Back to Journals » Journal of Multidisciplinary Healthcare » Volume 14
COVID-19 Prevention Practices and Associated Factors among Diabetes and HIV/AIDS Clients in South-Wollo Zone, Ethiopia: A Health Facility-Based Cross-Sectional Study
Authors Ademas A , Adane M , Keleb A , Berihun G , Lingerew M, Sisay T, Hassen S , Getachew M , Tesfaw G, Getaneh Feleke D , Addisu E , Berhanu L , Abebe M, Gizeyatu A, Abate H , Derso A
Received 21 June 2021
Accepted for publication 21 July 2021
Published 4 August 2021 Volume 2021:14 Pages 2079—2086
DOI https://doi.org/10.2147/JMDH.S325207
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 3
Editor who approved publication: Dr Scott Fraser
Ayechew Ademas,1 Metadel Adane,1 Awoke Keleb,1 Gete Berihun,1 Mistir Lingerew,1 Tadesse Sisay,1 Seada Hassen,1 Melaku Getachew,1 Getu Tesfaw,2 Dejen Getaneh Feleke,3 Elsabeth Addisu,4 Leykun Berhanu,1 Masresha Abebe,1 Adinew Gizeyatu,1 Habtemariam Abate,5 Atimen Derso1
1Department of Environmental Health, College of Medicine and Health Science, Wollo University, Dessie, Ethiopia; 2Department of Pharmacy, College of Health Science, Debre Tabor University, Debre Tabor, Ethiopia; 3Department of Pediatrics and Child Health Nursing, College of Health Science, Debre Tabor University, Debre Tabor, Ethiopia; 4Department of Public Health, College of Medicine and Health Science, Wollo University, Dessie, Ethiopia; 5Department of Environmental Health Regulatory Directorate, Addis Ababa Food Medicines and Health Care Control Authority, Addis Ababa, Ethiopia
Correspondence: Ayechew Ademas Email [email protected]
Background: Coronavirus-2019 (COVID-19) is affecting many people. Chronic patients are highly vulnerable to contracting an infection. Most people recover within a week, but chronic patients can face severe illness or death. The increasing of cases, complications, and mortality demands compulsory preventive measures. Therefore, this study was designed to identify major preventive practices and associated factors.
Methods: A facility-based cross-sectional study was employed from November to December 2020 among diabetics and HIV/AIDS follow-up clients in Dessie referral hospital. Data were entered into EpiData manager 4.6.0 version and exported to Statistical Package for Social Science (SPSS) version 25.0 for data cleaning and analysis. Logistic regression analysis was done and an adjusted odds ratio (AOR) with its 95% confidence interval (CI) was used for determining the strength of association.
Results: Data were collected from 426 participants with a mean age of 46.54 years. The overall poor prevention practice rate of COVID-19 among diabetic and HIV/AIDS follow-up clients was 68.8% 95% CI (64.6– 72.8%). Factors significantly associated with poor prevention practice were femaleness (AOR; 1.61; 95% CI; 1.03– 2.51), illiterate [AOR; 2.59; 95% CI; 1.33– 5.09]), family size greater than four (AOR; 2.06; 95% CI; 1.32– 3.23), absence of health professional in the household (AOR; 1.79; 95% CI; 1.13– 2.84), not having health insurance (AOR; 1.85; 95% CI; 1.18– 2.89) and urban residence (AOR; 0.38; 95% CI; 0.18– 0.79).
Conclusion: The overall proportion of prevention practice towards COVID-19 among diabetic and HIV/AIDS follow-up clients was poor. Illiteracy, having a family size greater than four, not having health professionals in the household, not having health insurance and urban residency were associated with poor prevention practices. Therefore, continuous health educations about good preventive behavioral practice should be enhanced by the health professionals.
