 (65.5%) with vasoconstrictive agents. Fifty-eight (98.3%) patients were administered with empirical antibiotic treatment. Glucocorticoids were given to 48 (81.4%) patients, Xuebijing to eight (13.6%) patients, immunoglobulin to 22 (37.3%) patients, and interferon (IFN) α-2b to 15 (25.4%) patients. Fifty-two (88.1%) received oseltamivir or arbidol, and/or ribavirin, six (10.2%) patients received lopinavir/ritonavir. One patient treated with ECMO was intravenously infused with the convalescent plasma (Table 2).
The comorbidities of the 59 patients are shown in Table 2. As many as 91.5% of patients developed one or more comorbidities. The common complications were ARDS (43, 72.9%), AKI (25, 42.4%), cardiac injury (38, 64.4%), arrhythmia (16, 27.1%), and so on. Up to March 8, 41 (69.5%) had died, the median interval from onset of symptoms to death was 25.0 (IQR=19–32) days (Table 2).
Prognostic-Related Factors of Critically Ill COVID-19 Pneumonia Patients
Tables 1–3 summarize the characteristics of patients. We found that more non-survival patients were of older age, had a smoking history, presented with fatigue and diarrhea, had several complications (including ADRS, AKI, cardiac injury, arrhythmia), received the approaches of MV, and had vasoconstrictive agents (P<0.05). The non-survivors had neutrophilia, more severe lymphocytopenia and thrombocytopenia, elevated D-dimer and FDP levels as well as IL8, lower T cell count, CD4+ T cells, and higher APACHE II and SOFA scores (P<0.05).
In single variant regression analysis, potential factors associated with death included age >70 years (hazard ratio [HR]=2.76; 95% CI=1.45–5.23), fatigue (HR=2.73, 95% CI=1.45–5.14), lymphopenia (HR=0.16, 95% CI=0.03–0.81), and thrombocytopenia (HR=1.00, 95% CI=0.99–1.00), higher interleukin 8 (HR=1.01, 95% CI=1.00–1.01) and organ injuries (acute kidney injury [HR=3.51, 95% CI=1.84–6.71], cardiac injury [HR=3.50, 95% CI=1.64–7.46] and arrhythmia [HR=3.45, 95% CI=1.79–6.66]). However, being aged over 70 (HR=2.76, 95% CI=1.45–5.23), having arrhythmia (HR=4.760, 95% CI=1.59–14.25), and a SOFA score above 4 (HR=5.16, 95% CI=1.29–20.55) were identified as risk factors for mortality of patients in multi-variant regression analysis (Table 4). The survival curves based on age, the time interval from the onset of symptoms to hospital admission, the complications, APACHE II, and SOFA score are shown in Figure 5.
 |
Table 4 Risk Factors for Death of Critically Ill Patients with COVID-19 Pneumonia |
Discussion
In this study, we reported on 59 critically ill patients with COVID-19 pneumonia. Up to March 8, 2020, 41 (69.5%) patients had died and 18 (30.5%) were alive. Among them, 44 (71.0%) patients were complicated with ARDS, and 52 (83.9%) required mechanical ventilation. Our data showed that the patients aged >70 years, with treatment delay, severe lymphopenia and thrombocytopenia, elevated interleukin 8, with acute kidney injury, cardiac injury, and arrhythmia, were at high risk of death. Among these factors, being aged over 70, arrhythmia, and a SOFA score above 4 were identified as independent risk factors for mortality of patients in multi-variant regression analysis.
