Back to Journals » International Journal of General Medicine » Volume 13
Could sST2 Predict Contrast-Induced Nephropathy in ST-Segment Elevation Myocardial Infarction?
Authors Avcı A , Somuncu MU, Can M, Akgul F
Received 21 October 2020
Accepted for publication 12 November 2020
Published 27 November 2020 Volume 2020:13 Pages 1297—1304
DOI https://doi.org/10.2147/IJGM.S287834
Checked for plagiarism Yes
Review by Single anonymous peer review
Peer reviewer comments 2
Editor who approved publication: Dr Scott Fraser
Ahmet Avcı,1 Mustafa Umut Somuncu,1 Murat Can,2 Ferit Akgul1
1Department of Cardiology, Faculty of Medicine, Zonguldak Bulent Ecevit University, Zonguldak, Turkey; 2Department of Biochemistry, Faculty of Medicine, Zonguldak Bulent Ecevit University, Zonguldak, Turkey
Correspondence: Ahmet Avcı
Department of Cardiology, Faculty of Medicine, Zonguldak Bulent Ecevit University, Bülent Ecevit Universitesi Tip Fakultesi Dekanligi Ibn-i Sina Kampusu, Esenköy/Kozlu, Zonguldak 67600, Turkey
Tel +90 505 548 2710
Email [email protected]
Background and Aim: One of the most worrying complications of primary percutaneous coronary interventions is contrast-induced nephropathy (CIN) that is associated with increased mortality and morbidity in myocardial infarction. In this study, we questioned whether soluble suppression of tumorigenesis-2 (sST2), which has thought to play a role in inflammatory processes, cardiac remodeling, and fibrosis could give an idea about the development of CIN in ST-elevation myocardial infarction (STEMI) patients.
Patients and Methods: This study is a cross-sectional observational study and includes 357 consecutive STEMI patients. Demographic features, medical history, laboratory parameters, and procedural characteristics were compared according to CIN’s development. The multivariate logistic regression analysis was selected to detect independent risk factors of CIN.
Results: In the study, 81 patients (22.7%) who developed CIN were identified. The concentration of sST2 in CIN (+) group was higher than that of CIN (-) group (40.6± 21.0 ng/mL vs 31.5± 13.0 ng/L, p< 0.001). Independent predictors of CIN development were diabetes mellitus (OR, 2.059; 95% CI, 1.093– 3.879; p=0.025), eGFR (OR, 0.983; 95% CI, 0.972– 0.995; p=0.006), lower systolic blood pressure (OR, 0.976; 95% CI, 0.960– 0.993; p=0.006), total procedure time (OR, 1.030; 95% CI, 1.011– 1.049; p=0.002), and sST2 (OR, 1.101; 95% CI; 1.046– 1.160; p< 0.001). Besides, the risk of developing CIN in the high sST2 group is 3.06 times higher than the low group sST2 group regardless of other risk factors.
Conclusion: sST2 levels on admission in STEMI patients are useful in predicting CIN development.
Keywords: contrast-induced nephropathy, soluble ST2, ST-elevation myocardial infarction, primary percutaneous coronary intervention
Introduction
Contrast-induced nephropathy (CIN), one of the major causes of iatrogenic acute renal failure, is a leading concern for physicians. If CIN occurs, the length of stay in the hospital would be longer, health care costs and mortality significantly would increase.1,2 In recent years, the primary percutaneous coronary intervention (PCI) had widely used in the treatment of ST-elevation myocardial infarction (STEMI) patients. Patients treated with primary PCI have a higher risk of developing CIN than patients with elective PCI.3 The possible mechanisms that cause CIN are vasoconstriction, medullary ischemia, oxidative stress, and direct toxic effects of contrast agents.4 Also, many studies have suggested that various factors associated with inflammation have a role in the development of CIN.1,5 The effective way for preventing CIN is still unknown. Therefore, there is a need for a more reliable and practical identification tool that can be used in CIN prediction.
sST2, secreted from cardiomyocytes, endotheliocytes, and fibroblasts in response to inflammatory signals, is a member of the interleukin-1 receptor family with transmembrane and soluble isoforms. sST2 secretion increases in response to myocardial damage in the first weeks after the onset of STEMI and increased sST2 is associated with increased myocardial fibrosis and adverse cardiovascular outcomes.6–8
The relationship between high sST2 levels and major cardiovascular events in STEMI patients is well known9; however, to our knowledge, the predictive value of sST2 on the CIN development has not been identified in STEMI patients to date. In this study, we investigated the relationship between sST2 and CIN development in STEMI patients.