Keywords: behavioral practice, COVID-19, Ethiopia, prevention
Introduction
COVID-19 is an acute respiratory disease affecting people with signs and symptoms of headache fever or feeling feverish/chills, cough, fatigue (tiredness), sore throat, shortness of breath, or difficulty breathing muscle pain, runny or stuffy nose, and body aches.1 Most people recover and improve from symptoms within one week without requiring medication. Nevertheless, it might cause severe illness or death to high-risk groups of people. Illnesses in this group of people may range from mild-severe and also may result in death. Admission to hospitals and mortality mainly occur in these high-risk groups of the community.
COVID-19 spread mainly via small respiratory droplets that are released from an infected person while sneezing, coughing, and talking. A person who is in proximate distant places may inhale the droplets or become infected through physical contacts like hugging or handshaking, followed by touching their nose or mouth. Individuals do not necessarily show symptoms to be considered contagious since COVID-19 can be transmitted by presymptomatic to symptomatic patients. COVID-19 cases are increasing and there is a likelihood of community transmission. The WHO recently modified the COVID-19 suspect case definition to include severe acute respiratory infection and advises testing of all severe acute respiratory illness (SARI) cases to tackle by early identification of cases and to quarantine them.2
Globally, an estimated annual epidemics result in about three to five million cases of severe illness and 290,000–650,000 respiratory deaths.3 In addition, according to the worldometer as of April 4, 2021 coronavirus cases reach 131,472,605 and results in 2,861,373 deaths globally. When the case of COVID-19 is severe, causes respiratory failure, pneumonia, acute respiratory distress syndrome, heart stroke or attack, severe inflammation, multiple organ failure, sepsis, and also death.4
According to a recent study, each year seasonal influenza results in the mortality of 650,000 people worldwide, and of these deaths, 72,000 occur in the WHO European countries.5 In Africa, about 16% of study subjects were positive out of 34,553 specimens tested for the influenza virus. Influenza type A virus was predominantly circulating, accounting for 62% of the positive sample specimens, of which influenza A(H3N2) subtype accounts 56%.6 A study conducted in Ethiopia using survey data which includes Severe Acute Respiratory Illness (SARI) and Influenza-like Illness (ILI) from 2012 to 2017 indicates that from a total of 5715 cases 20.4% of the survey has influenza viruses confirmed by the laboratory. Among SARI and ILI suspected case was 3% and 25% respectively.7 According to the Centers for Disease Control and Prevention (CDC), the disease of COVID-19 is more contagious in susceptible populations like people with chronic diseases and elder groups. The disease has also shown more super-spreading activity than the flu disease.4
In diabetic patients, higher blood sugar can create an environment where viruses are likely to thrive.8 This condition suppressed the immune system like that of HIV/AIDS and makes them less able to fight the coronavirus.9 Therefore, the increased impact of morbidity and mortality worldwide in chronic patients needs a compulsory preventive measure of COVID-19 since it is the cost-effective and main strategy to prevent the pandemic. Thus, the objective of this study was to determine the proportion of prevention practices and associated factors among chronic patients of diabetics and HIV/AIDS.
Methods and Materials
Study Design, Period, and Setting
A health facility-based cross-sectional study design was conducted in Dessie referral hospital from November to December 2020. Dessie city is the Zonal Administration center of South Wollo Zone, having five hospitals (one government) and four governmental health centers, and twelve private clinics. It has a large catchment population other than the South Wollo zone including clients from Oromia special zone, North Wollo zone, and Afar region.
Source and Study Population
The source population for the study includes all diabetes mellitus and HIV/AIDS chronic patients, attending regular follow up at Dessie referral hospital. All chronic patients attending the follow-up ward during the time of data collection period were the study population.
Inclusion and Exclusion Criteria
All diabetics and HIV/AIDS clients who have a regular follow-up within the study period were included and those under the age of eighteen years old were excluded from the study.