Up to now, no specific therapeutic drug has been found. So the treatments of patients with SARS-CoV-2 infection are mainly symptomatic treatments. Lopinavir/ritonavir was shown to be effective against SARS-CoV-2 in patients and in tissue culture.8 Six (10.2%) patients in this study received lopinavir/ritonavir and died, probably because of the severe lung pathology of the patients.9 IFN α-2b has been used successfully as a primary treatment and prophylaxis in severe acute respiratory syndrome (SARS) and Middle East respiratory syndrome (MERS).10 Our data demonstrated that IFN α-2b therapy might be an effective therapy for the critically ill COVID-19 patients. In this study there were 43 (72.9%) patients treated with non-invasive MV and 36 (61.0%) with invasive MV. Once the patients required the treatment with MV and/or vasoconstrictive agents, it reflected serious condition of patients, which predicted poor clinical outcomes of the patients. Convalescent plasma is a potential therapy for COVID-19.11 In this study, the treatment with convalescent plasma alleviated the severity of one patient with ECMO support, leading to the withdrawal of ECMO and intubation (data not shown).
As mentioned in previous studies, most patients (38, 64.4%) were men, with a M/F ratio of 1.80.2,4 Older age has been reported as an important independent predictor of mortality in SARS and MERS.12,13 Twenty-six (44.1%) patients were aged over 70 years, which was an independent risk factor for death (HR=3.323, 95% CI=1.124–9.823). Both SARS-CoV-2 and SARS-CoV spike proteins have a high degree of homology, whose spike proteins have strong binding affinity to human angiotensin-converting enzyme 2 (ACE2).14,15 ACE2 provides a pathway for SARS‐CoV‐2 to invade the body, increasing the chance of viral infection. The studies demonstrated that ACE2 expression is higher in young people than in elderly individuals and higher in females than in males.16,17 Moreover, ACE2 plays a protective role in acute lung injury and a lack of functional ACE2 in the lung might contribute to the pathogenesis of lung injury.18,19 These might illustrate partly that severely ill COVID‐19 patients were mostly elderly males. The manifestation of diarrhea was more frequent in the non-survivors (19, 46.3%) than survivors (3, 16.7%), though the difference was not statistically significant (P>0.05). Diarrhea is a common symptom in coronavirus infections, presenting in 30% of patients with MERS and 10.6% of patients with SARS.20,21 Highest ACE2 mRNA expression levels were detected in the intestinal epithelium.18 So fecal–oral transmission might be an important pathway to spread the virus.
Cardiac complications are common in patients with pneumonia. SARS-CoV-2 could also cause myocardial injury, as assessed by increased troponin I level accompanying increased cardiovascular symptoms in COVID-19 patients.2,4 It is unclear whether cardiac injury is directly caused by viral infection or is secondary to hypoxia and systemic inflammation. The putative SARS-CoV-2 receptor, ACE2, is expressed in cardiomyocytes and mural cells, particularly pericytes, which is significantly elevated in cardiomyocytes of patients with heart disease.22 Cardiac arrhythmias were one of the possible SARS-CoV-2 manifestations, in which sinus tachycardia was with an overall incidence of 72%, and sinus bradycardia was with an overall incidence of 14.9%.23,24 In the setting of the 2012 MERS‐CoV syndrome, the occurrence rates of cardiac arrhythmias and hepatic dysfunction were about 15.7% and 31.4%, respectively.25 In this series of critically ill patients, we described that 38 (64.4%) patients presented with cardiac injury, and 16 (27.1%) patients presented with arrhythmia, both of which were higher in non-survivors (P<0.0.5). And we further proved that arrhythmia was identified as an independent risk factor for mortality of critically ill patients with COVID-19 pneumonia. Moreover, we observed that no all patients with cardiac injury would develop cardiac arrhythmias, and vice versa.
APACHE II and SOFA score have been widely used to predict the outcome of the critically ill patients. These scores can be used to not only predict which patients are likely to develop sepsis but also to predict which patients can survive sepsis or not. And the early recognition of patients at risk of developing sepsis allows an appropriate approach that would be started in ICU admission and this may improve outcomes.26 Among them, SOFA score represents a simple and objective score for predicting sepsis and septic shock, and calculate both the number and the severity of organ dysfunction.27 The score has recently been validated in the prediction of short-term and long-term mortality of ICU patients.28 In the current study, we calculated the APACHE II and SOFA score at ICU admission. We demonstrated that both scores were significantly higher in non-survivors than survivors (P<0.05). The data of multi-variant regression analysis showed that the SOFA score could be used as a risk stratification tool to predict the possible mortality of critically ill patients with SARS-CoV-2 infection.