Patients and Methods
Study Population
We included 357 consecutive STEMI patients (308 male, 49 female) who were admitted to our hospital (Zonguldak Bulent Ecevit University Research and Training Hospital, Zonguldak, Turkey) between September 2017 and May 2018 and treated with primary PCI in a cross-sectional observational study. We excluded cancer patients, those with known infections, or patients with other systemic inflammatory conditions. ST-segment elevation in adjacent ≥2 leads (≥2 mm in precordial leads, ≥1mm in limb leads) or left-hand bundle branch block, ischemic type chest pain lasting more than 30 minutes, and serum creatine kinase-myocardial band and troponin levels of two or more elevation criteria had used as STEMI diagnostic criteria. Renal dysfunction (absolute serum creatinine increase of 0.3mg/dl or at least 1.5 times increase of basal creatinine) within 7 days after intravenous contrast administration defined as CIN, and 81 patients (22.7%) had CIN. Moreover, 22 of our patient population (15.1%) found to have CIN according to the contrast nephropathy criteria which was recommended by the Journal of the American College of Cardiology.10 Since there are more studies about the criteria we mentioned first in the literature, we created our study design according to these criteria. All included patients signed informed consent forms. The study had approved by the Ethics Committee and the institutional review board of Zonguldak Bulent Ecevit University Faculty of Medicine. This study complies with the declaration of Helsinki.
Laboratory and Clinical Assessment
Patient demographic features, medical history, medications, laboratory measurements recorded. Patients with fasting glucose level above 126 mg/dl or using antidiabetic medication defined as Diabetes mellitus (DM). Patients with systolic blood pressure above 140 mmHg or diastolic blood pressure above 90 mmHg or who had been using antihypertensive drugs for more than 2 weeks defined as hypertension. Hemoglobin below 12 mg/dl for women and hemoglobin below 13 mg/dl for men accepted as anemia. Below glomerular filtration rate (GFR) levels, 60 mL/min/1.73 m2 accepted as chronic renal failure. Blood samples had obtained when the patients were admitted to the emergency room before PCI. Five milliliters of peripheral venous blood were taken from all patients placed in EDTA-coated vacuum tubes. The blood samples were then separated by centrifugation and stored at −70 °C. We followed the manufacturer’s protocol and reached sST2 levels by measuring sandwich enzyme immunoassay using ELISA kits (Shanghai YL Biotech, Shanghai, China). Daily blood samples were taken from the patients to determine the changes in serum creatinine and to diagnose CIN.
Coronary Intervention Procedure
Coronary angiography (CA) was conducted through the femoral or radial artery. The primary PCI was performed using standard guiding catheters, guidewires, balloon catheters, and stents according to current guidelines. All PCI interventions were performed by experienced invasive cardiologists. The type of primary PCI and the amount of contrast were decided by the medical team. All patients received non-ionic, monomeric, low osmolar iodinated iohexol (Omnipaque 300 mg I/mL, Opakim A.Ş., Istanbul, Turkey) as an intravascular contrast material. Participants were treated according to the guidelines of the American Heart Association/American College of Cardiology Foundation. Contrast volume had defined as the contrast volume used during the CA and PCI. The time between the local anesthesia for vascular access and removal of all catheters had defined as the total procedure time.
Statistical Analysis
SPSS software version 21.0 for Windows (SPSS Inc. Chicago, Illinois, USA) was used for statistical analysis of the study. The normality of the distribution was evaluated using the Kolmogorov–Smirnov test. Descriptive analysis was reported as the mean and standard deviation for normally distributed variables. Also, it was presented as the median and interquartile range for the non-normal distribution. The categorical variables are expressed as numbers and percentages. The Student’s t-test was used for continuous variables with the normal distribution. Parameters without normal distribution were compared with the Mann–Whitney U-test. Categorical data were compared using the Chi-square test. We used logistic regression analysis to determine the prognostic factors on the development of CIN. Receiver operating characteristic (ROC) curve analysis was performed to determine the cut-off value of sST2 to predict CIN. A p-value of <0.05, a 95% confidence interval, and a 5% margin of error was considered statistically significant.