Sample Size Estimation and Sampling Procedure
A total of 426 study participants were determined by using the single population proportion formula.10 By considering the following assumptions of, n = the required minimum sample size, Zα/2 value at 95% CI, which is 1.96, p is the percentage of prevention practice for COVID-19 (50%) and d is the margin of error 5%. Then, adding a 10% non-response rate we get an estimated final sample size of 426. Finally, a consecutive sampling method was used to reach study subjects.
Outcome Measurement and Explanatory Variable
The dependent variable was the rate of prevention practice of COVID-19 which was computed from behavioral factors. The behavioral factors were comprised of 20 questions that were adapted from different literature.11,12 Those who reported above the mean score value were considered to have good prevention practice towards COVID-19. Explanatory variables consisted of socio-demographic and behavioral factors of the study participants.
Data Collection and Quality Assurance
A pretested, structured questionnaire adapted from WHO COVID-19 assessment tools and different pieces of literature was used to collect the data.11,12 The questionnaire includes socio-demographic and behavioral factors. To ensure the consistency of the tool, it was crafted in English originally, and converted into Amharic and back into English by a language expert who is fluent both in English and Amharic. Data was collected by three female BSc nurses through a face-to-face interview. The data collection process was supervised by an environmental health professional. To ensure the acquisition of reliable data both data collectors and supervisors were trained by the principal investigator for one day. Pre-tested was done on 21 similar subjects in nearby Haik health Center and made adjustments to the tool as necessary. Daily checks were done by the supervisors and principal investigator for completeness immediately before the data entry.
Data Management and Statistical Analysis
After checking data, it was coded and entered into the EpiData manager version 4.6.0 software and then exported to SPSS version 25.0 for data cleaning, processing, and analysis. Descriptive statistics like proportions and frequencies were done. Both bivariate and multivariable logistic regressions were done to determine associated factors.
First bivariate analysis was performed and variables having a p-value less than 0.25 were retained as a candidate variable for adjusted multivariable analysis. A backward stepwise method was used to select associated factors. In the adjusted model, AOR values with a 95% CI, to determine the strength of association and a p-value < 0.05 were taken to identify statistically significant factors. The model fitness was checked using a p-value of 0.22 from the Hosmer and Lemeshow test, which suggests a good model (if the p-value is >0.05). Multi-collinearity was checked using the standard error of the coefficient of the model with a value >2 and none of the variables was collinear.
Results
Socio-Demographic Characteristics of Study Subjects
The response rate was 100% (426) among diabetics and HIV/AIDS clients at Dessie referral hospital. The mean ages of the study participant were 46.54 years old and the majority 284 (66.7%) fall in the range of 36–55 years of age. Slightly more than half (52.1%) of the study respondents were female and the majority of 356 (83.6%) were from Amhara ethnicity. Regarding the educational level, 74 (17.4%) were illiterate and 144 (33.8%) had a primary level of education. Nearly half of the respondents 216 (50.7%) were residents of Dessie city and about 232 (54.5%) had a family member of four and above. In addition to this about 183 (43%) had health professionals in their household and 193 (45.3%) had health insurance (Table 1).
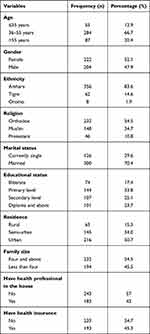 |
Table 1 Socio-Demographic Characteristics of Diabetic and HIV/AIDS Follow Up Clients in Dessie Referral Hospital from November to December 2020 |
Preventive Practice Measurement Indicators of COVID-19
About one-third, 142 (33.3%) of the study participants avoid respiratory illness sick people, about 175 (41.1%) of the study participants use hand sanitizer and/or disinfectants to avoid spreading and catching COVID-19. Besides this, about 188 (44.1) of participants seek treatment as soon as they are becoming ill and nearly a quarter of 110 (25.8%) of them gets regular exercise. Similarly, 260 (61.0%) and 179 (42.0%) of participants listen to advice from health professionals and media and use alternative non-touching greetings to prevent catching and spreading of COVID-19 respectively (Table 2).