This study had several limitations. First, this study had limited sample size, because only one hospital was included in this study. Second, it was a retrospective study. Not all patients were measured with all laboratory testing, especially the test of lymphocyte subsets. Their role in predicting death of the critically ill might be underestimated. Third, 37 (62.7%) patients did not receive the treatment in time, which might contribute to the poor clinical outcomes in some patients. A large-scale study of critically ill patients with COVID-19 pneumonia would be of benefit for the clinicians to identify the clinical characteristics and potential risk factors of the disease.
In conclusion, the mortality of critically ill COVID-19 pneumonia patients was high. Critically ill COVID-19 patients aged over 70, arrhythmia, and a SOFA score above 4 have a high risk of mortality, and need prior medical intervention.
Disclosure
The authors declare that there is no conflict of interest.
References
1. World Health Organization. Coronavirus disease 2019 (COVID-19): situation report—89. Available from: https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200418-sitrep-89-covid-19.pdf?sfvrsn=3643dd38_2.
2. Liu X, Zhou H, Zhou Y, et al. Risk factors associated with disease severity and length of hospital stay in COVID-19 patients. J Infect. 2020;81(1):e95–e97. doi:10.1016/j.jinf.2020.04.008
3. Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395(10223):497–506. doi:10.1016/S0140-6736(20)30183-5.
4. Yang X, Yu Y, Xu J, et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet Respir Med. 2020;8(5):475–481. doi:10.1016/S2213-2600(20)30079-5.
5. WHO. Clinical management of severe acute respiratory infection when novel coronavirus (2019-nCoV) infection is suspected interim guidance 28 january 2020. Available from: https://www.who.int/docs/default-source/coronaviruse/clinical-management-of-novel-cov.pdf?sfvrsn=bc7da517_6&download=true7.http://www.nhc.gov.cn/yzygj/s7653p/202003/46c9294a7dfe4cef80dc7f5912eb1989/files/ce3e6945832a438eaae415350a8ce964.pdf.
6. Available from: http://www.nhc.gov.cn/yzygj/s7653p/202003/46c9294a7dfe4cef80dc7f5912eb1989/files/ce3e6945832a438eaae415350a8ce964.pdf.
7. Ronco C, Bellomo R, Kellum JA. Acute kidney injury. Lancet. 2019;394(10212):1949–1964. doi:10.1016/S0140-6736(19)32563-2
8. Martinez MA. Compounds with therapeutic potential against novel respiratory 2019 coronavirus. Antimicrob Agents Chemother. 2020;64(5):e00399–20. doi:10.1128/AAC.00399-20
9. Sheahan TP, Sims AC, Leist SR, et al. Comparative therapeutic efficacy of remdesivir and combination lopinavir, ritonavir, and interferon beta against MERS-CoV. Nat Commun. 2020;11(1):222. doi:10.1038/s41467-019-13940-6
10. Khalid M, Al Rabiah F, Khan B, Al Mobeireek A, Butt TS, Al Mutairy E. Ribavirin and interferon-α2b as primary and preventive treatment for Middle East respiratory syndrome coronavirus: a preliminary report of two cases. Antivir Ther. 2015;20(1):87–91. doi:10.3851/IMP2792
11. Tu H, Tu S, Gao S, Shao A, Sheng J. The epidemiological and clinical features of COVID-19 and lessons from this global infectious public health event. J Infect. 2020;81(1):1–9. doi:10.1016/j.jinf.2020.04.011