Results
A total of 357 consecutive STEMI patients were treated with PCI were enrolled. The mean age was 59.45±11.88 years, and 298 patients (86.2%) were men. CIN occurred in 81 of 357 patients (22.7%). The patients were divided into two groups as CIN (+) group (n = 81) and CIN (-) group (n = 276). The basic clinical characteristics of the CIN (+) group and CIN (-) group are shown in Table 1. There was no statistical difference in risk factors other than DM, systolic blood pressure (SBP) and diastolic blood pressure (DBP). CIN (+) patients had lower SBP and DBP on admission (112.0 ±24.5 vs 132.0±22.8; p<0.001 and 64.6±12.8 vs 74.9±12.7; p<0.001). The percentage of patients with DM in the CIN (+) group was higher than in the CIN (-) group (37.0% vs 24.3%; p=0.023). Antiplatelet use before the procedure was 9.2%, while it was 99.7% after the procedure, and there was no difference between the group in terms of antiplatelet and other cardiac treatment.
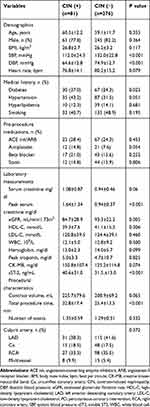 |
Table 1 Basic Clinical Characteristics of CIN (+) Group and CIN (−) Group |
Peak creatinine was significantly higher in patients with CIN (+) compared to the patients with CIN (-) (1.64±1.34 mg/L vs 0.94±0.37 mg/L; p<0.001). CIN (+) group had significantly lower eGFR when compared with the CIN (-) group (84.7±28.9 mL/min/1.73m2 vs 93.3±22.2 mL/min/1.73m2; p=0.005). There was no difference between the groups in terms of other laboratory findings. Besides, sST2 groups are compared in Table 1 in terms of process characteristics. There was no difference according to culprit coronary vessels and the number of stents. The procedure time was statistically longer in patients who developed CIN (32.8±17.4 min vs 25.4±13.5 min; p<0.001). The total amount of contrast media used during the procedure was higher in CIN (+) patients, but this difference was not statistically significant (225.7±79.6 mL vs 208±69.2 mL; p=0.065) (Table 1).
The median sST2 level of all patients was 36.6 ng/mL. Patients with CIN (+) had higher sST2 levels than patients with CIN (-) (40.6±21.0 ng/mL vs.31.5±13.0 ng/mL; p<0.001) (Table 1). And, 51.9% of CIN (+) patients had sST2 levels above the prognostic cut-off point of 35 ng/mL. Also, CIN (-) patients only had 28.6% with sST2 levels above 35 ng/mL, and the difference between the groups was statistically significant (p<0.001) (Figure 1).
 |
Figure 1 Numbers and percentages of CIN (+) and CIN (−) groups according to baseline sST2 cut-off value of >35 ng/mL. |
Statistical analysis of ROC showed that admission sST2 above a cut-off 32.1 ng/mL had 62.9% specificity with 64.2% sensitivity for the prediction of CIN development (area under the curve: 0.642, 95% confidence interval: 0.590–0.692; p<0.001) (Figure 2). DM, lower eGFR, lower SBP, total procedure time, and contrast volume were independent risk factors for the development of CIN in the logistic regression analysis. Additionally, the patient in the high sST2 group is three times more at risk for CIN development than the patient in the low sST2 group, regardless of other risk factors (Table 2). It was also investigated whether sST2 is an independent predictor of CIN according to the criteria suggested by the Journal of the American College of Cardiology, and sST2 was found statistically significant to predict CIN in regression analysis (OR: 2.802; 95% CI, 1.511–5.193; p<0.001).
 |
Table 2 Multivariate Logistic Regression Analysis for Potential Predictors of Contrast-Induced Nephropathy |
 |
Figure 2 Receiver operating characteristics curve showing the distinguishing ability of soluble ST2 level for contrast-induced nephropathy. |
Discussion
There were three major findings of this study. First, the levels of sST2 in CIN (+) patients were higher than that of CIN (-) patients. Second, DM, eGFR, SBP, total procedure time, and sST2 are independent predictors for the development of CIN in STEMI patients who are treated with PCI. Finally, regardless of other risk factors, patients with high sST2 levels on admission have 3.06 times more increased risk for CIN development than patients with low sST2 levels. This study is the first in the literature to identify such a relationship between sST2 and CIN in PCI-administered STEMI patients. This study proves that serum sST2 levels on admission are significantly associated with CIN and can be used to predict CIN development.