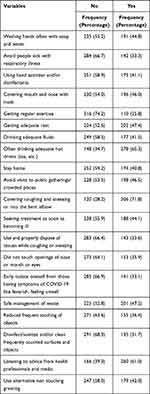 |
Table 2 Behavioral Preventive Practice Towards COVID-19 Among Diabetic and HIV/AIDS Follow Up Clients in Dessie Referral Hospital from November to December 2020 |
The Proportion of Prevention Practice Towards COVID-19
The overall mean prevention practice of COVID-19 was 8.69 with a standard deviation of 2.22. We also found that the overall proportion of poor prevention practice among diabetic and HIV/AIDS follow-up clients was 68.8% with 95% CI (64.6–72.8%) and the other 31.2% with 95% CI (27.2–35.4%) has good prevention practice towards COVID-19. Of those having good practices, females account for about 61.65% (Table 3).
 |
Table 3 Bivariate Analysis of Socio-Demographic Characteristics of Diabetic and HIV/AIDS Follow Up Clients in Dessie Referral Hospital from November to December 2020 |
Associated Factors for COVID-19 Prevention Practice
In the adjusted logistic regression analysis, the result revealed that; femaleness (AOR; 1.61; 95% CI; 1.03–2.51), illiterate [AOR; 2.59; 95% CI; 1.33–5.09], family size greater than four (AOR; 2.06; 95% CI; 1.32–3.23), not having health professional in the house (AOR: 1.79; 95% CI: 1.13–2.84), not having health insurance (AOR; 1.85; 95% CI; 1.18–2.89) and being in the rural area (AOR; 0.38; 95% CI; 0.183–0.79) were factors affecting good prevention practice towards catching and spreading of COVID-19 infection (Table 4).
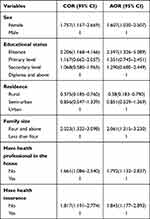 |
Table 4 Factors Associated with Poor Preventive Practice Among Diabetic and HIV/AIDS Follow Up Clients from Multivariable Logistic Regression |
Discussion
Our study result showed that handwashing practice (44.8%) is much lower than the finding in US adults (83.2%) to remove spreading or catching influenza during the 2015 to 2016 influenza seasons. Similarly, about 71.8% of our study participants cover coughing and sneezing or into the bent elbow which was lower than a finding in the US (80.0%).12 Also in this study 40.8%, stay home, 33.3% avoid respiratory illness sick people, and 44.1% seeking treatment as soon as becoming ill whereas in the US, 78.2% stay home if sick, 64.4% avoid respiratory illness sick people and 50.2% get treatment as soon as possible.12 Similar to our finding, in Hong Kong 46.65% wash their hands with water and soap frequently. However, those who wore face masks (46%) were higher than a finding in Hong Kong (21.5%).13
The overall good prevention practice (31.2%) of COVID-19 was in line with a study finding in Addis Ababa, Ethiopia.14 However, our study was different from a finding at Addis Zemen Hospital; Northwest Ethiopia which showed that 47.3% had poor preventive practice towards COVID-19.15 The difference may become due to study subjects and the tools used for measuring preventive practices. In addition to this, a study conducted among diabetes mellitus and hypertensive clients attending public health facilities in Ambo shows that 10.4% of study subjects had good COVID-19 prevention practice measures which are very low as compared to our study finding.16 Besides this, a study in Debre Tabor General Hospital, Ethiopia, showed that about 49.0% have good prevention practice towards COVID-19 which is higher than the finding of this study.17 However, 60.8% of study subjects in Cameron showed that good preventive practice for COVID-19.18
In this study, the odds of females to prevent COVID-19 was 1.607 times more likely as compared to males. But our result was not similar to the finding from a study done in Cameron.18 The difference may be due to the difference in the underlying medical conditions and the socio-demographic attributes of study subjects. Our result indicates that illiterate participants were one determinant factor associated with the poor preventive practice. Again this was supported by the finding from a study done among Chronic Patients at Addis Zemen Hospital, Ethiopia.16 Similarly, a study conducted among diabetic and hypertensive clients visiting community health facilities in Ambo, Ethiopia indicates that those who do not have formal education were associated with poor prevention practice.16