12. Choi KW, Chau TN, Tsang O, et al. Outcomes and prognostic factors in 267 patients with severe acute respiratory syndrome in Hong Kong. Ann Intern Med. 2003;139(9):715–723. doi:10.7326/0003-4819-139-9-200311040-00005
13. Ko JH, Park GE, Lee JY, et al. Predictive factors for pneumonia development and progression to respiratory failure in MERS-CoV infected patients. J Infect. 2016;73(5):468–475. doi:10.1016/j.jinf.2016.08.005
14. Li F, Li W, Farzan M, Harrison SC. Structure of SARS coronavirus Spike receptor-binding domain complexed with receptor. Science. 2005;309(5742):1864–1868. doi:10.1126/science.1116480
15. Deng L, Li C, Zeng Q, et al. Arbidol combined with LPV/r versus LPV/r alone against corona virus disease 2019: a retrospective cohort study. J Infect. 2020;81(1):e1–e5. doi:10.1016/j.jinf.2020.03.002
16. Xie X, Chen J, Wang X, Zhang F, Liu Y. Age- and gender-related difference of ACE2 expression in rat lung. Life Sci. 2006;78(19):
17. Soro-Paavonen A, Gordin D, Forsblom C, et al. Circulating ACE2 activity is increased in patients with type 1 diabetes and vascular complications. J Hypertens. 2012;30(2):
18. Jia H. Pulmonary angiotensin-converting enzyme 2 (ACE2) and inflammatory lung disease. Shock. 2016;46(3):239–248. doi:10.1097/SHK.0000000000000633
19. Busse LW, Chow JH, McCurdy MT, Khanna AK. COVID-19 and the RAAS-a potential role for angiotensin II? Crit Care. 2020;24(1):136. doi:10.1186/s13054-020-02862-1
20. Chan JF, Yuan S, Kok KH, et al. A familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: a study of a family cluster. Lancet. 2020;395(10223):514–523. doi:10.1016/S0140-6736(20)30154-9
21. Fan Y, Zhao K, Shi ZL, Zhou P. Bat coronaviruses in China. Viruses. 2019;11(3):210. doi:10.3390/v11030210
22. Nicin L, Abplanalp WT, Mellentin H, et al. Cell type-specific expression of the putative SARS-CoV-2 receptor ACE2 in human hearts. Eur Heart J. 2020;41(19):1804–1806. doi:10.1093/eurheartj/ehaa311
23. Yu CM, Wong RS, Wu EB, et al. Cardiovascular complications of severe acute respiratory syndrome. Postgrad Med J. 2006;82(964):
24. Kochi AN, Tagliari AP, Forleo GB, Fassini GM, Tondo C. Cardiac and arrhythmic complications in patients with COVID-19. J Cardiovasc Electrophysiol. 2020;31(5):1003–1008. doi:10.1111/jce.14479
25. Saad M, Omrani AS, Baig K, et al. Clinical aspects and outcomes of 70 patients with Middle East respiratory syndrome coronavirus infection: a single‐center experience in Saudi Arabia. Int J Infect Dis. 2014;29:
26. Basile-Filho A, Lago AF, Menegueti MG, et al. The use of APACHE II, SOFA, SAPS 3, C-reactive protein/albumin ratio, and lactate to predict mortality of surgical critically ill patients: a retrospective cohort study. Medicine (Baltimore). 2019;98(26):e16204. doi:10.1097/MD.0000000000016204
27. Innocenti F, Tozzi C, Donnini C, et al. SOFA score in septic patients: incremental prognostic value over age, comorbidities, and parameters of sepsis severity. Intern Emerg Med. 2018;13(3):405–412. doi:10.1007/s11739-017-1629-5
28. García-Gigorro R, Sáez-de la Fuente I, Marín Mateos H, Andrés-Esteban EM, Sanchez-Izquierdo JA, Montejo-González JC. Utility of SOFA and Δ-SOFA scores for predicting outcome in critically ill patients from the emergency department. Eur J Emerg Med. 2018;25(6):387–393. doi:10.1097/MEJ.0000000000000472
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.