PCI is widely used and very important in the treatment of STEMI patients.11 It provides normal blood flow in the culprit artery in STEMI patients, resulting in a regression of symptoms and a better prognosis.12 The development of CIN after PCI in STEMI patients is relatively rare, but could be a fatal complication when it develops.13 A sudden deterioration in renal function following the application of an iodinated contrast agent is known as CIN.14 The reduction in renal function is usually mild and peaks in 2–3 days. In general, renal functions return to their initial values within 1–3 weeks.14 The long-used universal definition of CIN is known as “an increase in creatinine level of ≥0.5 mg/dl, or 25% from baseline within 2–5 days after exposure to contrast”.15 Recently, the Kidney Disease Improving Global Outcomes (KDIGO) provided an updated definition, which is currently the most commonly used. KDIGO defines CIN as a creatinine level increase of ≥0.3 mg/dl (26.5 μmol/l) above initial value within 48 hours of contrast media exposure, or an increase of at least 1.5 times the initial value within 7 days.16 In previous studies, the reported incidence of CIN after PCI was observed between 2% and 25% of the patients.17 In our study, the incidence of CIN was observed in 22.7%. The high incidence of CIN in our study may be related to the risk profile of patients.
sST2 is a member of an interleukin-1 receptor group with transmembrane (ST2L) and soluble isoforms (sST2). Although sST2 is secreted by cardiomyocytes, vascular endothelial cells have also been shown to be a source of sST2.18 IL-33 has positive effects on myocardial functions, hypertrophy, apoptosis, and fibrosis. Circulating sST2 binds IL-33, thus, the positive effects of IL-33 have been removed.19 sST2 can be seen as a precursor of inflammation, remodeling, and fibrosis, and is recently considered to be a new and promising biomarker in the assessment of prognosis for heart failure, acute coronary syndrome, and cardiovascular disease.20 sST2 may be considered as a new prognostic marker because it has a poor association with known prognostic risk factors such as cardiac troponin, Killip class, C-reactive protein, and comorbidity.21 Moreover, sST2 plays a role in the induction of allergic inflammation. In particular, this pathway is thought to be one of the primary pathways responsible for allergic diseases such as asthma, atopic dermatitis, and autoimmune diseases such as rheumatoid arthritis.22 The IL-33/sST2 pathway has been shown to induce IL-6 and IL-8 and cause systemic inflammation in a study on smoke-induced COPD mice.23 Besides, in another study, the increase in IL 6 via ST2 receptor in human arterial endothelial cells may have a role in the pathophysiology of idiopathic pulmonary arterial hypertension.24 Moreover, most studies have described sST2 as an independent marker of renal function and hemodialysis.25 Guerchicoff et al investigated the relationship between acute renal injury and various biochemical parameters such as adiponectin, B-type natriuretic peptide (BNP), chemokine ligand, cystatin C, endothelial cell-selective and adhesion molecule in STEMI patients.26 Between these biomarkers, only BNP levels were found to be significant predictors for CIN development. Furthermore, heart failure patients with an initial sST2 value >35 ng/mL are considered to indicate poorer prognosis. When we group our study population based on this threshold value, 51.9% of CIN (+) patients had sST2 levels >35 ng/mL, while only 28.6% of CIN (−) patients had sST2 levels >35 ng/mL in our study and the difference between the groups was found statistically significant (p<0.001).
Iodized contrast media causes renal damage for many mechanical reasons; however, the causes of CIN development have been constantly questioned in clinical medicine. The contrast agent has a direct nephrotoxic effect on tubular epithelial cells, leading to osmotic nephrosis and reduced oxygen conduction. Besides, these agents induce the release of vasoactive molecules such as endothelin, adenosine, and reduce the availability of vasodilators such as prostaglandins and nitric oxide. Thus, it causes vasoconstriction and ischemic damage.27,28 These pathophysiological changes cause oxidative stress and increased cell damage. The renal medulla with relatively low partial oxygen pressure is particularly vulnerable to vascular changes.29 The other hypothesis for CIN is the hypoxic and toxic damage caused by reactive oxygen derivatives.30 It has been stated that during myocardial infarction (MI), kidney oxygenation may decrease, and subsequent medullar hypoxia may trigger oxidative stress.31 Moreover, vasoconstriction in the renal artery and consequent decrease in renal blood flow can also trigger CIN with a contrast agent.32 Damage after MI can trigger the production of sST2 from neighboring cells. As a protective mechanism, sST2 can be involved in the regulation of the synthesis of proinflammatory cytotoxins from macrophages to prevent uncontrolled inflammatory reactions.33 SST2, which rises rapidly with acute cardiovascular stress, maybe triggering these pathways. In addition, the increase in sST2 due to the pressure occurring in the heart wall may trigger these conditions in the renal pathways. In our study, parallel to the pathophysiological mechanisms mentioned above, patients with high sST2 levels were three times more likely to develop CIN than patients with low sST2 levels.