However, our study revealed that the rural residence was preventive to poor practice than the urban community and this was different from the finding in Addis Zemen Hospital.15 The reason might be due to the negligence of the urban population. Similarly, this finding was contrary to the study finding at Debre Tabor General Hospital, Ethiopia which showed that urban residency was associated with good preventive practice.17
We found that the odds of those who have a health professional in their household to have good preventive practice towards catching and spreading of COVID-19 among diabetic and HIV/AIDS clients were 1.79 times higher as compared to those who did not have a health professional. Similarly, the odds of households having health insurance toward having good prevention practice were 1.85 times higher as compared to those who do not have health insurance. But the finding among students in Ghana revealed that there is no difference between those who have and do not have health insurance.19 Our result also revealed that those who have a family size of ≥4 were 2.06 more likely to have good preventive practice towards COVID-19 infection than those who had a family size of less than four which was supported by finding in Southwest Ethiopia.20 However as a limitation, in this study we estimated only socio-demographic factors and we might miss controlling other variables which may affect the result.
Conclusion
In this study, there is an overall poor preventive practice among diabetic and HIV/AIDS follow-up clients towards COVID-19. We found that illiteracy, having a family size greater than four, not having health professionals in the household, not having health insurance and urban residency were associated with poor practice towards catching and spreading COVID-19 infection. Therefore, continuous health educations about good preventive behavior practices are critical to reducing the spread of these diseases among the community. Proper and regular handwashing with soap and water, avoid people sick with respiratory illness and public gathering, using hand sanitizer and/or disinfectants, covering mouth and nose with a mask, getting regular exercise and adequate rest, drinking adequate fluids, stay home and isolate themselves, seek treatment as soon as becoming ill and follow advice from health professionals and media are recommended to chronic patients of HIV/AIDS and diabetics for further improvement of prevention practice towards COVID-19.
Abbreviations
AOR, adjusted odds ratio; CI, confidence interval; COR, crude odds ratio; COVID-19, coronavirus- 2019; WHO, World Health Organization.
Data Sharing Statement
The data is available from the corresponding author upon request.
Ethics
All the study procedures and principles were conducted following the declaration of Helsinki. Ethical approval was obtained from the Institutional Ethical Review Committee of the College of Medicine and Health Sciences, Wollo University. The questions were proved not to affect the morale and personality of study participants. Informed verbal consent was taken for those who cannot read and write and written consent for those who can read and write after explaining why they were taking part in the research. Confidentiality was also ensured by using code numbers rather than names, data collectors interviewed people separately to retain the secrecy. Health education and advice were given about the benefits of good prevention practices to study subjects during data collection.
Acknowledgment
We acknowledge Wollo University and Dessie Referral Hospital for their permission and willingness to undertake the study. We also recognize and give much gratitude to study participants, data collectors as well as supervisors for their relentless participation and support.
Funding
There is no specific funding for this study.
Disclosure
The authors declare that they have no conflicts of interest for this work.
References
1. WHO. Coronavirus disease (COVID-19)_ similarities and differences with influenza [Internet]; 2021 [
2. WHO. WHO COVID-19 Case definition; 2020. Available from: https://www.who.int/publications/i/item/WHO-2019-nCoV-Surveillance_Case_Definition-2020.2.
3. WHO. Influenza (Seasonal) [Internet]; 2020. Available from: http://www.who.int/mediacentre/factsheets/fs211/en/.
4. AMS. COVID-19 and the Flu [Internet]. American Society For Microbiology; 2020. Available from: https://asm.org/Articles/2020/July/COVID-19-and-the-Flu.