Some risk factors claimed to be associated with CIN were examined in our study apart from sST2. The most prominent of these risk factors were DM, chronic renal failure (CRF), advanced age, and hemodynamic status at presentation. In our study, DM, CRF, and SBP at presentation were found as independent predictors of CIN. The high prevalence of kidney disease in DM patients may also have been effective in this situation. In terms of CRF, it was observed that GFR <30 mL/min/1.73 m2 increased CIN three times.34 We found that GFR <60 mL/min/1.73 m2 could predict the presence of CIN at a rate of 4.4 times. It has been reported that sSt2 levels are associated with diabetes and renal dysfunction in patients with heart failure who are hemodynamically stable.35 These diseases may trigger CIN through pathways similar to sST2. Also, in the Framingham Heart Study, it was stated that the sST2 level was associated with systolic blood pressure and diabetes in a healthy population. In previous studies, it was stated that the volume of contrast may affect renal function,11 but we did not find a difference between CIN (+) and CIN (−) groups according to contrast amount. Even so, minimizing the volume of contrast media used whenever possible are mandatory prevention measures.
There are several limitations to our study. First, the study included only STEMI patients in a single center with low population numbers. Secondly, we measured sST2 only once at the time on admission and did not observe potential changes in sST2 over time. Third, we do not know the long-term levels of sST2 after renal function has normalized in patients with contrast nephropathy. Moreover, inflammation markers such as C-reactive protein and erythrocyte sedimentation rate could not be added to the data because of the study design. Finally, since echocardiography was not performed in all patients, left ventricular systolic functions and the presence of heart failure could not be added to the statistical analysis.
Conclusion
CIN in STEMI patients is one of the most feared complications of primary percutaneous intervention. In this study, it was demonstrated for the first time that the sST2 levels at the admission of STEMI patients undergoing PCI were independently associated with the development of CIN. Evaluation of the sST2 level could help the clinician to take the necessary actions to prevent the development of CIN in STEMI patients. More studies are needed to understand the impact of sST2 on CIN development and to predict patients at high risk for CIN development.
Disclosure
The authors report no conflicts of interest for this work.
References
1. Duan C, Cao Y, Liu Y, et al. A new preprocedure risk score for predicting contrast-induced acute kidney injury. Can J Cardiol. 2017;33(6):714–723. doi:10.1016/j.cjca.2017.01.015
2. McCullough PA, Adam A, Becker CR, et al. CIN Consensus Working Panel. Epidemiology and prognostic implications of contrast-induced nephropathy. Am J Cardiol. 2006;98(6A):5K–13K.
3. Busch SV, Jensen SE, Rosenberg J, Gögenur I. Prevention of contrast-induced nephropathy in STEMI patients undergoing primary percutaneous coronary intervention: a systematic review. J Interv Cardiol. 2013;26(1):97–105. doi:10.1111/joic.12007
4. Wi J, Ko YG, Kim JS, et al. Impact of contrast-induced acute kidney injury with transient or persistent renal dysfunction on long-term outcomes of patients with acute myocardial infarction undergoing percutaneous coronary intervention. Heart. 2011;97(21):1753–1757. doi:10.1136/hrt.2010.218677
5. Goldenberg I, Matetzky S. Nephropathy induced by contrast media: pathogenesis, risk factors and preventive strategies. CMAJ. 2005;172(11):1461–1471. doi:10.1503/cmaj.1040847
6. Salvagno GL, Pavan C. Prognostic biomarkers in acute coronary syndrome. Ann Transl Med. 2016;4(13):258. doi:10.21037/atm.2016.06.36
7. Mueller T, Jaffe AS. Soluble ST2-analytical considerations. Am J Cardiol. 2015;115(7):8B–21B. doi:10.1016/j.amjcard.2015.01.035
8. Sabatine MS, Morrow DA, Higgins LJ, et al. Complementary roles for biomarkers of biomechanical strain ST2 and N-terminal prohormone B-type natriuretic peptide in patients with ST-elevation myocardial infarction. Circulation. 2008;117(15):1936–1944. doi:10.1161/CIRCULATIONAHA.107.728022
9. Somuncu MU, Akgun T, Cakir MO, et al. The elevated soluble ST2 predicts no-reflow phenomenon in ST-elevation myocardial infarction undergoing primary percutaneous coronary intervention. J Atheroscler Thromb. 2019;26(11):970–978. doi:10.5551/jat.48413
10. Mehran R, Aymong ED, Nikolsky E, et al. A simple risk score for prediction of contrast-induced nephropathy after percutaneous coronary intervention: development and initial validation. J Am Coll Cardiol. 2004;44:1393–1399.