5. WHO. WHO_Europe _ burden of influenza [Internet]; 2021. [
6. WHO. Influenza Surveillance In the WHO African Region [Internet]; 2017. Available from: http://www.afro.who.int/en/integrated-disease-surveillance/publications/4792-protocol-for-national-influenza-sentinel-surveillance.html.
7. Tadesse M, Mengesha M, Tayachew A, et al. Burden and seasonality of medically attended influenza-like illness (ILI) in Ethiopia, 2012 to 2017. BMC Infect Dis. 2020;20(1):1–13. doi:10.1186/s12879-020-4827-0
8. WHO. COVID-19: vulnerable and high-risk groups [Internet]; 2020. [
9. CDC. Treating Influenza (Flu). US Department of Health and Human Services Centers for Disease Control and Prevention [Internet]; 2016. Available from: https://www.cdc.gov/flu/pdf/freeresources/updated/treating-influenza.pdf.
10. Kelsey JL, Whittemore AS, Evans AS. Methods in Observational Epidemiology.
11. Srivastav A, Santibanez TA, Lu P, et al. Preventive behaviors adults report using to avoid catching or spreading influenza, United States, 2015–2016 influenza season. PLoS One. 2018;2:1–16.
12. WHO. Coronavirus disease (COVID-19) advice for the public [Internet]; 2020 [
13. Lau JTF, Griffiths S, Choi KC, Lin C. Prevalence of preventive behaviors and associated factors during the early phase of the H1N1 influenza epidemic. Am J Infect Control. 2010;38(5):374–380. doi:10.1016/j.ajic.2010.03.002
14. Tadesse T, Alemu T, Amogne G, Endazenaw G, Mamo E. Predictors of coronavirus disease 2019 (COVID-19) prevention practices using health belief model among employees in Addis Ababa, Ethiopia, 2020. Infect Drug Resist. 2020;13:3751–3761. doi:10.2147/IDR.S275933
15. Akalu Y, Ayelign B, Molla MD. Knowledge, attitude, and practice towards COVID-19 among chronic disease patients at Addis Zemen hospital, Northwest Ethiopia. Infect Drug Resist. 2020;13:1949–1960. doi:10.2147/IDR.S258736
16. Taye GM, Bose L, Beressa TB, et al. COVID-19 knowledge, attitudes, and prevention practices among people with hypertension and diabetes mellitus attending public health facilities in Ambo, Ethiopia. Infect Drug Resist. 2020;13:4203–4214. doi:10.2147/IDR.S283999
17. Tigabu Desie E, Tekalign Amera B, Sheganew Fetene T, Abraham Tsedalu A, Nigusie Selomon T, Chalie Marew T. Assessment of knowledge, practice and associated factors towards prevention of novel coronavirus among clients attending at Debre Tabor General Hospital, Debre Tabor Town, North West Ethiopia, 2020: institutional based cross-sectional study. J Community Med Heal Solut. 2020;1(1):26–34. doi:10.29328/journal.jcmhs.1001004
18. Id AN, Nkengazong L, Ambe LA, et al. Knowledge, attitudes, practices towards COVID 19 preventive measures and symptoms: a cross-sectional study during the exponential rise of the outbreak in Cameroon. PLoS Negl Trop Dis. 2020;24:1–15.
19. Apanga PA, Lettor IBK, Akunvane R. Practice of COVID-19 preventive measures and its associated factors among students in Ghana. Am J Trop Med Hyg. 2021;104(2):526–531.
20. Wondimu W, Ayenew AEM, Kidnau AW, et al. Factors associated with coronavirus disease 2019 prevention practices in three zones of southwest Ethiopia: community-based cross-sectional study. Int J Gen Med. 2020;13:1603–1611. doi:10.2147/IJGM.S288067
 © 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2021 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