11. Marenzi G, De Metrio M, Rubino M, et al. Acute hyperglycemia and contrast-induced nephropathy in primary percutaneous coronary intervention. Am Heart J. 2010;160(6):1170–1177. doi:10.1016/j.ahj.2010.09.022
12. Levine GN, Bates ER, Blankenship JC, et al. 2011 ACCF/AHA/SCAI guideline for percutaneous coronary intervention. A report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines and the Society for Cardiovascular Angiography and Interventions. J Am Coll Cardiol. 2011;58(24):2550–2583. doi:10.1016/j.jacc.2011.08.006
13. Liu Y, Chen JY, Huo Y, et al.; RESCIND group. Aggressive hydraTion in patients with ST-Elevation Myocardial infarction undergoing Primary percutaneous coronary intervention to prevenT contrast-induced nephropathy (ATTEMPT): study design and protocol for the randomized, controlled trial, the ATTEMPT, RESCIND 1 (First study for reduction of contraSt-induCed nephropathy followINg carDiac catheterization) trial. Am Heart J. 2016;172:88–95.
14. Ali A, Bhan C, Malik MB, Ahmad MQ, Sami SA. The prevention and management of contrast-induced acute kidney injury: a mini-review of the literature. Cureus. 2018;10(9):e3284.
15. Van der Molen AJ, Reimer P, Dekkers IA, et al. Post-contrast acute kidney injury- Part 1: definition, clinical features, incidence, role of contrast medium and risk factors: recommendations for updated ESUR Contrast Medium Safety Committee guidelines. Eur Radiol. 2018;28(7):2845–2855. doi:10.1007/s00330-017-5246-5
16. Fliser D, Laville M, Covic A, et al.; Ad-hoc working group of ERBP. A European Renal Best Practice (ERBP) position statement on the Kidney Disease Improving Global Outcomes (KDIGO) clinical practice guidelines on acute kidney injury: part 1: definitions, conservative management and contrast-induced nephropathy. Nephrol Dial Transplant. 2012;27(12):4263–4272. doi:10.1093/ndt/gfs375.
17. Firouzi A, Alemzadeh-Ansari MJ, Mohammadhadi N, et al. Association between the risks of contrast-induced nephropathy after diagnostic or interventional coronary management and the transradial and transfemoral access approaches. J Cardiovasc Thorac Res. 2020;12(1):51–55. doi:10.34172/jcvtr.2020.08
18. Bartunek J, Delrue L, van Durme F, et al. Nonmyocardial production of ST2 protein in human hypertrophy and failure is related to diastolic load. J Am Coll Cardiol. 2008;52(25):2166–2174. doi:10.1016/j.jacc.2008.09.027
19. Weinberg EO, Shimpo M, De Keulenaer GW, et al. Expression and regulation of ST2, an interleukin-1 receptor family member, in cardiomyocytes and myocardial infarction. Circulation. 2002;106(23):2961–2966. doi:10.1161/01.CIR.0000038705.69871.D9
20. Seo SM, Kim SH, Kim Y, Yoon HE, Shin SJ. Prognostic utility of soluble suppression of tumorigenicity 2 level as a predictor of clinical outcomes in incident hemodialysis patients. Int J Med Sci. 2018;15(7):730–737. doi:10.7150/ijms.23638
21. Jenkins WS, Roger VL, Jaffe AS, et al. Prognostic value of soluble ST2 after myocardial infarction: a community perspective. Am J Med. 2017;130(9):
22. Takatori H, Makita S, Ito T, Matsuki A, Nakajima H. Regulatory mechanisms of IL-33-ST2-mediated allergic inflammation. Front Immunol. 2018;9(9):2004. doi:10.3389/fimmu.2018.02004
23. Wu H, Yang S, Wu X, et al. Interleukin-33/ST2 signaling promotes production of interleukin-6 and interleukin-8 in systemic inflammation in cigarette smoke-induced chronic obstructive pulmonary disease mice. Biochem Biophys Res Commun. 2014;450(1):110–116. doi:10.1016/j.bbrc.2014.05.073
24. Shao D, Perros F, Caramori G, et al. Nuclear IL-33 regulates soluble ST2 receptor and IL-6 expression in primary human arterial endothelial cells and is decreased in idiopathic pulmonary arterial hypertension. Biochem Biophys Res Commun. 2014;451(1):8–14. doi:10.1016/j.bbrc.2014.06.111
25. Plawecki M, Morena M, Kuster N, et al. sST2 as a new biomarker of chronic kidney disease-induced cardiac remodeling: impact on risk prediction. Mediators Inflamm. 2018;2018:3952526. doi:10.1155/2018/3952526
26. Guerchicoff A, Stone GW, Mehran R, et al. Analysis of biomarkers for risk of acute kidney injury after primary angioplasty for acute ST-segment elevation myocardial infarction: results of the HORIZONS-AMI trial . Catheter Cardiovasc Interv. 2015;85(3):335–342. doi:10.1002/ccd.25620
27. Chandiramani R, Cao D, Nicolas J, Mehran R. Contrast-induced acute kidney injury. Cardiovasc Interv Ther. 2020;35(3):209–217. doi:10.1007/s12928-020-00660-8.
28. Azzalini L, Spagnoli V, Ly HQ. Contrast-induced nephropathy: from pathophysiology to preventive strategies. Can J Cardiol. 2016;32(2):247–255. doi:10.1016/j.cjca.2015.05.013
29. Heyman SN, Rosen S, Rosenberger C. Renal parenchymal hypoxia, hypoxia adaptation, and the pathogenesis of radiocontrast nephropathy. Clin J Am Soc Nephrol. 2008;3:288–296.
30. Heyman SN, Rosen S, Khamaisi M, Idée JM, Rosenberger C. Reactive oxygen species and the pathogenesis of radiocontrast-induced nephropathy. Invest Radiol. 2010;45:188–195.
31. Clanton TL. Hypoxia-induced reactive oxygen species formation in skeletal muscle. J Appl Physiol. 2007;102(6):2379–2388. doi:10.1152/japplphysiol.01298.2006
32. Tumlin JA, Wang A, Murray PT, Mathur VS. Fenoldopam mesylate blocks reductions in renal plasma flow after radiocontrast dye infusion: a pilot trial in the prevention of contrast nephropathy. Am Heart J. 2002;143:894–903.
33. Weir RA, Millar AM, Murphy GE, et al. Serum soluble ST2: a potential novel mediator in left ventricular and infarct remodeling after acute myocardial infarction. J Am Coll Cardiol. 2010;55(3):243–250. doi:10.1016/j.jacc.2009.08.047
34. Tsai TT, Patel UD, Chang TI, et al. Contemporary incidence, predictors, and outcomes of acute kidney injury in patients undergoing percutaneous coronary interventions: insights from the NCDR Cath-PCI registry. JACC Cardiovasc Interv. 2014;7(1):1–9. doi:10.1016/j.jcin.2013.06.016
35. Felker GM, Fiuzat M, Thompson V, et al. Soluble ST2 in ambulatory patients with heart failure: association with functional capacity and long-term outcomes. Circ Heart Fail. 2013;6(6):1172–1179. doi:10.1161/CIRCHEARTFAILURE.113.000207
 © 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
© 2020 The Author(s). This work is published and licensed by Dove Medical Press Limited. The full terms of this license are available at https://www.dovepress.com/terms.php and incorporate the Creative Commons Attribution - Non Commercial (unported, v3.0) License.
By accessing the work you hereby accept the Terms. Non-commercial uses of the work are permitted without any further permission from Dove Medical Press Limited, provided the work is properly attributed. For permission for commercial use of this work, please see paragraphs 4.2 and 5 of our Terms.
